Abstracts
Baru (Dipteryx alata Vog.) is an abundant legume in the Brazilian Savanna. Its nuts can be exploited sustainably using its protein and lipid fractions. This study aimed to analyze the proteins of the nuts present in the defatted flour and protein concentrate in terms of their functional properties, the profile of their fractions, and the in vitro digestibility. The flour was defatted with hexane and extracted at the pH of higher protein solubility to obtain the protein concentrate. The electrophoretic profile of the protein fractions was evaluated in SDS-PAGE gel. The functional properties of the proteins indicate the possibility of their use in various foods, like soybeans providing water absorption capacity, oil absorption capacity, emulsifying properties, and foamability. Globulins, followed by the albumins, are the major fractions of the flour and protein concentrate, respectively. Digestibility was greater for the concentrate than for the defatted flour.
Dipteryx alata; protein fractions; functional properties; solubility; in vitro digestibility
O baru (Dipteryx alata Vog.) é uma leguminosa abundante no Cerrado brasileiro, cuja castanha pode ser explorada através do uso sustentável para o aproveitamento das frações proteicas e lipídicas. Este trabalho teve como objetivo estudar as proteínas desta castanha, presentes na farinha desengordurada e no concentrado proteico, quanto as suas propriedades funcionais, ao perfil das frações proteicas e à digestibilidade in vitro. A farinha desengordurada com hexano foi submetida à extração no pH de maior solubilidade das proteínas, obtendo-se o concentrado proteico. O perfil eletroforético das frações proteicas foi avaliado em gel de SDS-PAGE. As propriedades funcionais indicaram a possibilidade de emprego em diversos alimentos, assim como a soja, conferindo capacidade de absorção de água, capacidade de absorção de óleo, propriedades emulsificantes e espumabilidade. As globulinas, seguidas das albuminas, são as frações majoritárias da farinha e do concentrado proteico, respectivamente. A digestibilidade foi superior no concentrado em relação à farinha desengordurada.
Dipteryx alata; frações proteicas; propriedades funcionais; solubilidade; digestibilidade in vitro
Study of the proteins in the defatted flour and protein concentrate of baru nuts (Dipteryx alata Vog)
Estudo das proteínas da farinha desengordurada e concentrado protéico de castanhas de baru (Dipteryx alata Vog)
Rita de Cássia Avellaneda GuimarãesI* * Corresponding author ; Simone Palma FavaroI; Antonio Camilo Arguelho VianaI; José Antônio Braga NetoIII; Valdir Augusto NevesII; Michael Robin HonerI
IProgram in Biotechnology, Universidade Católica Dom Bosco - UCDB, Av. Tamandaré, 6000, CP 100, CEP 79117-900, Campo Grande , MS, Brazil, e-mail: ritaaguimaraes@gmail.com
IIFood and Nutrition Department, School of Pharmaceutical Sciences, São Paulo State University - UNESP, Rod. Araraquara-Jaú, Km 01, CEP 14801-902, Araraquara, SP, Brazil
IIIDepartment of Food Technology and Public Health, Federal University of Mato Grosso do Sul - UFMS, University City, s/n, CP 549, CEP 79070-900, Campo Grande, MS, Brazil
ABSTRACT
Baru (Dipteryx alata Vog.) is an abundant legume in the Brazilian Savanna. Its nuts can be exploited sustainably using its protein and lipid fractions. This study aimed to analyze the proteins of the nuts present in the defatted flour and protein concentrate in terms of their functional properties, the profile of their fractions, and the in vitro digestibility. The flour was defatted with hexane and extracted at the pH of higher protein solubility to obtain the protein concentrate. The electrophoretic profile of the protein fractions was evaluated in SDS-PAGE gel. The functional properties of the proteins indicate the possibility of their use in various foods, like soybeans providing water absorption capacity, oil absorption capacity, emulsifying properties, and foamability. Globulins, followed by the albumins, are the major fractions of the flour and protein concentrate, respectively. Digestibility was greater for the concentrate than for the defatted flour.
Keywords:Dipteryx alata; protein fractions; functional properties; solubility; in vitro digestibility.
