Abstracts
The consumption of lutein is associated with the prevention and reduction of age-related macular degeneration. Its incorporation into Prato cheese as a yellowish food coloring is a valid alternative to increase the daily intake of this compound. However, part of the lutein added may be lost in the whey during the cheese making, or it can be degraded by light during storage, resulting in color changes reducing the sensory acceptance of the cheese. The objectives of this study were to determine the transference of the lutein (dye), added to the milk, in the whey, and cheese, to evaluate the effect of the lutein addition, light exposure, and storage time on the cheese color, and to verify the sensory acceptance of Prato cheese with addition of lutein. The lutein recovery of cheese was 95.25%. Color saturation (chrome) increased during storage time resulting in a cheese with more intense color, but there were no changes in the hue of the cheese. Adjusting the amount of lutein added to Prato cheese may lead to greater acceptance. The high recovery of lutein in the cheese and the fact that the hue remained unchanged during storage under light showed that the incorporation of lutein into Prato cheese is feasible from a technical point of view.
lutein; Prato cheese; color
A ingestão de luteína está associada à prevenção e redução da degeneração macular relacionada à idade e a sua adição em queijo Prato, como corante de tom amarelado, é uma alternativa válida para aumentar a ingestão diária desse composto. Entretanto, parte da luteína adicionada pode ser perdida no soro durante a fabricação do queijo e também, ser degradada pela ação da luz durante armazenamento, resultando em mudanças na cor dos queijos, diminuindo a aceitação sensorial. O objetivo do trabalho foi determinar a transferência do corante luteína, adicionado ao leite, no soro e queijo, avaliar o efeito da adição de luteína, exposição à luz e tempo de armazenamento na cor do queijo e verificar a aceitação sensorial do queijo com luteína. A recuperação de luteína no queijo foi de 95,25%. A saturação da cor aumentou com o tempo de armazenamento, resultando em queijos com coloração mais intensa, porém a tonalidade dos queijos não sofreu alteração. Ajustes no teor de luteína adicionado ao queijo Prato pode levar a uma maior aceitação. A alta recuperação de luteína no queijo e a manutenção da cor durante o armazenamento sob luz mostrou que a adição de luteína ao queijo Prato é viável do ponto de vista técnico.
luteína; queijo Prato; cor
ORIGINAL
Transference of lutein during cheese making, color stability, and sensory acceptance of prato cheese
Transferência de luteína na fabricação de queijo prato, estabilidade da cor e aceitação sensorial
Mirian Tiaki Kaneiwa Kubo; Diogo Maus; Ana Augusta Odorissi Xavier; Adriana Zerlotti Mercadante; Walkiria Hanada Viotto* * Corresponding author
Department of Food Technology, Faculty of Food Engineering, University of Campinas - UNICAMP, Rua Monteiro Lobato, 80, CEP 13083-862, Campinas, SP, Brazil, e-mail: walkiria@fea.unicamp.br
ABSTRACT
The consumption of lutein is associated with the prevention and reduction of age-related macular degeneration. Its incorporation into Prato cheese as a yellowish food coloring is a valid alternative to increase the daily intake of this compound. However, part of the lutein added may be lost in the whey during the cheese making, or it can be degraded by light during storage, resulting in color changes reducing the sensory acceptance of the cheese. The objectives of this study were to determine the transference of the lutein (dye), added to the milk, in the whey, and cheese, to evaluate the effect of the lutein addition, light exposure, and storage time on the cheese color, and to verify the sensory acceptance of Prato cheese with addition of lutein. The lutein recovery of cheese was 95.25%. Color saturation (chrome) increased during storage time resulting in a cheese with more intense color, but there were no changes in the hue of the cheese. Adjusting the amount of lutein added to Prato cheese may lead to greater acceptance. The high recovery of lutein in the cheese and the fact that the hue remained unchanged during storage under light showed that the incorporation of lutein into Prato cheese is feasible from a technical point of view.
Keywords: lutein; Prato cheese; color.
RESUMO
A ingestão de luteína está associada à prevenção e redução da degeneração macular relacionada à idade e a sua adição em queijo Prato, como corante de tom amarelado, é uma alternativa válida para aumentar a ingestão diária desse composto. Entretanto, parte da luteína adicionada pode ser perdida no soro durante a fabricação do queijo e também, ser degradada pela ação da luz durante armazenamento, resultando em mudanças na cor dos queijos, diminuindo a aceitação sensorial. O objetivo do trabalho foi determinar a transferência do corante luteína, adicionado ao leite, no soro e queijo, avaliar o efeito da adição de luteína, exposição à luz e tempo de armazenamento na cor do queijo e verificar a aceitação sensorial do queijo com luteína. A recuperação de luteína no queijo foi de 95,25%. A saturação da cor aumentou com o tempo de armazenamento, resultando em queijos com coloração mais intensa, porém a tonalidade dos queijos não sofreu alteração. Ajustes no teor de luteína adicionado ao queijo Prato pode levar a uma maior aceitação. A alta recuperação de luteína no queijo e a manutenção da cor durante o armazenamento sob luz mostrou que a adição de luteína ao queijo Prato é viável do ponto de vista técnico.
