Abstract
A 32 factorial design was employed to develop an in vitro digestion method for estimation of Fe bioaccessible fractions in cooked chicken meat. The effects of sample size and the in vitro bioaccessible fractions of this essential element were evaluated. A sample preparation method employing a microwave assisted digestion with dilute nitric acid was used prior to total Fe determination. For the bioacessibility studies, the optimized procedure employed 7.5 g of sample and 6% w/v of an acid pepsin solution. This procedure was applied to two kinds of chicken meat samples: breast and liver. Flame Atomic Absorption Spectrometry was used to determine total and bioaccessible (chyme or soluble portion) levels of iron in the samples. With respect to total Fe content, the bioaccessible fractions of Fe found in these samples were around 23% and 56 %, for breast and chicken liver, respectively. The chicken liver sample showed the highest total (400 ± 10 mg kg-1) and bioaccessible Fe contents (223 ± 18 mg kg-1) and stands out as a good source of this micronutrient.
Keywords:
iron bioaccessibility; factorial design; chicken meat; microwave assisted digestion; Flame Atomic Absorption Spectrometry
1 Introduction
Brazil is a country with intense agricultural activity and stands out in the international market in terms of the production and export of poultry products, especially chicken. According to the Brazilian Association of Animal Protein – ABPA (Associação Brasileira de Proteína Animal, 2015Associação Brasileira de Proteína Animal – ABPA. (2015). Relatório Anual. São Paulo: ABPA. Retrieved from http://abpa-br.com.br/files/publicacoes/c59411a243d6dab1da8e605be58348ac.pdf
http://abpa-br.com.br/files/publicacoes/...
), total Brazilian production of chicken meat stands at 12.69 million tons in 2014, of which, 32.3% was destined for exportation. Additionally, Brazilian exports of chicken products for the same year exceeded the exports of the United States of America, China and the European Community. Given the economic importance of this commodity to the Brazilian economy, studies to monitor the quality and/or nutritional characteristics of chicken meat are extremely relevant.
This poultry meat is also an important source of several nutrients for the humans namely proteins, amino acids, lipids and some trace elements such as Cu, Fe, Mn and Zn. Among these micronutrients, Fe content was investigated due to its high importance in overall health and nutrition. This essential element is found in animal tissues including muscle and viscera, as well as in plant, such as beans and other pulse crops (López & Martos, 2004López, M. A. A., & Martos, F. C. (2004). Iron availability: an updated review. International Journal of Food Sciences and Nutrition, 55(8), 597-606. http://dx.doi.org/10.1080/09637480500085820.
http://dx.doi.org/10.1080/09637480500085...
).
In food, Fe is found in two chemical forms: heme-iron or ferrous, Fe (II) and non-heme iron or ferric, Fe (III). Heme iron is present in beef, fish and poultry, especially in their muscle and viscera, and usually has a higher bioavailability than ferric iron because the former does not interact with absorption inhibitors compounds such as phytates and Ca present in the gastrointestinal tract (Azevedo & Chasin, 2003Azevedo, F. A., & Chasin, A. A. M. (2003). Metais: gerenciamento da toxicidade. São Paulo: Editora Atheneu.; Grotto, 2008Grotto, H. Z. W. (2008). Metabolismo do ferro: uma revisão sobre os principais mecanismos envolvidos em sua homeostase. Revista Brasileira de Hematologia e Hemoterapia, 30(5), 390-397. http://dx.doi.org/10.1590/S1516-84842008000500012.
http://dx.doi.org/10.1590/S1516-84842008...
).
Many biological processes are mediated by enzymes that require iron as a cofactor, including cytochromes for electron transport in the respiratory chain, cytochromes P450 involved in the detoxification of foreign substances in the liver, the synthesis of steroid hormones and bile acids, DNA synthesis, as well as signal controlling in some neurotransmitters such as serotonin and dopamine in the brain.; Furthermore, most of the iron in the organism is bound to hemoglobin in the blood and myoglobin in muscle tissue, which are responsible for oxygen transport and storage respectively (Azevedo & Chasin, 2003Azevedo, F. A., & Chasin, A. A. M. (2003). Metais: gerenciamento da toxicidade. São Paulo: Editora Atheneu.). Iron deficiency is considered a serious global health problem that affects a great part of the population, mostly in developing countries. In Brazil, The Brazillian Health Regulatory Agency, ANVISA, based on the World Health Organization`s recommendations, established the daily intake of this element for various population groups: adults (14 mg Fe/day), pregnant (27 mg Fe/day) and lactating (15 mg Fe/day) women who require higher values (Brasil, 2005Brasil, Agência Nacional de Vigilância Sanitária. (2005, September 23). Resolução RDC n.º 269, de 22 de setembro de 2005. Aprova o “Regulamento Técnico sobre a Ingestão Diária Recomendada (IDR) de Proteína, Vitaminas e Minerais”. Diário Oficial [da] República Federativa do Brasil.)
