Abstract
Ginkgo biloba extract (GbE) has been indicated as an efficient medicine for the treatment of diabetes mellitus type 2. It remains unclear if its effects are due to an improvement of the insulin signaling cascade, especially in obese subjects. The aim of the present study was to evaluate the effect of GbE on insulin tolerance, food intake, body adiposity, lipid profile, fasting insulin, and muscle levels of insulin receptor substrate 1 (IRS-1), protein tyrosine phosphatase 1B (PTP-1B), and protein kinase B (Akt), as well as Akt phosphorylation, in diet-induced obese rats. Rats were fed with a high-fat diet (HFD) or a normal fat diet (NFD) for 8 weeks. After that, the HFD group was divided into two groups: rats gavaged with a saline vehicle (HFD+V), and rats gavaged with 500 mg/kg of GbE diluted in the saline vehicle (HFD+Gb). NFD rats were gavaged with the saline vehicle only. At the end of the treatment, the rats were anesthetized, insulin was injected into the portal vein, and after 90s, the gastrocnemius muscle was removed. The quantification of IRS-1, Akt, and Akt phosphorylation was performed using Western blotting. Serum levels of fasting insulin and glucose, triacylglycerols and total cholesterol, and LDL and HDL fractions were measured. An insulin tolerance test was also performed. Ingestion of a hyperlipidic diet promoted loss of insulin sensitivity and also resulted in a significant increase in body adiposity, plasma triacylglycerol, and glucose levels. In addition, GbE treatment significantly reduced food intake and body adiposity while it protected against hyperglycemia and dyslipidemia in diet-induced obesity rats. It also enhanced insulin sensitivity in comparison to HFD+V rats, while it restored insulin-induced Akt phosphorylation, increased IRS-1, and reduced PTP-1B levels in gastrocnemius muscle. The present findings suggest that G. biloba might be efficient in preventing and treating obesity-induced insulin signaling impairment.
Ginkgo biloba extract; Obesity; Insulin tolerance; Body adiposity; Akt phosphorylation; Rats
Introduction
Obesity has become a major public health problem worldwide (11. Pereira-Lancha LO, Campos-Ferraz PL, Lancha AH Jr. Obesity: considerations about etiology, metabolism, and the use of experimental models. Diabetes Metab Syndr Obes 2012; 5: 75-87.,22. Pego-Fernandes PM, Bibas BJ, Deboni M. Obesity: the greatest epidemic of the 21st century? São Paulo Med J 2011; 129: 283-284, doi: 10.1590/S1516-31802011000500001.
https://doi.org/10.1590/S1516-3180201100...
). It has been suggested that high levels of abdominal adiposity and excess saturated fat intake promote obesity-related metabolic syndrome (33. Yang ZH, Miyahara H, Takeo J, Katayama M. Diet high in fat and sucrose induces rapid onset of obesity-related metabolic syndrome partly through rapid response of genes involved in lipogenesis, insulin signalling and inflammation in mice. Diabetol Metab Syndr 2012; 4: 32, doi: 10.1186/1758-5996-4-32.
https://doi.org/10.1186/1758-5996-4-32...
4. Trayhurn P. Adipose tissue in obesity - an inflammatory issue. Endocrinology 2005; 146: 1003-1005, doi: 10.1210/en.2004-1597.
https://doi.org/10.1210/en.2004-1597...
-55. Xu C, Yang X, Zu S, Han S, Zhang Z, Zhu G. Association between serum lipids, blood pressure, and simple anthropometric measures in an adult Chinese population. Arch Med Res 2008; 39: 610-617, doi: 10.1016/j.arcmed.2008.05.001.
https://doi.org/10.1016/j.arcmed.2008.05...
), and that the association between obesity, insulin resistance, and dyslipidemia potentiates the risk for cardiovascular diseases (66. Hilal Y, Acar TN, Koksal E, Gezmen KM, Akbulut G, Bilici S, et al. The association of anthropometric measurements and lipid profiles in Turkish hypertensive adults. Afr Health Sci 2011; 11: 407-413.).
Ginkgo biloba extract (GbE; a standardized preparation named EGb 761, which is obtained from the leaves of G. biloba) is one of the plant extracts most used in therapeutics (77. Park YM, Won JH, Yun KJ, Ryu JH, Han YN, Choi SK, et al. Preventive effect of Ginkgo biloba extract (GBB) on the lipopolysaccharide-induced expressions of inducible nitric oxide synthase and cyclooxygenase-2 via suppression of nuclear factor-kappaB in RAW 264.7 cells. Biol Pharm Bull 2006; 29: 985-990, doi: 10.1248/bpb.29.985.
https://doi.org/10.1248/bpb.29.985...
). GbE (50:1) contains about 24% flavonoid glycosides, 6% terpenoids (such as the diterpenoids A, B, C, M, and J ginkgolides, and the sesquiterpene bilobalide), 7% proanthocyanidins, and organic acids of low molecular weight (88. DeFeudis FV, Papadopoulos V, Drieu K. Ginkgo biloba extracts and cancer: a research area in its infancy. Fundam Clin Pharmacol 2003; 17: 405-417, doi: 10.1046/j.1472-8206.2003.00156.x.
https://doi.org/10.1046/j.1472-8206.2003...
). Antioxidant, anti-inflammatory, vasodilator, and anti-edematogenic properties have been reported for this plant (99. Mehlsen J, Drabaek H, Wiinberg N, Winther K. Effects of a Ginkgo biloba extract on forearm haemodynamics in healthy volunteers. Clin Physiol Funct Imaging 2002; 22: 375-378, doi: 10.1046/j.1475-097X.2002.00445.x.
https://doi.org/10.1046/j.1475-097X.2002...
,1010. Rostoker G, Behar A, Lagrue G. Vascular hyperpermeability in nephrotic edema. Nephron 2000; 85: 194-200, doi: 10.1159/000045661.
https://doi.org/10.1159/000045661...
).
Studies have suggested that GbE intake by type 2 diabetes mellitus (T2DM) patients reduces circulatory diseases and improves the glycemic profile (1111. Kudolo GB. The effect of 3-month ingestion of Ginkgo biloba extract on pancreatic beta-cell function in response to glucose loading in normal glucose tolerant individuals. J Clin Pharmacol 2000; 40: 647-654, doi: 10.1002/j.1552-4604.2000.tb05991.x.
https://doi.org/10.1002/j.1552-4604.2000...
