Abstract
Chronic kidney disease (CKD) is a world-wide public health problem, with adverse outcomes of kidney failure, cardiovascular disease, and premature death. This finding has led to the hypothesis that earlier recognition of kidney disease and successful intervention may improve outcome. The National Kidney Foundation, through its Kidney Disease Outcomes Quality Initiative (K/DOQI), and other National institutions recommend glomerular filtration rate (GFR) for the definition, classification, screening, and monitoring of CKD. Blood creatinine clearance, the most widely used clinical marker of kidney function, is now recognized as an unreliable measure of GFR because serum creatinine is affected by age, weight, muscle mass, race, various medications, and extra-glomerular elimination. Cystatin C concentration is a new and promising marker for kidney dysfunction in both native and transplanted kidneys. Because of its low molecular weight, cystatin C is freely filtered at the glomerulus and is almost completely reabsorbed and catabolized, but not secreted, by tubular cells. Given these characteristics, cystatin C concentration may be superior to creatinine concentration in detecting chronic kidney disease. This review aims to evaluate from recent literature the clinical efficiency and relevance of these GFR markers in terms of screening CKD.
Kidney function; Glomerular filtration rate; Creatinine; Cystatin C; Measurement and calculation of GFR
Braz J Med Biol Res, June 2010, Volume 43(6) 528-536
Monitoring renal function: measured and estimated glomerular filtration rates - a review
 Correspondence and Footnotes
Correspondence and Footnotes
 J.V. Salgado1, F.A. Neves1, M.G. Bastos2, A.K. França3, D.J. Brito3, E.M. Santos3 and N. Salgado Filho4
J.V. Salgado1, F.A. Neves1, M.G. Bastos2, A.K. França3, D.J. Brito3, E.M. Santos3 and N. Salgado Filho4
1Departamento de Ciências Farmacêuticas, Faculdade de Ciências da Saúde, Universidade de Brasília, Brasília, DF, Brasil
2Universidade Federal de Juiz de Fora, Juiz de Fora, MG, Brasil
3Setor de Nefrologia, Hospital Universitário Presidente Dutra, 4Departamento de Medicina I, Faculdade de Medicina, Universidade Federal do Maranhão, São Luís, MA, Brasil
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Abstract
Chronic kidney disease (CKD) is a world-wide public health problem, with adverse outcomes of kidney failure, cardiovascular disease, and premature death. This finding has led to the hypothesis that earlier recognition of kidney disease and successful intervention may improve outcome. The National Kidney Foundation, through its Kidney Disease Outcomes Quality Initiative (K/DOQI), and other National institutions recommend glomerular filtration rate (GFR) for the definition, classification, screening, and monitoring of CKD. Blood creatinine clearance, the most widely used clinical marker of kidney function, is now recognized as an unreliable measure of GFR because serum creatinine is affected by age, weight, muscle mass, race, various medications, and extra-glomerular elimination. Cystatin C concentration is a new and promising marker for kidney dysfunction in both native and transplanted kidneys. Because of its low molecular weight, cystatin C is freely filtered at the glomerulus and is almost completely reabsorbed and catabolized, but not secreted, by tubular cells. Given these characteristics, cystatin C concentration may be superior to creatinine concentration in detecting chronic kidney disease. This review aims to evaluate from recent literature the clinical efficiency and relevance of these GFR markers in terms of screening CKD.
Key words: Kidney function; Glomerular filtration rate; Creatinine; Cystatin C; Measurement and calculation of GFR
Introduction
Introduction
Measures of renal function are important and commonly used in most clinical investigations. Their routine use is growing rapidly with the increasing incidence of chronic kidney disease (CKD) worldwide as a consequence population aging and the increasing incidence of type 2 diabetes mellitus (1). Consequently, patients with diabetes and hypertension are particularly affected by CKD. Most patients with CKD are identified or treated after a considerable delay. Early identification is critical to delay progression of CKD to end-stage renal disease and to reduce the cardiovascular co-morbidity associated with CKD. At the first meeting for the global definition and classification of CKD in 2004, the independent international Kidney Disease Improving Global Outcome organization (KDIGO) recommended two laboratory tests for an earlier detection of CKD. The tests are: estimation of glomerular filtration rate (GFR), which is the best estimate of renal function, using the simplified equation derived from the Modification of Diet in Renal Disease Study (MDRD), and determination of proteinuria, preferably microalbuminuria, corrected for urinary creatinine. On this basis, kidney damage is defined as structural or functional abnormalities of the kidney, with or without decreased GFR. Markers of kidney damage include proteinuria, abnormalities of urine sediment, abnormalities in imaging tests, or abnormalities of blood or urine composition specific for certain syndromes. CKD is also defined if the GFR is <60 mL·min-1·1.73 (m2)-1 for ≥3 months, with or without kidney damage. These recommendations have been accepted by the nephrological community (2,3).
Measurement of glomerular filtration rate
GFR cannot be measured directly in humans. Rather, it has to be determined by measuring the clearance of an ideal filtration marker, which has to be freely filtered at the level of the glomerulus. Thus, the molecular weight of such a marker has to be low and the compound must not bind to plasma proteins. The ideal marker must be able to achieve a stable plasma concentration, is not reabsorbed, secreted or metabolized by the kidney, is physiologically inert and does not alter renal function. Inulin, an uncharged polymer of fructose derived from plant tubers, fulfills these requirements. Thus, inulin may be regarded as the gold standard against which other filtration markers for estimating GFR must be compared. However, the use of inulin requires continuous intravenous infusion and multiple, timed urine collections. Furthermore, inulin measurement is expensive, cumbersome and not routinely available. Consequently, inulin is only used in research studies when very accurate estimation of renal function is necessary (4,5).
Several compounds labeled with radioisotopes have been used for the assessment of GFR. Among these are 125I-iothalamate, 99mTc-diethylenethiaminepenta-acetic acid (99mTc-DTPA) and 51Cr-ethylenediaminetetra-acetic acid (51Cr-EDTA). All these compounds have a low molecular weight and their plasma protein binding and extrarenal elimination are negligible. Thus, they fulfill some of the essential criteria for a renal filtration marker. The advantage of these substances is their ease of administration as well as the simplicity, accuracy and precision of their measurement. These properties enable the plasma concentration vs time curve after intravenous bolus injection to be measured easily. From the plasma concentration vs time curves obtained, the total body clearance can be calculated using pharmacokinetic models, usually a two-compartment model. Intrinsic to the use of radioactive substances is concern about radiation exposure, particularly in women of child-bearing age. For the substances mentioned above, the whole body radiation exposure achieved as a result of a single clearance measurement with the usual doses is lower than that received from a chest X-ray (4,5). Another important consideration is that the terminal elimination phase is significantly prolonged in advanced renal failure. In patients with moderate renal failure (GFR between 30 and 59 mL/min) samples are taken for up to 5 h after injection, whereas in patients with advanced renal failure samples are required for up to 24 h after injection (6).