Due to the critical role of iron in human nutrition, it is necessary to determine the total concentration in food as well as the quantity that is available for absorption in the gastrointestinal tract. This quantity is frequently denominated as the bioaccessible fraction (Peixoto et al., 2013Peixoto, R. R. A., Mazon, E. A. M., & Cadore, S. (2013). Estimation of the bioaccessibility of metallic elements in chocolate drink powder using an in vitro digestion method and spectrometric techniques. Journal of the Brazilian Chemical Society, 24, 884-890. http://dx.doi.org/10.5935/0103-5053.20130111.
http://dx.doi.org/10.5935/0103-5053.2013...
). Most studies, however, involve the determination of total iron in food stuffs and cannot provide this important piece of nutritional information.
The bioaccessibility of an element can be realized by in vivo tests, using experimental animals or even humans, or in vitro experiments. Ethical concerns and higher costs have been a considerable limitation for in vivo studies (Peixoto et al., 2013Peixoto, R. R. A., Mazon, E. A. M., & Cadore, S. (2013). Estimation of the bioaccessibility of metallic elements in chocolate drink powder using an in vitro digestion method and spectrometric techniques. Journal of the Brazilian Chemical Society, 24, 884-890. http://dx.doi.org/10.5935/0103-5053.20130111.
http://dx.doi.org/10.5935/0103-5053.2013...
). In such cases, many in vitro methods can be found in the literature to obtain bioaccessible and bioavailable fractions for metals from foodstuffs (Miller et al., 1981Miller, D. D., Schricker, B. R., Rasmussen, R. R., & Van Campen, D. (1981). An in vitro method for estimation of iron availability from meals. The American Journal of Clinical Nutrition, 34(10), 2248-2256.; Luten et al., 1996Luten, J., Crews, H., Flynn, A., Van Dael, P., Kastenmayer, P., Hurrell, R., Deelstra, H., Shen, L.-H., Fairweather-Tait, S., Hickson, K., Farré, R., Schlemmer, U., & Frøhlich, W. (1996). Interlaboratory trial on the determination of the in vitro iron dialysability from food. Journal of the Science of Food and Agriculture, 72(4), 415-424. http://dx.doi.org/10.1002/(SICI)1097-0010(199612)72:4<415::AID-JSFA675>3.0.CO;2-X.
http://dx.doi.org/10.1002/(SICI)1097-001...
; Cámara et al., 2005Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009.
http://dx.doi.org/10.1016/j.foodchem.200...
; Purchas et al., 2006Purchas, R. W., Busboom, J. R., & Wilkinson, B. H. P. (2006). Changes in the forms of iron and in concentrations of taurine, carnosine, coenzyme Q(10), and creatine in beef longissimus muscle with cooking and simulated stomach and duodenal digestion. Meat Science, 74(3), 443-449. http://dx.doi.org/10.1016/j.meatsci.2006.03.015.
http://dx.doi.org/10.1016/j.meatsci.2006...
; Kulkarni et al., 2007Kulkarni, S. D., Acharya, R., Rajurkar, N. S., & Reddy, A. V. R. (2007). Evaluation of bioaccessibility of some essential elements from wheatgrass (Triticum aestivum L.) by in vitro digestion method. Food Chemistry, 103(2), 681-688. http://dx.doi.org/10.1016/j.foodchem.2006.07.057.
http://dx.doi.org/10.1016/j.foodchem.200...
; Hur et al., 2011Hur, S. J., Lim, B. O., Decker, E. A., & McClements, D. J. (2011). In vitro human digestion models for food applications. Food Chemistry, 125(1), 1-12. https://doi.org/10.1016/j.foodchem.2010.08.036.
https://doi.org/10.1016/j.foodchem.2010....
; Ramos et al., 2012Ramos, A., Cabrera, M. C., & Saadoun, A. (2012). Bioaccessibility of Se, Cu, Zn, Mn and Fe, and heme iron content in unaged and aged meat of Hereford and Braford steers fed pasture. Meat Science, 91(2), 116-124. http://dx.doi.org/10.1016/j.meatsci.2012.01.001.
http://dx.doi.org/10.1016/j.meatsci.2012...
). In these studies, a great challenge is to simulate all physiological conditions within the human organism as well as the sequence of events that occur during the digestion of food. Factors such as temperature, peristaltic movements, pH, residence time and enzymatic composition of digestive fluids (gastric juice, bile and duodenal fluid) are extremely important to reproduce in vitro all the events that occur in vivo (Hur et al., 2011Hur, S. J., Lim, B. O., Decker, E. A., & McClements, D. J. (2011). In vitro human digestion models for food applications. Food Chemistry, 125(1), 1-12. https://doi.org/10.1016/j.foodchem.2010.08.036.
https://doi.org/10.1016/j.foodchem.2010....
).
