Abstract
The increased marketing of olive oil in Brazil has intensified legal requirements to ensure regulation of this product. The measurement of the specific extinction at 270 nm (E 270) and content of stigmastadiene can be used to assess the presence of refined oils in virgin olive oil. During the vegetable oil refining process, compounds with conjugated double bonds are generated from unsaturated fatty acids that absorb at 270 nm and sterols, such as stigmasta-3,5-diene. To compare these parameters, seven samples of extra virgin olive oil and three samples of olive oil (blend of virgin and refined) were analyzed. Among the samples analyzed, four extra virgin samples had levels of stigmastadiene and E 270 higher than expected, among which two were adulterated with seed oil (rich in linoleic acid) and the other two with olive pomace oil. The results demonstrate the higher sensitivity of stigmastadiene to determine the presence of the refined oil in virgin olive oil and good agreement with determining E 270. The latter technique is a simple, quick, and low cost method of determination that can be easily implemented in laboratories to assist in the screening and regulation of olive oils sold in Brazil.
adulteration; quality control; legislation
Stigmastadiene and specific extitntion (270 nm) to evaluate the presence of refined oils in virgin olive oil commercialized in Brazil
Sabria Aued-Pimentel* * Corresponding author ; Simone Alves Da Silva; Emy Takemoto; Cristiane Bonaldi Cano
Núcleo de Química, Física e Sensorial, Centro de Alimentos, Instituto Adolfo Lutz - IAL, Av. Doutor Arnaldo, 355, CEP 01246-902, São Paulo, SP, Brasil, e-mail: spimente@ial.sp.gov.br
ABSTRACT
The increased marketing of olive oil in Brazil has intensified legal requirements to ensure regulation of this product. The measurement of the specific extinction at 270 nm (E 270) and content of stigmastadiene can be used to assess the presence of refined oils in virgin olive oil. During the vegetable oil refining process, compounds with conjugated double bonds are generated from unsaturated fatty acids that absorb at 270 nm and sterols, such as stigmasta-3,5-diene. To compare these parameters, seven samples of extra virgin olive oil and three samples of olive oil (blend of virgin and refined) were analyzed. Among the samples analyzed, four extra virgin samples had levels of stigmastadiene and E 270 higher than expected, among which two were adulterated with seed oil (rich in linoleic acid) and the other two with olive pomace oil. The results demonstrate the higher sensitivity of stigmastadiene to determine the presence of the refined oil in virgin olive oil and good agreement with determining E 270. The latter technique is a simple, quick, and low cost method of determination that can be easily implemented in laboratories to assist in the screening and regulation of olive oils sold in Brazil.
Keywords: adulteration; quality control; legislation.
1 Introduction
Mediterranean countries, such as Spain, Italy, and Greece are the main producers of olive oil. The trade of this product significantly contributes to the growth of the GDP (gross domestic product) in these countries. Worldwide production volume is approximately 2 - 3% of total oils, but it corresponds to roughly15% of the monetary value of total oil production (APARICIO, 2003). The Brazilian domestic demand for olive oil is met by imported oil, and Brazil is one of the leading importers in the world (CONSEJO..., 2011). The increased marketing of olive oil combined with the persistent problem of adulteration of products commercialized in Brazil (AUED-PIMENTEL et al., 2008; MODOLO, 2007) has led the government to set legal requirements to ensure regulation of this product. Additionally, the government aims to make the official laboratories qualified to control the olive oil commercialized in Brazil (BRASIL, 2012).
Oil extracted from olives has unique characteristics compared to other vegetable oils, such as exceptional sensory and nutritional attributes. Limited production of olive oil has led to high market value (DIEGO et al., 2008). However, oils of different grade and quality can be obtained from the fruit of the olive (APARICIO, 2003).