Palavras-chave: luteína; queijo Prato; cor.
1 Introduction
Carotenoids are natural pigments known for their coloring properties and biological functions (VAN DEN BERG et al., 2000). They are synthesized by plants, and, therefore, their presence in animals is attributed to the ingestion of food (OLSON; KRINSKY, 1995; YOUNG; LOWE, 2001). Lutein (3,3-dihydroxy-α-carotene) is the second most prevalent carotenoid in the human body (KHACHIK; BEECHER; SMITH, 1995) accumulating mainly in the macula, which is the central part of the retina responsible for the visual acuity of human eye (LANDRUM et al., 1999).
Aging is a natural process that begins at birth and involves several physiological changes such as alterations of digestive function and physical and sensory perceptions (GUIMARAES, 2004). One of the deficiencies resulting from advancing age, age-related macular degeneration (ARMD), affects 25% of the population over 75 years old in Brazil and is the leading cause of irreversible blindness in the elderly in Western countries (FINE et al., 2000). The ingestion of 6 mg per day of lutein leads to a 43% lower risk for ARMD (SEDDON et al., 1994). Lutein may also contribute to a significant reduction in oxidative stress and DNA damage, which can affect the development of cancer cells in the human body (SERPELONI et al., 2010).
Lutein supplementation (capsules or tablets) has been used for prevention and treatment of ARMD. Lutein can be found in dark-green leafy vegetables such as spinach, broccoli, and collard greens in low concentrations ranging from 4 to 5 mg/100 g (RODRIGUEZ-AMAYA et al., 2008); thus, it is necessary the consumption of five daily servings of vegetables to achieve the daily recommended intake of lutein (PERRY; RASMUSSEN; JOHNSON, 2009). With increasing concerns regarding human health, the incorporation of lutein as an ingredient of beverages and foods is quite convenient and attractive.
Prato cheese is one of the most popular cheeses in Brazil, and it is produced in all regions of the country with annual average production of about 150 000 tonnes (ASSOCIAÇÃO..., 2010). It is a ripened, medium-moisture, semi-hard cheese with a mild flavor and pale yellow color, which is given by annatto, a natural dye.
The replacement of annatto with lutein in cheese manufacturing may be an option to increase the intake of this functional compound in the diet, helping to reduce the risk associated with ARMD. Due to the high cost of the commercial lutein dyes, it is important to account for the distribution of lutein added to milk, in the whey, and cheese. Therefore, the objectives of this study were to evaluate the distribution of lutein added to the milk on the characteristics of the cheese and whey, as well as to investigate changes in the cheese color with time during refrigerated storage and to verify the sensory acceptance of Prato cheese with addition of lutein.
2 Materials and methods
2.1 Lutein and emulsifier
The liquid emulsion of lutein (20% Lutein FS, DSM Nutritional Products, Basel, Switzerland) added to the cheeses is a food grade product consisting of crystals of lutein (20%) and zeaxanthin (about 1.0%), extracted from Tagetes erecta and dispersed in corn oil.
The emulsifier used was polysorbate 80 (Alkest TW 80 K, Oxiteno S/A, São Paulo, Brazil) with HLB (hydrophilic-lipophilic balance) around 15 (GRIFFIN, 1949).
2.2 Cheese making
The cheeses were produced in 4 replicates, and the first three processes were used for monitoring the content of lutein and color during storage. The fourth replicate cheeses were used to evaluate the sensory acceptance and purchase intention.
Raw milk (Atilatte, Itatiba, SP, Brazil) was pasteurized in a plate heat exchanger (Eurocal-18, Fischer Term. Ind. Com Ltda, São Paulo, Brazil) at 72 ºC/15 s, cooled to 5 ºC, and stored overnight at 5 ºC. The experiment was performed in the pilot plant of the Laboratory of Dairy Products - DTA/FEA/UNICAMP.
The pasteurized milk was divided into two equal parts of approximately 150 liters for each treatment. One of the portions was used to produce the control cheese without lutein. The other portion was used to produce the cheese with lutein (0.04%) and emulsifier (0.06% polysorbate 80). The order of cheese making was previously randomized.