RESUMO
O baru (Dipteryx alata Vog.) é uma leguminosa abundante no Cerrado brasileiro, cuja castanha pode ser explorada através do uso sustentável para o aproveitamento das frações proteicas e lipídicas. Este trabalho teve como objetivo estudar as proteínas desta castanha, presentes na farinha desengordurada e no concentrado proteico, quanto as suas propriedades funcionais, ao perfil das frações proteicas e à digestibilidade in vitro. A farinha desengordurada com hexano foi submetida à extração no pH de maior solubilidade das proteínas, obtendo-se o concentrado proteico. O perfil eletroforético das frações proteicas foi avaliado em gel de SDS-PAGE. As propriedades funcionais indicaram a possibilidade de emprego em diversos alimentos, assim como a soja, conferindo capacidade de absorção de água, capacidade de absorção de óleo, propriedades emulsificantes e espumabilidade. As globulinas, seguidas das albuminas, são as frações majoritárias da farinha e do concentrado proteico, respectivamente. A digestibilidade foi superior no concentrado em relação à farinha desengordurada.
Palavras-chave:Dipteryx alata; frações proteicas; propriedades funcionais; solubilidade; digestibilidade in vitro.
Introduction
Brazil has a large number of native plant species and, although little explored, some fruits have been proved to be excellent sources of nutrients. The species Dipteryx alata Vog. is a native tree of the Brazilian Cerrado, belonging to the Leguminosae, popularly known as baru, cumaru or cumbaru (JUNQUEIRA; FAVARO, 2006). The nut of this fruit, rich in proteins and lipids, has a very pleasant taste, and its consumption has expanded beyond the borders of the Brazilian Savanna. The employment of protein fraction as a food ingredient can improve the value attributed to this fruit. However, very little information about baru nuts properties is available (TOGASHI; SGARBIERI, 1995).
The use of defatted flours and protein concentrates in foods requires investigation to better understand their behavior in different types of food systems (FONTANARI et al., 2007).The production of protein concentrates obtained from vegetable sources is of great interest to industry due to their wide application in food products (SÁNCHES-VIOQUE et al., 1999).
The direct use of defatted flours as functional ingredients play an important role in industries because of their lower production cost compared to that of protein concentrates. The use of these flours depends on their performance as functional ingredients and their behavior in particular food systems (WANG et al., 2000).
The biodisponibility of amino acids depends on the digestibility of proteins. Low protein digestibility is usually presented by the seeds of legumes (NEVES et al., 2004).
In order to contribute to a better use of baru nuts, this study evaluated the functional properties, the profile of the principal protein fractions, and the in vitro digestibility of the defatted flour and protein concentrate.
2 Materials and methods
2.1 Raw material
The fruits were collected from more than three trees in three different areas in the city of Campo Grande, Mato Grosso do Sul State, Brazil. The defatted flour and concentrate were obtained from different extraction methods.
The nuts were removed and dried in an air oven at 70 ºC for 24 hours to facilitate peeling. The cleaned nuts were ground in a grinder and the flour was sifted. The flour was defatted with hexane in a Soxhlet extractor.
The protein content of the whole flour, defatted flour, and protein concentrate was determined by the micro-Kjeldhal method with a conversion factor of 6.25 (ASSOCIATION..., 1997). Ash content was determined using a muffle furnace at 550 ºC to a fixed mass, and the moisture content of the defatted flour was determined by drying in an oven at 105 ºC (ASSOCIATION..., 1997).
Monosaccharides were separated in the defatted flour by the extraction of reducing sugars such as glucose; disaccharides in non-reducing sugars such as sucrose, and polysaccharides such as starch were extracted (INSTITUTO..., 1985). The sugars were assayed as reducing sugars by spectrophotometric method (NELSON, 1944).
2.2 Preparation of protein concentrate
The protein concentrate was obtained by extracting the proteins in a solution (m/v 1 defatted flour: 20 pH 10 solution), pH of maximum solubility. This solution was centrifuged at 3000 rpm for 20 minutes. The supernatant was dried by spray draying (Büchi - Model B290, with an entrance temperature of the sample of 165 ºC, 100% aspiration, and an injection pump pressure of 25%).
2.3 Extraction and fractionation of principal proteins of baru nuts
The extraction and fractionation of the baru proteins were carried out according to the method described by Sathe and Salunkhe (1981). A sequential extraction was performed obtaining soluble proteins in salt, globulin, albumin, prolamin, glutelin, and an insoluble residue and a dialyzable fraction. These fractions were dried, and the protein content was quantified by the micro-Kjeldhal method (ASSOCIATION..., 1997).
2.4 Sodium dodecyl sulfate polyacrylamide gel electrophoresis
The electrophoretic profile of the protein fractions in the defatted flour and the protein concentrate was obtained in polyacrylamide gel using a discontinuous system (LAEMLLI, 1970). The gels were dyed by immersion into a solution of 0.025% Coomassie Blue.