Although they are adequate methods, they are less accurate than two- and multiple-sample methods and may lead to a substantial systematic error in GFR of up to 10-15 mL/min (4,5). 99mTc-DTPA is a chelate of DTPA and 99mTc. The urinary clearance of 99mTC-DTPA is similar to the urinary clearance of 125I-iothalamate. However, plasma clearance of 99mTc-DTPA exceeds the urinary clearance of the compound by a large and variable amount. Furthermore, the plasma clearance of 99mTc-DTPA exceeds the urinary clearance of inulin. These data suggest that extrarenal elimination of 99mTc-DTPA occurs, thus limiting its use as a marker of glomerular filtration. 51Cr-EDTA is a chelate of 51Cr and EDTA. Plasma clearance of 51Cr-EDTA measured after intravenous bolus injection of the compound exceeds its urinary clearance by an average of 6 mL/min. This may be the result of extrarenal elimination of the substance. The error induced by this phenomenon is particularly relevant at low levels of GFR. No difference was found between the plasma clearance of 51Cr-EDTA and of 99mTc-DTPA (4,5).
On the other hand, Medeiros et al. (7) recently showed that plasma clearance of 51Cr-EDTA (51Cr-EDTA-Cl) is a precise and accurate measurement of GFR in renal transplant recipients compared with renal inulin clearance (In-Cl). Using the Bland and Altman statistical approach to quantify the agreement between In-Cl and 51Cr-EDTA-Cl, these investigators found that two plasma samples for 51Cr-EDTA-Cl taken at 4 and 8 h or at 4 and 6 h presented narrow limits of agreement (-8.5 to +14.1 and -8.9 to +14.3) and a difference (bias) of 2.8 and 2.7 mL/min, respectively (7).
125I-iothalamate has been used as a glomerular filtration marker both in normal subjects and in patients with various degrees of renal insufficiency. In a comparative study, the plasma clearance of 125I-iothalamate was significantly higher than the clearance of 51Cr-EDTA. The difference in clearance was reduced by pretreatment of the patients with probenecid, suggesting that tubular secretion of 125I-iothalamate occurs. Additional studies of the urinary clearances measured during constant infusion showed that the urinary clearance of unlabeled iothalamate substantially exceeded the urinary clearance of inulin in healthy volunteers (8). These data suggest that in humans renal tubular secretion of iothalamate involves an organic anion transport system. This assumption is supported by the finding that the urinary clearance of 125I-iothalamate exceeds the inulin clearance in subjects with normal kidney function (9). Thus, 125I-iothalamate appears not to be an ideal marker of glomerular filtration. Iothalamate may also be used without the radioactive label. Its plasma levels can then be assessed by high performance liquid chromatography (10) or X-ray fluorescence (11). Taken together, the available radioisotopic filtration markers are inferior to inulin for the estimation of GFR. However, plasma clearances determined using these compounds may be sufficiently accurate for clinical practice and for the follow-up of GFR over time.
Creatinine
Serum creatinine (SCr) concentration is the most widely used test of renal function outside specialist hospital settings. Nevertheless, several drawbacks have been identified when SCr is used as a marker of GFR: first, proximal tubular cell secretion plays an important role in creatinine elimination with declining GFR. Therefore, SCr concentrations can be within the normal range even with a GFR around 60 mL·min-1·1.73 (m2)-1 suggesting the existence of a so-called creatinine blind range (12). Second, the endogenous production of creatinine is primarily determined by muscle mass and dietary intake, which probably accounts for the variations in SCr levels observed among different age, geographic, ethnic, and racial groups (13). Additionally, creatinine production varies considerably intra- and inter-individually. This explains the high inter-individual variability of SCr, which precludes a reliable estimation of kidney function from a single creatinine measurement without additional patient data (12). As a result, SCr has poor sensitivity for renal failure or mild renal dysfunction, particularly in children, malnourished or elderly patients and severely ill patients, with consequent under-recognition of renal impairment (14,15).
In addition to these problems related to creatinine metabolism, creatinine measurement is still technically imprecise. Several different methods have been developed, with the Jaffé method being the most commonly used. This assay is hindered by the interference of non-creatinine chromogens (e.g., acetic acid, acetone, pyruvate, glucose, ascorbic acid, bilirubin) of up to 20%. With the development of enzymatic assays, the problem of interferences has been reduced but not eliminated. The development of a reference method using isotope dilution mass spectrometry has facilitated the measurement of true creatinine (16). However, this method is available in only a few highly specialized laboratories.
Creatinine clearance (CrCl) is calculated by determining creatinine concentration in timed urine collections and simultaneously in blood using the following equation: CrCl (mL/min) = [urinary creatinine (mg/dL) x urine volume (mL/min)] / [SCr (mg/dL)]. CrCl is considered to be the clinical gold standard endogenous method, better than determination of SCr levels, although it is necessary to remember the tubular secretion of creatinine, which may increase creatinine excretion more than 2-fold with declining GFR. Additionally, the measurement of creatinine clearance requires a timed urine collection, which has proven to be laborious, inconvenient and prone to collection failure, especially in the elderly and children (17).
Equations estimating GFR based on creatinine
To circumvent the practical difficulties of CrCl and to compensate for interindividual variation in creatinine production, several equations have been developed, which include anthropometric factors such as gender, age, ethnicity, and body size. On the whole, these formulae were calculated by regression analysis in large reference populations predominantly consisting of patients with chronic kidney disease and reduced GFR, who underwent a gold standard measurement of GFR. In this setting, the sensitivity to detect CKD may be markedly improved. Two formulae are widely used in clinical practice for adults: the Cockcroft-Gault (C-G) equation (18) and, more recently, the equation developed by the MDRD (19).
The C-G formula was developed in 1973 on the basis of data from 249 patients with CrCl from 30 to 130 mL/min. It actually estimates CrCl and is not normalized to 1.73 m2 body surface area. The C-G equation was developed using an outdated Jaffé assay for creatinine measurement (18) and should be considered with caution when calculated using current creatinine measurements, which more closely reflect true creatinine. Additionally, the C-G formula also leads to an underestimation of GFR in the elderly (20).