The use of an emulsifier was necessary due to the presence of high amounts of oil in this emulsion because milk contains about 87.3% water and 3.9 % lipids (WALSTRA; JENNESS, 1984). The choice of the emulsifier was based on its hydrophilic-lipophilic balance (HLB) value. Lecithin, modified lecithin, and polysorbate 80, with increasing HLB values, were tested, and polysorbate 80 had the lowest loss of lutein in the whey.
The milk of each treatment was transferred to the 150 L automatic cheese vat Queijomatic (Biasinox, Lambeth, Brazil), with a heating jacket steam, and heated to 35 ºC. The Lyofast MOS 062 E (SACCO Brazil, Campinas, Brazil), consisting of Streptococcus thermophilus, Lactococcus lactis subsp. lactis and/or subsp. cremoris, was added to milk in the proportion of 1% of the milk weight. Subsequently, calcium chloride (200 ppm) was added, and a lutein emulsion was used in the cheeses with dye. Rennet powder HA LA-1175 (Chr. Hansen, Valinhos, Brazil) was used in an amount sufficient to coagulate the milk in 35 minutes.
After milk coagulation, the gel was cut into cubes with sides of about 1.0 cm. Slow stirring was applied for about 15 minutes (first mixing), followed by an increasing agitation and further heating of the curd/whey mixture, 1 ºC every 2-3 minutes up to 42 ºC. Next, the whey was drained and the curd was distributed into 1 kg rectangular plastic molds and pressed. Pressing was done as follows: 20 psi/15 min, 40 psi/30 min and 50 psi/90 min at room temperature using a vertical pneumatic press (Biasinox, Lambari, MG, Brazi).
After pressing was completed, the cheeses were placed in a 20% NaCl solution for a period of 12 hours at 5º C. After remaining for 48 hours at 12 ºC, the cheeses were weighed, vacuum packed in bags (Spelbag MB-Spel Embalagens Ltda., Sao Paulo, Brazil) composed of coextruded polyolefin/polyamide. Once packed, the cheeses were equally distributed into a BOD incubator (MA 415, Marconi, Piracicaba, Brazil) at 5 ºC and stored for 56 days in the presence and absence of fluorescent light (1000 ± 100 lux). The fluorescent lamp in the BOD was set in a way that the incidence of light was perpendicular to the shelves where the cheeses were stored, in conditions similar to those found in supermarkets. The cheeses were inverted every 7 days during the storage period.
2.3 Sampling
Milk and whey
The milk sample (∼500 mL) was collected after mixing the milk with a stainless steel stirrer. After draining, the total volume of whey was collected in a 100 L tank, stirred, and the sample was then collected.
Prato cheese
The cheeses were chosen randomly to compose the samples. Cheese composition was evaluated five days after cheese making. Slices of about 0.5 cm from the sides, top, and bottom were cut and discarded. The middle portions of the cheeses were cut into smaller cubes and crushed in a multiprocessor to produce 2-3 mm particles. The material obtained was packed in plastic containers without free space and stored at 5 ºC (up to 2 d prior to analyses).
For the instrumental color analysis, 1 cm slices were cut from the central part of the cheese. The remaining cheese was ground and used for determination of lutein content. The samples were packed in plastic containers without free space, wrapped in aluminum foil, and kept refrigerated until analysis.
2.4 Physicochemical composition of milk, whey, and cheese
The pH values of the samples were determined by the potentiometric method (ASSOCIATION..., 1995; method 935.17); titratable acidity was determined by titrating a sample with N/9 sodium hydroxide (Dornic solution) in the presence of phenolphthalein indicator (ASSOCIATION..., 1995; method 947.05); the total solids were determined gravimetrically in a forced air circulation drying oven at 100 ºC for 24 hours (ASSOCIATION..., 1995; method 925.23); fat contents of milk and whey were determined by ethereal extraction according to Monjonier (ASSOCIATION..., 1995; method 989.05), and the fat content of the cheeses were determined by Gerber method (BRITISH..., 1989); the ash content was determined by incineration at 550º C/12 h (ASSOCIATION..., 1995; method 935.42). The analysis of salt content of the cheeses was carried out by the Volhard method (RICHARDSON, 1985). Total nitrogen was determined by the Kjeldahl method (ASSOCIATION..., 1995; method 991.20), and the nitrogen values were multiplied by the factor 6.38 to obtain the equivalent amount of protein. All determinations were carried out in triplicate.
2.5 Quantification of lutein
The total carotenoids found in milk added with lutein correspond to the sum of carotenoids naturally present in milk fat and in the lutein dye. Therefore, the content of lutein in milk was obtained from the difference between the total carotenoids of milk with and without added lutein dye. The contents of lutein in the whey and cheese were also calculated in the same way.
