Abstract
Gene therapy is an alternative treatment for genetic lung disease, especially monogenic disorders such as cystic fibrosis. Cystic fibrosis is a severe autosomal recessive disease affecting one in 2500 live births in the white population, caused by mutation of the cystic fibrosis transmembrane conductance regulator (CFTR). The disease is classically characterized by pancreatic enzyme insufficiency, an increased concentration of chloride in sweat, and varying severity of chronic obstructive lung disease. Currently, the greatest challenge for gene therapy is finding an ideal vector to deliver the transgene (CFTR) to the affected organ (lung). Adeno-associated virus is the most promising viral vector system for the treatment of respiratory disease because it has natural tropism for airway epithelial cells and does not cause any human disease. This review focuses on the basic properties of adeno-associated virus and its use as a vector for cystic fibrosis gene therapy.
Gene therapy; Adeno-associated virus; Cystic fibrosis; Cystic fibrosis transmembrane conductance regulator; Vectors
Abstract
Introduction
References
Acknowledgments
Braz J Med Biol Res, November 2011, Volume 44(11) 1097-1104
Adeno-associated virus for cystic fibrosis gene therapy
S.V. Martini1, P.R.M. Rocco2 and  Correspondence and Footnotes
Correspondence and Footnotes
 M.M. Morales1
M.M. Morales1
1Laboratório de Fisiologia Celular e Molecular and 2Laboratório de Investigação Pulmonar, Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, Rio de Janeiro, RJ, Brasil
 Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Correspondence and Footnotes
Gene therapy is an alternative treatment for genetic lung disease, especially monogenic disorders such as cystic fibrosis. Cystic fibrosis is a severe autosomal recessive disease affecting one in 2500 live births in the white population, caused by mutation of the cystic fibrosis transmembrane conductance regulator (CFTR). The disease is classically characterized by pancreatic enzyme insufficiency, an increased concentration of chloride in sweat, and varying severity of chronic obstructive lung disease. Currently, the greatest challenge for gene therapy is finding an ideal vector to deliver the transgene (CFTR) to the affected organ (lung). Adeno-associated virus is the most promising viral vector system for the treatment of respiratory disease because it has natural tropism for airway epithelial cells and does not cause any human disease. This review focuses on the basic properties of adeno-associated virus and its use as a vector for cystic fibrosis gene therapy.
Key words: Gene therapy; Adeno-associated virus; Cystic fibrosis; Cystic fibrosis transmembrane conductance regulator; Vectors
Gene therapy emerged in the 1980s as an alternative treatment for genetic diseases. It is based on the transfer of functional genes to replace or supplement defective genes. Gene transfer involves the entry of some genetic material (DNA, RNA or oligonucleotides) into target cells (1,2). In practice, the gene of interest (also called a transgene) must be delivered to the cell by a vector, which carries a molecule of DNA or RNA. The transgene can be delivered into the cell by the vector. The method of delivery differs depending on the type of treatment and organ/tissue to be targeted.
Gene therapy is an important alternative for the treatment of monogenic diseases that affect the lungs and has the advantage of easy access to lung cells by bronchoscopy, nebulizer or nasal spray. Among the genetic-based lung diseases, cystic fibrosis (CF) is a lethal autosomal recessive genetic disease caused by mutations in the CF transmembrane conductance regulator (CFTR). Mutations in the CFTR gene may result in defective processing of its protein and alter the function and regulation of this channel. Mutations are associated with different symptoms, including pancreatic insufficiency, bile duct obstruction, infertility in males, high Cl- level in sweat, intestinal obstruction, nasal polyp formation, chronic sinusitis, mucus dehydration, and chronic Pseudomonas aeruginosa and Staphylococcus aureus lung infection, responsible for 90% of deaths in patients with CF (3).
The strengths of gene therapy for CF are: a) it is a monogenic disease (CFTR mutation), b) the organ most affected is the lung, where the cells can be easily accessed, and c) it is a progressive disease with a virtually normal phenotype at birth, offering an advantageous therapeutic window (4,5).
However, there is no treatment for correcting the dysfunction of the chloride channel; existing therapies only alleviate the symptoms. Therefore, gene therapy has become a promising option because, due to easy access to cells in the airways, it is possible to insert the correct CFTR gene and restore the expression of the chloride channel in the cell membranes. Reports of the first patients with CF to receive CFTR gene therapy were published in 1993, and since then there have been more than 20 clinical trials of gene transfer agents (6).
Meanwhile, the major challenge in gene therapy is the choice of vector for safe delivery of the transgene to patient cells. The ideal vector for gene therapy must have certain characteristics, such as: a) the ability to deliver the gene of interest; the vector must have adequate capacity to carry the necessary genetic information and be undetectable by the immune system; b) be non-inflammatory; c) be safe for the human host even in the setting of lung inflammation; d) have a long duration of transgene expression. The vector must be able to maintain the levels of target gene expression required to correct the disease phenotype or be safely readministered (7,8).
Gene therapy uses gene transfer systems, both viral and nonviral, as vectors. Among the many gene transfer systems being investigated, we highlight the viral vectors such as those based on the genome of adeno-associated virus (AAV). Currently, 12 human serotypes of AAV (AAV1-12) have been described, many of which have distinct cell and tissue tropism, potentially creating the option to generate a variety of different vector classes from this viral genus (9-12).
AAV has emerged as a potential vector in gene therapy for CF because of a number of theoretical advantages: a) AAV has natural tropism for airway epithelial cells; b) AAV elicits little or no inflammatory response; c) AAV generally results in stable expression; d) site-specific integration of AAV does not activate the possible oncogenes and the inserted gene can be maintained for a relatively long time in the host cell genome and stably expressed in vivo (13-15).
Adeno-associated virus
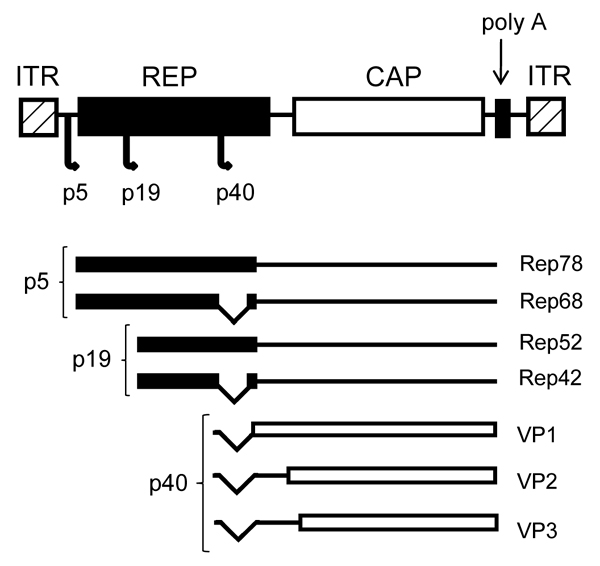
AAV is a non-pathogenic parvovirus, with single-stranded DNA, a genome of approximately 4.7 kb, not enveloped and has icosahedric conformation. AAV was first discovered in 1965 as a contaminant of adenovirus preparations (16). AAV belongs to the Dependovirus genus and Parvoviridae family, requiring helper functions from either herpes virus or adenovirus for replication (17). In the absence of helper virus, AAV can set up latency by integrating into human chromosome 19 at the 19q13.4 location. The AAV genome consists of two open reading frames (ORF), one for each of two AAV genes, Rep and Cap (18). The AAV DNA ends have a 145-bp inverted terminal repeat (ITR), and the 125 terminal bases are palindromic, leading to a characteristic T-shaped hairpin structure (19,20).