The MDRD study equation was presented in 1999 on the basis of data from 1628 adult patients with CKD. It estimates GFR adjusted for body surface area and was proposed by Levey et al. (19) using age, gender, ethnicity, SCr, urea nitrogen, and albumin. By avoiding inclusion of weight, the formula is less prone to errors from fluid overload and obesity. Subsequently, in 2000 a simplified version of the original MDRD equation was suggested, which performed as well as the original, but required only SCr, age, gender, and ethnicity (for African-American individuals) (21). With the exception of ethnicity, the other variables required are normally provided routinely when a sample is submitted to the clinical laboratory. It is therefore much easier for laboratories to report estimated GFR using this formula.
However, certain patient groups were not well represented in the MDRD study sample. The MDRD study predominantly included Caucasian, non-diabetic patients with a stable GFR of about 40 mL·min-1·1.73 (m2)-1 (19). Therefore, clearance measurements are still required for groups who were underrepresented in the original MDRD sample in order to fully validate the formula for all patients. These include: patients at extremes of age and body size; severely malnourished or obese patients; patients with skeletal muscle diseases, paraplegia, or quadriplegia, vegetarians, and those with rapidly changing kidney function as in acute kidney injury (19,22). The MDRD and C-G equations are inappropriate to estimate GFR in healthy individuals and have been shown to clearly underestimate GFR above approximately 60 mL·min-1·1.73 (m2)-1 and to markedly overestimate GFR below approximately 20 mL·min-1·1.73 (m2)-1 and in nephrotic range proteinuria (23-25).
Recently, Cirillo et al. (26) demonstrated that the accuracy of the MDRD formula tends to decrease in the elderly and/or in patients with body mass index <21 and >30 when compared to C-G. Both MDRD and C-G equations have been reported to be less accurate in populations without chronic kidney disease, such as young patients with type 1 diabetes without microalbuminuria and potential kidney donors (27,28). Some studies report the higher accuracy of the MDRD equation (26) while others report similar performance of MDRD and C-G equations (29).
In the MDRD study, creatinine was determined and calibrated by the kinetic Jaffé method and, similar to the C-G equation, a substantial error is introduced when the MDRD equation is used with different creatinine assays or standards. Calibration of serum creatinine assays to adjust for these differences is not standardized across laboratories, leading to substantial variation in values reported among laboratories, and, consequently, in error of the estimative of GFR by MDRD and C-G equations (30). The simplified MDRD equation was recalculated with SCr measurements calibrated for an enzymatic assay traceable to an isotope dilution mass spectrometry assay (traceable) and was found to provide an accurate GFR in patients with CKD, better than the C-G equation. However, differences between measured and estimated GFR were greater when the estimated GFR was 60 mL·min-1·1.73 (m2)-1 or greater (31).
Recently, Levey et al. (32) developed a new equation, the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, to predict GFR from SCr in adults by using a database pooled from 8254 participants in 10 studies. They showed that the CKD-EPI equation is more accurate than the commonly used MDRD equation and has lower bias, mainly for estimated GFR greater than 60 mL·min-1·1.73 (m2)-1, although precision remains suboptimal. However, the CKD-EPI equation has some limitations such as the fact that few participants had GFR in the normal range and were older than 70 years, with incomplete data about clinical conditions and medications that might affect SCr independent of GFR. In addition, this equation is more complex than the MDRD equation and does not overcome the limitations of serum creatinine, which is dependent on muscle mass, dietary intake, and extrarenal elimination (Table 1) (32).
Nevertheless, the simplified MDRD equation has been included as the primary GFR marker in the Practice Guidelines for Chronic Kidney Disease, published in 2002 by the Kidney Disease Outcomes Quality Initiative of the National Kidney Foundation (K/DOQI) and the more recent KDIGO guidelines. Moreover, the staging of CKD proposed by the K/DOQI and KDIGO is based on GFR estimated by the simplified MDRD (Table 2) (2,33).
Cystatin C as a marker of GFR
Since 1985, cystatin C (CysC) has been described as a promising endogenous marker of GFR for both adults and children. It has a low molecular weight (13 kDa) and is a member of the superfamily of cysteine protease inhibitors. The gene for CysC is expressed in all nucleated cells, has the characteristics of a housekeeping gene and is thought to be produced and secreted at a constant rate (34). The function of CysC seems to be to protect connective tissue from destruction by intracellular enzymes. It may also have an antibacterial or antiviral function (35). CysC is a nonglycosylated protein that is freely filtered at the glomerulus level and almost completely reabsorbed and catabolized, but not secreted, by the tubular epithelial cells. Since only small amounts are excreted into the urine, its urinary clearance cannot be measured. Therefore, the blood concentration of CysC depends almost entirely on the GFR and is not substantially affected by diet or nutritional status (34). Also, CysC is independent of lean muscle mass, age or gender, and does not have a circadian rhythm (15,34,36). Because of these properties, many investigators have proposed that serum concentrations of CysC could serve as an improved index of GFR compared with SCr (34).
Nonetheless, some circumstances can interfere with serum CysC levels. Large doses of glucocorticoids (500 mg methylprednisolone) have been shown to increase serum CysC concentration (37). On the other hand, low or medium doses of glucocorticoids (20 to 60 mg/day prednisone) do not affect serum CysC concentration (38). Additionally, thyroid function has an impact on CysC production since, as compared with the euthyroid state, CysC levels are lower in the hypothyroid and higher in the hyperthyroid state (39). Therefore, thyroid function has to be considered when CysC is used as a marker of renal function. Moreover, more recently it was demonstrated that serum levels of CysC increase independently of GFR in patients with malignancies (40).
It is interesting to note that, contrary to creatinine measurement, which is limited by interferences with other chromogens, cystatin C determination does not have any interfering factors, except excessively elevated rheumatoid factor in vitro (41). Knight et al. (42) found that CysC concentrations were significantly associated with increased age, male gender, increased weight and height, current smoking, and higher C-reactive protein levels, even after adjustment for creatinine clearance (42). Nevertheless, it is known that the use of CrCl rather than GFR as the gold standard in this study (42) limits the credibility of its conclusions. This is because the study could not distinguish whether CysC or CrCl was the better measure of GFR. Regarding reference values, there are no relevant differences between men and women. In healthy children, CysC concentration stabilizes from the second year of life and the reference range is identical to that of adults. Higher CysC concentrations reflect maturation of glomerular filtration in neonates and are related to decreased kidney function in the elderly (34). CysC may identify the gradient of kidney function among persons who do not meet conventional definitions of clinical kidney disease. Therefore, the term "preclinical kidney disease" was proposed to represent persons with estimated GFR >60 mL/min, but who have abnormal concentrations of CysC (≥1.0 mg/L) (43).