Higher quality olive oil is known as extra virgin and is obtained from the first cold pressing of fresh, healthy fruit. Good quality olive oils with greater acidity are classified as virgin olive oil. Lower quality categories simply include olive oil, which is a blend of virgin and refined olive oil. Lampante oil is a virgin olive oil not fit for human consumption because it has an undesirable flavor originating from poor quality olives. Olive pomace oil (orujo) is extracted from olive cakes by solvent extraction. Both lampante and olive pomace oil must be refined to become fit for human consumption (APARICIO, 2003; BRASIL, 2012; INTERNATIONAL..., 2010; CODEX..., 2009). Refining of vegetable oils consists of neutralization, bleaching and deodorization. This process can remove free fatty acids, soap, pesticides, and other organic contaminants, such as polycyclic aromatic hydrocarbons (PAH), from poor quality oils. The bleaching step with diatomaceous earth fully absorbs these compounds (ANTONOPOULOS et al., 2006). On the other hand, the refining process can lead to a significant reduction of desirable components in the oils, such as tocopherols, phenolic compounds and desmethylsterols that are beneficial to human health.
The category that comprises olive oil and olive pomace oil depends on the type of raw material, extraction process, and technological procedures applied. Categorization of oil is a difficult task that often requires the use of multiple analytical tests (APARICIO, 2003; AUED-PIMENTEL, 1991; AUED-PIMENTEL et al., 2008; CODEX..., 2009; MANCINI-FILHO; TAKEMOTO; AUED-PIMENTEL, 2007).
Among the analytical methods available, determining the specific extinction at 270 nm (E 270) and the content of steroidal hydrocarbons (steradienes) can be used evaluate the presence of refined oils in virgin olive oil. During the refining process, compounds with conjugated double bonds can be formed from both unsaturated fatty acids linked to glycerol and dehydration of desmethylsterols from the unsaponifiable fraction. In the first case, oxidation of polyunsaturated fatty acids (e.g. linoleic acid) by the action of bleaching earth takes place facilitating the formation of the conjugated double bonds. (AUED-PIMENTEL; ALMEIDA-GONÇALVES; MANCINI-FILHO, 1993; MITCHEL; KRAYBILL, 1942; O'CONNOR et al., 1949). The presence of these compounds in vegetable oils can be detected by increased ultraviolet absorption at 270 nm (APARICIO, 2003; AUED-PIMENTEL; ALMEIDA-GONÇALVES; MANCINI-FILHO, 1993). In addition, a small fraction of desmethylsterols also undergo dehydration reactions during the refining processes, resulting in unsaturated hydrocarbons. These compounds have a steroidal nucleus, and the stigmasta-3,5-diene, derived from the dehydration of beta-sitosterol, is the most abundant. (CAMACHO; RUIZ-MÉNDEZ; CONSTANTE, 1999; GORDON; FIRMAN, 2001; MATTHÄUS, 2008). Thus, beta-sitosterol produces stigmasta-3,5-diene, campesterol produces campesta-3,5-diene and stigmasterol produces stigmastan-3,5,22-triene. Detection of these traces compounds formed from sterols during the bleaching step indicates the presence of refined oils in virgin olive oils (DIEGO et al., 2008; MODOLO, 2007).
The quantification of stigmasta-3,5-diene and other steroidal hydrocarbons, commonly known as steradienes, is an established tool for the recognition of refining (in particular bleaching) of edible oils. Evidence that an edible vegetable oil was subjected to a refining process, such as the presence of stigmastadiene, is nowadays of a great interest due to the growing market of virgin and cold-pressed specialty oils (MATTHÄUS, 2008; OURRACH et al., 2012).
Low amounts of stigmastadiene have been found in oils labeled as non-refined or cold-pressed olive oil (less than 0.15 mg kg - 1). Neither the typical production of any oil (virgin olive oil or crude vegetable oils) nor long-term storage has been shown to produce measurable amounts of stigmastadiene (PIIRONEN et al., 2000; GROB; ARTHO; MARIANI, 1992; CERT et al., 1994; SCHULTE, 1994).