,1212. Kudolo GB, Delaney D, Blodgett J. Short-term oral ingestion of Ginkgo biloba extract (EGb 761) reduces malondialdehyde levels in washed platelets of type 2 diabetic subjects. Diabetes Res Clin Pract 2005; 68: 29-38, doi: 10.1016/j.diabres.2004.08.007.
https://doi.org/10.1016/j.diabres.2004.0...
). Reduced glycemia was also reported in streptozotocin-induced diabetic rats (1313. Tanaka S, Han LK, Zheng YN, Okuda H. [Effects of the flavonoid fraction from Ginkgo biloba extract on the postprandial blood glucose elevation in rats]. Yakugaku Zasshi 2004; 124: 605-611, doi: 10.1248/yakushi.124.605.
https://doi.org/10.1248/yakushi.124.605...
). It has been suggested that these beneficial effects could result from a significant stimulation of pancreatic beta-cell function/insulin production (1111. Kudolo GB. The effect of 3-month ingestion of Ginkgo biloba extract on pancreatic beta-cell function in response to glucose loading in normal glucose tolerant individuals. J Clin Pharmacol 2000; 40: 647-654, doi: 10.1002/j.1552-4604.2000.tb05991.x.
https://doi.org/10.1002/j.1552-4604.2000...
) and from a significant decrease in glycated hemoglobin levels of T2DM patients (1414. Kudolo GB, Wang W, Javors M, Blodgett J. The effect of the ingestion of Ginkgo biloba extract (EGb 761) on the pharmacokinetics of metformin in non-diabetic and type 2 diabetic subjects - a double blind placebo-controlled, crossover study. Clin Nutr 2006; 25: 606-616, doi: 10.1016/j.clnu.2005.12.012.
https://doi.org/10.1016/j.clnu.2005.12.0...
). Despite the evidence for a positive effect of GbE on glycemic homeostasis, data in the literature are scarce about the action of GbE on obesity-related insulin intolerance.
Thus, it is possible to speculate that GbE might also improve insulin insensitivity on the obesity-related impaired insulin signaling cascade. In this context, the aim of the present study was to evaluate the effect of GbE on the amelioration of several parameters related to insulin tolerance in diet-induced obese rats.
Material and Methods
The present study was approved by the Ethics Committee on Animal Research of the Universidade Federal de São Paulo (Process #0321/11). All efforts were made to minimize suffering.
Animals
Male Wistar rats (2 months old) were randomly assigned to a normal fat diet (NFD) group or a high-fat diet (HFD) group. The NFD group was fed standard pelleted laboratory chow (Nuvilab¯, Brazil), which provided 35.9% of energy as carbohydrate, 22.7% as protein, and 4.5% as fat, while the HFD group was fed a highly fat-enriched diet, which was prepared by adding to the standard chow, 28% (w/w) fat, 10% (w/w) sucrose, 20% (w/w) casein, to obtain the protein content of the control diet, and butylated hydroxytoluene in the amount of 0.4% (w/w) of additional oil. This provided 19.5% of energy as carbohydrate, 23.2% as protein, and 57.3% as fat. Table 1 shows the macronutrient and fatty acid compositions of the diets.
Animals were housed four to a cage and maintained in controlled conditions of lighting (12:12-h light-dark, lights on at 6:00 am) and temperature (23±1°C), with free access to food and water. The NFD group remained on its respective diet for the entire study, whereas, after 8 weeks, the HFD animals were subdivided into two subgroups, according to the phytotherapy treatment described below.
Phytotherapy treatment
GbE was obtained from Southern Anhui Dapeng (China). GbE contained flavone glycosides (26.12%), terpenoids (6.86%), ginkgolide A (2.20%), ginkgolide B (1.11%), ginkgolide C (1.05%), and bilobalide (2.50%).
Phytotherapy treatment was performed for 14 days. The HFD group was subdivided into two groups: HFD+Gb and HFD+V. The HFD+Gb group was gavaged daily with 500 mg/kg GbE diluted in 2 mL 0.9% saline (as vehicle), while the HFD+V and NFD groups were gavaged with 2 mL of the vehicle alone.
Food intake and body weight measurements
Both body weight and 24-h food intake were measured weekly from 2 to 4 months of age. During the phytotherapy treatment, animals were housed individually, and individual 24-h food intake and body weight were measured weekly. Daily food intake was calculated as the difference between the amount of food offered and the food remaining after 24 h.
Serum parameters and weight of fat pads
Rats were decapitated after a 6-h fasting period. Blood was collected, centrifuged at 1258 g for 15 min at 4°C, and the serum was stored at −80°C until analyses. Glucose, triacylglycerol (TAG), total cholesterol, LDL-cholesterol, and HDL-cholesterol concentrations were determined by enzymatic colorimetric methods using commercial kits (Labtest Diagnóstica, Brazil). Insulin levels were measured using an ELISA kit (Millipore¯, USA). The homeostasis model assessment estimate of insulin receptor (HOMA-IR) was calculated from fasting insulin (µU/mL) and fasting glucose (mmol/L), as follows: HOMA-IR = (insulin×glucose)/22.5.
Retroperitoneal, mesenteric, and epididymal adipose tissue depots were dissected and weighed immediately.
Insulin tolerance test (ITT)
Tail blood samples were collected before (0 min) and 5, 10, 15, 20, 25, and 30 min after an intraperitoneal injection of regular insulin (2 U/kg body weight, Humulin¯ Human Insulin, Eli Lilly do Brasil Ltda., Brazil). A blood drop was added to reactive strips, and glucose levels were measured using a glucometer (One Touch Ultra, Johnson & Johnson, USA).
Western blot analyses
The rats were anesthetized with sodium thiopental (80 mg/kg body weight, intraperitoneally). The abdominal cavity was opened, the portal vein was exposed, and 6 µg of insulin was injected. Gastrocnemius muscle was removed before and 90 s after the infusion of insulin and homogenized in 1.0 mL of solubilization buffer (100 mM Tris, pH 7.5, 0.1 mg/mL aprotinin, 2 mM phenylmethylsulfonylfluoride, 10 mM sodium orthovanadate, 100 mM sodium fluoride, 10 mM sodium pyrophosphate, and 10 mM EDTA). Triton X-100 was added to a final concentration of 10%. Samples were centrifuged at 16,000 g for 40 min and equal amounts of proteins were separated using 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis. Proteins were then transferred to nitrocellulose membranes by a semi-dry transfer apparatus (p-Akt/Akt: protein kinase B, IR: insulin receptor, and PTP-1B: protein tyrosine phosphatase 1B) or by a wet transfer apparatus (IRS-1 and IRS-2: insulin receptor substrates 1 and 2; Bio-Rad, USA).