Several cross-sectional studies have shown that CysC has greater sensitivity in detecting mildly reduced GFR in CKD than creatinine and other low molecular weight proteins (44). Serum CysC concentration increases already with mildly reduced GFR of 70 to 90 mL/min, i.e., in the "creatinine-blind range" (45). In addition, recent longitudinal studies have reported that CysC concentrations increase earlier in acute kidney injury in the intensive care unit, in cardiac surgery, coronary angiography, after liver transplantation, cisplatin chemotherapy, following uninephrectomy, and in progressive diabetic nephropathy (46). The performance of CysC as an estimator of GFR has been evaluated in large diverse populations such as geriatric patients (20), pediatric patients with various renal diseases (34), renal transplant recipients, type I and type II diabetics, patients with mild to moderate CKD of non-diabetic origin (44), patients with severe liver or neuromuscular disease, cancer patients, and women with preeclampsia (34,46).
Many investigators have reported the divergence between elevated CysC concentrations compared to creatinine in elderly patients. Hojs et al. (47), studying 144 older patients, found a significant correlation between 51Cr-EDTA clearance and SCr, serum CysC, the reciprocal of serum creatinine (C-G) and the reciprocal of serum CysC, as well as CrCl. However, they found that the correlation between 51Cr-EDTA clearance and serum CysC was significantly better than the correlation with SCr (P < 0.05). Likewise, the correlation between 51Cr-EDTA clearance and the reciprocal of serum CysC was superior to that with the reciprocal of serum creatinine (P < 0.003) and calculated CrCl (P < 0.003). These results show that serum CysC is a more reliable marker of GFR in the elderly than SCr or CrCl (47). Several investigators have also concluded that CysC performed better than creatinine and even better than equations estimating GFR based on creatinine as a GFR marker, although others have found no difference (44).
Schuck et al. (48), in a study of patients with CKD (GFR <40 mL·min-1·1.73 (m2)-1), did not observe the superiority of CysC over SCr, even though these patients had a significant increase in tubular secretion of creatinine from residual functioning nephrons. Therefore, to investigate whether CysC has any advantage over SCr, Dharnidharka et al. (49) performed a meta-analysis that summarized 46 articles published up to December 31, 2001, plus eight unpublished abstracts containing data on nearly 4500 subjects. Their findings confirmed that serum CysC is clearly superior to SCr for the detection of impaired GFR in cross-sectional studies. Roos et al. (44) performed a systematic review (January 1984-February 2006) considering only cross-sectional, cohort and case-control studies. They compared the diagnostic accuracy of CysC with that of SCr in 2007 patients from 27 study population groups with renal dysfunction between 60 and 90 mL/min. The results indicated that CysC had higher pooled sensitivity (81%) and similar specificity (88%) compared to SCr (69 and 88%, respectively). In this meta-analysis, the diagnostic accuracy of impaired renal function favored CysC (44).
Interestingly, although CysC measurement is probably accepted to be superior to creatinine to measure GFR, an important limitation of the use of CysC in routine assessment of kidney function is that CysC reference ranges vary widely when different cystatin assays are used. There are two major methods to measure CysC: particle-enhanced immunoturbidimetric immunoassay (PETIA) and immunonephelometric immunoassay (PENIA). Another method, ELISA kit, has a poor performance in terms of the analytical coefficient of variation compared to PETIA and PENIA (50).
Equations estimating GFR based on cystatin C
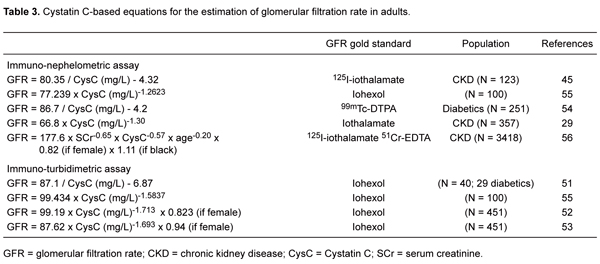
Several groups have recently developed equations to calculate GFR from serum CysC using approaches similar to those described for creatinine. In contrast to the MDRD equation, which was calculated from a large population in a multicenter study, CysC-based equations were created and validated in smaller samples using different gold standard measurements for GFR. Table 3 highlights some CysC-based equations for adults (29,45,51-56).
Many investigators have compared CysC-based equations to the C-G and the simplified MDRD equations. The vast majority, when comparing bias, precision, accuracy, and correlation, found CysC-based equations to be superior to creatinine-based equations as estimates of GFR (45,52,53), while some reported no difference in the performance of CysC- and creatinine-based equations (29,54). Recently, in order to improve the estimated kidney function, estimated GFR equations were developed, which incorporate both SCr and CysC. These equations, including serum CysC in combination with SCr, age, gender and race, performed better compared to the creatinine- and CysC-based equations regarding to bias, precision and accuracy (57-59). In this setting, Stevens et al. (56) developed new equations to estimate GFR using serum CysC alone or serum CysC and SCr in combination with demographic variables. The individuals were screened for 3 CKD studies in the United States (N = 2980) and a clinical population was studied in Paris, France (N = 438). GFR was measured using urinary clearance of 125I-iothalamate in the US studies and 51Cr-EDTA in the Paris study. The equations were developed by using linear regression with log GFR, and the best-fitting equation included both creatinine and cystatin as well as age, gender and race (Table 3) (56). The studies also showed that serum CysC alone estimates GFR as accurately as SCr levels adjusted for age, gender and race. However, in another study, Urbaniak et al. (60) did not find any improvement in predicting GFR using this combination.
Conclusion
We conclude that serum CysC has greater sensitivity in detecting reduced GFR in CKD than serum creatinine. However, further studies are necessary to compare CysC concentrations and CysC-based equations with MDRD equation and to clarify which one can better detect small reductions in kidney function within the normal range. This is because their diagnostic accuracies are very similar. The determination of plasma CysC levels is more expensive than routine plasma creatinine determination and the absence of very significant advantages could explain its limited use in daily clinical practice. Therefore, before these CysC-based equations are included in routine clinical practice, they require standardization of the CysC assay, availability of cost-effective assays, further investigation in multiple clinical situations and prospective studies to determine whether the combination of creatinine and CysC in one prediction formula may yield superior results.
Creatinine-based equations for the estimation of glomerular filtration rate (GFR) in adults.
References
1. Meguid El Nahas A, Bello AK. Chronic kidney disease: the global challenge. Lancet 2005; 365: 331-340.
2. Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2005; 67: 2089-2100.