2.5 Functional properties
Protein solubility is a functional property derived from the protein-water interaction and is usually the first property investigated. Solubility was determined according to the method described by Canella (1978) with modifications.
Defatted flour was dispersed (1:20 m/v) in distilled water, and the pH values were adjusted with NaOH or HCl to reach the range from 1 to 12. The final volume of all solutions was made up to 50 mL. The suspensions were stirred for 1 hour at 4 ºC and centrifuged at 3000 rpm for 20 minutes. The supernatant was collected, and the protein content determined by the micro-Kjeldhal method, as described above. The solubility was calculated by the Equation 1 and expressed in percentage:
where: Psuperntant is the concentration of protein in supernatant (mg.mL-1); Wtsample is the sample weight (mg) and Psample is the content of the protein in the sample (%).
Water and oil absorption capacities
Water (WAC) and oil (OAC) absorption capacities were determined according to the method of LIN et al. (1974) and calculated by the difference between the total volume and the free volume.
Foam capacity and foam stability
Foam capacity was determined according to the method of Paredes-Lópes et al. (1991) adjusting the pH at 5 and 7. It was calculated as the percentage of volume increase after blending.
Foam stability was obtained by the difference between the initial and the decreased volume after 5, 10, 30, and 60 minutes after foam formation (PAREDES et al., 1991).
Emulsifying activity and emulsion stability
Emulsifying activity was determined by the method of Yasumatsu et al. (1972). A suspension was prepared with protein samples and soybean oil followed by mechanical emulsification. Emulsifying activity was obtained as the ratio between the emulsified layer and the total volume expressed as percentage. The emulsion was centrifuged, and the emulsion stability was considered as the volume of emulsified layer remaining in relation to the volume of initial layer.
All functional properties were determined in triplicate for each repetition.
2.6 In vitro digestibility
The in vitro digestibility of the defatted flour and protein concentrate was obtained in triplicate following the methods of Akeson and Staman (1964). The hydrolyzed soluble nitrogen content was determined by the micro-Kjeldahl method, and the degree of digestibility (%H) was obtained considering the total and soluble nitrogen produced after enzymatic digestion.
2.7 Statistical analysis
The results are expressed as the mean value ± standard deviation (SD) of three repetitions, and the analyses were carried out with three replications. The data were statistically analyzed using analysis of variance, and the averages were compared by the Tukey's test. Significant differences were determined at the p < 0.05 level. In order to present the dispersion of data, the coefficients of variation (CV) were showed.
3 Results and discussion
3.1 Characteristics of the flour and protein concentrate
The protein content in whole flour was 28.45 ± 0.43% on wet basis (4.96% moisture). Many other legumes have high levels of proteins, such a peanuts (Arachis hypogaea L.) with 30% protein, beans (Vicia faba L.), 27.7%, chickpeas (Cicer arietinum L.) 23.9%, lentils (Lens culinaris) 27.6%, peas (Pisum sativum) 22.8%, white lupine (Lupinus albus ) 39.4%, cowpea (Vigna unguiculata) 29.32%, pigeon pea (Cajanus cajan(L. Milsp) 28.1%, and the common bean (Phaseolus vulgaris) 20% (COMAI et al., 2007).
Defatted flour of baru nuts presented the following proximate composition on wet basis (%): moisture 4.26 ± 0.08, protein 49.01 ± 1.37, lipids 7.37 ± 0.10, reducing sugars such as glucose 0.11 ± 0.003, non-reducing sugars such as sucrose 4.97 ± 0.55, polyssaccharides such as starch 6.06 ± 0.79, and ashes 4.91 ± 0.04.
The ash content of the defatted flour was close to that found by Donadel e Ferreira (1999) in the defatted flour of the common bean (P. vulgaris) at 5.06%, but it was higher than that found by Shoshima et al. (2005) in cowpeas (Vigna unguiculata L. Walp) with a content of 3.59%. The glucose values in the defatted flour (0.11%) are about ten times lower than those found by Junqueira and Favaro (2006) in the whole flour. Higher moisture contents were found in defatted flours of other species, such as pigeon pea (Cajanus caja (L) Millsp.) with 12.15% (MIZUBUTI et al., 2000) and whole soybean meal with 9.56% (MENDES et al., 2007).