Studies with respect to the bioaccessibility of mineral elements in various types of food are plentiful in literature (Miller et al., 1981Miller, D. D., Schricker, B. R., Rasmussen, R. R., & Van Campen, D. (1981). An in vitro method for estimation of iron availability from meals. The American Journal of Clinical Nutrition, 34(10), 2248-2256.; Luten et al., 1996Luten, J., Crews, H., Flynn, A., Van Dael, P., Kastenmayer, P., Hurrell, R., Deelstra, H., Shen, L.-H., Fairweather-Tait, S., Hickson, K., Farré, R., Schlemmer, U., & Frøhlich, W. (1996). Interlaboratory trial on the determination of the in vitro iron dialysability from food. Journal of the Science of Food and Agriculture, 72(4), 415-424. http://dx.doi.org/10.1002/(SICI)1097-0010(199612)72:4<415::AID-JSFA675>3.0.CO;2-X.
http://dx.doi.org/10.1002/(SICI)1097-001...
; Cámara et al., 2005Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009.
http://dx.doi.org/10.1016/j.foodchem.200...
; Purchas et al., 2006Purchas, R. W., Busboom, J. R., & Wilkinson, B. H. P. (2006). Changes in the forms of iron and in concentrations of taurine, carnosine, coenzyme Q(10), and creatine in beef longissimus muscle with cooking and simulated stomach and duodenal digestion. Meat Science, 74(3), 443-449. http://dx.doi.org/10.1016/j.meatsci.2006.03.015.
http://dx.doi.org/10.1016/j.meatsci.2006...
; Kulkarni et al., 2007Kulkarni, S. D., Acharya, R., Rajurkar, N. S., & Reddy, A. V. R. (2007). Evaluation of bioaccessibility of some essential elements from wheatgrass (Triticum aestivum L.) by in vitro digestion method. Food Chemistry, 103(2), 681-688. http://dx.doi.org/10.1016/j.foodchem.2006.07.057.
http://dx.doi.org/10.1016/j.foodchem.200...
; Hur et al., 2011Hur, S. J., Lim, B. O., Decker, E. A., & McClements, D. J. (2011). In vitro human digestion models for food applications. Food Chemistry, 125(1), 1-12. https://doi.org/10.1016/j.foodchem.2010.08.036.
https://doi.org/10.1016/j.foodchem.2010....
; Maulvault et al., 2011Maulvault, A. L., Machado, R., Afonso, C., Lourenço, H. M., Nunes, M. L., Coelho, I., Langerholc, T., & Marques, A. (2011). Bioaccessibility of Hg, Cd and As in cooked black scabbard fish and edible crab. Food and Chemical Toxicology, 49(11), 2808-2815. http://dx.doi.org/10.1016/j.fct.2011.07.059.
http://dx.doi.org/10.1016/j.fct.2011.07....
; Koch et al., 2011Koch, I., Moriarty, M., House, K., Sui, J. & Cullen, W. R. Saper R. B, Reimer K. J. (2011). Bioaccessibility of lead and arsenic in traditional Indian medicines. Science of the total environment, 409(21), 4545-4552. http://doi.org/10.1016/j.scitotenv.2011.07.059. PMid:21864885
http://doi.org/10.1016/j.scitotenv.2011....
; Ramos et al., 2012Ramos, A., Cabrera, M. C., & Saadoun, A. (2012). Bioaccessibility of Se, Cu, Zn, Mn and Fe, and heme iron content in unaged and aged meat of Hereford and Braford steers fed pasture. Meat Science, 91(2), 116-124. http://dx.doi.org/10.1016/j.meatsci.2012.01.001.
http://dx.doi.org/10.1016/j.meatsci.2012...
, Peixoto et al., 2013Peixoto, R. R. A., Mazon, E. A. M., & Cadore, S. (2013). Estimation of the bioaccessibility of metallic elements in chocolate drink powder using an in vitro digestion method and spectrometric techniques. Journal of the Brazilian Chemical Society, 24, 884-890. http://dx.doi.org/10.5935/0103-5053.20130111.
http://dx.doi.org/10.5935/0103-5053.2013...
; Stelmach et al., 2014Stelmach, E., Pohl, P., & Szymczycha-Madeja, A. (2014). Evaluation of the bioaccessability of Ca, Fe, Mg and Mn in ground coffee infusions by in vitro gastrointestinal digestion. Journal of the Brazilian Chemical Society, 25, 1993-1999. http://dx.doi.org/10.5935/0103-5053.20140183.
http://dx.doi.org/10.5935/0103-5053.2014...
; Silva et al., 2015Silva, E. N., Heerdt, G., Cidade, M., Pereira, C. D., Morgon, N. H., & Cadore, S. (2015). Use of in vitro digestion method and theoretical calculations to evaluate the bioaccessibility of Al, Cd, Fe and Zn in lettuce and cole by inductively coupled plasma mass spectrometry. Microchemical Journal, 119, 152-158. http://dx.doi.org/10.1016/j.microc.2014.12.002.
http://dx.doi.org/10.1016/j.microc.2014....
; Pereira et al., 2016Pereira, C. C., Silva, E. N., Souza, A. O., Vieira, M. A., Ribeiro, A. S., & Cadore, S. (2016). Evaluation of the bioaccessibility of minerals from blackberries, raspberries, blueberries and strawberries. Journal of Food Composition and Analysis. In press. http://dx.doi.org/10.1016/j.jfca.2016.12.001.
http://dx.doi.org/10.1016/j.jfca.2016.12...