The Rep gene is transcribed from promoters p5 and p19 into four Rep proteins (Rep78, Rep68, Rep52, and Rep40), which have important roles in the life cycle of the virus. Proteins Rep78 and Rep68 are encoded by the mRNA transcribed from promoter p5. These proteins are essential for viral DNA replication, transcription and control of site-specific integration. The two smaller proteins Rep52 and Rep40 are generated by the mRNA transcribed from promoter p19. These proteins are involved in the formation of a single-stranded viral genome for packaging and viral integration (21-23). The Cap gene encodes three viral capsid proteins: VP1 (735 amino acids, ~90 kDa), VP2 (598 amino acids, ~72 kDa) and VP3 (533 amino acids, ~60 kDa), which form the viral capsid of 60 subunits, at the ratio of 1:1:10. The three capsid proteins are translated from the mRNA transcribed from the promoter p40 (Figure 1) (20,22).
The ITRs are the only sequences necessary for packaging and viral replication, and they have the following functions: a) they serve as a primer for synthesizing a new strand of DNA (24); b) they possess a binding site for Rep78 and Rep68 proteins, which have helicase and endonuclease activity (25); c) they have a terminal resolution site (trs) identical to the sequence of chromosome 19, which serves as a sequence of integration of the viral genome (25,26).
Despite the high prevalence of AAV in the human population, there is no known disease associated with AAV in humans or other species, making it an ideal vector for gene transfer (27,28). Although the wild-type AAV, in the presence of the Rep gene, has the ability to integrate DNA into the host cell genome in a 4-kb region on chromosome 19 (q13.4), this feature disappears in the vectors (recombinant AAV), which are extracted from all viral genes. Thus, in most cases AAV remains episomal, but transcriptionally active (29). Another advantage of AAV vectors is the fact that they are capable of infecting a large number of cell types (proliferating and differentiating) with the same efficiency (30,31). Moreover, AAV vectors induce a relatively weak immune response, allowing re-administration in many cases (7).
However, one limitation of AAV vectors is their relatively small genome. Studies have shown that the ideal size for packaging a viral genome is 4100-4900 bp (32,33). Other studies suggest that packaging becomes very inefficient when the size exceeds 4900-5000 bp. Consequently, this becomes a problem for genes with large coding sequences, such as CFTR, which has a cDNA of 4450 bp (34). The AAV vector contains two ITR and enhancer/promoter elements and therefore the expression cassette can easily surpass the limits of packaging. Therefore, strategies are being developed to avoid this limitation for CF gene therapy. Some studies have used short sequences of endogenous AAV promoters (with or without proteasome inhibitors to increase expression) (35-37). Other studies have produced AAV vectors with a CFTR cDNA with a deletion at the N terminal (38), the regulatory domain (39), and missing the first four transmembrane domains of CFTR (40). These deletions did not alter the function of the CFTR chloride channel (38-40).
Adeno-associated virus serotype 2
Among different AAV serotypes, adeno-associated virus type 2 (AAV2) is the most studied. AAV2 gains entry into target cells by using the cellular receptor heparan sulfate proteoglycan (HSPG) and cells that do not express HSPG show resistance to infection with AAV2 (41). In addition, AAV2 uses a co-receptor for internalization into the cell via endocytosis. Five receptors have been identified: αVβ5 integrin (42), fibroblast growth factor receptor 1 (43), hepatocyte growth factor receptor (44), αVβ1 integrin (45), and laminin receptor (46). AAV, in general, binds to its receptors and rapidly undergoes endocytosis in clathrin-coated vesicles. For successful AAV infection in gene therapy, after endocytosis, the AAV vector should escape from endocytic vesicles, enter the nucleus through the nuclear pore complex and promote transgene expression, such as CFTR (Figure 2) (13,47).
AAV2 vectors have rapidly gained importance in CF gene therapy applications. AAV2 vectors have been constructed in an attempt to deliver the CFTR gene to airway epithelial cells and to restore the expression of CFTR protein. Efficient expression from recombinant AAV-associated cystic fibrosis transmembrane conductance regulator vectors (rAAV-CFTR) was observed in the airways of rabbits and monkeys for up to 6 months after a single bronchoscopic delivery (1010 particles of vector) (34,48). Another study demonstrated that repeated (two doses) bronchoscopic airway delivery of rAAV-CFTR vectors (5 x 109 DNase-resistant particles, DRP) in rabbit lung resulted in incremental transgene expression without detectable toxicity. No significant inflammatory responses to repeated airway exposure to rAAV2-CFTR vector expression were observed, showing successful redelivery (49). These studies provide the preclinical data necessary for the initiation of a clinical trial of rAAV in patients with CF.
A phase I clinical trial using AAV2 containing CFTR cDNA was performed in 10 patients with CF. The vector was administered into respiratory epithelial cells of the maxillary sinus. In this trial, there was no obvious inflammatory response and gene transfer was observed in 0.1 to 1 copies of the vector per cell, obtained from biopsy 2 weeks after the administration of the vector. The persistence of gene transfer was maintained for 41 days in one patient and for 10 weeks in another (50-52).
A further clinical trial used single doses of the AAV2 vector encoding the human CFTR cDNA (dose ranging from 1010 to 1013 DRP) delivered by aerosol or nebulization to the lungs of 12 patients with CF. The maximum gene transfer was observed at 0.6 and 0.1 vector copies per brushed cell (collected via bronchoscopy) after 14 and 30 days, respectively, after nebulization of 1013 DRP, and this declined to nearly undetectable levels by day 90. Furthermore, it was not possible to detect vector-derived mRNA expression by the polymerase chain reaction (PCR) after 90 days of viral administration. This phase I trial demonstrated that a single dose of AAV-CFTR by aerosol was safe and well tolerated by patients with CF with mild lung disease (53). Similar results were observed in a third phase I trial. Doses of rAAV2 vector expressing full-length human CFTR (6 x 104 to 2 x 1012 DRP) were administered intranasally and endobronchially to 25 patients with CF. Gene transfer measured by PCR was very low. The poor mRNA CFTR expression observed in these studies was attributed to the lack of an efficient promoter region within the construction of the viral DNA. Another important observation in both studies was the increase in neutralizing AAV2 antibodies in the serum of patients with CF who received the vector in these clinical trials. The increase of these antibodies could also be the cause of the reduced efficiency of gene transfer of CFTR. The inflammatory process itself can prevent the transduction of the vector, which raises concerns about possible reduced effectiveness in repeat dosing (14,36).
In addition, AAV has been administrated repeatedly to the lung in two trials conducted by the same group. In the first of these, AAV2 vectors containing CFTR cDNA (tgAAVCF; 1013 viral particles or placebo) were administered in three doses at intervals of 30 days by aerosol to the lungs of 37 subjects with CF. Improvements in induced-sputum interleukin-8 and interleukin-10 and FEV1 (forced expiratory flow in 1 s) were observed. Gene transfer but not gene expression was detected in a subset of six tgAAVCF subjects who underwent bronchoscopy, and the delivery of tgAAVCF was associated with an increase in AAV2-neutralizing antibodies. The second study also delivered repeated doses of tgAAVCF to 102 subjects with CF using the same protocols (54). Unhappily, no significant improvement in lung function (FEV1) was observed in patients who received the viral vector compared with placebo controls (54,55).
Based on these preclinical and clinical trials, a number of key limitations of AAV2 vector technology were recognized: a) low abundance of the AAV2 receptor and co-receptors on the apical surface of the airways; b) relative inefficiency of the AAV-ITR promoter; c) inactivation of rAAV2 particles by extracellular barriers, including elastase, alpha defensins, and other substances; d) short duration of gene transfer associated with rapid turnover of the CF airway epithelium with the subsequent loss of rAAV genomes, and e) the development of neutralizing antibodies by patients limiting the efficiency or repeated administrations of the vector (56).