Nonspecific protein binding to the nitrocellulose was reduced by preincubating the membrane overnight at 4°C in blocking buffer (1% bovine serum albumin, 1 M Tris, pH 7.5, 5 M NaCl, 0.02% Tween-20). Membranes were then incubated overnight with the primary antibody against phospho-Akt (Ser-473 9271S, Cell Signaling, USA), IR (sc-711), IRS-1 (sc-559), IRS-2 (sc-8299), and PTP-1B (sc-1718), obtained from Santa Cruz Biotechnology (USA). All membranes were then incubated with specific secondary antibody conjugated with horseradish peroxidase (Cell Signaling 7074S for membranes incubated with IR, IRS-1, and IRS2; Sigma-Aldrich, USA, A-51420 for membranes incubated with PTP-1B) followed by chemiluminescence detection (Amersham Biosciences, USA). The same membranes used for phospho-Akt were stripped and reprobed with the antibody against Akt (Cell Signaling 9272S), followed by incubation with specific secondary antibody conjugated with horseradish peroxidase (Cell Signaling 7074S). The membranes used for IR, IRS-1, IRS-2, Akt, and PTP-1B were stripped and reprobed with the antibody against β-tubulin (Cell Signaling 2146S).
Quantitative analysis was performed with the Scion Image software (Scion Corp., USA). In all experiments, at least one sample from each group was analyzed in parallel, and the results are reported as percentage change relative to the NFD group.
Statistical analyses
Statistical analyses were performed using PASW Statistics version 19 (SPSS Inc., USA) with the level of statistical significance set at P<0.05. Comparisons between two groups were performed by the Student t-test. Comparisons among three groups were analyzed by one-way ANOVA followed by the Tukey HSD test for multiple comparisons. Nonparametrical data were analyzed by the Kruskal-Wallis test followed by the Dunnet T3 test for multiple comparisons among all groups. Two-way ANOVA (time vs phytotherapy treatment) and the Tukey HSD post hoc analysis were used for the comparison of glucose levels in response to ITT.
Results
Food intake and body weight during an 8-week period of diet-induced obesity development
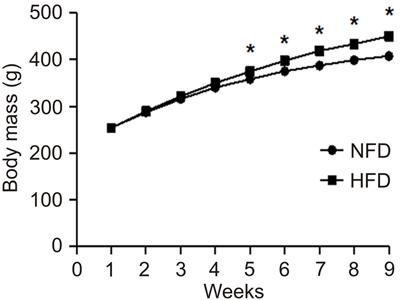
As can be seen in Figure 1, HFD rats presented a significant increase in body mass compared to the NFD group at 5 (5.34%, P=0.023), 6 (6.92%, P=0.004), 7 (8.70%, P=0.002), 8 (9.37%, P<0.001), and 9 (10.78%, P<0.001) weeks of hyperlipidic diet obesity induction.
Body mass of normal fat diet (NFD; n=16) and high-fat diet (HFD; n=71) groups during obesity-induction period. *P<0.05 vs NFD (Student t-test).
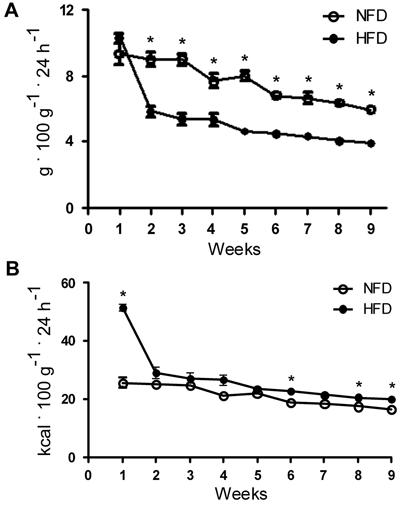
Figure 2A shows that, from the second to ninth week, the HFD group presented a significantly lower meal consumption (P<0.001) compared to the NFD group, eating approximately 35% less. In relation to energy intake, it can be observed in Figure 2B that the HFD group during the first week presented a significant increase of 50% (P<0.001) compared to the NFD group. However, from the second week, the increase only reached statistical significance at 6 (21.06%, P=0.008), 8 (17.61%, P=0.033), and 9 weeks (22.17%, P=0.012).
A, Food intake (g·100 g-1·24 h-1) and B, energy intake (kcal·100 g-1·24 h-1) of normal fat diet (NFD; n=16) and high-fat diet (HFD; n=71) groups during obesity-induction period. *P<0.05 vs NFD (Student t-test).
Food intake and body adiposity in response to phytotherapy treatment
The cumulative food intake during the first 13 days of phytotherapy treatment is illustrated at Figure 3A. The HFD+V group demonstrated a significant reduction in food intake compared to the NFD group (45.54%, P<0.001), while the HFD+Gb group showed a greater significant reduction of 55.33% (P<0.001) compared to the NFD group. It is interesting to note in Figure 3A that the HFD+Gb group ingested 18% less than the HFD+V group (P<0.001). In relation to energy intake, it can be observed that the HFD+Gb group also presented a significant reduction of 18% compared to the HFD+V group (P<0.001).
A, Cumulative food intake (g) and B, cumulative energy intake (kcal/100 g) of normal fat diet (NFD; n=12), high-fat diet plus vehicle (HFD+V; n=25), and high-fat diet plus Ginkgo biloba (HFD+Gb; n=20) groups during phytotherapy treatment. *P<0.05 vs NFD, #P<0.05 vs HFD+V (one-way ANOVA followed by the Tukey HSD test).
Figure 4 shows that the HFD+V group presented a significant increase (P<0.001) of retroperitoneal, epididymal, and mesenteric adipose tissue depots compared to the NFD group. In addition, the HFD+Gb group presented a significant reduction of retroperitoneal (27.94%, P<0.010) and epididymal (14.20%, P<0.05) adipose tissue compared to the HFD+V group.
Mass weight (g) of retroperitoneal, epididymal and mesenteric white adipose tissue depots of normal fat diet (NFD; n=16), high-fat diet plus vehicle (HFD+V; n=11), and high-fat diet plus Ginkgo biloba (HFD+Gb; n=9) groups after phytotherapy. *P<0.05 vs NFD, #P<0.05 vs HFD+V (one-way ANOVA followed by the Tukey HSD test).