) but are particularly rare concerning chicken products. Despite the amount of in vitro bioaccessibility studies, there is one paper that suggests a consensus (Minekus et al., 2014Minekus, M., Alminger, M., Alvito, P., Ballance, S., Bohn, T., Bourlieu, C., Carrière, F., Boutrou, R., Corredig, M., Dupont, D., Dufour, C., Egger, L., Golding, M., Karakaya, S., Kirkhus, B., Le Feunteun, S., Lesmes, U., Macierzanka, A., Mackie, A., Marze, S., McClements, D. J., Ménard, O., Recio, I., Santos, C. N., Singh, R. P., Vegarud, G. E., Wickham, M. S. J., Weitschies, W., & Brodkorb, A. (2014). A standardised static in vitro digestion method suitable for food - an international consensus. Food & Function, 5(6), 1113-1124. http://dx.doi.org/10.1039/c3fo60702j.
http://dx.doi.org/10.1039/c3fo60702j...
) for this type of experiment to be applied to food matrices (foods in general). However, especially for chicken meat, there is still no available results regarding this procedure. One important parameter relates to pepsin concentrations, the principal proteolytic enzyme in the stomach. As such, it is necessary to determine if the experimental conditions utilized in these in vitro studies achieve conditions as similar as possible to in vivo situations.
Furthermore, very few works on this topic are found in the Brazilian literature (Nascimento et al., 2010Nascimento, A. N., Naozuka, J., & Oliveira, P. V. (2010). In vitro evaluation of Cu and Fe bioavailability in cashew nuts by off-line coupled SEC–UV and SIMAAS. Microchemical Journal, 96(1), 58-63. http://dx.doi.org/10.1016/j.microc.2010.01.016.
http://dx.doi.org/10.1016/j.microc.2010....
; Peixoto et al., 2013Peixoto, R. R. A., Mazon, E. A. M., & Cadore, S. (2013). Estimation of the bioaccessibility of metallic elements in chocolate drink powder using an in vitro digestion method and spectrometric techniques. Journal of the Brazilian Chemical Society, 24, 884-890. http://dx.doi.org/10.5935/0103-5053.20130111.
http://dx.doi.org/10.5935/0103-5053.2013...
; Silva et al., 2015Silva, E. N., Heerdt, G., Cidade, M., Pereira, C. D., Morgon, N. H., & Cadore, S. (2015). Use of in vitro digestion method and theoretical calculations to evaluate the bioaccessibility of Al, Cd, Fe and Zn in lettuce and cole by inductively coupled plasma mass spectrometry. Microchemical Journal, 119, 152-158. http://dx.doi.org/10.1016/j.microc.2014.12.002.
http://dx.doi.org/10.1016/j.microc.2014....
; Pereira et al., 2016Pereira, C. C., Silva, E. N., Souza, A. O., Vieira, M. A., Ribeiro, A. S., & Cadore, S. (2016). Evaluation of the bioaccessibility of minerals from blackberries, raspberries, blueberries and strawberries. Journal of Food Composition and Analysis. In press. http://dx.doi.org/10.1016/j.jfca.2016.12.001.
http://dx.doi.org/10.1016/j.jfca.2016.12...
) In this sense, it is important the development of studies regarding the bioaccessible fractions of nutrients in food widely and routinely consumed in Brazil, such as chicken meat. Additionally, the contents of a mineral micronutrient such as iron can be influenced by several factors in meat such as the breed, age, sex and nourishment of the animal (Olivio, 2008Olivio, N. (2008). Mercado Mundial de Carnes. Criciúma: Editora Varela.). In this context, routine analysis and monitoring of chicken meat produced and commercialized in Brazil is relevant to evaluate some nutritional aspects related to its consume such as iron bioaccessible fractions. The aim of the present work was to develop an in vitro method for the estimation of the bioaccessible fraction of iron in chicken meat after a home cooking procedure and to determine, using a 32 factorial design approach, which variables affects these values.
2 Materials and methods
2.1 Apparatus and reagents
All materials were washed in neutral soap (Prolab), soaked in 10 % (v/v) nitric acid for 24 hours and then washed with deionized water prior to use. Deionized water was obtained with a Mili-Q System (Millipore, Bedford, MA, USA).
A 1,000 µg mL-1 Fe solution (Chemis High Purity, Jundiai, SP, Brazil) was used to prepare standard solutions with concentrations between 0.5 and 4.0 mg L-1.
A microwave oven from Berghof (model SpeedWave Four, Eningen, BW, Germany) was used for the acid digestion employing HNO3 65% m/v P.A. (Vetec, Rio de Janeiro, RJ, Brazil), for total iron determination.
The materials used for cooking the samples were a stove (Atlas, Instruterm, São Paulo, SP, Brazil), liquefied gas (Supergasbras, model P13, Betim, MG, Brazil), an infrared digital thermometer (Instrutherm, TI 860, São Paulo, SP, Brazil), garlic (Oishii, São Paulo, SP, Brazil), kitchen salt (Cisne, Cabo Frio, RJ, Brazil), soybean oil (Soya, Rio Grande, RS, Brazil), a handcrafted iron skillet and a wooden spoon, bought at local grocery store.