In agreement with previous studies, Lassance et al. (57) showed that readministration of the rAAV2 vector containing the DNA of green fluorescence protein (GFP; three doses of 4 x 109 particles) caused a mild inflammatory response that affected the airways and lung parenchyma in mice. Histological findings showed that after the first dose it was possible to detect changes in lung parenchyma. The percentage of normal areas and collapsed areas decreased and increased, respectively, in all groups of animals that received the viral vector intratracheally. However, these morphological changes were not sufficient to induce changes in lung function (57).
Several strategies to improve the efficiency of this promising viral vector are under investigation. One is to explore the use of other AAV serotypes. Early work showed that serotypes AAV1, AAV5 and AAV6 are better able to transfer to lung cells compared to AAV2. This is probably due to the reduced number of receptors for AAV2 on the apical surface of lung epithelial cells (58). Changes in the viral genome containing the coding portion of the CFTR gene have also been made in an attempt to improve the ability for expression; for example, inclusion of certain elements and use of other transcriptional promoter regions showed an improvement in transcription ability (59).
Due to the low transduction observed in lung cells after administration of the AAV2 vector, the AAV5 vector has also been studied for gene therapy of the lungs.
Adeno-associated virus serotype 5
There is evidence that the AAV5 vector is more efficient for CF gene therapy than AAV2. Zabner et al. (38) suggested that AAV5 can increase gene delivery 50 times compared to AAV2 in human airway epithelium. Corroborating this study, Walters et al. (60) showed that the apical membrane of airway epithelium contains high-affinity receptors for AAV5, in contrast to AAV2, which has its receptors located in the basolateral membrane. AAV5 requires sialic acid (2,3-linked sialic acid) to connect to cell and gene transfer. Sialic acid is the most common terminal glucosyl residue of mammalian proteins, and is present not only in airway epithelium but also in many tissues (60). Other studies have shown that expression of the platelet-derived growth factor receptor (PDGFR-α-polypeptide) was highly associated with AAV5-mediated transduction and both PDGFR-α and PDGFR-β were confirmed as receptors for AAV5 (61).
With regard to the AAV vector, it is known that neutralizing antibodies against AAV2 are most prevalent in the general population with neutralizing antibodies against AAV5 (62). Moreover, neutralizing antibodies did not limit the transfer of the transgene by AAV5 in mice first exposed to AAV2 (63).
In addition, to improve the gene transfer of AAV vectors for CF gene therapy, the activities of different promoters, cytomegalovirus (CMV), Rous sarcoma virus and cytomegalovirus promoter conjugated/β-actin (CB), were evaluated in CF bronchial cells. The CB promoter proved to be the most efficient. Also, the capsids of the AAV serotypes rAAV2 and rAAV5 were compared and rAAV5 was shown to be significantly more efficient at transferring the transgene than rAAV2. The rAAV5 vector-CB carrying the CFTR gene with a deletion at the N terminal (AAV5-CB-Δ264CFTR) was able to correct defective chloride transport in vitro in a CF bronchial cell line, as well as the hyper-inflammatory lung phenotype in Pseudomonas-agarose bead challenged CF mouse lungs in vivo (64). The AAV5-CB-Δ264CFTR vector has been further tested in non-human primates in a study that utilized vector lung delivery of AAV5-ΔCFTR or AAV5-GFP by aerosol in macaques. The studies demonstrated safe and efficient delivery to the lungs (1.24 x 105 copies of CFTR/µg DNA) with the absence of inflammation compared to untreated macaques (65).
In 2005, Ostedgaard et al. (39) conducted experiments to overcome the limited capacity of the AAV genome; they deleted a portion of the CFTR-regulatory domain (CFTRΔR) and the CMV promoter. The deleted elements (promoter and CFTRΔR) and the ITR were packaged in rAAV5 and applied to the apical surface of differentiated CF airway epithelia. Two to 4 weeks later, the AAV5 vectors partially corrected the chloride transport, defective in CF airway epithelium, providing the first evidence that a single AAV vector administered by itself can correct the CF Cl- transport defect in differentiated CF airway epithelia.
However, several doses are needed to maintain the transgene expression in the airways, since the permanence of transgene expression is limited. Martini et al. (66), using rAAV5-GFP, demonstrated that rAAV5 (4 x 1011 particles) is safe for gene transfer to mouse lungs. Immunohistochemistry results showed that re-administration (3 doses) of the rAAV5 vector leads to sustained expression of the reporter gene (GFP) in the lungs of treated animals. Also, repeated administration of rAAV5 did not induce an inflammatory process, apoptosis, or mechanical or morphometric changes in the lungs. These results agree with previous studies suggesting that AAV5 is a good vector for CF gene therapy.
Other possible AAV vectors
Other viral vectors that have shown efficiency in gene transfer to the lung are the AAV subtypes 6 and 9. Halbert et al. (63) observed that the AAV6 vector is more efficient in gene transfer to mouse airway cells that the AAV2 vector. Corroborating this study, Limberis et al. (67) suggested that AAV6, among the AAV serotypes 1-9, is the most efficient for gene transfer to human ciliated epithelial cells. AAV9 has also been shown to transduce airways remarkably well (68).
With the aim of improving the transduction of rAAV vectors, newer vectors are often cross-packaged or pseudotyped with different capsid versions. Usually, the AAV2 genome containing the ITRs is packaged into capsids from different serotypes in order to exploit the altered tropism of the capsid. Several of these, including AAV1, AAV5, and AAV10, are substantially superior to AAV2 for efficient in vivo transduction of the rodent lung (8,12).
Auricchio et al. (69) evaluated the potential of pseudotyped AAV-mediated gene transfer to the lung. They showed that AAV vectors containing the capsid from serotype 5 transduce lung epithelial cells more efficiently than AAV2/1 and AAV2/2, and that AAV2/5 can be readministered non-invasively. A similar study observed the distribution of two pseudotyped vectors, AAV2/10 and AAV2/11. Systemic injection of AAV2/10 led to persistent transduction in the murine liver, heart, muscle, lung, kidney, and uterus, whereas AAV2/11 resulted in efficient transduction of the muscle, kidney, spleen, lung, heart, and stomach (31). In agreement, an early study used rAAV2/9 (1010 genome copies per mouse) vectors expressing the luciferase gene in surfactant protein B (SPB)-/- mice to test the efficiency of this vector in an inflamed lung (the vector was applied directly to the lung lobes). This study demonstrated stable long-term expression using an AAV2/9-based vector, suggesting that AAV2/9 would be an excellent candidate for gene therapy of pulmonary disease (70).
Future directions
Although significant progress has been made in the use of AAV vectors for CF gene therapy, several developments are likely to enhance the potential usefulness of this system.
The efficiency of rAAV transduction is dependent on the interaction of the virus with the host cell, which includes binding to cell receptors, to overcome the intracellular barriers that limit nuclear accumulation of the virus. The viral capsid is an essential element that influences the events of extracellular recognition of specific receptors and the process of intracellular trafficking. As a result, the viral capsid plays a crucial role in cell tropism, the kinetics of transduction efficiency and transgene expression. Regulating these properties can improve the efficiency and safety of gene therapy (71).
One strategy used to improve the efficiency of adenovirus vector was associated with the construction of a mutant AAV2 vector. Wu et al. (72) showed that insertion of the serpin receptor ligand into the N-terminal regions of VP1 or VP2 can change the tropism of AAV2 and increase the transduction efficiency for the human lung cell line IB3. In agreement with this study, other groups evaluated the transduction and expression efficiencies of several AAV serotypes and AAV2 capsid mutants with specific pulmonary targeting ligands in the mouse lung and compared intranasal delivery with intratracheal lung delivery. It was observed that rAAV8 was the most efficient serotype in expressing α1-antitrypsin (AAT) in the lung among all the serotypes and mutants tested (73).