The protein content (dry basis) of the whole flour increased by 72% after the lipid extraction, while the concentrate increased by 93%, reaching 57.65 ± 4.92%. The method used to obtain the concentrate used in this study was more efficient than that used for soybeans by Singh et al. (2008), in which the whole flour with 41% of protein produced a concentrate with 66.2%, corresponding to an increase in protein of 61%.
3.2 Fractioning of the principal baru proteins
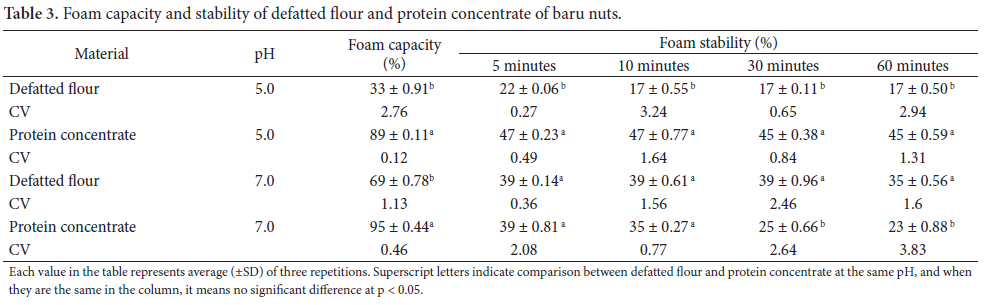
The defatted baru nut flour has a protein composition similar to that found in the majority of legumes, and salt soluble proteins, globulins, and albumins are its major fractions (Table 1). The third most abundant fraction was glutelin and the fraction prolamin was present in much reduced quantities. Other legumes may be taken as an example with a profile similar to that of the baru, such as the cowpea (Vigna unguiculata L.) which contains 41.99% globulin, 10.11% albumin, and 7.81% glutelin (SHOSHIMA et al., 2005). On the other hand, in the species Vigna aconitifolia L., the second principal group is the glutelin fraction (27.83 ± 0.27%), followed by albumins (5.06 ± 0.27%) (SATHE; VENKATACHALAN, 2007).
In the protein concentrate, the globulin fraction increased and the albumin decreased, while the glutelins remained practically constant. The prolamin fraction was not detected in the concentrate. The dialyzable fraction, 9.27% in the flour, increased to 22.81% in the concentrate, representing the efficient soluble protein extraction of low molecular weight in salt. The final residue decreased to 10.94% in the defatted flour and 2.12% in the concentrate.
Baru presents a protein profile similar to that of the majority of the legumes although some species show quite different proportions between the main fractions. As an example, the protein isolate from the velvet bean, Mucuna pruriens which contains albumin (87%) and glutelin (22.1%) as the main fraction (AEBOWALE et al., 2007).
3.3 SDS-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
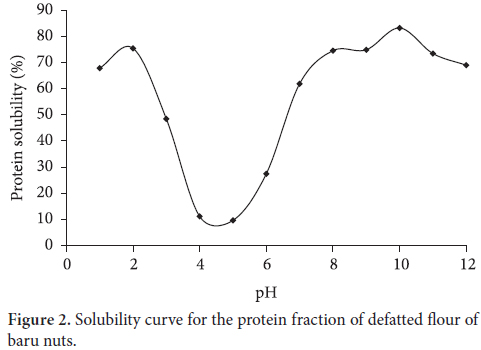
The electrophoretic profile of the principal protein fractions of the defatted flour of the baru nuts is shown in Figure 1.
In the salt soluble extract, in which the globulin and albumin fractions are concentrated (1), marked bands can be seen with molecular weights between 97 and 66 kDa.
A conspicuous band can be seen at about 45 kDa. Bands with molecular weights of less than 29 kDa can also be seen in this extract; the last band has a low molecular weight of about 14 kDa.
In the globulin fraction, a weak band can be seen at 97 kDa, but bands around 66-45 kDa can clearly be seen. A number of bands also occur at less than 29 kDa, probably corresponding to oleosins (HUANG, 1996). Bands can also be seen at less than 14.4 kDa.
The defatted flour and the globulin fraction of baru were similar in SDS-PAGE electrophoresis, with a major band estimated at between 66-45 kDa and various bands between 97 and 29 kDa. In the globulin fraction of the defatted flour, bands could be seen between 66-45 kDa, similar to those found in a study on defatted bean flour (54-67 kDa). Bands less than 97 kDa were also seen in the defatted baru flour confirming the marked similarity between the results.
In the albumin fraction (3), a band of high molecular weight was seen at 97 kDa. Less marked bands can also be seen, especially around 66-29 kDa. There were no defined bands in the glutelin fraction (4).