Insulin sensitivity
Figure 5 depicts the results from the ITT test. It can be seen that the HFD+V group presented elevated glycemia levels compared to the NFD group at 5, 10, 20, 25, and 30 min (24.2, 26.7, 35.8%, P<0.01; 58.4 and 55.2%, P<0.001, respectively). The HFD+Gb group presented similar glycemia levels compared to the NFD group while presenting a significant decrease compared to the HFD+V group at 5, 10, 25, and 30 min (−18.0%, P<0.05; −22.9%, P<0.01; −30.0%, P<0.01; and −27.6%, P<0.05, respectively).
Insulin tolerance test (ITT) of normal fat diet (NFD; n=11), high-fat diet plus vehicle (HFD+V; n=6), and high-fat diet plus Ginkgo biloba (HFD+Gb; n=6) groups after phytotherapy treatment. *P<0.05 vs NFD; #P<0.05 vs HFD+Gb (two-way ANOVA - time vs phytotherapy treatment - followed by Tukey HSD post hoc analysis).
Akt phosphorylation levels
Akt phosphorylation levels are shown in Figure 6. It can be seen that the insulin injection significantly increased Akt phosphorylation in gastrocnemius muscle by 1322% (P=0.034) compared to basal levels (Figure 6A). It is interesting to note, in Figure 6B, that insulin-induced Akt phosphorylation (HFD+V+) was impaired by the ingestion of a high-fat diet, since no change in relation to basal levels was observed (HFD+V−). On the other hand, in Figure 6C, it can be seen that prolonged administration of GbE promoted a significant increase of 426.2% (P=0.029) in insulin-induced Akt phosphorylation in relation to basal levels.
Akt phosphorylation levels in gastrocnemius muscle of A, NFD- (basal, n=5) and NFD+ (insulin-stimulated, n=5); B, HFD+V- (basal, n=6) and HFD+V+ (insulin-stimulated, n=6); and C, HFD+Gb- (basal, n=6) and HFD+Gb+ (insulin-stimulated, n=6) groups after phytotherapy treatment. Akt: protein kinase B; NFD: normal fat diet; HFD: high-fat diet; V: vehicle (saline); Gb: Ginkgo biloba. *P<0.05 vs basal (Student t-test).
IR, IRS-1, IRS-2, Akt, and PTP-1B levels
GbE treatment promoted a significant increase of 347.8% (P=0.0458) in IRS-1 levels with a concomitant reduction of 51.9% (P=0.0373) in PTP-1B levels compared to the HFD+V group (Figures 7 and 8, respectively). No differences were observed in IR, IRS-2, and Akt levels among all groups (data not shown).
IRS-1 levels in gastrocnemius muscle of NFD (n=10), HFD+V (n=7), and HFD+Gb (n=9) groups after phytotherapy treatment. IRS-1: insulin receptor substrate 1; NFD: normal fat diet; HFD: high-fat diet; V: vehicle (saline); Gb: Ginkgo biloba. #P<0.05 vs HFD+V (one-way ANOVA followed by the Tukey HSD test).
PTP-1B levels in gastrocnemius muscle of NFD (n=10), HFD+V (n=9), and HFD+Gb (n=8) groups after phyttherapy treatment. PTP-1B: protein tyrosine phosphatase 1B; NFD: normal fat diet; HFD: high-fat diet; V: vehicle (saline); Gb: Ginkgo biloba. #P<0.05 vs HFD+V (one-way ANOVA followed by the Tukey HSD test).
Biochemical parameters
All biochemical parameters are reported in Table 2. Glycemia was 21% higher in the HFD+V group compared to the NFD group (P<0.01). No differences were observed in the HFD+Gb group. In addition, GbE treatment promoted a three-fold increase of fasting insulinemia levels compared to the NFD group (P=0.048), while no differences were observed in the HFD+V group.
In comparison to the NFD group, the HFD+V group presented a significant increase in TAG plasma levels (17.90%, P=0.013), while the HFD+Gb group was not different. In addition, the HDF+V group presented a tendency to increased total cholesterol (16.43%, P=0.058) compared to the NFD group, while GbE treatment protected against changes in lipid profile. No significant differences were observed in LDL cholesterol and HDL cholesterol plasma levels.
Discussion
The present study demonstrated that high-fat intake caused a significant increase in both energy intake and body mass gain, especially from the sixth week during the obesity development period. In addition, high-fat diet intake promoted obesity in HFD+V rats, as can be observed by the significant increase in body adiposity. This effect might have resulted from the high energy intake. In agreement with our findings, Lladó et al. (1515. Lladó I, Rodriguez-Cuenca S, Pujol E, Monjo M, Estrany ME, Roca P, et al. Gender effects on adrenergic receptor expression and lipolysis in white adipose tissue of rats. Obes Res 2002; 10: 296-305, doi: 10.1038/oby.2002.41.
https://doi.org/10.1038/oby.2002.41...
) described increased retroperitoneal adipose tissue accumulation in rats fed a cafeteria diet. The authors suggested that it could be a consequence of impaired balance of alpha-2/beta-3 adrenergic receptors, which resulted in reduced lipolysis.
An interesting finding of the present study was the observation that GbE promoted a significant reduction in retroperitoneal and epididymal adipose tissue depots compared to the HFD+V group. The reduced food intake presented by the HFD+Gb group might have resulted in a reduction of body adiposity. Otherwise, the reduced body adiposity of HFD+Gb rats might be due to the lipolytic effect of GbE. In fact, Saponara and Bosisio (1616. Saponara R, Bosisio E. Inhibition of cAMP-phosphodiesterase by biflavones of Ginkgo biloba in rat adipose tissue. J Nat Prod 1998; 61: 1386-1387, doi: 10.1021/np970569m.
https://doi.org/10.1021/np970569m...
) demonstrated that the G. biloba biflavones stimulated lipolysis via inhibition of the cAMP-phosphodiesterase enzymatic complex in rat epididymal adipose tissue. In addition, another study showed evidence that the GbE biflavones stimulated lipolysis in cultures of 3T3-L1 adipocytes (1717. Dell'Agli M, Bosisio E. Biflavones of Ginkgo biloba stimulate lipolysis in 3T3-L1 adipocytes. Planta Med 2002; 68: 76-79, doi: 10.1055/s-2002-19876.
https://doi.org/10.1055/s-2002-19876...