For iron in vitro bioacessibility evaluation the following materials were used: a thermostatic bath (SOLAB, model Dubnoff SL-157/22, Piracicaba, SP, Brazil), centrifuge (Hettich Technology Universal model 320, Tuttlingen, BH, Germany), a 10 mL disposable syringe (Descarpack Slip, Joinville, SC, Brazil), pH indicator paper (J. Prolab, São Paulo, SP, Brazil), a 0.45 µm, surface modified PTFE filter (Millex LCR, Darmstadt, HE, Germany), and the following solutions: Reagent grade HCl 38%, NaHCO3 (Isofar, Duque de Caxias, RJ, Brazil), NaOH (Vetec, São Paulo, SP, Brazil), Pepsin from porcine gastric mucosa (Sigma Aldrich, St. Louis, MO, EUA), Pancreatin from porcine pancreas (Sigma Aldrich, St. Louis. A Thermo Scientific atomic absorption spectrometer, model SOLAAR M5 (Thermo Scientific, China) equipped with deuterium background correction were used for all iron determinations. An iron hollow cathode lamp was used as the radiation source (248.3 nm) with 10 mA of electric current. Other instrumental conditions were a bandpass of 0.2 nm, burner height of 7.0 cm and a gas mixture of air/C2H2 (C2H2 rate: 1.2 L min-1).
2.2 Acquisition and cooking of samples
Meat samples acquired in this work were chicken breast and chicken liver 1,000 g of chicken breast were acquired at two different local markets (Chicken breast A and B), totaling 2,000 g. Chicken breast A was used for the 32 factorial design and Chicken Breast B for evaluation of the bioaccessibility method. In this specific case, skin, visible fat and bones were manually removed with a stainless steel knife, and then the samples were cut into small pieces. Furthermore, an amount of 1,000 g of chicken livers were purchased at a local market and cut into small pieces before the cooking procedure.
In order to evaluate the bioaccessibility of iron in chicken meat, we proceeded with a domestic cooking simulation procedure in the laboratory under controlled conditions. For this purpose, approximately 200 g (randomly selected) of each type of meat sample was spiced with about 4 g of kitchen salt and 3 g of crushed garlic. Then, approximately 10 g of soybean oil was added to the iron pot, which was heated on a domestic stove up to 250 °C. This temperature was measured with an infrared digital thermometer. After that, each spiced sample was separately added to the iron pot and cooked for about 15 minutes, with two tap water additions, totaling a volume of 140 mL. After this step, the cooked samples were prepared for analysis. Residual liquids and cooked samples were transferred to a domestic blender and crushed. The Fe content were also measured in the blank solution containing both the water and the spices employed to the cook procedure and used to determine background Fe concentration The cooked samples were dried in a vacuum oven at (70 ± 1) °C for 72 h, milled again and then placed in plastic flasks that were stored at room temperature (± 25 °C).
Between cooking of the different sample types, the iron pot was washed with neutral soap, tap water and dried at room temperature. Abrasive products were avoided.
2.3 32 Factorial design
Since the studies in the literature does not provide a standard method for determining the factors that significantly affect iron bioaccessibility in chicken meat, especially in terms of pepsin concentrations in gastric stage of the digestion, in this work we evaluated how the variables “sample mass” and “pepsin concentration” affect the iron bioaccessibility fractions. In this context, the concentration of pancreatin and bile salt solutions were fixed at (2.5 and 0.4 w/v%, respectively) as reported by several works (Miller et al., 1981Miller, D. D., Schricker, B. R., Rasmussen, R. R., & Van Campen, D. (1981). An in vitro method for estimation of iron availability from meals. The American Journal of Clinical Nutrition, 34(10), 2248-2256.; Cámara et al, 2005Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009.
http://dx.doi.org/10.1016/j.foodchem.200...
; Hur et al., 2011Hur, S. J., Lim, B. O., Decker, E. A., & McClements, D. J. (2011). In vitro human digestion models for food applications. Food Chemistry, 125(1), 1-12. https://doi.org/10.1016/j.foodchem.2010.08.036.
https://doi.org/10.1016/j.foodchem.2010....
) and the sample mass (X1) and pepsin concentration (X2) were evaluated at three different levels, using a 32 factorial design (View item 1 in Supplementary Material for detailed information).
11 random experiments were conducted, in triplicate at the central point, to evaluate experimental error and the statistical significance of the variables. The assessed levels for pepsin concentrations were based on the method developed by Miller et al., 1981 (central point), the most cited work on the subject, and other papers that used 6% w/v (Cámara et al., 2005Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009.
http://dx.doi.org/10.1016/j.foodchem.200...
; Kulkarni et al., 2007Kulkarni, S. D., Acharya, R., Rajurkar, N. S., & Reddy, A. V. R. (2007). Evaluation of bioaccessibility of some essential elements from wheatgrass (Triticum aestivum L.) by in vitro digestion method. Food Chemistry, 103(2), 681-688. http://dx.doi.org/10.1016/j.foodchem.2006.07.057.
http://dx.doi.org/10.1016/j.foodchem.200...