The Codex Alimentarius standard and the International Olive Oil Council (IOOC) trade agreement have established the maximum value of stigmastadiene to be 0.15 ppm in virgin olive oil (CODEX..., 2009; INTERNATIONAL..., 2010; BRASIL, 2012). The official analytical method used for the measurement of this compound is laborious, time consuming, and it involves multiple steps with considerable consumption of solvents prior to analysis by gas chromatography (INTERNATIONAL..., 2001b). On the other hand, the determination of specific extinction at 270 nm is rapid and simple (INSTITUTO..., 2005).
The present study aimed to quantify trace amounts of stigmastadiene in commercial olive oils and compare two different analytical methods (stigmasta-3,5-diene content and E 270 values) to assess the presence of refined oils. Fatty acid composition and difference in ECN 42 were also analyzed to assess the authenticity of the oils (APARICIO, 2003; AUED-PIMENTEL et al., 2008).
2 Materials and Methods
2.1 Chemicals and reagents
The following reagents were used: cholesta-3,5-diene (>95% purity; Sigma Aldrich Chemical Co, St. Louis, MO, USA), a mixture of 37 certified methyl esters of fatty acids ranging from C4 to C24 (Supelco, Bellefonte, Pa), a mixture of cis/trans methyl esters of fatty acid isomers of 18:2 and 18:3 (Sigma Aldrich Chemical Co, St. Louis, MO, USA), silica gel 60 (70-230 mesh ASTM, Merck, Darmstadt, Germany), nanograden-hexane (>95% purity; Mallinkrodt, St. Louis, MO, USA), spectroscopic grade cyclohexane (VetecQuímicaFina, Rio de Janeiro, RJ, Brazil), acetone and acetonitrile chromatography grade (J.T.Baker, USA). All other reagents (96% ethanol, potassium hydroxide, sodium sulphate anhydrous, ethyl ether and methanol) were of analytical grade.
2.2 Samples
Ten commercial olive oils were included in the analysis, of which seven were labeled as extra virgin (A, B, C, D. F, G, and I) and three (E, H, and J) as olive oil, indicating a blend of virgin and refined olive oil. Six samples (A, B, G, H, I, and J) were collected by the sanitation authority of the State of São Paulo, Brazil, and sent for analysis due to suspicion of fraud.
The reference sample consisted of 80% refined olive oil, 10% sunflower oil (high stearic and high oleic acids), and 10% refined olive pomace oil. It was provided by the IOOC, with an assigned value for stigmastadiene, and it was also analyzed for an analytical quality control.
The samples were coded as follows: EVOL, extra virgin olive oil; VOl, virgin olive oil; ROL, refined olive oil; OL, olive oil; ROPOL, refined olive pomace oil; OPOL, olive pomace oil.
2.3 Determination of stigmastadiene
One mL of a standard solution of cholesta-3,5-diene (0.3 mg mL - 1) was added to approximately 20.0 g of virgin olive oil (INTERNATIONAL..., 2001b) or 1.0 g of olive oil (i.e. blend of virgin and refined olive oil) (INTERNATIONAL..., 2001c). Extra virgin olive oil samples were previous saponified, and the unsaponifiable matter was extracted with n-hexane, as described in COI/T.20/Doc. nº 11 (INTERNATIONAL..., 2001b).
The steroidal hydrocarbon fraction of the unsaponifiable matter of virgin olive oils or direct olive oil samples, which probably contain more than 4 ppm of stigmatadines (INTERNATIONAL..., 2001c), was isolated through separation in a silica gel column with approximately 70 mL n-hexane eluent. The first 30 mL of eluent was discarded, and the remaining 40 mL, which was supposed to contain the hydrocarbon fraction of interest, was collected and evaporated to complete drying. The residue was dissolved in n-hexane and subjected to analysis in a gas chromatograph (Shimadzu, Model 17A) with a flame ionization detector (GC-FID) and equipped with a fused silica column containing a 5% phenyl methyl silicon stationary phase (30 m × 0.25 mm × 0.25 µm) (DB-5, J&W Scientific, USA). The setting conditions of the gas chromatograph were as follows: 235 °C (6 minutes) rising by2 °C min - 1 up to 285 °C, carrier gas: hydrogen, pressure: 187kPa, split ratio 1:10, injector temperature: 300 °C, and detector, 320 °C. Quantification and identification were performed relative to the internal standard (cholesta-3,5-diene) (INTERNATIONAL..., 2001b).