3. KDOQI. KDOQI clinical practice guidelines and clinical practice recommendations for diabetes and chronic kidney disease. Am J Kidney Dis 2007; 49 (2 Suppl 2): S12-154.
4. Blaufox MD, Aurell M, Bubeck B, Fommei E, Piepsz A, Russell C, et al. Report of the Radionuclides in Nephrourology Committee on renal clearance. J Nucl Med 1996; 37: 1883-1890.
5. Fleming JS, Zivanovic MA, Blake GM, Burniston M, Cosgriff PS. Guidelines for the measurement of glomerular filtration rate using plasma sampling. Nucl Med Commun 2004; 25: 759-769.
6. Tett SE, Kirkpatrick CM, Gross AS, McLachlan AJ. Principles and clinical application of assessing alterations in renal elimination pathways. Clin Pharmacokinet 2003; 42: 1193-1211.
7. Medeiros FS, Sapienza MT, Prado ES, Agena F, Shimizu MH, Lemos FB, et al. Validation of plasma clearance of 51Cr-EDTA in adult renal transplant recipients: comparison with inulin renal clearance. Transpl Int 2009; 22: 323-331.
8. Odlind B, Hallgren R, Sohtell M, Lindstrom B. Is 125I iothalamate an ideal marker for glomerular filtration? Kidney Int 1985; 27: 9-16.
9. Perrone RD, Steinman TI, Beck GJ, Skibinski CI, Royal HD, Lawlor M, et al. Utility of radioisotopic filtration markers in chronic renal insufficiency: simultaneous comparison of 125I-iothalamate, 169Yb-DTPA, 99mTc-DTPA, and inulin. The Modification of Diet in Renal Disease Study. Am J Kidney Dis 1990; 16: 224-235.
10. Boschi S, Marchesini B. High-performance liquid chromatographic method for the simultaneous determination of iothalamate and o-iodohippurate. J Chromatogr 1981; 224: 139-143.
11. Effersoe H, Rosenkilde P, Groth S, Jensen LI, Golman K. Measurement of renal function with iohexol. A comparison of iohexol, 99mTc-DTPA, and 51Cr-EDTA clearance. Invest Radiol 1990; 25: 778-782.
12. Perrone RD, Madias NE, Levey AS. Serum creatinine as an index of renal function: new insights into old concepts. Clin Chem 1992; 38: 1933-1953.
13. Jones CA, McQuillan GM, Kusek JW, Eberhardt MS, Herman WH, Coresh J, et al. Serum creatinine levels in the US population: third National Health and Nutrition Examination Survey. Am J Kidney Dis 1998; 32: 992-999 [Erratum, Am J Kidney Dis 2000; 35: 179].
14. Swedko PJ, Clark HD, Paramsothy K, Akbari A. Serum creatinine is an inadequate screening test for renal failure in elderly patients. Arch Intern Med 2003; 163: 356-360.
15. Baxmann AC, Ahmed MS, Marques NC, Menon VB, Pereira AB, Kirsztajn GM, et al. Influence of muscle mass and physical activity on serum and urinary creatinine and serum cystatin C. Clin J Am Soc Nephrol 2008; 3: 348-354.
16. Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, et al. Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin Chem 2006; 52: 5-18.
17. Horio M. Assessment of renal function - Up-to-date. Clinic All-round 2006; 55: 1203-1208.
18. Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31-41.
19. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999; 130: 461-470.
20. Burkhardt H, Bojarsky G, Gretz N, Gladisch R. Creatinine clearance, Cockcroft-Gault formula and cystatin C: estimators of true glomerular filtration rate in the elderly? Gerontology 2002; 48: 140-146.
21. Levey AS, Greene T, Kusek J, Beck G. A simplified equation to predict glomerular filtration rate from serum creatinine. J Am Soc Nephrol 2000; 11: 155A.
22. Jafar TH, Schmid CH, Levey AS. Serum creatinine as marker of kidney function in South Asians: a study of reduced GFR in adults in Pakistan. J Am Soc Nephrol 2005; 16: 1413-1419.
23. Poggio ED, Nef PC, Wang X, Greene T, Van Lente F, Dennis VW, et al. Performance of the Cockcroft-Gault and modification of diet in renal disease equations in estimating GFR in ill hospitalized patients. Am J Kidney Dis 2005; 46: 242-252.
24. Branten AJ, Vervoort G, Wetzels JF. Serum creatinine is a poor marker of GFR in nephrotic syndrome. Nephrol Dial Transplant 2005; 20: 707-711.
25. Rule AD, Gussak HM, Pond GR, Bergstralh EJ, Stegall MD, Cosio FG, et al. Measured and estimated GFR in healthy potential kidney donors. Am J Kidney Dis 2004; 43: 112-119.
26. Cirillo M, Anastasio P, De Santo NG. Relationship of gender, age, and body mass index to errors in predicted kidney function. Nephrol Dial Transplant 2005; 20: 1791-1798.
27. Lin J, Knight EL, Hogan ML, Singh AK. A comparison of prediction equations for estimating glomerular filtration rate in adults without kidney disease. J Am Soc Nephrol 2003; 14: 2573-2580 [Erratum, J Am Soc Nephrol 2005; 16: 2814].
28. Ibrahim H, Mondress M, Tello A, Fan Y, Koopmeiners J, Thomas W. An alternative formula to the Cockcroft-Gault and the modification of diet in renal diseases formulas in predicting GFR in individuals with type 1 diabetes. J Am Soc Nephrol 2005; 16: 1051-1060.
29. Rule AD, Bergstralh EJ, Slezak JM, Bergert J, Larson TS. Glomerular filtration rate estimated by cystatin C among different clinical presentations. Kidney Int 2006; 69: 399-405.
30. Van Biesen W, Vanholder R, Veys N, Verbeke F, Delanghe J, De Bacquer D, et al. The importance of standardization of creatinine in the implementation of guidelines and recommendations for CKD: implications for CKD management programmes. Nephrol Dial Transplant 2006; 21: 77-83.
31. Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med 2006; 145: 247-254.
32. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF III, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604-612.
33. National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification and stratification. Am J Kidney Dis 2002; 39 (2 Suppl 1): S1-S266.
34. Filler G, Bokenkamp A, Hofmann W, Le Bricon T, Martinez-Bru C, Grubb A. Cystatin C as a marker of GFR - history, indications, and future research. Clin Biochem 2005; 38: 1-8.