).
As expected, a high-fat diet intake promoted a significant increase in plasma TAG levels while GbE treatment protected against diet-induced hypertriglyceridemia. It has been reported that GbE treatment did not alter plasma TAG levels (1212. Kudolo GB, Delaney D, Blodgett J. Short-term oral ingestion of Ginkgo biloba extract (EGb 761) reduces malondialdehyde levels in washed platelets of type 2 diabetic subjects. Diabetes Res Clin Pract 2005; 68: 29-38, doi: 10.1016/j.diabres.2004.08.007.
https://doi.org/10.1016/j.diabres.2004.0...
13. Tanaka S, Han LK, Zheng YN, Okuda H. [Effects of the flavonoid fraction from Ginkgo biloba extract on the postprandial blood glucose elevation in rats]. Yakugaku Zasshi 2004; 124: 605-611, doi: 10.1248/yakushi.124.605.
https://doi.org/10.1248/yakushi.124.605...
14. Kudolo GB, Wang W, Javors M, Blodgett J. The effect of the ingestion of Ginkgo biloba extract (EGb 761) on the pharmacokinetics of metformin in non-diabetic and type 2 diabetic subjects - a double blind placebo-controlled, crossover study. Clin Nutr 2006; 25: 606-616, doi: 10.1016/j.clnu.2005.12.012.
https://doi.org/10.1016/j.clnu.2005.12.0...
15. Lladó I, Rodriguez-Cuenca S, Pujol E, Monjo M, Estrany ME, Roca P, et al. Gender effects on adrenergic receptor expression and lipolysis in white adipose tissue of rats. Obes Res 2002; 10: 296-305, doi: 10.1038/oby.2002.41.
https://doi.org/10.1038/oby.2002.41...
16. Saponara R, Bosisio E. Inhibition of cAMP-phosphodiesterase by biflavones of Ginkgo biloba in rat adipose tissue. J Nat Prod 1998; 61: 1386-1387, doi: 10.1021/np970569m.
https://doi.org/10.1021/np970569m...
17. Dell'Agli M, Bosisio E. Biflavones of Ginkgo biloba stimulate lipolysis in 3T3-L1 adipocytes. Planta Med 2002; 68: 76-79, doi: 10.1055/s-2002-19876.
https://doi.org/10.1055/s-2002-19876...
-1818. Drieu K, Vranckx R, Benassayad C, Haourigi M, Hassid J, Yoa RG, et al. Effect of the extract of Ginkgo biloba (EGb 761) on the circulating and cellular profiles of polyunsaturated fatty acids: correlation with the anti-oxidant properties of the extract. Prostaglandins Leukot Essent Fatty Acids 2000; 63: 293-300, doi: 10.1054/plef.2000.0217.
https://doi.org/10.1054/plef.2000.0217...
). However, an important effect of GbE observed in the present study was the significant reduction in visceral adiposity, which might have helped to decrease plasma TAG levels. Similar to our hypothesis, Goldberg et al. (1919. Goldberg Y, Boaz M, Matas Z, Goldberg I, Shargorodsky M. Weight loss induced by nutritional and exercise intervention decreases arterial stiffness in obese subjects. Clin Nutr 2009; 28: 21-25, doi: 10.1016/j.clnu.2008.10.001.
https://doi.org/10.1016/j.clnu.2008.10.0...
) and Fayh et al. (2020. Fayh AP, Lopes AL, da Silva AM, Reischak-Oliveira A, Friedman R. Effects of 5% weight loss through diet or diet plus exercise on cardiovascular parameters of obese: a randomized clinical trial. Eur J Nutr 2013; 52: 1443-1450, doi: 10.1007/s00394-012-0450-1.
https://doi.org/10.1007/s00394-012-0450-...
) observed in obese subjects a significant reduction of total cholesterol, LDL-cholesterol, and TAG levels after a weight loss of 8% and 5% of their body mass, respectively. It is possible that the effect presented by GbE might occur only in a process of adiposity loss induced by the treatment.
Our results also demonstrated that high-fat intake decreased insulin sensitivity and resulted in hyperglycemia, indicating that excess saturated fat intake might disrupt peripheral insulin activity. Such an effect is supported by a study, which demonstrated the development of peripheral insulin resistance in rats fed a high-fat lard-enriched diet during a 4-week period (2121. Tremblay F, Lavigne C, Jacques H, Marette A. Defective insulin-induced GLUT4 translocation in skeletal muscle of high fat-fed rats is associated with alterations in both Akt/protein kinase B and atypical protein kinase C (zeta/lambda) activities. Diabetes 2001; 50: 1901-1910, doi: 10.2337/diabetes.50.8.1901.
https://doi.org/10.2337/diabetes.50.8.19...
). On the other hand, GbE treatment protected against hyperglycemia and improved insulin sensitivity, since HFD+Gb rats presented similar results of glycemia and insulin sensitivity compared to the NFD group. In addition, GbE promoted a significant 3-fold increase in fasting insulin plasma levels compared to HFD+V. In fact, other studies have demonstrated similar effects, since GbE was able to enhance insulin synthesis in T2DM individuals as well as in a culture of pancreatic beta-cells of mice (2222. Kudolo GB. The effect of 3-month ingestion of Ginkgo biloba extract (EGb 761) on pancreatic beta-cell function in response to glucose loading in individuals with non-insulin-dependent diabetes mellitus. J Clin Pharmacol 2001; 41: 600-611, doi: 10.1177/00912700122010483.
https://doi.org/10.1177/0091270012201048...
,2323. Vasseur M, Jean T, DeFeudis FV, Drieu K. Effects of repeated treatments with an extract of Ginkgo biloba (EGb 761), bilobalide and ginkgolide B on the electrical activity of pancreatic beta cells of normal or alloxan-diabetic mice: an ex vivo study with intracellular microelectrodes. Gen Pharmacol 1994; 25: 31-46, doi: 10.1016/0306-3623(94)90007-8.
https://doi.org/10.1016/0306-3623(94)900...
).
It has been reported that insulin sensitivity might be enhanced by reduction in body adiposity (2424. Buettner R, Scholmerich J, Bollheimer LC. High-fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity 2007; 15: 798-808, doi: 10.1038/oby.2007.608.
https://doi.org/10.1038/oby.2007.608...