2.4 Determination of ultraviolet absorbency (specific extinction) at 270 nm
Specific extinction was determined by first dissolving the oil samples in spectroscopic grade (1%) cyclohexane and then measuring ultraviolet absorbance at 270 nm using a HP 8453 spectrophotometer (INSTITUTO..., 2005).
2.5 Fatty acid determination by gas chromatography analysis
Fatty acid composition of oils was determined as previously described in Métodos Físicos e Químicos (INSTITUTO..., 2005). Fatty acid methyl esters were prepared with a methanolic KOH solution (2 mol L-1) and injected into a GC-FID (Shimadzu, Model 17A, Japan) equipped with a fused silica column with a cyano propyl siloxane stationary phase (100 m x 0.25 mm x 0.25 µm) (CP 7420, Varian, USA). The following conditions were applied: injector and detector temperature: 250 °C; oven programming temperatures: 45 °C (1 minute) rising by 13 °C min - 1 up to 175 °C, then rising by 4 °C min - 1 up to 215 °C for 35 minutes; carrier gas: hydrogen; and column pressure: 175 kPa; split ratio of 1:50. Separated components were identified by co-injection with standards and comparison of the absolute retention time. Fatty acids were quantified using an area normalization technique.
2.6 Difference between observed and expected triacylglycerol content with ECN 42
Triacylglycerols were separated and quantified using a high performance liquid chromatograph with a refractive index detector, according to the standard COI/T.20/ Doc. nº 20 (INTERNATIONAL..., 2001a). The samples were subjected to prior purification in a column with silica gel 60 to remove oxidation products. A computer program (CORTESI, ROVELLINI, FEDELI, 1990) was used to calculate the difference between the observed triacylglycerol content determined by high performance liquid chromatography and the expected triacylglycerol content based on the composition of fatty acids. The following conditions were used: HPLC, (Shimadzu) consisting of modules: LC-10AD pump, Rheodyne injector with a looping of 20 µL, refractive index detector RID-10AD, communication module CBM-20A, and software LC Solution for data storage and system control. A C-18 reverse phase column (250 mm × 4.6 mm × 5 µm) (Varian, USA); mobile phase: acetone:acetonitrile (1:1), flow 1.15 mL min - 1 was also used.
3 Results and discussion
Stigmasta-3,5-diene content, E 270 values, and the ECN 42 difference observed in commercial olive oils and the reference values obtained from Codex Stan 33 (CODEX..., 2009) are summarized in Table 1.
Table 2 shows the reference and measured fatty acid composition of commercial olive oil samples (CODEX..., 2009).
A generally characteristic fatty acid profile of olive oil was observed for most samples, with the exception of sample G. Specifically, linoleic acid content in sample G was approximately 28%, while the established limit for linoleic acid in olive oil is 21% (CODEX..., 2009), indicating that a seed oil rich in linoleic acid was added during preparation. Sample I contained 1% of linoleic acid, which is the limit established in the Brazilian Normative Instruction n°1 of Ministério da Agricultura, Pecuária e Abastecimento, the current law for classification, quality, and identity parameters for olive oil commercialized in Brazil (BRASIL, 2012). In the current Codex Alimentarius standard for olive oil and pomace olive oil, the limit for linoleic acid was not establish due to lack of a consensus among countries (CODEX..., 2009). Based on fatty acid composition alone, sample I would be considered a virgin olive oil.