Milk and whey
Measurements of carotenoids in milk and whey with lutein dye were performed according to the method adapted from Hulshof et al. (2006). One gram of sample, 0.25 mL of ammonium acetate, and 1 mL 25% ethanol were stirred with a vortex mixer for 30 seconds, followed by further addition of 2 mL diethyl ether, and 2 mL petroleum ether and vortexed for 2 minutes. The mixture was centrifuged for 2 minutes at 3000 x g, and the ether layer was collected. The extraction and centrifugation procedures were repeated 4 times. The extracts were transferred to a separatory funnel containing 30 mL of ethyl ether and petroleum ether in the ratio of 2:1(v/v) and washed with distilled water. The ether phase was collected, dried on rotary evaporator (temperature < 35 ºC), and the extract was dissolved in a known volume of ethanol to measure absorbance at 445 nm with an Agilent® 8453 UV-visible spectrophotometer (Santa Clara, USA). Total carotenoid concentration was calculated according to the Lambert-Beer law (Equation 1):
where, C is total carotenoid concentration expressed as lutein; A is absorbance at 445 nm, and  = 2550 is the absorptivity coefficient of lutein in ethanol (BRITTON, 1995).
= 2550 is the absorptivity coefficient of lutein in ethanol (BRITTON, 1995).
Forty five grams of sample were used to determine the total carotenoids in milk and whey without added lutein. Since β-carotene is the major carotenoid of milk (CALDERÓN et al., 2007), the extract was dissolved in petroleum ether, and the absorbance was measured at 450 nm. The content of total carotenoids was calculated according to Equation 1, considering  = 2590 as the absorptivity coefficient of β-carotene in petroleum ether (BRITTON, 1995).
= 2590 as the absorptivity coefficient of β-carotene in petroleum ether (BRITTON, 1995).
Cheese
A new method developed by Xavier (2012) was used for determination of the carotenoids in cheese added with lutein. This validated method showed to be linear in the range from 0.05 to 1.15 mg lutein/g cheese, with limit of detection of 0.02 mg/g and limit of quantification of 0.06 mg/g. Good repeatability (relative standard deviation of 2.85%) and high recovery values, between 93 and 98%, were also observed in the developed method. The sample of cheese (0.20 ± 0.01 g) was weighed into a porcelain capsule, and the same amount of hyflosupercel was added. The extraction was performed with ethyl acetate by maceration with a pistil. The organic phase was transferred to a glass tube and centrifuged at 3500 g for 5 minutes at 20 ºC. The supernatant was collected and transferred to flasks. The process of extraction and centrifugation was repeated five times to achieve complete extraction of carotenoids. For the partition step, the combined organic phases were transferred to a separatory funnel containing a mixture of ethyl ether/petroleum ether (2:1, v/v), washed with 4 L distilled water, and collected in the flasks. Anhydrous sodium sulfate was added to remove traces of the remaining water from the washing step, and the solvent was completely evaporated in a rotary evaporator (temperature < 35 ºC). The extract was dissolved in a known volume of ethanol, and the carotenoid concentration was determined as previously described for the milk added with lutein milk and whey.
For extraction of carotenoids from the cheese without lutein, a 15 g sample was centrifuged at 20,000 g for 30 minutes to separate the fat. The supernatant was collected and 5 mL petroleum ether was added to the mass of cheese and centrifuged again. This procedure was performed three times; the supernatants were combined, and the solvent volume was reduced using a rotary evaporator (temperature < 35 ºC) to enable measurement in a 10 mL volumetric flask with petroleum ether. The absorbance of the extract was measured at 450 nm, and the carotenoid content was calculated using the same equation for milk and whey without added lutein.
2.6 Recovery of lutein
The lutein content of the cheese and whey was calculated using Equation 2 as follows:
The recovery percentage of lutein (% R) was calculated as the weight of cheese or whey multiplied by its percentage of lutein and divided by the total weight of lutein present in the milk and multiplied by 100, according to Equation 3:
where, sample refers to whey or cheese.
The total recovery (TR) of lutein was calculated according to Equation 4:
The total recovery of lutein is not exactly 100% due to experimental cumulative errors in both chemical analysis and weight measurements. However, the values of total lutein must be close to 100%. In this study, lutein recoveries in the whey and in the cheeses were standardized to 100% to neutralize the small differences from one tank to another.