). It is possible that the reduction of both food intake and body adiposity observed in the HFD+Gb group resulted in protection against the effects of high-fat intake on insulin signaling impairment. Despite the fact that literature reports are scarce concerning the role of GbE on central control of food intake, it has been demonstrated that GbE affects the serotonergic system. In rats submitted to stress by immobilization, Shah et al. (2525. Shah ZA, Sharma P, Vohora SB. Ginkgo biloba normalises stress-elevated alterations in brain catecholamines, serotonin and plasma corticosterone levels. Eur Neuropsychopharmacol 2003; 13: 321-325, doi: 10.1016/S0924-977X(03)00005-1.
https://doi.org/10.1016/S0924-977X(03)00...
) reported that GbE restored serotonin levels in the central nervous system (whole brain). Since serotonin plays a pivotal role in the central control of energy homeostasis, inhibiting food intake and stimulating energy expenditure, it is possible that the observed inhibitory effect of GbE on food intake might have occurred, at least in part, due to a stimulatory role on hypothalamic serotonin release. It is also possible that GbE might interact with other central mechanisms involved in the complex process of energy homeostasis. Such a hypothesis must be tested in further studies in order to describe the pathways involved in GbE-induced reduction of food intake.
Remarkably, the present study also showed evidence of an impairment of Akt phosphorylation induced by insulin in the gastrocnemius muscle of HFD+V rats. Thus, it is well documented that obesity and high-fat consumption reduce both IRS-1 levels and IRS-1 phosphorylation in insulin-dependent tissues, disrupting the insulin signaling cascade as evidenced by reduced glucose uptake via GLUT4 (2626. Figueiredo-Silva AC, Panserat S, Kaushik S, Geurden I, Polakof S. High levels of dietary fat impair glucose homeostasis in rainbow trout. J Exp Biol 2012; 215: 169-178, doi: 10.1242/jeb.063933.
https://doi.org/10.1242/jeb.063933...
,2727. Youngren JF, Paik J, Barnard RJ. Impaired insulin-receptor autophosphorylation is an early defect in fat-fed, insulin-resistant rats. J Appl Physiol 2001; 91: 2240-2247.). On the other hand, GbE treatment increased Akt phosphorylation and IRS-1 while it reduced PTP-1B levels in gastrocnemius muscle compared to non-treated obese rats, indicating that G. biloba plays a beneficial role against insulin signaling impairment induced by obesity. It is noteworthy that Akt phosphorylation is an essential step required for GLUT4 translocation to the plasma membrane and consequently for glucose uptake by insulin-dependent tissues, and PTP-1B has been related to reduced IR tyrosine phosphorylation and, consequently, insulin signaling impairment (2828. Elchebly M, Payette P, Michaliszyn E, Cromlish W, Collins S, Loy AL, et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 1999; 283: 1544-1548, doi: 10.1126/science.283.5407.1544.
https://doi.org/10.1126/science.283.5407...
,2929. Abdul-Ghani MA, DeFronzo RA. Pathogenesis of insulin resistance in skeletal muscle. J Biomed Biotechnol 2010; 2010: 476279doi: http://dx.doi.org/10.1155/2010/476279.
http://dx.doi.org/10.1155/2010/476279...
). The results mentioned earlier demonstrate, for the first time, a direct beneficial effect of GbE on obesity-related insulin insensitivity.
Finally, it is important to consider that, despite several potent hypoglycemic agents available for the treatment of T2DM, some evidence points to the important side effects related to these drugs. A cohort study involving diabetic patients without prior stroke showed that individuals who had used rosiglitazones, a type of thiazolidinedione (TZD), for 10 years presented an increased risk of ischemic stroke and heart failure (3030. Lu CJ, Sun Y, Muo CH, Chen RC, Chen PC, Hsu CY. Risk of stroke with thiazolidinediones: a ten-year nationwide population-based cohort study. Cerebrovasc Dis 2013; 36: 145-151, doi: 10.1159/000353679.
https://doi.org/10.1159/000353679...
). Furthermore, the long-term use of rosiglitazones has been associated with bone loss and fracture risk (3131. Betteridge DJ. Thiazolidinediones and fracture risk in patients with Type 2 diabetes. Diabet Med 2011; 28: 759-771, doi: 10.1111/j.1464-5491.2010.03187.x.
https://doi.org/10.1111/j.1464-5491.2010...
). In addition, a review (3232. Campbell IW. Comparing the actions of older and newer therapies on body weight: to what extent should these effects guide the selection of antidiabetic therapy? Int J Clin Pract 2010; 64: 791-801, doi: 10.1111/j.1742-1241.2009.02292.x.
https://doi.org/10.1111/j.1742-1241.2009...
) reported a positive correlation between sulfonylurea/TZD and weight gain. It is important to note that GbE has been described as protecting against congestive heart failure, becoming one of the most common complementary therapies used by patients with cardiovascular diseases (3333. El-Boghdady NA. Increased cardiac endothelin-1 and nitric oxide in adriamycin-induced acute cardiotoxicity: protective effect of Ginkgo biloba extract. Indian J Biochem Biophys 2013; 50: 202-209.,3434. Yeh GY, Davis RB, Phillips RS. Use of complementary therapies in patients with cardiovascular disease. Am J Cardiol 2006; 98: 673-680, doi: 10.1016/j.amjcard.2006.03.051.
https://doi.org/10.1016/j.amjcard.2006.0...
). GbE has also been recognized as protecting against bone loss, reducing the progress of osteoporosis (3535. Lucinda LM, Vieira BJ, Oliveira TT, Sa RC, Peters VM, Reis JE, et al. Evidences of osteoporosis improvement in Wistar rats treated with Ginkgo biloba extract: a histomorphometric study of mandible and femur. Fitoterapia 2010; 81: 982-987, doi: 10.1016/j.fitote.2010.06.014.
https://doi.org/10.1016/j.fitote.2010.06...
). Taking into consideration that the present study demonstrated beneficial effects of GbE on body adiposity and glucose metabolism, we suggest that GbE might be a good alternative to other hypoglycemic therapies, since it also protects against important side effects related to the conventional therapy for T2DM.