35. Randers E, Erlandsen EJ. Serum cystatin C as an endogenous marker of the renal function - a review. Clin Chem Lab Med 1999; 37: 389-395.
36. Cimerman N, Brguljan PM, Krasovec M, Suskovic S, Kos J. Twenty-four hour variations of cystatin C and total cysteine proteinase inhibitory activity in sera from healthy subjects. Clin Chim Acta 2000; 291: 89-95.
37. Risch L, Herklotz R, Blumberg A, Huber AR. Effects of glucocorticoid immunosuppression on serum cystatin C concentrations in renal transplant patients. Clin Chem 2001; 47: 2055-2059.
38. Bokenkamp A, van Wijk JA, Lentze MJ, Stoffel-Wagner B. Effect of corticosteroid therapy on serum cystatin C and beta2-microglobulin concentrations. Clin Chem 2002; 48: 1123-1126.
39. Fricker M, Wiesli P, Brandle M, Schwegler B, Schmid C. Impact of thyroid dysfunction on serum cystatin C. Kidney Int 2003; 63: 1944-1947.
40. Nakai K, Kikuchi M, Fujimoto K, Kaneko Y, Omori S, Nakai K, et al. Serum levels of cystatin C in patients with malignancy. Clin Exp Nephrol 2008; 12: 132-139.
41. Lamb E, Stowe H. Rheumatoid factor can interfere with cystatin C measurement. Ann Clin Biochem 2003; 40: 195-196.
42. Knight EL, Verhave JC, Spiegelman D, Hillege HL, De Zeeuw D, Curhan GC, et al. Factors influencing serum cystatin C levels other than renal function and the impact on renal function measurement. Kidney Int 2004; 65: 1416-1421.
43. Shlipak MG, Praught ML, Sarnak MJ. Update on cystatin C: new insights into the importance of mild kidney dysfunction. Curr Opin Nephrol Hypertens 2006; 15: 270-275.
44. Roos JF, Doust J, Tett SE, Kirkpatrick CM. Diagnostic accuracy of cystatin C compared to serum creatinine for the estimation of renal dysfunction in adults and children - a meta-analysis. Clin Biochem 2007; 40: 383-391.
45. Hoek FJ, Kemperman FA, Krediet RT. A comparison between cystatin C, plasma creatinine and the Cockcroft and Gault formula for the estimation of glomerular filtration rate. Nephrol Dial Transplant 2003; 18: 2024-2031.
46. Herget-Rosenthal S, Bokenkamp A, Hofmann W. How to estimate GFR-serum creatinine, serum cystatin C or equations? Clin Biochem 2007; 40: 153-161.
47. Hojs R, Bevc S, Antolinc B, Gorenjak M, Puklavec L. Serum cystatin C as an endogenous marker of renal function in the elderly. Int J Clin Pharmacol Res 2004; 24: 49-54.
48. Schuck O, Teplan V, Jabor A, Stollova M, Skibova J. Glomerular filtration rate estimation in patients with advanced chronic renal insufficiency based on serum cystatin C levels. Nephron Clin Pract 2003; 93: c146-c151.
49. Dharnidharka VR, Kwon C, Stevens G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am J Kidney Dis 2002; 40: 221-226.
50. Laterza OF, Price CP, Scott MG. Cystatin C: an improved estimator of glomerular filtration rate? Clin Chem 2002; 48: 699-707.
51. Tan GD, Lewis AV, James TJ, Altmann P, Taylor RP, Levy JC. Clinical usefulness of cystatin C for the estimation of glomerular filtration rate in type 1 diabetes: reproducibility and accuracy compared with standard measures and iohexol clearance. Diabetes Care 2002; 25: 2004-2009.
52. Grubb A, Bjork J, Lindstrom V, Sterner G, Bondesson P, Nyman U. A cystatin C-based formula without anthropometric variables estimates glomerular filtration rate better than creatinine clearance using the Cockcroft-Gault formula. Scand J Clin Lab Invest 2005; 65: 153-162.
53. Grubb A, Nyman U, Bjork J, Lindstrom V, Rippe B, Sterner G, et al. Simple cystatin C-based prediction equations for glomerular filtration rate compared with the modification of diet in renal disease prediction equation for adults and the Schwartz and the Counahan-Barratt prediction equations for children. Clin Chem 2005; 51: 1420-1431.
54. MacIsaac RJ, Tsalamandris C, Thomas MC, Premaratne E, Panagiotopoulos S, Smith TJ, et al. Estimating glomerular filtration rate in diabetes: a comparison of cystatin-C- and creatinine-based methods. Diabetologia 2006; 49: 1686-1689.
55. Larsson A, Malm J, Grubb A, Hansson LO. Calculation of glomerular filtration rate expressed in mL/min from plasma cystatin C values in mg/L. Scand J Clin Lab Invest 2004; 64: 25-30.
56. Stevens LA, Coresh J, Schmid CH, Feldman HI, Froissart M, Kusek J, et al. Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD. Am J Kidney Dis 2008; 51: 395-406.
57. Rigalleau V, Beauvieux MC, Lasseur C, Chauveau P, Raffaitin C, Perlemoine C, et al. The combination of cystatin C and serum creatinine improves the monitoring of kidney function in patients with diabetes and chronic kidney disease. Clin Chem 2007; 53: 1988-1989.
58. Ma YC, Zuo L, Chen JH, Luo Q, Yu XQ, Li Y, et al. Improved GFR estimation by combined creatinine and cystatin C measurements. Kidney Int 2007; 72: 1535-1542.
59. Tidman M, Sjostrom P, Jones I. A comparison of GFR estimating formulae based upon s-cystatin C and s-creatinine and a combination of the two. Nephrol Dial Transplant 2008; 23: 154-160.
60. Urbaniak J, Weyde W, Smolska D, Zagocka E, Klak R, Kusztal M, et al. S-cystatin C formulae or combination of s-cystatin C and s-creatinine formulae do not improve prediction of GFR. Nephrol Dial Transplant 2008; 23: 2425-2426.
Acknowledgments
Research supported by FAPEMA.
 Address for correspondence: J.V. Salgado, Rua dos Angelins, Qd. 10 c 30, São Francisco, 65076-030 São Luís, MA, Brasil. Fax: +55-98-2109-8026. E-mail: jvlsalgado@yahoo.com.br
Address for correspondence: J.V. Salgado, Rua dos Angelins, Qd. 10 c 30, São Francisco, 65076-030 São Luís, MA, Brasil. Fax: +55-98-2109-8026. E-mail: jvlsalgado@yahoo.com.br
Received June 10, 2009. Accepted April 20, 2010. Available online May 14, 2010. Published June 11, 2010.