2.7 Color measurement
The color of the cheeses was evaluated using the CIELAB system in a colorimeter Color Quest XE (Hunter Associates Laboratory, Reston, Virginia, USA) considering the parameters L* = lightness, a* = red-green, and b* =yellow-blue. The measurements were performed in reflectance mode with the specular component included using illuminant D65 and an observation angle of 10º. The white standard parameters were L*, a*, and b* of 92.03, -0.88, and 0.63, respectively. The color was determined over the surface of a slice with 1 cm thick (BUFFA et al., 2001; LURUEÑA-MARTINEZ et al., 2010). Three readings were performed in the inner part of the cheeses. The instrumental color analysis was performed on days 0, 14, 28, 42, and 56 during refrigerated storage at 5 ºC. The values of C* = chrome (color saturation), h = hue angle (hue), and ΔE* = total color difference were calculated according to Equations 5, 6, and 7, respectively.
2.8 Sensory evaluation
After 30 days of refrigerated storage, the cheeses were subjected to sensory evaluation for consumer acceptance and purchase intention applied to 103 untrained assessors. Initially, the consumers evaluated flavor and texture of commercial Prato cheese (with annatto dye) and Prato cheese with and without lutein. The sessions were performed in isolated sensory evaluation booths under red light so that no color could influence the responses of the assessors. The consumers were provided with water and unsalted crackers for palate cleansing. The same samples were then evaluated for appearance and color attributes, and the sessions were carried out under white light. The cheeses at room temperature were randomly served in portions of 20 g in a disposable white plate coded with a three-digit number.
The attributes color, appearance, flavor, and texture were evaluated using a 9-point hedonic scale, where 1 = dislike extremely, 5 = neither liked/disliked, and 9 = liked extremely (STONE; SIDEL, 1993). Purchase intention was measured on a five-point "definitely would buy" to "definitely would not buy" scale, where 1 = definitely would not buy and 5 = definitely would buy (MEILGAARD et al., 2006).
Prior to the sensory evaluation, a questionnaire on the consumption of Prato cheese was presented to consumers to obtain their free and informed consent for participation in the research. The implementation of the sensory analysis was approved by the Ethics Committee in Research of UNICAMP (CEP opinion no. 858/2009) and the National Information System on Research Ethics by the Certificate of Presentation to Ethics Appreciation (CAAE No 0675.0.146.000 -09).
2.9 Experimental design and statistical analysis
A completely randomized split-split-plot design with three replications was used to evaluate the color of the cheeses. The factor studied was the addition of lutein, and the presence of light was treated as a sub portion and time storage as sub portion of the light factor. The differences between the treatments and their interactions were evaluated by ANOVA. The factors and/or interactions with p values ≤ 0.05 were considered significantly different.
The results of lutein recovery in whey and cheese and the sensory acceptability of the cheese were analyzed by analysis of variance (ANOVA) using the Tukey test to determine differences between means. The results of purchase intention were evaluated using a bar histogram.
3 Results and discussion
3.1 Composition of milk
The pH (6.7) and acidity (0.15% lactic acid) values meet the standards for milk of good quality obtained under hygienic conditions. The fat content of milk added with lutein (3.23%) was slightly higher than that of milk without lutein addition (3.16%), but this fact was expected since the lutein emulsion used in this study presented 96.05 % fat content. As a result, the total solids of the milk added with lutein were slightly higher (11.80%) and the protein content was lower (3.02%) than those found in the control milk, 11.65% and 3.11%, respectively.
3.2 Whey composition
The mean composition of the whey obtained from Prato cheese with or without lutein was within the normal range values considering the variability in the milk composition. The contents of total solids, ash, and protein were similar to those found by Silveira and Abreu (2003) for Prato cheese whey.
The mean composition of the whey obtained from the cheeses with or without lutein performed within the normal range values. The variability observed may be due to the different values found in milk composition.
3.3 Composition of Prato cheese
The results presented in Table 1 show that the composition of Prato cheese is in accordance with the Technical Regulation of Identity and Quality of Cheeses (BRASIL, 1996). This regulation classifies Prato cheese as a fat cheese, which must present 45 to 59.9% fat in dry matter (F/DM) and mean values of moisture varying from 36 to 45.9%.
The Prato cheese with lutein had higher fat content on a dry matter basis than the Prato cheese without lutein, evidencing the higher fat content of the milk with lutein. Cheese manufacturing is a process of concentration of milk fat and protein; therefore, the higher content of the content of fat on dry matter (FDM) in the cheese added with lutein results from milk fat concentration. The cheeses also had salt content (Table 1) in the range of 1.6-1.9%, in agreement with the previously reported findings for Prato cheese (FURTADO; LOURENÇO NETO, 1994).
3.4 Recovery of lutein
The study on the distribution of lutein in cheese manufacture is very important since lutein is an expensive ingredient and, therefore, it is important to determine its recovery in the cheese to evaluate possible losses of this ingredient in the whey.