In conclusion, the present study demonstrated that short-term GbE treatment significantly reduced food intake and body adiposity while it protected against hyperglycemia and dyslipidemia in diet-induced obese rats. G. biloba also enhanced insulin sensitivity, IRS-1, and Akt phosphorylation while it reduced PTP-1B levels in gastrocnemius muscle compared to nontreated obese rats. It is noteworthy that such effects were observed in rats that had been fed a hyperlipidic diet. Taking into consideration that most obese people resist adhering to a program of nutritional reeducation, GbE therapy might be very helpful for avoiding the development of comorbidities in those patients. These findings suggest that G. biloba might be an efficient therapy to prevent and/or treat obesity-induced insulin signaling impairment, and warrants additional studies to better understand the complex mechanisms involved in GbE hypoglycemic effects.
Acknowledgments
The authors are grateful for the valuable support given by Suzete Maria Cerutti, Luciana Chagas Caperuto, Aparecida Emiko Hirata, Joel Machado Jr., and João Henrique Ghilard Lago. Research supported by FAPESP (2011/06375-0) and CAPES.
References
-
1Pereira-Lancha LO, Campos-Ferraz PL, Lancha AH Jr. Obesity: considerations about etiology, metabolism, and the use of experimental models. Diabetes Metab Syndr Obes 2012; 5: 75-87.
-
2Pego-Fernandes PM, Bibas BJ, Deboni M. Obesity: the greatest epidemic of the 21st century? São Paulo Med J 2011; 129: 283-284, doi: 10.1590/S1516-31802011000500001.
» https://doi.org/10.1590/S1516-31802011000500001 -
3Yang ZH, Miyahara H, Takeo J, Katayama M. Diet high in fat and sucrose induces rapid onset of obesity-related metabolic syndrome partly through rapid response of genes involved in lipogenesis, insulin signalling and inflammation in mice. Diabetol Metab Syndr 2012; 4: 32, doi: 10.1186/1758-5996-4-32.
» https://doi.org/10.1186/1758-5996-4-32 -
4Trayhurn P. Adipose tissue in obesity - an inflammatory issue. Endocrinology 2005; 146: 1003-1005, doi: 10.1210/en.2004-1597.
» https://doi.org/10.1210/en.2004-1597 -
5Xu C, Yang X, Zu S, Han S, Zhang Z, Zhu G. Association between serum lipids, blood pressure, and simple anthropometric measures in an adult Chinese population. Arch Med Res 2008; 39: 610-617, doi: 10.1016/j.arcmed.2008.05.001.
» https://doi.org/10.1016/j.arcmed.2008.05.001 -
6Hilal Y, Acar TN, Koksal E, Gezmen KM, Akbulut G, Bilici S, et al. The association of anthropometric measurements and lipid profiles in Turkish hypertensive adults. Afr Health Sci 2011; 11: 407-413.
-
7Park YM, Won JH, Yun KJ, Ryu JH, Han YN, Choi SK, et al. Preventive effect of Ginkgo biloba extract (GBB) on the lipopolysaccharide-induced expressions of inducible nitric oxide synthase and cyclooxygenase-2 via suppression of nuclear factor-kappaB in RAW 264.7 cells. Biol Pharm Bull 2006; 29: 985-990, doi: 10.1248/bpb.29.985.
» https://doi.org/10.1248/bpb.29.985 -
8DeFeudis FV, Papadopoulos V, Drieu K. Ginkgo biloba extracts and cancer: a research area in its infancy. Fundam Clin Pharmacol 2003; 17: 405-417, doi: 10.1046/j.1472-8206.2003.00156.x.
» https://doi.org/10.1046/j.1472-8206.2003.00156.x -
9Mehlsen J, Drabaek H, Wiinberg N, Winther K. Effects of a Ginkgo biloba extract on forearm haemodynamics in healthy volunteers. Clin Physiol Funct Imaging 2002; 22: 375-378, doi: 10.1046/j.1475-097X.2002.00445.x.
» https://doi.org/10.1046/j.1475-097X.2002.00445.x -
10Rostoker G, Behar A, Lagrue G. Vascular hyperpermeability in nephrotic edema. Nephron 2000; 85: 194-200, doi: 10.1159/000045661.
» https://doi.org/10.1159/000045661 -
11Kudolo GB. The effect of 3-month ingestion of Ginkgo biloba extract on pancreatic beta-cell function in response to glucose loading in normal glucose tolerant individuals. J Clin Pharmacol 2000; 40: 647-654, doi: 10.1002/j.1552-4604.2000.tb05991.x.
» https://doi.org/10.1002/j.1552-4604.2000.tb05991.x -
12Kudolo GB, Delaney D, Blodgett J. Short-term oral ingestion of Ginkgo biloba extract (EGb 761) reduces malondialdehyde levels in washed platelets of type 2 diabetic subjects. Diabetes Res Clin Pract 2005; 68: 29-38, doi: 10.1016/j.diabres.2004.08.007.
» https://doi.org/10.1016/j.diabres.2004.08.007 -
13Tanaka S, Han LK, Zheng YN, Okuda H. [Effects of the flavonoid fraction from Ginkgo biloba extract on the postprandial blood glucose elevation in rats]. Yakugaku Zasshi 2004; 124: 605-611, doi: 10.1248/yakushi.124.605.
» https://doi.org/10.1248/yakushi.124.605 -
14Kudolo GB, Wang W, Javors M, Blodgett J. The effect of the ingestion of Ginkgo biloba extract (EGb 761) on the pharmacokinetics of metformin in non-diabetic and type 2 diabetic subjects - a double blind placebo-controlled, crossover study. Clin Nutr 2006; 25: 606-616, doi: 10.1016/j.clnu.2005.12.012.
» https://doi.org/10.1016/j.clnu.2005.12.012 -
15Lladó I, Rodriguez-Cuenca S, Pujol E, Monjo M, Estrany ME, Roca P, et al. Gender effects on adrenergic receptor expression and lipolysis in white adipose tissue of rats. Obes Res 2002; 10: 296-305, doi: 10.1038/oby.2002.41.
» https://doi.org/10.1038/oby.2002.41 -
16Saponara R, Bosisio E. Inhibition of cAMP-phosphodiesterase by biflavones of Ginkgo biloba in rat adipose tissue. J Nat Prod 1998; 61: 1386-1387, doi: 10.1021/np970569m.
» https://doi.org/10.1021/np970569m -
17Dell'Agli M, Bosisio E. Biflavones of Ginkgo biloba stimulate lipolysis in 3T3-L1 adipocytes. Planta Med 2002; 68: 76-79, doi: 10.1055/s-2002-19876.