) and 26% w/v concentrations (Purchas et al., 2006Purchas, R. W., Busboom, J. R., & Wilkinson, B. H. P. (2006). Changes in the forms of iron and in concentrations of taurine, carnosine, coenzyme Q(10), and creatine in beef longissimus muscle with cooking and simulated stomach and duodenal digestion. Meat Science, 74(3), 443-449. http://dx.doi.org/10.1016/j.meatsci.2006.03.015.
http://dx.doi.org/10.1016/j.meatsci.2006...
) low (-1) and high (+1) levels, respectively.
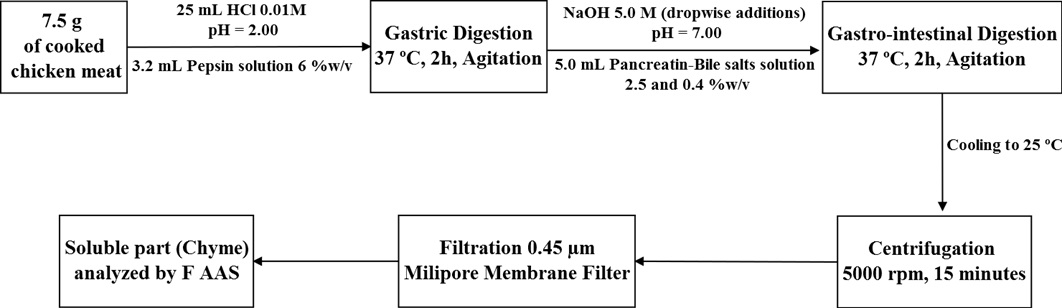
Chicken breast sample (A) subjected to same home cooking procedure as described above was used for multivariate analysis. The samples (2.5; 5.0 and 7.5 g) were weighed and mixed with 25.00 mL of 0.01 M HCl solution. The pH was adjusted to 2.0 by drop wise additions of 1.0M HCl and 3.2 mL of pepsin (in 0.1M HCl) solution, at different concentrations (6, 16 and 26% w/v). This step was used to simulate of the gastric stage of human digestion. These mixtures were then incubated in a thermostatic bath with mild agitation (level 5) at 37 °C for 2 hours.
After this time, the pH of the solutions were adjusted to 7.0 by drop wise additions of NaOH 5.0 M solution and 5.0 mL of pancreatin and bile salts (in 0.1M NaHCO3) solution. This second step constituted the gastro-intestinal digestion simulation. These solutions were incubated again for more 2 hours, being submitted to the same heating and agitation conditions described before.
At the end of the simulated digestion, the obtained digests were cooled to room temperature to reduce the enzymatic action and were then centrifuged at 5000 rpm for 15 minutes. After centrifugation, the soluble (chyme) and insoluble (pellet) digested fractions were separated. Next, the soluble portion was filtered through a 0.45 µm Millipore® membrane filter and stored in decontaminated plastic flasks, under refrigeration (0-4 ºC) for 12 h, until analysis by FAAS.
In this case, the spectrometer calibration was performed between 0.50-4.0 mg L-1 using aqueous iron solutions in 1 % v/v HNO3. To evaluate the fit of the model curve, ANOVA tests were performed. Microsoft Office® Excel 2010 software was used for the statistical analysis while Origin Pro® 8.0 was used for plotting the curve.
2.4 Total iron determination
In order to determine total iron levels in the chicken meat samples, approximately 250 mg of dried breast or liver samples were mixed with 6.00 mL of 7.0 M HNO3 solution and left for 30 minutes in predigestion. Then, the acidified samples were digested in a microwave oven under the following heating conditions: 200 ºC, ramp 15 min, hold time 30 min (Power = 1200 W). After cooling, the samples were transferred to 25.00 mL volumetric flasks and the volume was made up with deionized water. This procedure was also applied to a reference material (NIST 1546-Meat Homogenate) to evaluate the accuracy of the procedure.
2.5 Figures of merit
The accuracy of the developed method was evaluated by analysis of a reference material (NIST 1546-Meat Homogenate). Precision was assessed by monitoring the residual standard deviation (RSD) and the limits of detection (LOD) and quantification (LOQ) were also calculated according to IUPAC recommendations (Harris, 2005Harris, D. (2005). Análise química quantitativa (6. ed.). Rio de Janeiro: LTC.). To provide a measure for the sensitivity of the analyte under selected conditions the characteristic concentration approach was calculated (Welz, 1999Welz, B. (1999). Atomic absorption spectrometry (3rd ed.). New York: Wiley-VCH.). Linear work range and linearity were also determined using parametric statistical tools.
3 Results and discussions
3.1 Iron determination: statistics of the analytical curve
The equation of the best fit linear regression curve was y = 0.03483(±0.0003)Fe + 0.0048(±0.0008) using the standard linear least squares method. Statistical assumptions, within 95% confidence interval such as, lack of fit and residues normality were evaluated. ANOVA was applied to verify the fit model of the Fe calibration curve. If the model is well adjusted, the quadratic average will reflect just the random errors (Harris, 2005Harris, D. (2005). Análise química quantitativa (6. ed.). Rio de Janeiro: LTC.; Barros et al., 2007Barros, B. No, Scarmino, I. S., & Bruns, R. E. (2007). Como fazer experimentos (3. ed.). Campinas: Editora Unicamp.). (View item 2 in Supplementary Material for detailed information).