Mutations in the tyrosine residues on the surface of the viral capsid, highly conserved residues in the AAV serotypes, were also performed. Tyrosine phosphorylation serves as a signal for ubiquitination of AAV particles, leading to subsequent targeting for proteasome-mediated vector degradation before reaching the nucleus. Consequently, mutation of capsid tyrosine residues from AAV particles is predicted to allow the vectors to escape the proteasome degradation pathway and thus promote more vector genome delivery to the nucleus and more transgene expression. Previous studies have demonstrated that point mutations in surface-exposed capsid tyrosine residues in AAV showed great efficiency in gene transfer to human cells in vitro, and mouse hepatocytes and retina in vivo (71,74,75).
Another strategy that is being tested is the use of agents that modulate intracellular enzymes (76) to prevent degradation of AAV2 within the host cell, somehow optimizing gene transfer. The use of these factors has been shown to increase the expression of the reporter transgene about 1000 times (76).
Conclusion
Great advances have been made in learning about the mechanism of AAV gene transfer to improve its transduction. AAV remains a promising delivery system for CF gene therapy mainly because of its lack of pathogenicity and immunogenicity in the airways. However, more data are needed regarding the effectiveness of transgene delivery of different serotype, pseudotyped and mutated AAV vectors.
Map of the wild-type adeno-associated virus (AAV) genome. REP and CAP genes flanked by inverted terminal repeats (ITR), three promoters (p5, p19, and p40), four Rep proteins involved in viral replication (Rep78, Rep68, Rep52, and Rep42), and three Cap proteins involved in capsid formation (VP1-3).
Cell entry and trafficking of the adeno-associated virus (AAV) vector. The AAV vector enters the cell by endocytosis in a receptor-mediated manner, through clathrin-coated pits. After endocytosis, the AAV vector should escape from the endosome, enter the nucleus through the nuclear pore complex and promote transgene expression (cystic fibrosis transmembrane conductance regulator, CFTR).
1. Shimotohno K, Temin HM. Formation of infectious progeny virus after insertion of herpes simplex thymidine kinase gene into DNA of an avian retrovirus. Cell 1981; 26: 67-77.
2. Wei CM, Gibson M, Spear PG, Scolnick EM. Construction and isolation of a transmissible retrovirus containing the src gene of Harvey murine sarcoma virus and the thymidine kinase gene of herpes simplex virus type 1. J Virol 1981; 39: 935-944.
3. Morales MM, Capella MA, Lopes AG. Structure and function of the cystic fibrosis transmembrane conductance regulator. Braz J Med Biol Res 1999; 32: 1021-1028.
4. Cohen-Haguenauer O, Boiron M. [Gene therapy: facts and prospects]. Pathol Biol 1992; 40: 5-13.
5. Flotte TR, Laube BL. Gene therapy in cystic fibrosis. Chest 2001; 120: 124S-131S.
6. Davies JC, Alton EW. Gene therapy for cystic fibrosis. Proc Am Thorac Soc 2010; 7: 408-414.
7. Flotte TR. Recombinant adeno-associated virus vectors for cystic fibrosis gene therapy. Curr Opin Mol Ther 2001; 3: 497-502.
8. Mueller C, Flotte TR. Gene therapy for cystic fibrosis. Clin Rev Allergy Immunol 2008; 35: 164-178.
9. Schultz BR, Chamberlain JS. Recombinant adeno-associated virus transduction and integration. Mol Ther 2008; 16: 1189-1199.
10. Blacklow NR, Hoggan MD, Rowe WP. Serologic evidence for human infection with adenovirus-associated viruses. J Natl Cancer Inst 1968; 40: 319-327.
11. Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci U S A 2002; 99: 11854-11859.
12. Rabinowitz JE, Rolling F, Li C, Conrath H, Xiao W, Xiao X, et al. Cross-packaging of a single adeno-associated virus (AAV) type 2 vector genome into multiple AAV serotypes enables transduction with broad specificity. J Virol 2002; 76: 791-801.
13. Daya S, Berns KI. Gene therapy using adeno-associated virus vectors. Clin Microbiol Rev 2008; 21: 583-593.
14. Flotte TR, Zeitlin PL, Reynolds TC, Heald AE, Pedersen P, Beck S, et al. Phase I trial of intranasal and endobronchial administration of a recombinant adeno-associated virus serotype 2 (rAAV2)-CFTR vector in adult cystic fibrosis patients: a two-part clinical study. Hum Gene Ther 2003; 14: 1079-1088.
15. Kotin RM, Siniscalco M, Samulski RJ, Zhu XD, Hunter L, Laughlin CA, et al. Site-specific integration by adeno-associated virus. Proc Natl Acad Sci U S A 1990; 87: 2211-2215.
16. Atchison RW, Casto BC, Hammon WM. Adenovirus-associated defective virus particles. Science 1965; 149: 754-756.
17. Goodman S, Xiao X, Donahue RE, Moulton A, Miller J, Walsh C, et al. Recombinant adeno-associated virus-mediated gene transfer into hematopoietic progenitor cells. Blood 1994; 84: 1492-1500.
18. Hermonat PL, Labow MA, Wright R, Berns KI, Muzyczka N. Genetics of adeno-associated virus: isolation and preliminary characterization of adeno-associated virus type 2 mutants. J Virol 1984; 51: 329-339.
19. Hauswirth WW, Berns KI. Origin and termination of adeno-associated virus DNA replication. Virology 1977; 78: 488-499.
20. Lu Y. Recombinant adeno-associated virus as delivery vector for gene therapy - a review. Stem Cells Dev 2004; 13: 133-145.
21. Balague C, Kalla M, Zhang WW. Adeno-associated virus Rep78 protein and terminal repeats enhance integration of DNA sequences into the cellular genome. J Virol 1997; 71: 3299-3306.
22. Grimm D. Production methods for gene transfer vectors based on adeno-associated virus serotypes. Methods 2002; 28: 146-157.
23. Buning H, Ried MU, Perabo L, Gerner FM, Huttner NA, Enssle J, et al. Receptor targeting of adeno-associated virus vectors. Gene Ther 2003; 10: 1142-1151.
24. Russell DW, Kay MA. Adeno-associated virus vectors and hematology. Blood 1999; 94: 864-874.
25. Young SM Jr, McCarty DM, Degtyareva N, Samulski RJ. Roles of adeno-associated virus Rep protein and human chromosome 19 in site-specific recombination. J Virol 2000; 74: 3953-3966.
26. Young SM Jr, Samulski RJ. Adeno-associated virus (AAV) site-specific recombination does not require a Rep-dependent origin of replication within the AAV terminal repeat. Proc Natl Acad Sci U S A 2001; 98: 13525-13530.
27. Erles K, Sebokova P, Schlehofer JR. Update on the prevalence of serum antibodies (IgG and IgM) to adeno-associated virus (AAV). J Med Virol 1999; 59: 406-411.
28. Halbert CL, Miller AD, McNamara S, Emerson J, Gibson RL, Ramsey B, et al. Prevalence of neutralizing antibodies against adeno-associated virus (AAV) types 2, 5, and 6 in cystic fibrosis and normal populations: Implications for gene therapy using AAV vectors. Hum Gene Ther 2006; 17: 440-447.
29. Duan D, Sharma P, Yang J, Yue Y, Dudus L, Zhang Y, et al. Circular intermediates of recombinant adeno-associated virus have defined structural characteristics responsible for long-term episomal persistence in muscle tissue. J Virol 1998; 72: 8568-8577.
30. Davidson BL, Stein CS, Heth JA, Martins I, Kotin RM, Derksen TA, et al. Recombinant adeno-associated virus type 2, 4, and 5 vectors: transduction of variant cell types and regions in the mammalian central nervous system. Proc Natl Acad Sci U S A 2000; 97: 3428-3432.