Considering the difference in ECN42 in the samples A, B, G, and I, the values were greater than those of extra virgin olive oil, in accordance with Codex Standard 33 (CODEX..., 2009) (Table 1). The limit for the ECN 42 difference for the extra virgin olive oil is 0.20, but the value obtained for samples A and B were almost 0.40, which is within the expected range for olive pomace oil and refined olive pomace oil (i.e. oils with poor quality). As for the olive pomace oil, the value of ECN 42 difference is higher than that of virgin olive oil (0.40-0.50). It has been shown that the contribution of various parts of the fruit influences oil composition, reflecting an increase in trilinolein (LLL) and, consequently, the value of ECN 42 difference (SYNOURI et al., 1995). Samples G and I had values greater than 0.50, indicating the presence of a seed oil rich in linoleic acid, such as soybean. Based on these results, it became quite evident that the ECN 42 difference is a much more sensitive parameter than the fatty acid composition to detect the addition of rich linoleic seed oils in the commercial samples G and I (Tables 1 and 2).
Six samples labeled as extra virgin olive oil showed specific extinction values at 270 nm within the range of the category specified on the label (Table 1). A value higher than 1.00 was observed in four samples (A, B, G, and I), which is far greater than the 0.22, the maximum value established for extra virgin olive oil. According to Codex Stand 33, E 270 values greater than 1.00 are only expected for refined olive oil (ROL), refined olive pomace oil (ROPOL), and olive pomace oil (OPOL), which is a blend of refined olive pomace oil and virgin olive oil.
The content of stigmastadiene in three samples (C, D, and F) was below the established limit for extra virgin olive oil (0.15 mg kg - 1). Olive oil samples E, H, and J contained 26.4, 24.9 and 41.7 mg kg - 1, respectively, indicating the presence of refined olive oil, which was expected given that this category of olive oil (a blend of virgin and refined olive oil) (PIIRONEN et al., 2000; GROB; ARTHO; MARIANI, 1992; CERT et al., 1994; SCHULTE, 1994). The extra virgin olive oil samples A, B, G, and I had very high levels of stigmastadiene and very high readings for E 270 (Table 1).
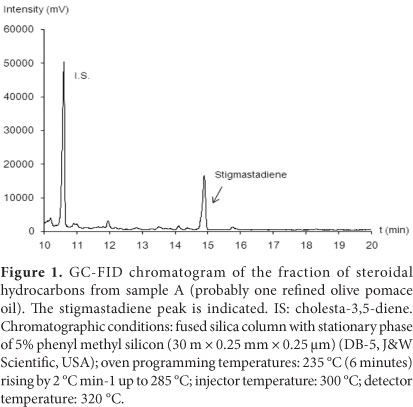
Figure 1 shows the chromatogram of the isolated fraction of steroidal hydrocarbons in the refined oil and the stigmastadiene peak. The stigmasta-3,5-diene goes with small quantities of an isomer (stigmasta-2,4-diene), and usually both originate a single chromatographic peak, as observed in this study. Nevertheless, if the column is too polar or shows a great resolving power, the isomer may appear as a small peak just before the stigmasta-3,5-diene peak. In this case, the two areas must be summed. To ensure the analytical quality of stigmastadiene quantitation, a sample with reference values was analyzed. The reference sample COI 47/10 contained 32.67 ± 9.57 mg kg - 1 stigmastadiene, and 35.26 mg kg - 1 (mean of two determinations) was quantified in our laboratory using the described methods.
Stigmastadiene levels between 2 to 135 mg kg - 1have been reported in commercial fats and margarines or between 2 to 45 mg kg - 1 in refined olive oils. For refined pomace olive oils, stigmastadiene contents can reach up to 200 mg kg - 1. Samples A and B had very high amounts of stigmastadiene. These values are similar to the values described in the literature for refined pomace olive oils (PIIRONEN et al., 2000; GROB; ARTHO; MARIANI, 1992; CERT et al., 1994; SCHULTE, 1994).