3.2 Samples analysis: determination of total Fe concentration
Analytical data for both cooked chicken meat and NIST 1546 reference material (Meat Homogenate- a mixture of both chicken and pork meat) are given in Table 1. Chicken liver presents higher total iron levels than the samples of chicken breast and these experimental values are in agreement with those reported elsewhere (Franco, 1992Franco, G. (1992). Tabela de composição química dos alimentos. (9. ed.). São Paulo: Editora Atheneu.).
Total Fe concentrations in chicken meat samples and reference material obtained after microwave acid digestion (N=3).
The total concentrations were obtained from three independent experiments and the quoted uncertainties correspond to a 95 % confidence interval. The good agreement between the determined and certified values for iron concentrations indicates that the method presents adequate accuracy, with an analytical error less than 5 %.
The instrumental and method LOD and LOQ, the fit of the analytical curve and the characteristic concentrations for the analyte were also evaluated. The method LOD and LOQ were equal to 1.2 and 3.5 mg kg-1, respectively, and were smaller than those obtained by Souza et al. (2013)Souza, S. N. P., Nascentes, C. C., & Costa, L. M. (2013). Validation of a microwave-assisted digestion procedure of pate samples using diluted HNO3 for Fe and Zn determination by FS FAAS. Analytical Methods, 5(22), 6411-6415. http://dx.doi.org/10.1039/c3ay41343h.
http://dx.doi.org/10.1039/c3ay41343h...
, that employed diluted nitric acid solution (3.5 M) for the determination of Fe in pâté samples.
The characteristic concentration obtained for the acid digestion method, 0.12 mg L-1, is slightly higher than a reference value (W) found for this element (0.09 mg L-1). On the other hand, all the measurements presented an adequate RSD, with values below 5 % in most cases.
3.3 32 Factorial design
Table 2 shows the codified matrix containing the factors, experimental levels and the response data obtained for the iron bioaccessible fractions, for each experiment related to the 32 factorial design employed.
Optimization of the Fe bioaccessible fractions in cooked chicken meat breast: variables and their levels according to a 32 factorial design.
The iron bioaccessible fraction was obtained from Equation 1 and was calculated based on the iron concentration that was extracted from cooked chicken meat samples via the in vitro digestion method. Thus, the soluble Fe content represents the concentration of Fe present in the chyme, while the total content represents the Fe concentration obtained in the samples after the acid digestion.
In a factorial design, it is assumed that the experimental variables and their interactions influence the obtained response. The data were treated based upon matrix operations using the matrix coefficients coded X, and the response, Fe bioaccessible content (mg kg-1), as the Y vector (See item 1 in Supplementary Material for detailed information). The equation of the response surface for the Fe bioaccessible content as a function of the selected variables is described by Equation 2, where m is the sample mass (g; X1) and P is the pepsin concentration (% w/v; X2).
The fit model was evaluated by ANOVA and no evidence of lack of fit at 95% confidence interval was observed. (See item 1 in Supplementary Material for detailed information). Thus, this model can be used to predict the optimum factors for the determination of the Fe bioaccessible fraction in chicken meat, taking into account the surface response study (Figure 1).
Surface response for Fe bioaccessible content as a function of sample mass and pepsin concentration.
This optimization aims to simulate, in vitro, human digestion which is a complex biological system. As such, the maximal analytical response for iron was not of primary concern. Our principal objective was to discover conditions that reproduce this complex process in terms of analyte availability using concentrations of the enzymes encountered physiologically in the gastrointestinal medium.
The best conditions were selected by comparing the experimental results with iron bioavailability data reported in the literature, the range between 20-30 % (Grotto, 2008Grotto, H. Z. W. (2008). Metabolismo do ferro: uma revisão sobre os principais mecanismos envolvidos em sua homeostase. Revista Brasileira de Hematologia e Hemoterapia, 30(5), 390-397. http://dx.doi.org/10.1590/S1516-84842008000500012.
http://dx.doi.org/10.1590/S1516-84842008...
), which is in agreement with the results provided by some of our experimental conditions. Therefore, combining this information with analysis costs and considering the fact that the cost of pepsin is not negligible, the conditions of Experiment 3 (X1 = 7.5 g and X2 = 6 % w/v) were selected as the best to determine the iron bioaccessible fractions in cooked chicken breast. The pepsin concentration employed in this experiment is compatible with those utilized in others works that simulate human digestion (Cámara et al., 2005Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009.
http://dx.doi.org/10.1016/j.foodchem.200...
; Kulkarni et al., 2007Kulkarni, S. D., Acharya, R., Rajurkar, N. S., & Reddy, A. V. R. (2007). Evaluation of bioaccessibility of some essential elements from wheatgrass (Triticum aestivum L.) by in vitro digestion method. Food Chemistry, 103(2), 681-688. http://dx.doi.org/10.1016/j.foodchem.2006.07.057.
http://dx.doi.org/10.1016/j.foodchem.200...