31. Kwon I, Schaffer DV. Designer gene delivery vectors: molecular engineering and evolution of adeno-associated viral vectors for enhanced gene transfer. Pharm Res 2008; 25: 489-499.
32. Fulmer SB, Schwiebert EM, Morales MM, Guggino WB, Cutting GR. Two cystic fibrosis transmembrane conductance regulator mutations have different effects on both pulmonary phenotype and regulation of outwardly rectified chloride currents. Proc Natl Acad Sci U S A 1995; 92: 6832-6836.
33. Dong JY, Fan PD, Frizzell RA. Quantitative analysis of the packaging capacity of recombinant adeno-associated virus. Hum Gene Ther 1996; 7: 2101-2112.
34. Flotte TR, Afione SA, Conrad C, McGrath SA, Solow R, Oka H, et al. Stable in vivo expression of the cystic fibrosis transmembrane conductance regulator with an adeno-associated virus vector. Proc Natl Acad Sci U S A 1993; 90: 10613-10617.
35. Zhang LN, Karp P, Gerard CJ, Pastor E, Laux D, Munson K, et al. Dual therapeutic utility of proteasome modulating agents for pharmaco-gene therapy of the cystic fibrosis airway. Mol Ther 2004; 10: 990-1002.
36. Lee TW, Matthews DA, Blair GE. Novel molecular approaches to cystic fibrosis gene therapy. Biochem J 2005; 387: 1-15.
37. Flotte TR, Afione SA, Solow R, Drumm ML, Markakis D, Guggino WB, et al. Expression of the cystic fibrosis transmembrane conductance regulator from a novel adeno-associated virus promoter. J Biol Chem 1993; 268: 3781-3790.
38. Zabner J, Seiler M, Walters R, Kotin RM, Fulgeras W, Davidson BL, et al. Adeno-associated virus type 5 (AAV5) but not AAV2 binds to the apical surfaces of airway epithelia and facilitates gene transfer. J Virol 2000; 74: 3852-3858.
39. Ostedgaard LS, Rokhlina T, Karp PH, Lashmit P, Afione S, Schmidt M, et al. A shortened adeno-associated virus expression cassette for CFTR gene transfer to cystic fibrosis airway epithelia. Proc Natl Acad Sci U S A 2005; 102: 2952-2957.
40. Cebotaru L, Vij N, Ciobanu I, Wright J, Flotte T, Guggino WB. Cystic fibrosis transmembrane regulator missing the first four transmembrane segments increases wild type and DeltaF508 processing. J Biol Chem 2008; 283: 21926-21933.
41. Summerford C, Samulski RJ. Membrane-associated heparan sulfate proteoglycan is a receptor for adeno-associated virus type 2 virions. J Virol 1998; 72: 1438-1445.
42. Summerford C, Bartlett JS, Samulski RJ. AlphaVbeta5 integrin: a co-receptor for adeno-associated virus type 2 infection. Nat Med 1999; 5: 78-82.
43. Qing K, Mah C, Hansen J, Zhou S, Dwarki V, Srivastava A. Human fibroblast growth factor receptor 1 is a co-receptor for infection by adeno-associated virus 2. Nat Med 1999; 5: 71-77.
44. Kashiwakura Y, Tamayose K, Iwabuchi K, Hirai Y, Shimada T, Matsumoto K, et al. Hepatocyte growth factor receptor is a coreceptor for adeno-associated virus type 2 infection. J Virol 2005; 79: 609-614.
45. Asokan A, Hamra JB, Govindasamy L, Agbandje-McKenna M, Samulski RJ. Adeno-associated virus type 2 contains an integrin alpha5beta1 binding domain essential for viral cell entry. J Virol 2006; 80: 8961-8969.
46. Akache B, Grimm D, Pandey K, Yant SR, Xu H, Kay MA. The 37/67-kilodalton laminin receptor is a receptor for adeno-associated virus serotypes 8, 2, 3, and 9. J Virol 2006; 80: 9831-9836.
47. Bartlett JS, Wilcher R, Samulski RJ. Infectious entry pathway of adeno-associated virus and adeno-associated virus vectors. J Virol 2000; 74: 2777-2785.
48. Conrad CK, Allen SS, Afione SA, Reynolds TC, Beck SE, Fee-Maki M, et al. Safety of single-dose administration of an adeno-associated virus (AAV)-CFTR vector in the primate lung. Gene Ther 1996; 3: 658-668.
49. Beck SE, Jones LA, Chesnut K, Walsh SM, Reynolds TC, Carter BJ, et al. Repeated delivery of adeno-associated virus vectors to the rabbit airway. J Virol 1999; 73: 9446-9455.
50. Wagner JA, Messner AH, Moran ML, Daifuku R, Kouyama K, Desch JK, et al. Safety and biological efficacy of an adeno-associated virus vector-cystic fibrosis transmembrane regulator (AAV-CFTR) in the cystic fibrosis maxillary sinus. Laryngoscope 1999; 109: 266-274.
51. Wagner JA, Moran ML, Messner AH, Daifuku R, Conrad CK, Reynolds T, et al. A phase I/II study of tgAAV-CF for the treatment of chronic sinusitis in patients with cystic fibrosis. Hum Gene Ther 1998; 9: 889-909.
52. Wagner JA, Reynolds T, Moran ML, Moss RB, Wine JJ, Flotte TR, et al. Efficient and persistent gene transfer of AAV-CFTR in maxillary sinus. Lancet 1998; 351: 1702-1703.
53. Aitken ML, Moss RB, Waltz DA, Dovey ME, Tonelli MR, McNamara SC, et al. A phase I study of aerosolized administration of tgAAVCF to cystic fibrosis subjects with mild lung disease. Hum Gene Ther 2001; 12: 1907-1916.
54. Moss RB, Rodman D, Spencer LT, Aitken ML, Zeitlin PL, Waltz D, et al. Repeated adeno-associated virus serotype 2 aerosol-mediated cystic fibrosis transmembrane regulator gene transfer to the lungs of patients with cystic fibrosis: a multicenter, double-blind, placebo-controlled trial. Chest 2004; 125: 509-521.
55. Moss RB, Milla C, Colombo J, Accurso F, Zeitlin PL, Clancy JP, et al. Repeated aerosolized AAV-CFTR for treatment of cystic fibrosis: a randomized placebo-controlled phase 2B trial. Hum Gene Ther 2007; 18: 726-732.
56. Flotte TR. Adeno-associated virus-based gene therapy for inherited disorders. Pediatr Res 2005; 58: 1143-1147.
57. Lassance RM, Passaro CP, Martini SV, Castiglione RC, Gutierrez TM, Abreu SC, et al. Does the use of recombinant AAV2 in pulmonary gene therapy damage lung function? Respir Physiol Neurobiol 2008; 160: 91-98.
58. Duan D, Yue Y, Yan Z, McCray PB Jr, Engelhardt JF. Polarity influences the efficiency of recombinant adenoassociated virus infection in differentiated airway epithelia. Hum Gene Ther 1998; 9: 2761-2776.
59. Wang D, Fischer H, Zhang L, Fan P, Ding RX, Dong J. Efficient CFTR expression from AAV vectors packaged with promoters - the second generation. Gene Ther 1999; 6: 667-675.
60. Walters RW, Yi SM, Keshavjee S, Brown KE, Welsh MJ, Chiorini JA, et al. Binding of adeno-associated virus type 5 to 2,3-linked sialic acid is required for gene transfer. J Biol Chem 2001; 276: 20610-20616.
61. Di Pasquale G, Davidson BL, Stein CS, Martins I, Scudiero D, Monks A, et al. Identification of PDGFR as a receptor for AAV-5 transduction. Nat Med 2003; 9: 1306-1312.