The results in Table 2 show a recovery of 95.19% lutein in the cheese, in contrast to only 4.81% recovery in the whey. This demonstrates that most of the commercial lutein dye added to the milk was retained in the curd and thus available for consumption. The higher recovery of lutein in the cheese and the lower recovery in the whey showed that lutein losses were minimal in the whey, determining the economic feasibility of using lutein as an ingredient in Prato cheese. This fact was already expected since Prato cheese is a protein-fat concentrate, and the commercial lutein dye chosen was fat-soluble.
3.5 Color stability
Color is an important criterion used to evaluate the quality of the cheese since this parameter is considered by consumers when making purchasing decisions. Statistical evaluation of the effect of lutein addition, light exposure, and storage time on color parameters of Prato cheese is presented in Table 3.
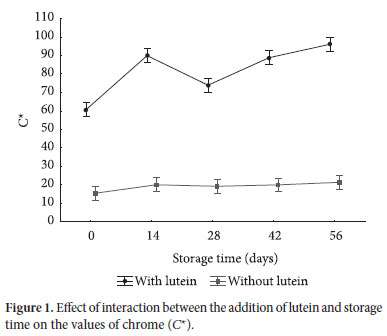
The addition of lutein (p = 0.000) and storage time (p = 0.000) significantly influenced the parameter chrome (C*). Moreover, the interaction between the addition of lutein and storage time was also highly significant (p = 0.000). When the interaction of two variables has a significant influence on the parameter analyzed, it means that they are not independent, and therefore should be analyzed together (interaction effect). Figure 1 shows the effect of interaction between the addition of lutein and storage time on the C* values.
The values of C*, which represent color saturation degree and intensity, show that the cheese with lutein (57.4 at time 0) had a more intense color than the cheese without lutein (14.4 at time 0). The C* values increased during storage for the cheese with lutein and were particularly pronounced between zero and 14 days (81.0) of storage. The increase in color intensity over time is probably due to the evaporation of water from the cheese during storage. The presence or absence of light during storage did not influence the parameter C* (p = 0.099).
The effect of the addition of lutein on the cheese color may be evidenced by the tonality, which is represented by the hue parameter (h). Light exposure (p = 0.293) and the storage period (p = 0.557) did not affect the h values. On the other hand, the addition of lutein significantly influenced h (0.000), as can be seen in Figure 2.
The cheese without lutein presented h value of 93º, which indicates yellow color, whereas the cheese added with lutein (63º) had a more orange color, corresponding to the first quadrant of the three-dimensional diagram of colors between 0º (red) and 90º (yellow).
The interaction between the addition of lutein and storage time (0.000) was significant for the parameter ΔE*, which indicates total color change. The effect of this interaction on the parameter ΔE* is shown in Figure 3. The highest color variation was found at day 14 of storage (ΔE* = 32.7), mainly for the cheeses with lutein. Light exposure during storage did not influence the parameter ΔE* (p = 0.979).
The ΔE* value, considering the time zero as the reference for each treatment, confirms that the difference in color between the cheese with and without lutein is visible to the naked eye. According to Francis and Clydesdale (1975), the differences become perceptible to the human eye if the ΔE* value is less than two units.
Therefore, we conclude that at the end of the storage period the tonality of the cheeses did not change and color saturation increased over time. The cheeses had more intense and vivid color, probably due to moisture loss during storage.
3.6 Sensory acceptance
The mean scores of flavor, texture, appearance, and color of Prato cheese with and without lutein and commercial Prato cheese are presented in Table 4.
There was no difference between samples with respect to the attribute texture. These results are similar to those reported by Jones, Aryana and Losso (2005), who evaluated Cheddar cheeses made with lutein addition.
The addition of lutein did not affect the flavor of the cheeses. However, the cheeses with and without lutein were scored lower for the attribute flavor than the commercial cheese (Table 4).
With regard to the attributes appearance and color, the cheeses with lutein had lower sensory scores than the commercial cheese. The low acceptance of this cheese can be attributed to the more intense yellow-orange color than that of cheeses produced with annatto dye, traditionally used in commercial Prato cheeses. In this study, the concentration of lutein used for cheese manufacturing was higher than that of the annatto dye employed commercially to prevent possible losses of lutein in the whey and to ensure that the amount of lutein incorporated into the cheese would be enough to promote beneficial health effects.