Based on E 270 values and on the content of stigmastadiene, poor quality oils or refined olive pomace oil were probably added to samples A and B. These results were corroborated by the values of ECN42 difference found.
Pomace olive oil, which is extracted using high heat and solvent, is the main by-product from olive oil processing. Production and consumption of refined olive pomace oil have been increasing due to mill technological advances (ANTONOPOULOS et al., 2006; MÉNDEZ; MORAL, 2006). However, problems could occur because of the quality of raw material. The high moisture content of olive cake may also require an additional drying step, and toxic PAH may be formed (GUILLÉN; SOPELANA; PALENCIA, 2004). The consumption of this type of oil is a major risk to public health. In addition to the toxic compounds that can be formed, significant quantities of stigmastadiene can also be formed. It is a consequence of the demand for more drastic conditions of the refining process, requiring greater quantity and quality of active carbon and bleaching earth used (ANTONOPOULOS et al., 2006).
A variety of factors, such as bleaching temperature or the amount of earth and its activity, affect the reaction of stigmastadienes formation. The drastic refining conditions of olive pomace oils can generate stigmastadiene quantities up to 200 mg kg - 1 (PIIRONEN et al., 2000; GROB; ARTHO; MARIANI, 1992; CERT et al., 1994; SCHULTE, 1994).
Trans fatty acid (TFA) content is also a good indicator of the presence of refined oils in virgin olive oil (APARICIO, 2003). Among edible vegetable oils, olive oil is the only one with established limits for trans fatty acid levels. Extra virgin olive oil, which was not subjected to the industrial process of refining, has a 0.05% maximum acceptable value of trans isomer of octadecanoicacid (18:1). Furthermore, the sum of octadecadienoic and octadecatrienoic trans fatty acid isomers (18:2 and 18:3) should not exceed 0.05%. For olive oil, which is a mixture of refined and virgin olive oil, 0.20% is the limit for 18:1and 0.30% is the limit for the sum of 18:2 and 18:3 trans isomers (CODEX..., 2009).
Samples A, B, G, and I (labeled as extra virgin olive oil) had levels of TFA above established limits. Drastic refining processes also promote the formation of TFA (AUED-PIMENTEL et al., 2009). In samples A and B, the contents of both octadecaenoic acid and the sum of 18:2 and 18:3 trans isomers were higher than 0.05%, whereas only the sum of 18:2 and 18:3 trans isomers in samples G and I exceeded the established limits (Table 2).
4 Conclusions
Four extra virgin olive oil samples included in the current analysis showed levels of stigmastadiene and E 270 values higher than expected for this category of olive oil. Among these oils, two were adulterated with seed oil rich in linoleic acid, while two others were adulterated with olive pomace oil based on the ECN 42 difference.
The results showed that stigmastadiene quantitation is more sensitive for the detection of refined oils in virgin olive oils than measuring E 270. However, both parameters showed good agreement. The measurement of E 270 is a simple, fast, and inexpensive method that can be easily implemented in Brazilian laboratories to ensure regulation of commercial virgin olive oils. E 270 measurement can be used as a screwing method to sort and control the quality of olive oils. However, complete assessment of the quality and identity of the different types of olive oil should include multiple analytical techniques, such as the ECN 42 difference, fatty acid composition, desmethylesterols, and wax content.
The high content of stigmastadiene found in the two samples is indicative of poor quality oils. These commercial olive oils pose a risk to the health of the consumers due to the possible presence and/or formation of toxic compounds, such as polycyclic aromatic hydrocarbons and trans fatty acids.
Received 2/7/2013
Accepted 6/14/2013 (005997)
- ANTONOPOULOS, K. et al. Olive oil and pomace olive oil processing. Grasas y Aceites, v. 57, n. 1, p. 56-67, 2006.