62. Hildinger M, Auricchio A, Gao G, Wang L, Chirmule N, Wilson JM. Hybrid vectors based on adeno-associated virus serotypes 2 and 5 for muscle-directed gene transfer. J Virol 2001; 75: 6199-6203.
63. Halbert CL, Rutledge EA, Allen JM, Russell DW, Miller AD. Repeat transduction in the mouse lung by using adeno-associated virus vectors with different serotypes. J Virol 2000; 74: 1524-1532.
64. Sirninger J, Muller C, Braag S, Tang Q, Yue H, Detrisac C, et al. Functional characterization of a recombinant adeno-associated virus 5-pseudotyped cystic fibrosis transmembrane conductance regulator vector. Hum Gene Ther 2004; 15: 832-841.
65. Fischer AC, Smith CI, Cebotaru L, Zhang X, Askin FB, Wright J, et al. Expression of a truncated cystic fibrosis transmembrane conductance regulator with an AAV5-pseudotyped vector in primates. Mol Ther 2007; 15: 756-763.
66. Martini SV, Fagundes SS, Schmidt AC, Avila M, Ornellas DS, Ribas VT, et al. Does the use of recombinant AAV5 in pulmonary gene therapy lead to lung damage? Respir Physiol Neurobiol 2009; 168: 203-209.
67. Limberis MP, Vandenberghe LH, Zhang L, Pickles RJ, Wilson JM. Transduction efficiencies of novel AAV vectors in mouse airway epithelium in vivo and human ciliated airway epithelium in vitro. Mol Ther 2009; 17: 294-301.
68. Limberis MP, Wilson JM. Adeno-associated virus serotype 9 vectors transduce murine alveolar and nasal epithelia and can be readministered. Proc Natl Acad Sci U S A 2006; 103: 12993-12998.
69. Auricchio A, O"Connor E, Weiner D, Gao GP, Hildinger M, Wang L, et al. Noninvasive gene transfer to the lung for systemic delivery of therapeutic proteins. J Clin Invest 2002; 110: 499-504.
70. Pfeifer C, Aneja MK, Hasenpusch G, Rudolph C. Adeno-associated virus serotype 9-mediated pulmonary transgene expression: effect of mouse strain, animal gender and lung inflammation. Gene Ther 2011. doi:10.1038/gt.2011.42.
71. Petrs-Silva H, Dinculescu A, Li Q, Min SH, Chiodo V, Pang JJ, et al. High-efficiency transduction of the mouse retina by tyrosine-mutant AAV serotype vectors. Mol Ther 2009; 17: 463-471.
72. Wu P, Xiao W, Conlon T, Hughes J, Agbandje-McKenna M, Ferkol T, et al. Mutational analysis of the adeno-associated virus type 2 (AAV2) capsid gene and construction of AAV2 vectors with altered tropism. J Virol 2000; 74: 8635-8647.
73. Liqun Wang R, McLaughlin T, Cossette T, Tang Q, Foust K, Campbell-Thompson M, et al. Recombinant AAV serotype and capsid mutant comparison for pulmonary gene transfer of alpha-1-antitrypsin using invasive and noninvasive delivery. Mol Ther 2009; 17: 81-87.
74. Markusic DM, Herzog RW, Aslanidi GV, Hoffman BE, Li B, Li M, et al. High-efficiency transduction and correction of murine hemophilia B using AAV2 vectors devoid of multiple surface-exposed tyrosines. Mol Ther 2010; 18: 2048-2056.
75. Zhong L, Li B, Mah CS, Govindasamy L, Agbandje-McKenna M, Cooper M, et al. Next generation of adeno-associated virus 2 vectors: point mutations in tyrosines lead to high-efficiency transduction at lower doses. Proc Natl Acad Sci U S A 2008; 105: 7827-7832.
76. Duan D, Yue Y, Yan Z, Yang J, Engelhardt JF. Endosomal processing limits gene transfer to polarized airway epithelia by adeno-associated virus. J Clin Invest 2000; 105: 1573-1587.
Research supported by PRONEX-FAPERJ, CNPq, and FAPERJ.
 Address for correspondence: M.M. Morales, Laboratório de Fisiologia Celular e Molecular, Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, 21941-902 Rio de Janeiro, RJ, Brasil. Fax: +55-21-2280-8193. E-mail: mmorales@biof.ufrj.br
Address for correspondence: M.M. Morales, Laboratório de Fisiologia Celular e Molecular, Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, 21941-902 Rio de Janeiro, RJ, Brasil. Fax: +55-21-2280-8193. E-mail: mmorales@biof.ufrj.br
Received May 30, 2011. Accepted September 13, 2011. Available online September 23, 2011. Published November 14, 2011.
The Brazilian Journal of Medical and Biological Research is partially financed by
All the contents of this journal, except where otherwise noted, is licensed under a Creative Commons Attribution License
- 1. Shimotohno K, Temin HM. Formation of infectious progeny virus after insertion of herpes simplex thymidine kinase gene into DNA of an avian retrovirus. Cell 1981; 26: 67-77.
- 2. Wei CM, Gibson M, Spear PG, Scolnick EM. Construction and isolation of a transmissible retrovirus containing the src gene of Harvey murine sarcoma virus and the thymidine kinase gene of herpes simplex virus type 1. J Virol 1981; 39: 935-944.
- 3. Morales MM, Capella MA, Lopes AG. Structure and function of the cystic fibrosis transmembrane conductance regulator. Braz J Med Biol Res 1999; 32: 1021-1028.
- 4. Cohen-Haguenauer O, Boiron M. [Gene therapy: facts and prospects]. Pathol Biol 1992; 40: 5-13.
- 5. Flotte TR, Laube BL. Gene therapy in cystic fibrosis. Chest 2001; 120: 124S-131S.
- 6. Davies JC, Alton EW. Gene therapy for cystic fibrosis. Proc Am Thorac Soc 2010; 7: 408-414.
- 7. Flotte TR. Recombinant adeno-associated virus vectors for cystic fibrosis gene therapy. Curr Opin Mol Ther 2001; 3: 497-502.
- 8. Mueller C, Flotte TR. Gene therapy for cystic fibrosis. Clin Rev Allergy Immunol 2008; 35: 164-178.
- 9. Schultz BR, Chamberlain JS. Recombinant adeno-associated virus transduction and integration. Mol Ther 2008; 16: 1189-1199.
- 10. Blacklow NR, Hoggan MD, Rowe WP. Serologic evidence for human infection with adenovirus-associated viruses. J Natl Cancer Inst 1968; 40: 319-327.
- 11. Gao GP, Alvira MR, Wang L, Calcedo R, Johnston J, Wilson JM. Novel adeno-associated viruses from rhesus monkeys as vectors for human gene therapy. Proc Natl Acad Sci U S A 2002; 99: 11854-11859.
- 12. Rabinowitz JE, Rolling F, Li C, Conrath H, Xiao W, Xiao X, et al. Cross-packaging of a single adeno-associated virus (AAV) type 2 vector genome into multiple AAV serotypes enables transduction with broad specificity. J Virol 2002; 76: 791-801.
- 13. Daya S, Berns KI. Gene therapy using adeno-associated virus vectors. Clin Microbiol Rev 2008; 21: 583-593.
- 14. Flotte TR, Zeitlin PL, Reynolds TC, Heald AE, Pedersen P, Beck S, et al. Phase I trial of intranasal and endobronchial administration of a recombinant adeno-associated virus serotype 2 (rAAV2)-CFTR vector in adult cystic fibrosis patients: a two-part clinical study. Hum Gene Ther 2003; 14: 1079-1088.