In the profile of the defatted flour of the hyacinth bean (Lablab purpureus L.), according to the SDS-PAGE, the predominance of three large polypeptides was seen (molecular mass 51-64 kDa) in the composition of the proteins (VENKATACHALAM; SATHE, 2007).
In the gel of defatted baru flour, a marked accumulation of bands can be seen in fractions 1, 2, and 3 with molecular weights greater from 45 to 97 kDa, similar to the profile found in the defatted flour of the Val bean.
In the electrophoretic gel of the protein concentrate, there is a smaller accumulation of bands with molecular weights above 45 kDa (Figure 1b) when compared to the defatted flour.
The salt soluble extract (1) shows protein bands with molecular weight between 97 and 66 kDa, as well as a band around 45 and 29 kDa with an accumulation of protein bands. The last band of low molecular weight is at 14.4 kDa.
Bands of 97 and 66 kDa and less marked bands were detected in the albumin fraction (3). In the glutelin fraction (4), no bands could be distinguished.
Comparing the two baru protein gels in the globulin fraction (2), two bands below 66 kDa that were found in the flour were not detected in the concentrate. In the fraction for albumin (3), the occurrence of a band at 97 kDa could be seen, but it was less dense than that of the defatted flour; other less marked bands were observed below 66 kDa. No bands were observed in the glutelin fraction (4).
3.4 Functional properties
Solubility of the protein fraction
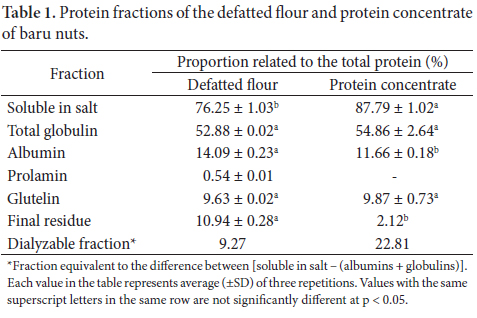
Baru proteins show peaks of solubility in both alkaline and acid pH (Figure 2). The region of least solubility occurs in the interval of pH 4 to 5. Maximum solubility was reached at pH 10, with 83% solubility of the protein fraction. Other solubility peaks were observed at pH 2 and pH 8, both with 75% of extraction. Above pH 10, a decrease in solubility was observed.
Like baru, various other seeds show alkaline pH values as the condition of maximum solubility. The protein concentrate of cupuaçu (Theobroma grandiflorum Schum) shows a maximum solubility of 65% at pH 10, with a low extraction at acid pH, reaching 25% solubility at pH 2 (CARVALHO, 2006). Solubility above 70% at pH 9 was observed in the protein concentrate of pigeon peas (Cajanus cajan L.) (MIZUBUTI, 2000). Among the pH values studied, the author states that pH 5 showed the lowest protein solubility, slightly more than 20%, and he explains that this pH value is near the isoelectric point of protein isolates from amaranth (4.6) resulting in aggregation and precipitation of the great part of the proteins (MIZUBUTI, 2000).
Protein solubility is an important factor for the optimization of functional properties. A more soluble product is also more easily formulated for some foods. Hence, almost all concentrates and isolates are neutralized and sold as proteinates. The major form is sodium, potassium, and calcium proteinate, but other proteinates are available (WOLF; COWAN, 1971).
Water and oil absorption capacities, emulsifying activity, and emulsion stability
The results obtained for the water and oil absorption capacities of defatted flour were 225.98 ± 0.91% and 199.80±0.93%, respectively, and for the protein concentrate they were 193.84 ± 0.89% and 205.28 ± 0.78%, respectively (
Table 2).
Although the value of the functional properties may be quite variable, depending on the technique and conditions of the assays, it can be inferred that the performance of the proteins in the defatted baru flour was similar to that of soybean (223%, Sosulski (1976) and 175% Sosulski; Mccurdy (1987)) for the water absorption capacity. Oil absorption in the defatted flour was lower to the data in the literature for soybean 130% (SOSULSKI, 1976) and 56% (SOSULSKI; MCCURDY, 1987). On the other hand, the baru concentrate showed lower water absorption capacity since soybean concentrate reached the value of 331% (SOSULSKI, 1976). Oil capacity absorption was practically the same in both concentrates indicating that baru could substitute for soybean as a functional ingredient in products in which an increase in WAC and OAC is required.