» https://doi.org/10.1055/s-2002-19876 -
18Drieu K, Vranckx R, Benassayad C, Haourigi M, Hassid J, Yoa RG, et al. Effect of the extract of Ginkgo biloba (EGb 761) on the circulating and cellular profiles of polyunsaturated fatty acids: correlation with the anti-oxidant properties of the extract. Prostaglandins Leukot Essent Fatty Acids 2000; 63: 293-300, doi: 10.1054/plef.2000.0217.
» https://doi.org/10.1054/plef.2000.0217 -
19Goldberg Y, Boaz M, Matas Z, Goldberg I, Shargorodsky M. Weight loss induced by nutritional and exercise intervention decreases arterial stiffness in obese subjects. Clin Nutr 2009; 28: 21-25, doi: 10.1016/j.clnu.2008.10.001.
» https://doi.org/10.1016/j.clnu.2008.10.001 -
20Fayh AP, Lopes AL, da Silva AM, Reischak-Oliveira A, Friedman R. Effects of 5% weight loss through diet or diet plus exercise on cardiovascular parameters of obese: a randomized clinical trial. Eur J Nutr 2013; 52: 1443-1450, doi: 10.1007/s00394-012-0450-1.
» https://doi.org/10.1007/s00394-012-0450-1 -
21Tremblay F, Lavigne C, Jacques H, Marette A. Defective insulin-induced GLUT4 translocation in skeletal muscle of high fat-fed rats is associated with alterations in both Akt/protein kinase B and atypical protein kinase C (zeta/lambda) activities. Diabetes 2001; 50: 1901-1910, doi: 10.2337/diabetes.50.8.1901.
» https://doi.org/10.2337/diabetes.50.8.1901 -
22Kudolo GB. The effect of 3-month ingestion of Ginkgo biloba extract (EGb 761) on pancreatic beta-cell function in response to glucose loading in individuals with non-insulin-dependent diabetes mellitus. J Clin Pharmacol 2001; 41: 600-611, doi: 10.1177/00912700122010483.
» https://doi.org/10.1177/00912700122010483 -
23Vasseur M, Jean T, DeFeudis FV, Drieu K. Effects of repeated treatments with an extract of Ginkgo biloba (EGb 761), bilobalide and ginkgolide B on the electrical activity of pancreatic beta cells of normal or alloxan-diabetic mice: an ex vivo study with intracellular microelectrodes. Gen Pharmacol 1994; 25: 31-46, doi: 10.1016/0306-3623(94)90007-8.
» https://doi.org/10.1016/0306-3623(94)90007-8 -
24Buettner R, Scholmerich J, Bollheimer LC. High-fat diets: modeling the metabolic disorders of human obesity in rodents. Obesity 2007; 15: 798-808, doi: 10.1038/oby.2007.608.
» https://doi.org/10.1038/oby.2007.608 -
25Shah ZA, Sharma P, Vohora SB. Ginkgo biloba normalises stress-elevated alterations in brain catecholamines, serotonin and plasma corticosterone levels. Eur Neuropsychopharmacol 2003; 13: 321-325, doi: 10.1016/S0924-977X(03)00005-1.
» https://doi.org/10.1016/S0924-977X(03)00005-1 -
26Figueiredo-Silva AC, Panserat S, Kaushik S, Geurden I, Polakof S. High levels of dietary fat impair glucose homeostasis in rainbow trout. J Exp Biol 2012; 215: 169-178, doi: 10.1242/jeb.063933.
» https://doi.org/10.1242/jeb.063933 -
27Youngren JF, Paik J, Barnard RJ. Impaired insulin-receptor autophosphorylation is an early defect in fat-fed, insulin-resistant rats. J Appl Physiol 2001; 91: 2240-2247.
-
28Elchebly M, Payette P, Michaliszyn E, Cromlish W, Collins S, Loy AL, et al. Increased insulin sensitivity and obesity resistance in mice lacking the protein tyrosine phosphatase-1B gene. Science 1999; 283: 1544-1548, doi: 10.1126/science.283.5407.1544.
» https://doi.org/10.1126/science.283.5407.1544 -
29Abdul-Ghani MA, DeFronzo RA. Pathogenesis of insulin resistance in skeletal muscle. J Biomed Biotechnol 2010; 2010: 476279doi: http://dx.doi.org/10.1155/2010/476279
» http://dx.doi.org/10.1155/2010/476279 -
30Lu CJ, Sun Y, Muo CH, Chen RC, Chen PC, Hsu CY. Risk of stroke with thiazolidinediones: a ten-year nationwide population-based cohort study. Cerebrovasc Dis 2013; 36: 145-151, doi: 10.1159/000353679.
» https://doi.org/10.1159/000353679 -
31Betteridge DJ. Thiazolidinediones and fracture risk in patients with Type 2 diabetes. Diabet Med 2011; 28: 759-771, doi: 10.1111/j.1464-5491.2010.03187.x.
» https://doi.org/10.1111/j.1464-5491.2010.03187.x -
32Campbell IW. Comparing the actions of older and newer therapies on body weight: to what extent should these effects guide the selection of antidiabetic therapy? Int J Clin Pract 2010; 64: 791-801, doi: 10.1111/j.1742-1241.2009.02292.x.
» https://doi.org/10.1111/j.1742-1241.2009.02292.x -
33El-Boghdady NA. Increased cardiac endothelin-1 and nitric oxide in adriamycin-induced acute cardiotoxicity: protective effect of Ginkgo biloba extract. Indian J Biochem Biophys 2013; 50: 202-209.
-
34Yeh GY, Davis RB, Phillips RS. Use of complementary therapies in patients with cardiovascular disease. Am J Cardiol 2006; 98: 673-680, doi: 10.1016/j.amjcard.2006.03.051.
» https://doi.org/10.1016/j.amjcard.2006.03.051 -
35Lucinda LM, Vieira BJ, Oliveira TT, Sa RC, Peters VM, Reis JE, et al. Evidences of osteoporosis improvement in Wistar rats treated with Ginkgo biloba extract: a histomorphometric study of mandible and femur. Fitoterapia 2010; 81: 982-987, doi: 10.1016/j.fitote.2010.06.014.
» https://doi.org/10.1016/j.fitote.2010.06.014
-
First published online July 25, 2014.
Publication Dates
-
Publication in this collection
25 July 2014 -
Date of issue
Sept 2014
History
-
Received
3 Sept 2013 -
Accepted
14 Apr 2014