- 15. Kotin RM, Siniscalco M, Samulski RJ, Zhu XD, Hunter L, Laughlin CA, et al. Site-specific integration by adeno-associated virus. Proc Natl Acad Sci U S A 1990; 87: 2211-2215.
- 16. Atchison RW, Casto BC, Hammon WM. Adenovirus-associated defective virus particles. Science 1965; 149: 754-756.
- 17. Goodman S, Xiao X, Donahue RE, Moulton A, Miller J, Walsh C, et al. Recombinant adeno-associated virus-mediated gene transfer into hematopoietic progenitor cells. Blood 1994; 84: 1492-1500.
- 18. Hermonat PL, Labow MA, Wright R, Berns KI, Muzyczka N. Genetics of adeno-associated virus: isolation and preliminary characterization of adeno-associated virus type 2 mutants. J Virol 1984; 51: 329-339.
- 19. Hauswirth WW, Berns KI. Origin and termination of adeno-associated virus DNA replication. Virology 1977; 78: 488-499.
- 20. Lu Y. Recombinant adeno-associated virus as delivery vector for gene therapy - a review. Stem Cells Dev 2004; 13: 133-145.
- 21. Balague C, Kalla M, Zhang WW. Adeno-associated virus Rep78 protein and terminal repeats enhance integration of DNA sequences into the cellular genome. J Virol 1997; 71: 3299-3306.
- 22. Grimm D. Production methods for gene transfer vectors based on adeno-associated virus serotypes. Methods 2002; 28: 146-157.
- 23. Buning H, Ried MU, Perabo L, Gerner FM, Huttner NA, Enssle J, et al. Receptor targeting of adeno-associated virus vectors. Gene Ther 2003; 10: 1142-1151.
- 24. Russell DW, Kay MA. Adeno-associated virus vectors and hematology. Blood 1999; 94: 864-874.
- 25. Young SM Jr, McCarty DM, Degtyareva N, Samulski RJ. Roles of adeno-associated virus Rep protein and human chromosome 19 in site-specific recombination. J Virol 2000; 74: 3953-3966.
- 26. Young SM Jr, Samulski RJ. Adeno-associated virus (AAV) site-specific recombination does not require a Rep-dependent origin of replication within the AAV terminal repeat. Proc Natl Acad Sci U S A 2001; 98: 13525-13530.
- 27. Erles K, Sebokova P, Schlehofer JR. Update on the prevalence of serum antibodies (IgG and IgM) to adeno-associated virus (AAV). J Med Virol 1999; 59: 406-411.
- 28. Halbert CL, Miller AD, McNamara S, Emerson J, Gibson RL, Ramsey B, et al. Prevalence of neutralizing antibodies against adeno-associated virus (AAV) types 2, 5, and 6 in cystic fibrosis and normal populations: Implications for gene therapy using AAV vectors. Hum Gene Ther 2006; 17: 440-447.
- 29. Duan D, Sharma P, Yang J, Yue Y, Dudus L, Zhang Y, et al. Circular intermediates of recombinant adeno-associated virus have defined structural characteristics responsible for long-term episomal persistence in muscle tissue. J Virol 1998; 72: 8568-8577.
- 30. Davidson BL, Stein CS, Heth JA, Martins I, Kotin RM, Derksen TA, et al. Recombinant adeno-associated virus type 2, 4, and 5 vectors: transduction of variant cell types and regions in the mammalian central nervous system. Proc Natl Acad Sci U S A 2000; 97: 3428-3432.
- 31. Kwon I, Schaffer DV. Designer gene delivery vectors: molecular engineering and evolution of adeno-associated viral vectors for enhanced gene transfer. Pharm Res 2008; 25: 489-499.
- 32. Fulmer SB, Schwiebert EM, Morales MM, Guggino WB, Cutting GR. Two cystic fibrosis transmembrane conductance regulator mutations have different effects on both pulmonary phenotype and regulation of outwardly rectified chloride currents. Proc Natl Acad Sci U S A 1995; 92: 6832-6836.
- 33. Dong JY, Fan PD, Frizzell RA. Quantitative analysis of the packaging capacity of recombinant adeno-associated virus. Hum Gene Ther 1996; 7: 2101-2112.
- 34. Flotte TR, Afione SA, Conrad C, McGrath SA, Solow R, Oka H, et al. Stable in vivo expression of the cystic fibrosis transmembrane conductance regulator with an adeno-associated virus vector. Proc Natl Acad Sci U S A 1993; 90: 10613-10617.
- 35. Zhang LN, Karp P, Gerard CJ, Pastor E, Laux D, Munson K, et al. Dual therapeutic utility of proteasome modulating agents for pharmaco-gene therapy of the cystic fibrosis airway. Mol Ther 2004; 10: 990-1002.
- 36. Lee TW, Matthews DA, Blair GE. Novel molecular approaches to cystic fibrosis gene therapy. Biochem J 2005; 387: 1-15.
- 37. Flotte TR, Afione SA, Solow R, Drumm ML, Markakis D, Guggino WB, et al. Expression of the cystic fibrosis transmembrane conductance regulator from a novel adeno-associated virus promoter. J Biol Chem 1993; 268: 3781-3790.
- 38. Zabner J, Seiler M, Walters R, Kotin RM, Fulgeras W, Davidson BL, et al. Adeno-associated virus type 5 (AAV5) but not AAV2 binds to the apical surfaces of airway epithelia and facilitates gene transfer. J Virol 2000; 74: 3852-3858.
- 39. Ostedgaard LS, Rokhlina T, Karp PH, Lashmit P, Afione S, Schmidt M, et al. A shortened adeno-associated virus expression cassette for CFTR gene transfer to cystic fibrosis airway epithelia. Proc Natl Acad Sci U S A 2005; 102: 2952-2957.
- 40. Cebotaru L, Vij N, Ciobanu I, Wright J, Flotte T, Guggino WB. Cystic fibrosis transmembrane regulator missing the first four transmembrane segments increases wild type and DeltaF508 processing. J Biol Chem 2008; 283: 21926-21933.
- 41. Summerford C, Samulski RJ. Membrane-associated heparan sulfate proteoglycan is a receptor for adeno-associated virus type 2 virions. J Virol 1998; 72: 1438-1445.
- 42. Summerford C, Bartlett JS, Samulski RJ. AlphaVbeta5 integrin: a co-receptor for adeno-associated virus type 2 infection. Nat Med 1999; 5: 78-82.
- 43. Qing K, Mah C, Hansen J, Zhou S, Dwarki V, Srivastava A. Human fibroblast growth factor receptor 1 is a co-receptor for infection by adeno-associated virus 2. Nat Med 1999; 5: 71-77.
- 44. Kashiwakura Y, Tamayose K, Iwabuchi K, Hirai Y, Shimada T, Matsumoto K, et al. Hepatocyte growth factor receptor is a coreceptor for adeno-associated virus type 2 infection. J Virol 2005; 79: 609-614.
- 45. Asokan A, Hamra JB, Govindasamy L, Agbandje-McKenna M, Samulski RJ. Adeno-associated virus type 2 contains an integrin alpha5beta1 binding domain essential for viral cell entry. J Virol 2006; 80: 8961-8969.
- 46. Akache B, Grimm D, Pandey K, Yant SR, Xu H, Kay MA. The 37/67-kilodalton laminin receptor is a receptor for adeno-associated virus serotypes 8, 2, 3, and 9. J Virol 2006; 80: 9831-9836.
- 47. Bartlett JS, Wilcher R, Samulski RJ. Infectious entry pathway of adeno-associated virus and adeno-associated virus vectors. J Virol 2000; 74: 2777-2785.
- 48. Conrad CK, Allen SS, Afione SA, Reynolds TC, Beck SE, Fee-Maki M, et al. Safety of single-dose administration of an adeno-associated virus (AAV)-CFTR vector in the primate lung. Gene Ther 1996; 3: 658-668.