Baru has higher WAC and OAC values than other legumes. The water absorption capacity for the defatted flour of peas and fava beans was 78 and 72%, respectively, and the values found for oil were 41 and 47% (SOSULSKI; MCCURDY, 1987).
For defatted baru flour, the emulsifying activity was 51.00 ± 0.76%, and the emulsion stability was 50.0 ± 0.47% (Table 3). The protein concentrate showed similar values to those of the defatted flour for both emulsifying activity and emulsion stability.
The emulsifying activity of the protein concentrate of the "carioca" variety of the bean (Phaseolus vulgaris) was very close to that of baru, 50.16% (DONADEL; FERREIRA, 1999), while the emulsion stability of this bean was lower than that of baru, reaching 38%.
Foam formation and stability
The major foam formations were found at pH 7 (Table 3), independently of the material evaluated for baru nuts. The maximum of 95% foam formation was found for the concentrate at pH 7, which is three times that of the foam formed with defatted flour at pH 5. The increase in the proportion of protein in the concentrate appears to influence positively the formation capacity and stability of baru foam due to the presence of soluble sugars, which increase viscosity.
Greater stability (twice as much) was observed at pH 7 for the flour in relation to pH 5, while at more acid levels a higher foam stability was observed remaining practically constant during the 120 minutes of observation.
In contrast, the foam formation capacity of the winged bean (Psophocarpus tetragonolobus (L.) DC) showed a 76% volume increase at pH 4.6 and 150% at pH 9.8 for the concentrate, while for the defatted flour, foam formation reached 52% at pH 6.0 (NARAYANA; NARASINGA, 1982).
Soybean (Glicina max (L.) Merril) flour and protein concentrate had an increase of 70 and 170%, respectively, in the volume of foam (LIN et al., 1974).
Protein concentrates of the "carioca" variety of the common bean showed a low foam stability at 30, 60, 90, and 12 minutes with values of 1.28, 3.18. 4.43, and 6.63%, respectively (DONADEL; FERREIRA, 1999).
3.5 In vitro digestibility
The in vitro digestibility of defatted baru flour (Table 4) was similar to that found by Shoshima et al. (2005), 54.62%, who studied the proteins of whole cowpea flour. The value for the baru protein concentrate was very similar to that found for the defatted flour of white lupine seeds (Lupinus albus L.) (NEVES et al., 2006) (Table 5).
In Brazil, beans (Phaseolus vulgaris) are the most consumed legume; they are used as a source of vegetable protein, but baru proteins show better digestibility. The bean varieties Rico-23, Rosinha G2, Carioca, and Pirantã-1 had in vitro digestibility for raw beans of 39.2, 45.2, 43.9, and 45, respectively (ANTUNES et al., 1995). After cooking, digestibility was increased to 60.5, 66.2, 66.8, and 65.5. Beans of the genetic lines ESAL 647, ESAL 654, ESAL 655, and ESAL 516 showed digestibility of 29.53, 26.17, 18.03 and 26.97, respectively (MESQUITA et al., 2007). The low digestibility of the raw beans is attributed to the activity of protease inhibitors, which reduces the activity of digestive enzymes. The heat treatment of the bean, cooking process, inactivates the protease inhibitors with a beneficial effect on digestibility,
Both the defatted flour and the protein concentrate of baru nuts underwent a pretreatment with temperature around 70 ºC, which was used to facilitate the peeling of the nuts, and 54 ºC in the Soxhlet lipid extraction. The protein concentrate underwent another heating of 165 ºC during spray drying. Hence, the digestibility of baru may have been increased by heating, especially in the concentrate. Changes in the composition of the principal fractions and in the electrophoretic profile indicate that there was a reduction in the molecular mass and this may have contributed to the increase in digestibility of the concentrate due to the improved activity of digestive enzymes.
4 Conclusions
Defatted flour had a protein content of 49%, which increased to 55% in the concentrate. The optimum solubility for the proteins was found at pH 10, with 83% extraction.
The water and oil absorption capacities were similar to those of soybean suggesting the applicability of baru proteins to foods.
In the protein concentrate, the main capacity for foam formation was at pH 7 with 95% although the foam stability was greater for the periods observed at pH 5. This suggests the application of the concentrate at pH 5 in more acid foods. However, the defatted flour showed optimum performance for foam formation at pH 7, and thus it could be used in foods with pH closer to neutral.
The globulin fraction was predominant in both defatted flour and protein concentrate, followed by the fractions of albumin and glutelin.
The digestibility of the defatted flour and the protein concentrate was 58.42 and 65.9%, respectively.