The Brazilian Journal of Medical and Biological Research is partially financed by
- 1. Meguid El Nahas A, Bello AK. Chronic kidney disease: the global challenge. Lancet 2005; 365: 331-340.
- 2. Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 2005; 67: 2089-2100.
- 4. Blaufox MD, Aurell M, Bubeck B, Fommei E, Piepsz A, Russell C, et al. Report of the Radionuclides in Nephrourology Committee on renal clearance. J Nucl Med 1996; 37: 1883-1890.
- 5. Fleming JS, Zivanovic MA, Blake GM, Burniston M, Cosgriff PS. Guidelines for the measurement of glomerular filtration rate using plasma sampling. Nucl Med Commun 2004; 25: 759-769.
- 6. Tett SE, Kirkpatrick CM, Gross AS, McLachlan AJ. Principles and clinical application of assessing alterations in renal elimination pathways. Clin Pharmacokinet 2003; 42: 1193-1211.
- 7. Medeiros FS, Sapienza MT, Prado ES, Agena F, Shimizu MH, Lemos FB, et al. Validation of plasma clearance of 51Cr-EDTA in adult renal transplant recipients: comparison with inulin renal clearance. Transpl Int 2009; 22: 323-331.
- 8. Odlind B, Hallgren R, Sohtell M, Lindstrom B. Is 125I iothalamate an ideal marker for glomerular filtration? Kidney Int 1985; 27: 9-16.
- 9. Perrone RD, Steinman TI, Beck GJ, Skibinski CI, Royal HD, Lawlor M, et al. Utility of radioisotopic filtration markers in chronic renal insufficiency: simultaneous comparison of 125I-iothalamate, 169Yb-DTPA, 99mTc-DTPA, and inulin. The Modification of Diet in Renal Disease Study. Am J Kidney Dis 1990; 16: 224-235.
- 10. Boschi S, Marchesini B. High-performance liquid chromatographic method for the simultaneous determination of iothalamate and o-iodohippurate. J Chromatogr 1981; 224: 139-143.
- 11. Effersoe H, Rosenkilde P, Groth S, Jensen LI, Golman K. Measurement of renal function with iohexol. A comparison of iohexol, 99mTc-DTPA, and 51Cr-EDTA clearance. Invest Radiol 1990; 25: 778-782.
- 12. Perrone RD, Madias NE, Levey AS. Serum creatinine as an index of renal function: new insights into old concepts. Clin Chem 1992; 38: 1933-1953.
- 13. Jones CA, McQuillan GM, Kusek JW, Eberhardt MS, Herman WH, Coresh J, et al. Serum creatinine levels in the US population: third National Health and Nutrition Examination Survey. Am J Kidney Dis 1998; 32: 992-999 [Erratum, Am J Kidney Dis 2000; 35: 179].
- 14. Swedko PJ, Clark HD, Paramsothy K, Akbari A. Serum creatinine is an inadequate screening test for renal failure in elderly patients. Arch Intern Med 2003; 163: 356-360.
- 15. Baxmann AC, Ahmed MS, Marques NC, Menon VB, Pereira AB, Kirsztajn GM, et al. Influence of muscle mass and physical activity on serum and urinary creatinine and serum cystatin C. Clin J Am Soc Nephrol 2008; 3: 348-354.
- 16. Myers GL, Miller WG, Coresh J, Fleming J, Greenberg N, Greene T, et al. Recommendations for improving serum creatinine measurement: a report from the Laboratory Working Group of the National Kidney Disease Education Program. Clin Chem 2006; 52: 5-18.
- 17. Horio M. Assessment of renal function - Up-to-date. Clinic All-round 2006; 55: 1203-1208.
- 18. Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31-41.
- 19. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999; 130: 461-470.
- 20. Burkhardt H, Bojarsky G, Gretz N, Gladisch R. Creatinine clearance, Cockcroft-Gault formula and cystatin C: estimators of true glomerular filtration rate in the elderly? Gerontology 2002; 48: 140-146.
- 21. Levey AS, Greene T, Kusek J, Beck G. A simplified equation to predict glomerular filtration rate from serum creatinine. J Am Soc Nephrol 2000; 11: 155A.
- 22. Jafar TH, Schmid CH, Levey AS. Serum creatinine as marker of kidney function in South Asians: a study of reduced GFR in adults in Pakistan. J Am Soc Nephrol 2005; 16: 1413-1419.
- 23. Poggio ED, Nef PC, Wang X, Greene T, Van Lente F, Dennis VW, et al. Performance of the Cockcroft-Gault and modification of diet in renal disease equations in estimating GFR in ill hospitalized patients. Am J Kidney Dis 2005; 46: 242-252.
- 24. Branten AJ, Vervoort G, Wetzels JF. Serum creatinine is a poor marker of GFR in nephrotic syndrome. Nephrol Dial Transplant 2005; 20: 707-711.
- 25. Rule AD, Gussak HM, Pond GR, Bergstralh EJ, Stegall MD, Cosio FG, et al. Measured and estimated GFR in healthy potential kidney donors. Am J Kidney Dis 2004; 43: 112-119.
- 26. Cirillo M, Anastasio P, De Santo NG. Relationship of gender, age, and body mass index to errors in predicted kidney function. Nephrol Dial Transplant 2005; 20: 1791-1798.
- 27. Lin J, Knight EL, Hogan ML, Singh AK. A comparison of prediction equations for estimating glomerular filtration rate in adults without kidney disease. J Am Soc Nephrol 2003; 14: 2573-2580 [Erratum, J Am Soc Nephrol 2005; 16: 2814].
- 28. Ibrahim H, Mondress M, Tello A, Fan Y, Koopmeiners J, Thomas W. An alternative formula to the Cockcroft-Gault and the modification of diet in renal diseases formulas in predicting GFR in individuals with type 1 diabetes. J Am Soc Nephrol 2005; 16: 1051-1060.
- 29. Rule AD, Bergstralh EJ, Slezak JM, Bergert J, Larson TS. Glomerular filtration rate estimated by cystatin C among different clinical presentations. Kidney Int 2006; 69: 399-405.
- 30. Van Biesen W, Vanholder R, Veys N, Verbeke F, Delanghe J, De Bacquer D, et al. The importance of standardization of creatinine in the implementation of guidelines and recommendations for CKD: implications for CKD management programmes. Nephrol Dial Transplant 2006; 21: 77-83.
- 31. Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med 2006; 145: 247-254.
- 32. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF III, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009; 150: 604-612.