3.7 Purchase intention
Figure 4 shows the histogram of purchase intention of Prato cheese with and without lutein in relation to flavor and texture attributes. Commercial Prato cheese got the highest purchase intention score (75.7% definitely/possibly buy), followed by the Prato cheese without lutein (66% definitely/possibly buy) and Prato cheese with lutein, which presented 47.6% purchase intention. Some assessors reported that residual bitterness of Prato cheese with lutein may probably explain the lower purchase intention score. This result was also found previously for Cheddar cheese colored with lutein (JONES; ARYANA; LOSSO, 2005). The liquid emulsion of lutein has a bitter taste that the assessors detected in the sensory analysis, probably due to the large amount of dye added.
The purchase intention histogram for the attributes appearance and color (Figure 5) shows that 87.4% of the consumers would certainly buy/possibly buy the commercial cheese, whereas 19.5% would definitely or probably buy cheese with lutein. According to the comments by the panelists, the main cause of rejection of the cheese added with lutein was due to its intense yellow-orange color, which is a color associated to Cheddar cheese not to Prato cheese.
An alternative to increase the sensory acceptance and purchase intention of the cheese with lutein is to reduce the amount of added lutein since the one used in this study is higher than the dose of 6 mg per day recommended for preventing and reducing risks of ARMD. The addition of a lower amount of lutein is likely to decrease the cheese bitter aftertaste and reduce the intensity of the yellow-orange color resulting in a color similar to the characteristic color of the commercial Prato cheese.
4 Conclusion
The incorporation of lutein dye into Prato cheese was feasible from a technical and economical point of view since the lutein had a high recovery in this cheese. The light exposure did not change the cheese color during storage although the color intensity increased over time in the cheeses added with lutein. Reducing the amount of lutein added to the cheese may be a good option to increase the sensory acceptance and purchase intention of Prato cheese with lutein.
Acknowledgements
The authors are grateful to the Brazilian foundations CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) and FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) for the financial support.
Received 8/8/2012
Accepted 16/10/2012 (00T5834)
- ASSOCIATION OF OFFICIAL ANALYTICAL CHEMISTS - AOAC. Official methods of analysis 15th ed. Washington: AOAC, 1995.
- ASSOCIAÇÃO BRASILEIRA DAS INDÚSTRIAS DE QUEIJO - ABIQ. Produção brasileira de queijos São Paulo: ABIQ, 2010. Disponível em: <www.scotconsultoria.com.br>. Acesso em: 10 jul. 2012.
- BRASIL. Ministério da Agricultura, Pecuária e Abastecimento. Portaria nş 146, de 07 de março de 1996. Regulamento Técnico de Identidade e Qualidade de Queijos. Diário Oficial da República Federativa do Brasil, Brasília, DF, 11 mar. 1996. Seção 1, n. 172, p. 47-48.
- BRITTON, G. UV/visible spectroscopy. In: BRITTON, S.; LIAANSEN-JENSEN; H. Pfander Carotenoids: Spectroscopy. Basel: Birkhuaser, 1995. v. 1B.
- BRITISH STANDARDS INSTITUTION. Determination of fat content of milk and milk products (Gerber methods) Methods. London: British Standards Institution, 1989. 12 p.
- BUFFA, M. N. et al. Changes in textural, microstructural, and colour characteristics during ripening of cheeses made from raw, pasteurized or high-pressure-treated goats' milk. International Dairy Journal, v. 11, p. 927-934, 2001. http://dx.doi.org/10.1016/S0958-6946(01)00141-8
- CALDERÓN, F. et al. Variations in carotenoids, vitamins A and E, and color in cow's plasma and milk during late pregnancy and the first three months of lactation. Journal of Dairy Science, v. 90, n. 5, p. 2335-2346, 2007. PMid:17430936. http://dx.doi.org/10.3168/jds.2006-630
- FINE, S. L. et al. Age-related macular degeneration. Journal of Medicinal Chemistry, v. 342, p. 483-492, 2000.
- FURTADO, M. M.; LOURENÇO NETO, J. P. M. L. Tecnologia de Queijos São Paulo: Dipemar, 1994. cap. 15: Minas Frescal, p. 73-75. Manual Técnico para a Produção Industrial de Queijos.
- FRANCIS, J. F.; CLYDESDALE, F. M. Food colorimetr y, theory and application New York: Vans Nostrand Reinhold/AVI. 1975.
- GRIFFIN, W. C. Classification of surface- active agents by HLB. Journal of Cosmetic Chemistry, v. 1, p. 311-326, 1949.
- GUIMARAES, F. A. Desenvolvimento de leite pasteurizado para pessoas idosas: identificação de necessidades, e expectativas de segmentos de mercado e utilização da Matriz de Qualidade. 2004. Tese (Mestrado)-Universidade Federal de Viçosa, Minas Gerais, 2004.