The process used to obtain the protein concentrate may have hindered the attainment of better digestibility due to the formation of peptides with lower molecular mass.
Received 07/03/2010
Accepted 28/02/2012 (004707)
- AEBOWALE, Y. A. et al. Isolation, fractionation and characterisation of proteins from Mucuna bean Food Chemistry, v. 104, p. 287‑299, 2007. http://dx.doi.org/10.1016/j.foodchem.2006.11.050
- AKESON, W.; STAHMAN, M. A. A pepsin pancreatin digest index protein quality evaluation. Journal of Food and Nutrition, v. 83, p. 257-260, 1964.
- ANTUNES, P. L. et al. Valor nutricional de feijão (Phaseolus vulgaris, L.), cultivares rico 23, carioca, pirata-1 e rosinha-G2. Revista Brasileira de Agrociência, v. 1, p. 12-18, 1995.
- ASSOCIATION OF OFFICIAL ANALYTICAL CHEMISTS - AOAC. Official methods of analysis of the Association of Official Analytical. Chemists. 16. ed. Washington: AOAC, 1997.
- CANELLA, M. Whipping properties of sunflower protein dispersions. Food Science and Technology, vol. 12, p. 95-101, 1978.
- CARVALHO, A. V. et al. Physico chequical properties of the flour, protein concentrate, and protein isolate of the cupuassu (Thebroma grandiflorum Schum) seed. Journal of Food Science, v. 71, p. 573‑578, 2006. http://dx.doi.org/10.1111/j.1750-3841.2006.00156.x
- COMAI, S. et al. Protein and non-protein (free and protein‑bound) tryptophan in legume seeds. Food Chemistry, v. 103, p. 657‑661, 2007. http://dx.doi.org/10.1016/j.foodchem.2006.07.045
- DONADEL, M. E.; FERREIRA, S. H. P. Propriedades funcionais de concentrado protéico de feijão envelhecido. Ciência e Tecnologia de Alimentos, v. 19, p. 1-13, 1999.
- FONTANARI, G. G. et al. Protein isolate of guava seed (Psidium guajava): functional properties of characterization. Ciência e Tecnologia de Alimentos, v. 27, p. 73-79, 2007. http://dx.doi.org/10.1590/S0101-20612007000500013
- HUANG, A. H. C. Oleosins and oil bodies in seeds of other organs, Plant Pphysiology. In: INSTITUTO ADOLFO LUTZ - IAL. Normas analíticas do Instituto Adolfo Lutz. São Paulo: IAL,1996. v. 110, p. 1055-1061.
- INSTITUTO ADOLFO LUTZ - IAL. Normas analíticas do Instituto Adolfo Lutz. São Paulo: IAL,1985.
- JUNQUEIRA, D. R.; FAVARO, S. P. Caracterização físico-química e processamento de castanhas de baru (Dipteryx alata Vog.). In: ENCONTRO DE INICIAÇÃO CIENTÍFICA DA UNIVERSIDADE CATÓLICA DOM BOSCO - UCDB, 8., 2006, Campo Grande. Cadernos de Resumos.. UCDB, 2006. 27 p.
- LAEMLLI, U. K. Cleavage of structural proteins during assembly of the head of bacteriophage. Nature, v. 227, p. 680-85, 1970. PMid:5432063. http://dx.doi.org/10.1038/227680a0
- LIN, M. J. Y. et al. Certain properties of sunflower meal products. Journal of Food Science, v. 39, p. 368-370, 1974. http://dx.doi.org/10.1111/j.1365-2621.1974.tb02896.x
- MENDES, F. Q. et al. Digestibilidade protéica e caracterização bromatológica de linhagens de soja com ausência ou presença do inibidor de tripsina kunitz e das isozimas lipoxigenases. Bioscience Journal, v. 23, p. 14-21, 2007.
- MESQUITA, F. R. et al. Linhagens de feijão (Phaseolus vulgaris L.): Composição química e digestibilidade protéica. Ciência Agrotécnica, v. 31, p. 1114-1121, 2007.
- MIZUBUTI, I. et al. Propriedades funcionais da farinha e concentrado protéico de feijão guandu (Cajanus cajan(I.)Millsp). Archivos Latioamericanos de Nutrición, v. 50, p. 274-280, 2000. PMid:11347298.