- APARICIO, R. Manual del aceite de oliva. In: APARICIO, R.; HARWOOD, J. (Ed.). Autentificación del aceite de oliva Madri: Mundi-prensa, 2003. cap. 14, p. 475-496.
- AUED-PIMENTEL, S. Avaliação do grau discriminatório de parâmetros analíticos do azeite de oliva. 1. Aplicação da espectrometria derivada. 1991. 223 f. Dissertação (Mestrado em Ciência de Alimentos)-Faculdade de Ciências Farmacêuticas, Universidade de São Paulo, São Paulo, 1991.
- AUED-PIMENTEL, S.; ALMEIDA-GONÇALVES, M. I.; MANCINI-FILHO, J. Aplicação da espectrofotometria derivada na avaliação da qualidade do azeite de oliva. Ciência e Tecnologia de Alimentos, v. 13, n. 2, p. 121-131, 1993.
- AUED-PIMENTEL, S. et al. Determinação da diferença entre o valor real e o teórico do triglicerídeo ECN 42 para a detecção de adulteração em azeites de oliva comercializados no Brasil. Química Nova, v. 31, n. 1, p. 31-34, 2008. http://dx.doi.org/10.1590/S0100-40422008000100006
- AUED-PIMENTEL, S. et al. Ácidos graxos trans em óleos vegetais poli-insaturados refinados comercializados no Estado de São Paulo, Brasil. Ciência e Tecnologia de Alimentos, v. 29, p.646-51, 2009. http://dx.doi.org/10.1590/S0101-20612009000300030
- BRASIL. Instrução Normativa nº 1, de 30 de janeiro de 2012. Estabelecer o regulamento técnico do azeite de oliva e do óleo de bagaço de oliva. Diário Oficial da União República Federativa do Brasil, Brasília, DF, 01 fev. 2012. Seção 1, n. 23, p. 5-8.
- CAMACHO, M. L.; RUIZ-MÉNDEZ, V.; CONSTANTE, E. G. Changes in olive oil components during deodorization and/or physical refining at the pilot plant scale using nitrogen as stripping gas. Lipid Science, v. 101, n. 1, p. 38-43, 1999. http://dx.doi.org/10.1002/(SICI)1521-4133(19991)101:1<38::AID-LIPI38>3.0.CO;2-3
- CERT, A. et al. Formation of stigmasta-3,5-diene in vegetable oils. Food Chemistry, v. 49, n. 3, p. 287-293, 1994. http://dx.doi.org/10.1016/0308-8146(94)90173-2
- CODEX ALIMENTARIUS. Codex Standards for olive oils, and olive pomace oils Roma: FAO/WHO, 1981. Codex Stan 33. Revisions in 1989, 2003. Amendment in 2009.
- CONSEJO OLEICOLA INTERNACIONAL. Argentina. Desarrolo, perspectivas y potencial de la industria olivícola local. Olivae, v. 116, p. 36-43, 2011.
- CORTESI, N.; ROVELLINI, P.; FEDELI, E. Triglyceride of natural oils. Note 1. Rivista Italiana Delle Sostanze Grasse, v. 67, n. 2, p. 69-73,1990.
- DIEGO, L. et al. Virgin olive oil - Chemical implications on quality and health. European Journal Lipid Science and Technology, v. 110, n. 6, p. 602-607, 2008.
- GORDON, M. H.; FIRMAN, C. Effects of heating and bleaching on formation of stigmastadienes in olive oil. Journal of the Science of Food and Agriculture, v. 81, n. 15, p. 1530-1532, 2001. http://dx.doi.org/10.1002/jsfa.986
- GROB, K.; ARTHO, A.; MARIANI, C. Determination of raffination of edible oils and fats by olefinic degradation products of sterols and squalene, using coupled LCGC. Fat Science Technology, v. 94, n. 10, p. 394-400, 1992.