- HULSHOF, P. J. M. et al. Variation in retinol and carotenoid content of milk and milk products in The Netherlands. Journal of Food Composition and Analysis, v. 19, n. 1, p. 67-75, 2006. http://dx.doi.org/10.1016/j.jfca.2005.04.005
- JONES, S. T.; ARYANA, K. J.; LOSSO, J. N. Storage stabiliy of lutein during ripening of cheddar cheese. Journal of Dairy Science, v. 88, p. 1661-1670, 2005. http://dx.doi.org/10.3168/jds.S0022-0302(05)72838-1
- KHACHIK, F.; BEECHER, G. R.; SMITH, J. C. Lutein, lycopene, and their oxidative metabolites in chemoprevention of cancer. Journal of Cellular Biochemistry, v. 22, p. 236-246, 1995. http://dx.doi.org/10.1002/jcb.240590830
- LANDRUM, J. T. et al. Carotenoids in human retina. Pure and Applied Chemistry, v. 71, p. 2237-2244, 1999. http://dx.doi.org/10.1351/pac199971122237
- LURUEÑA-MARTINEZ, M. A. et al. The influence of breed on the organoleptic characteristics of Zamorano sheep raw milk cheese and its assessment by instrumental analysis. International Journal of Dairy Technology, v. 63, p. 216-220, 2010. http://dx.doi.org/10.1111/j.1471-0307.2010.00565.x
- MEILGAARD, M.; CIVILLE, G. V.; CARR, B. T. Sensory Evaluation Techniques 4th ed. CRC Press: Boca Raton, FL, 2006. 448 p.
- OLSON, J. A.; KRINSKY, N. I. Introduction: the colorful fascinating world of the carotenoids: important physiologic modulators. FASEB J, 1995. cap. 9, p. 1547-1550.
- PERRY, A.; RASMUSSEN, H.; JOHNSON, E. J. Xanthophyll (lutein, zeaxanthin) content in fruits, vegetables and corn and egg products. Journal of Food Composition and Analysis, v. 22, p. 9-15, 2009. http://dx.doi.org/10.1016/j.jfca.2008.07.006
- RICHARDSON, G. H. (Ed.). Standard methods for examination of dairy products 15th ed. Washington: American Public Health Association, 1985. p. 340-342.
- RODRIGUEZ-AMAYA, D. B. et al. Updated Brazilian database on food carotenoids: Factors affecting carotenoid composition. Journal of Food Composition and Analysis, v. 21, p. 445-463, 2008. http://dx.doi.org/10.1016/j.jfca.2008.04.001
- SEDDON, J. M. et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Journal of the American Medical Association, v. 272, p. 1413-1420, 1994. PMid:7933422. http://dx.doi.org/10.1001/jama.1994.03520180037032
- SERPELONI, J. M. et al. Lutein improves antioxidant defense in vivo and protects against DNA damage and chromosome instability induced by cisplatin. Archives of Toxicology, v. 84, p. 811-822, 2010. PMid:20683581. http://dx.doi.org/10.1007/s00204-010-0576-y
- SILVEIRA, P. R.; ABREU, L. R. Rendimento e composição físico-química do Queijo prato elaborado com leite pasteurizado pelo sistema HTST e injeção direta de vapor. Ciência e Agrotecnologia, v. 27, n. 6, p. 1340-1347, 2003. http://dx.doi.org/10.1590/S1413-70542003000600019
- STONE, H.; SIDEL, J. L. Sensory evaluation practices New York: Academic Press, 1993. 308 p.
- VAN DEN BERG, H. et al. The potential for the improvement of carotenoid levels in foods and the likely systemic effects. Journal of the Science of Food and Agriculture, v. 80, p. 880-912, 2000. http://dx.doi.org/10.1002/(SICI)1097-0010(20000515)80:7<880::AID-JSFA646>3.0.CO;2-1
- WALSTRA, P.; JENNESS, R. Dairy chemistry and physics Nova York: John Wiley & Sons, 1984.
- XAVIER, A. A. O. Carotenoides em leite e produtos lácteos adicionados de corante luteína: métodos analíticos, estabilidade e bioacessibilidade in vitro 2012. 132 f. Tese (Doutorado em Ciência de Alimentos)-Faculdade de Engenharia de Alimentos, Universidade Estadual de Campinas, Campinas, 2012.
- YOUNG, A. J.; LOWE, G. M. Antioxidant and prooxidant properties of carotenoids. Archives of Biochemistry and Biophysics, v. 385, p. 20-27, 2001. PMid:11361018. http://dx.doi.org/10.1006/abbi.2000.2149
Publication Dates
-
Publication in this collection
06 Mar 2013 -
Date of issue
Feb 2013
History
-
Received
08 Aug 2012 -
Accepted
16 Oct 2012