- 33. National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification and stratification. Am J Kidney Dis 2002; 39 (2 Suppl 1): S1-S266.
- 34. Filler G, Bokenkamp A, Hofmann W, Le Bricon T, Martinez-Bru C, Grubb A. Cystatin C as a marker of GFR - history, indications, and future research. Clin Biochem 2005; 38: 1-8.
- 35. Randers E, Erlandsen EJ. Serum cystatin C as an endogenous marker of the renal function - a review. Clin Chem Lab Med 1999; 37: 389-395.
- 36. Cimerman N, Brguljan PM, Krasovec M, Suskovic S, Kos J. Twenty-four hour variations of cystatin C and total cysteine proteinase inhibitory activity in sera from healthy subjects. Clin Chim Acta 2000; 291: 89-95.
- 37. Risch L, Herklotz R, Blumberg A, Huber AR. Effects of glucocorticoid immunosuppression on serum cystatin C concentrations in renal transplant patients. Clin Chem 2001; 47: 2055-2059.
- 38. Bokenkamp A, van Wijk JA, Lentze MJ, Stoffel-Wagner B. Effect of corticosteroid therapy on serum cystatin C and beta2-microglobulin concentrations. Clin Chem 2002; 48: 1123-1126.
- 39. Fricker M, Wiesli P, Brandle M, Schwegler B, Schmid C. Impact of thyroid dysfunction on serum cystatin C. Kidney Int 2003; 63: 1944-1947.
- 40. Nakai K, Kikuchi M, Fujimoto K, Kaneko Y, Omori S, Nakai K, et al. Serum levels of cystatin C in patients with malignancy. Clin Exp Nephrol 2008; 12: 132-139.
- 41. Lamb E, Stowe H. Rheumatoid factor can interfere with cystatin C measurement. Ann Clin Biochem 2003; 40: 195-196.
- 42. Knight EL, Verhave JC, Spiegelman D, Hillege HL, De Zeeuw D, Curhan GC, et al. Factors influencing serum cystatin C levels other than renal function and the impact on renal function measurement. Kidney Int 2004; 65: 1416-1421.
- 43. Shlipak MG, Praught ML, Sarnak MJ. Update on cystatin C: new insights into the importance of mild kidney dysfunction. Curr Opin Nephrol Hypertens 2006; 15: 270-275.
- 44. Roos JF, Doust J, Tett SE, Kirkpatrick CM. Diagnostic accuracy of cystatin C compared to serum creatinine for the estimation of renal dysfunction in adults and children - a meta-analysis. Clin Biochem 2007; 40: 383-391.
- 45. Hoek FJ, Kemperman FA, Krediet RT. A comparison between cystatin C, plasma creatinine and the Cockcroft and Gault formula for the estimation of glomerular filtration rate. Nephrol Dial Transplant 2003; 18: 2024-2031.
- 46. Herget-Rosenthal S, Bokenkamp A, Hofmann W. How to estimate GFR-serum creatinine, serum cystatin C or equations? Clin Biochem 2007; 40: 153-161.
- 47. Hojs R, Bevc S, Antolinc B, Gorenjak M, Puklavec L. Serum cystatin C as an endogenous marker of renal function in the elderly. Int J Clin Pharmacol Res 2004; 24: 49-54.
- 48. Schuck O, Teplan V, Jabor A, Stollova M, Skibova J. Glomerular filtration rate estimation in patients with advanced chronic renal insufficiency based on serum cystatin C levels. Nephron Clin Pract 2003; 93: c146-c151.
- 49. Dharnidharka VR, Kwon C, Stevens G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am J Kidney Dis 2002; 40: 221-226.
- 50. Laterza OF, Price CP, Scott MG. Cystatin C: an improved estimator of glomerular filtration rate? Clin Chem 2002; 48: 699-707.
- 51. Tan GD, Lewis AV, James TJ, Altmann P, Taylor RP, Levy JC. Clinical usefulness of cystatin C for the estimation of glomerular filtration rate in type 1 diabetes: reproducibility and accuracy compared with standard measures and iohexol clearance. Diabetes Care 2002; 25: 2004-2009.
- 52. Grubb A, Bjork J, Lindstrom V, Sterner G, Bondesson P, Nyman U. A cystatin C-based formula without anthropometric variables estimates glomerular filtration rate better than creatinine clearance using the Cockcroft-Gault formula. Scand J Clin Lab Invest 2005; 65: 153-162.
- 53. Grubb A, Nyman U, Bjork J, Lindstrom V, Rippe B, Sterner G, et al. Simple cystatin C-based prediction equations for glomerular filtration rate compared with the modification of diet in renal disease prediction equation for adults and the Schwartz and the Counahan-Barratt prediction equations for children. Clin Chem 2005; 51: 1420-1431.
- 54. MacIsaac RJ, Tsalamandris C, Thomas MC, Premaratne E, Panagiotopoulos S, Smith TJ, et al. Estimating glomerular filtration rate in diabetes: a comparison of cystatin-C- and creatinine-based methods. Diabetologia 2006; 49: 1686-1689.
- 55. Larsson A, Malm J, Grubb A, Hansson LO. Calculation of glomerular filtration rate expressed in mL/min from plasma cystatin C values in mg/L. Scand J Clin Lab Invest 2004; 64: 25-30.
- 56. Stevens LA, Coresh J, Schmid CH, Feldman HI, Froissart M, Kusek J, et al. Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD. Am J Kidney Dis 2008; 51: 395-406.
- 57. Rigalleau V, Beauvieux MC, Lasseur C, Chauveau P, Raffaitin C, Perlemoine C, et al. The combination of cystatin C and serum creatinine improves the monitoring of kidney function in patients with diabetes and chronic kidney disease. Clin Chem 2007; 53: 1988-1989.
- 58. Ma YC, Zuo L, Chen JH, Luo Q, Yu XQ, Li Y, et al. Improved GFR estimation by combined creatinine and cystatin C measurements. Kidney Int 2007; 72: 1535-1542.
- 59. Tidman M, Sjostrom P, Jones I. A comparison of GFR estimating formulae based upon s-cystatin C and s-creatinine and a combination of the two. Nephrol Dial Transplant 2008; 23: 154-160.
- 60. Urbaniak J, Weyde W, Smolska D, Zagocka E, Klak R, Kusztal M, et al. S-cystatin C formulae or combination of s-cystatin C and s-creatinine formulae do not improve prediction of GFR. Nephrol Dial Transplant 2008; 23: 2425-2426.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
07 June 2010 -
Date of issue
June 2010
History
-
Received
10 June 2009 -
Accepted
20 Apr 2010