- 49. Beck SE, Jones LA, Chesnut K, Walsh SM, Reynolds TC, Carter BJ, et al. Repeated delivery of adeno-associated virus vectors to the rabbit airway. J Virol 1999; 73: 9446-9455.
- 50. Wagner JA, Messner AH, Moran ML, Daifuku R, Kouyama K, Desch JK, et al. Safety and biological efficacy of an adeno-associated virus vector-cystic fibrosis transmembrane regulator (AAV-CFTR) in the cystic fibrosis maxillary sinus. Laryngoscope 1999; 109: 266-274.
- 51. Wagner JA, Moran ML, Messner AH, Daifuku R, Conrad CK, Reynolds T, et al. A phase I/II study of tgAAV-CF for the treatment of chronic sinusitis in patients with cystic fibrosis. Hum Gene Ther 1998; 9: 889-909.
- 52. Wagner JA, Reynolds T, Moran ML, Moss RB, Wine JJ, Flotte TR, et al. Efficient and persistent gene transfer of AAV-CFTR in maxillary sinus. Lancet 1998; 351: 1702-1703.
- 53. Aitken ML, Moss RB, Waltz DA, Dovey ME, Tonelli MR, McNamara SC, et al. A phase I study of aerosolized administration of tgAAVCF to cystic fibrosis subjects with mild lung disease. Hum Gene Ther 2001; 12: 1907-1916.
- 54. Moss RB, Rodman D, Spencer LT, Aitken ML, Zeitlin PL, Waltz D, et al. Repeated adeno-associated virus serotype 2 aerosol-mediated cystic fibrosis transmembrane regulator gene transfer to the lungs of patients with cystic fibrosis: a multicenter, double-blind, placebo-controlled trial. Chest 2004; 125: 509-521.
- 55. Moss RB, Milla C, Colombo J, Accurso F, Zeitlin PL, Clancy JP, et al. Repeated aerosolized AAV-CFTR for treatment of cystic fibrosis: a randomized placebo-controlled phase 2B trial. Hum Gene Ther 2007; 18: 726-732.
- 56. Flotte TR. Adeno-associated virus-based gene therapy for inherited disorders. Pediatr Res 2005; 58: 1143-1147.
- 57. Lassance RM, Passaro CP, Martini SV, Castiglione RC, Gutierrez TM, Abreu SC, et al. Does the use of recombinant AAV2 in pulmonary gene therapy damage lung function? Respir Physiol Neurobiol 2008; 160: 91-98.
- 58. Duan D, Yue Y, Yan Z, McCray PB Jr, Engelhardt JF. Polarity influences the efficiency of recombinant adenoassociated virus infection in differentiated airway epithelia. Hum Gene Ther 1998; 9: 2761-2776.
- 59. Wang D, Fischer H, Zhang L, Fan P, Ding RX, Dong J. Efficient CFTR expression from AAV vectors packaged with promoters - the second generation. Gene Ther 1999; 6: 667-675.
- 60. Walters RW, Yi SM, Keshavjee S, Brown KE, Welsh MJ, Chiorini JA, et al. Binding of adeno-associated virus type 5 to 2,3-linked sialic acid is required for gene transfer. J Biol Chem 2001; 276: 20610-20616.
- 61. Di Pasquale G, Davidson BL, Stein CS, Martins I, Scudiero D, Monks A, et al. Identification of PDGFR as a receptor for AAV-5 transduction. Nat Med 2003; 9: 1306-1312.
- 62. Hildinger M, Auricchio A, Gao G, Wang L, Chirmule N, Wilson JM. Hybrid vectors based on adeno-associated virus serotypes 2 and 5 for muscle-directed gene transfer. J Virol 2001; 75: 6199-6203.
- 63. Halbert CL, Rutledge EA, Allen JM, Russell DW, Miller AD. Repeat transduction in the mouse lung by using adeno-associated virus vectors with different serotypes. J Virol 2000; 74: 1524-1532.
- 64. Sirninger J, Muller C, Braag S, Tang Q, Yue H, Detrisac C, et al. Functional characterization of a recombinant adeno-associated virus 5-pseudotyped cystic fibrosis transmembrane conductance regulator vector. Hum Gene Ther 2004; 15: 832-841.
- 65. Fischer AC, Smith CI, Cebotaru L, Zhang X, Askin FB, Wright J, et al. Expression of a truncated cystic fibrosis transmembrane conductance regulator with an AAV5-pseudotyped vector in primates. Mol Ther 2007; 15: 756-763.
- 66. Martini SV, Fagundes SS, Schmidt AC, Avila M, Ornellas DS, Ribas VT, et al. Does the use of recombinant AAV5 in pulmonary gene therapy lead to lung damage? Respir Physiol Neurobiol 2009; 168: 203-209.
- 67. Limberis MP, Vandenberghe LH, Zhang L, Pickles RJ, Wilson JM. Transduction efficiencies of novel AAV vectors in mouse airway epithelium in vivo and human ciliated airway epithelium in vitro Mol Ther 2009; 17: 294-301.
- 68. Limberis MP, Wilson JM. Adeno-associated virus serotype 9 vectors transduce murine alveolar and nasal epithelia and can be readministered. Proc Natl Acad Sci U S A 2006; 103: 12993-12998.
- 69. Auricchio A, O"Connor E, Weiner D, Gao GP, Hildinger M, Wang L, et al. Noninvasive gene transfer to the lung for systemic delivery of therapeutic proteins. J Clin Invest 2002; 110: 499-504.
- 70. Pfeifer C, Aneja MK, Hasenpusch G, Rudolph C. Adeno-associated virus serotype 9-mediated pulmonary transgene expression: effect of mouse strain, animal gender and lung inflammation. Gene Ther 2011. doi:10.1038/gt.2011.42.
- 71. Petrs-Silva H, Dinculescu A, Li Q, Min SH, Chiodo V, Pang JJ, et al. High-efficiency transduction of the mouse retina by tyrosine-mutant AAV serotype vectors. Mol Ther 2009; 17: 463-471.
- 72. Wu P, Xiao W, Conlon T, Hughes J, Agbandje-McKenna M, Ferkol T, et al. Mutational analysis of the adeno-associated virus type 2 (AAV2) capsid gene and construction of AAV2 vectors with altered tropism. J Virol 2000; 74: 8635-8647.
- 73. Liqun Wang R, McLaughlin T, Cossette T, Tang Q, Foust K, Campbell-Thompson M, et al. Recombinant AAV serotype and capsid mutant comparison for pulmonary gene transfer of alpha-1-antitrypsin using invasive and noninvasive delivery. Mol Ther 2009; 17: 81-87.
- 74. Markusic DM, Herzog RW, Aslanidi GV, Hoffman BE, Li B, Li M, et al. High-efficiency transduction and correction of murine hemophilia B using AAV2 vectors devoid of multiple surface-exposed tyrosines. Mol Ther 2010; 18: 2048-2056.
- 75. Zhong L, Li B, Mah CS, Govindasamy L, Agbandje-McKenna M, Cooper M, et al. Next generation of adeno-associated virus 2 vectors: point mutations in tyrosines lead to high-efficiency transduction at lower doses. Proc Natl Acad Sci U S A 2008; 105: 7827-7832.
- 76. Duan D, Yue Y, Yan Z, Yang J, Engelhardt JF. Endosomal processing limits gene transfer to polarized airway epithelia by adeno-associated virus. J Clin Invest 2000; 105: 1573-1587.
Correspondence and Footnotes
Publication Dates
-
Publication in this collection
01 Nov 2011 -
Date of issue
Nov 2011
History
-
Accepted
13 Sept 2011 -
Received
30 May 2011