- GUILLÉN, M. D.; SOPELANA, P.; PALENCIA, G. Polycyclic Aromatic Hydrocarbons and Olive Pomace Oil. Journal of Agricultural and Food Chemistry, v. 52, n. 7, p. 2123-2132, 2004. PMid:15053562. http://dx.doi.org/10.1021/jf035259q
- INSTITUTO ADOLFO LUTZ - IAL. Métodos físico-químicos para análise de alimentos. 4. ed. Brasília: ANVISA, 2005. 1018 p.
- INTERNATIONAL OLIVE OIL COUNCIL - IOOC. Calculation of the difference between the actual and theoretical ECN 42 triglyceride content. IOOC, 2001a. COI/T.20/ Doc. n. 20.
- INTERNATIONAL OLIVE OIL COUNCIL - IOOC. Determination of stigmastadienes in vegetable oils IOOC, 2001b. COI/T.20/Doc. n. 11.
- INTERNATIONAL OLIVE OIL COUNCIL - IOOC. Determination of sterenes in refined vegetable oils IOOC, 2001c. COI/T.20/Doc. n. 16, rev.1.
- INTERNATIONAL OLIVE OIL COUNCIL - IOOC. Trade Standard Applying to Olive Oil and Olive-Pomace Oil IOOC, 2010. COI/T.15/NC n. 3, Rev. n. 5.
- MANCINI-FILHO J.; TAKEMOTO E.; AUED-PIMENTEL, S. Parâmetros de identidade e qualidade de óleos e gorduras. In: ALMEIDA-MURADIAN, L. B.; PENTEADO, M. V. C. (Ed.). Vigilância sanitária: tópicos sobre legislação e análise de alimentos. Rio de Janeiro: Guanabara Koogan, 2007. cap. 6, p. 81-107.
- MATTHÄUS, B. Virgin grape seed oil: Is it really a nutritional highlight? European Journal Lipid Science and Technology, v. 110, n. 6, p. 645-650, 2008. http://dx.doi.org/10.1002/ejlt.200700276
- MÉNDEZ, M. V. R.; MORAL, P. S. Production of pomace olive oil. Grasas Y Aceites, v. 57, n. 1, 2006.
- MITCHEL, J. H.; KRAYBILL, H. R. Formation of conjugated material during bleaching of vegetable oils. Journal American Oil Chemist's Society, v. 64, n. 4, p. 988-994, 1942. http://dx.doi.org/10.1021/ja01256a069
- MODOLO, J. S. Quantificação de estigmastadienos em azeite de oliva (Olea europaea L.) por cromatografia em fase gasosa 2007. 160 f. Dissertação (Mestrado em Tecnologia de Alimentos)-Faculdade de Engenharia de Alimentos, Universidade Estadual de Campinas, Campinas, 2007.
- O'CONNOR, R. T. et al. The influence of processing on the spectral properties of vegetable oils. Journal American Oil Chemist's Society, v. 26, n. 12, p. 710-718, 1949. http://dx.doi.org/10.1007/BF02903179
- OURRACH, I. et al. Detection of argan oil adulterared with vegetable oils: new markers. Grasas Y Aceites, v. 63, n. 4, 2012.
- PIIRONEN V. et al. Plant sterols: biosynthesis, biological function and their importance to human nutrition. Journal Science Food Agriculture, v. 80, n. 7, p. 939-966, 2000. http://dx.doi.org/10.1002/(SICI)1097-0010(20000515)80:7<939::AID-JSFA644>3.0.CO;2-C
- SCHULTE, E. Determination of edible fat refining by HPLC of D 3,5-steradienes. Fat Science Technology, v. 96, n. 4, p. 124-128, 1994.
- SYNOURI, S. et al. Influence of certain factors on the composition of olive-pomace oils. Part I: triglycerides and fatty acids. Rivista Italiana Delle Sostanze Grasse, v. 72, n. 11, p. 483-491, 1995.
Publication Dates
-
Publication in this collection
20 Aug 2013 -
Date of issue
Sept 2013
History
-
Received
02 July 2013 -
Accepted
14 June 2013