- NARAYANA, K.; NARASINGA, R. Functional properties of raw and heat processed winged bean (Psocarphus tetragonolobus) flour. Journal of Food Science, v. 47, p. 1534-1538,1982. http://dx.doi.org/10.1111/j.1365-2621.1982.tb04976.x
- NELSON, N. A. A photometric adaptation of Somogyi method for the determination of glucose. Journal of Biological Chemistry, v. 135, p. 136-175, 1944.
- NEVES, V. A. et al. Caracterização e hidrólise in vitro da globulina principal de grão-de-bico (Cicer arietinum L.), var. IAC-Marrocos. Ciência e Tecnologia de Alimentos, v. 24, p. 139-145, 2004. http://dx.doi.org/10.1590/S0101-20612004000100025
- NEVES, V. A. et al. Isolamento da globulina majoritária, digestibilidade in vivo e in vitro das proteínas do tremoço-doce (Lupinus albus L.), var. multolupa. Ciência e Tecnologia de Alimentos, v. 26, p. 832‑840, 2006. http://dx.doi.org/10.1590/S0101-20612006000400019
- PAREDES-LÓPES, O. et al. Chickpea protein isolates: physicochemical, functional and nutritional caracterization. Journal of Food Science, v. 56, p. 726-729, 1991. http://dx.doi.org/10.1111/j.1365-2621.1991.tb05367.x
- SATHE, S. K.; SALUNKHE, D. K. Solubilization and eletrophoretic characterization of the Great Northern bean (Phaseolus vulgaris L.) proteins. Journal of Food Science, v. 46, p. 82-87, 1981. http://dx.doi.org/10.1111/j.1365-2621.1981.tb14536.x
- SATHE, S. K.; VENKATACHALAM, M. Fractionation and biochemical characterization of moth bean (Vigna aconitifolia L.) proteins. Science Direct, n. 40, p. 600-610, 2007.
- SHOSHIMA, A. H. R. et al. Digestibilidade in vitro das proteínas do caupi (Vigna unguiculata L. Walp) "Var. Br-14 Mulato:" Efeito dos fatores antinutricionais. Brazilian Journal of Food Technology, v. 4, p. 299-304, 2005.
- SÁNCHES-VIOQUE, R. et al. Protein isolates from chickpea (Cicer arietinum L.): chemical composition, functional properties and protein characterization. Food Chemistry, v. 64, p. 237-243, 1999. http://dx.doi.org/10.1016/S0308-8146(98)00133-2
- SINGH, P. et al. Functional and Edible uses of soy protein products. Comprehensive reviews in food science and food safety, v. 7, p. 14-28, 2008. http://dx.doi.org/10.1111/j.1541-4337.2007.00025.x
- SOSULSKI, F. Functional properties of rapeseed flours concentrades and isolate. Journal of Food Science, v. 41, p. 1349-1352, 1976. http://dx.doi.org/10.1111/j.1365-2621.1976.tb01168.x
- SOSULSKI, F.; MCCURDY, A. R. Functionality of flours, protein fractions and isolate from field peas and faba bean. Journal of Food Science, v. 52, p. 1010-1014, 1987. http://dx.doi.org/10.1111/j.1365-2621.1987.tb14263.x
- TOGASHI, M.; SGARBIERI, V. C. Avaliação nutricional da proteína e do óleo de sementes de baru (Dipteryxalata Vog.). Ciência e Tecnologia de Alimentos, v. 15, p. 65-69, 1995.
- VENKATACHALAM, M.; SATHE, S. K. Val bean (Lablab purpureus L.) proteins:composition and biochemical properties. Journal of the Science of Food and Agriculture, v. 87, p. 1539-1549, 2007. http://dx.doi.org/10.1002/jsfa.2885
- WANG, S. H.; FERNANDES, S. M.; CABRAL, L. C. Solubilidade de nitrogênio, dispersibilidade de proteína e propriedades emulsificantes dos extratos hidrossolúveis desidratados de arroz e soja. Ciência e Tecnologia Alimentos, v. 20, n. 1, p. 12-17, 2000. http://dx.doi.org/10.1590/S0101-20612000000100003
- WOLF, W. J.; COWAN, J. C. Soybeans as a food source. CRC Critical Reviews in Food Technology, v. 1, p. 81-158, 1971.
- YASUMATSU, K. et al. Whipping and emulsifying properties of soybean products. Agricultural and Biological Chemistry, v. 36, p. 719-727, 1972. http://dx.doi.org/10.1271/bbb1961.36.719
Publication Dates
-
Publication in this collection
07 June 2012 -
Date of issue
Sept 2012
History
-
Received
07 Mar 2010 -
Accepted
28 Feb 2012