).
The selected conditions were applied to determine the bioaccessible Fe content in other cooked chicken meat samples. Chicken Breast is one of the most consumed poultry products in Brazil. Chicken liver, also used for preparation of other products such as pâté and sausages also is of significant importance (Bressan et al., 2004Bressan, M. C., Vieira, J. O., Faria, P. B., Vicente, J. No, Andrade, P. L., & Figueiredo, E. E. S. (2004). História,aspectos econômicos, obtenção e ciência da carne. Lavras: Fundação de Apoio ao Ensino, Pesquisa e Extensão – FAEPE.). The optimized in vitro method to evaluate Fe bioaccessibility in chicken breast is depicted in Figure 2.
Schematic representation of optimized procedure for in vitro determination of Fe bioaccessibility in chicken meat.
In the absence of a CRM (Certified Reference Material) to evaluate the accuracy of bioaccessible methods and considering that chicken meat has a complex matrix, the standard additions method was applied for calibration, which was made in triplicate. Characteristic concentration was calculated to estimate the sensitivity. LOD and LOQ values were also determined. Table 3 shows the figures of merit evaluated for determination of the Fe bioaccessible fraction. A good linearity (R2Chicken Liver = 0.9991 and R2Chicken Breast B = 0.9976) was observed along the working range used for both chicken meat samples.
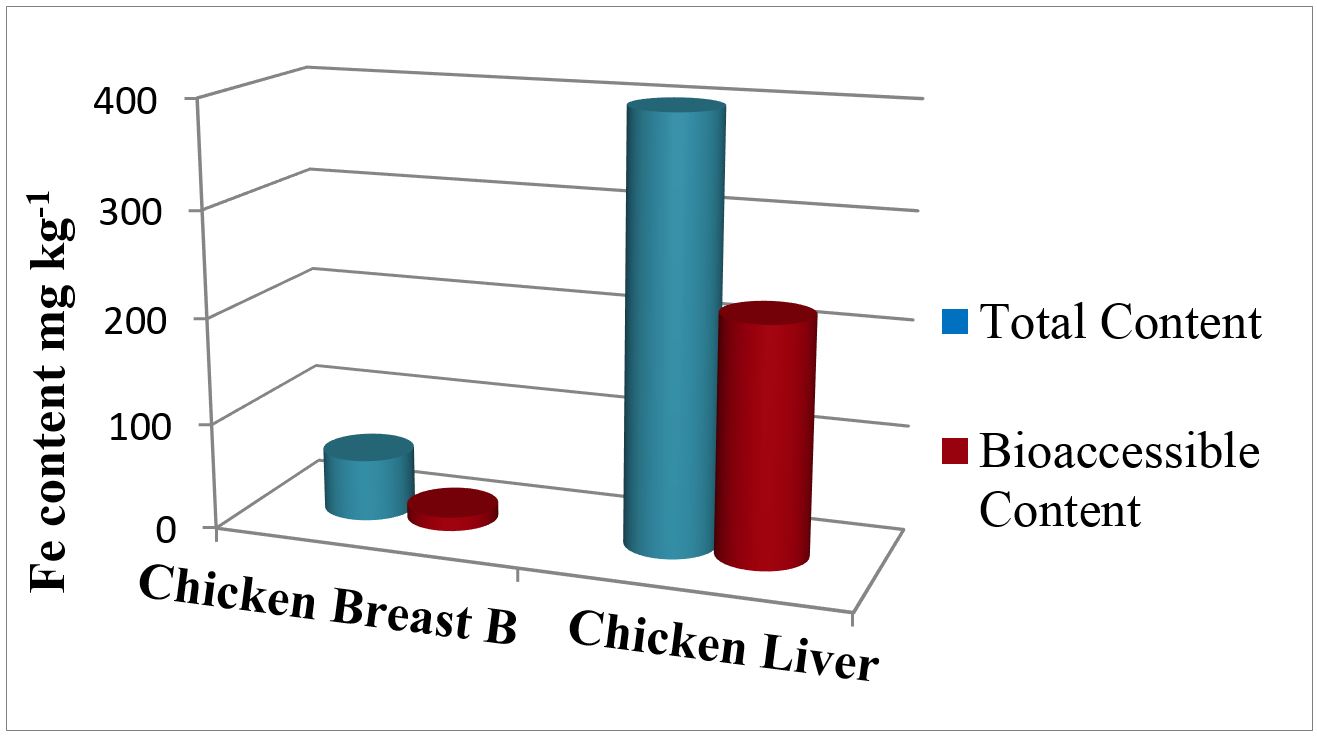
The values obtained for LOD, LOQ and characteristic mass for the Fe bioaccessible fractions were acceptable but higher than those obtained in the acid digestion method. This can be related to the chyme composition, which is a medium with a higher content of dissolved solids, present as organic material, inorganic salts and enzymes. This high content of dissolved solids may have an effect on the sample aspiration and analysis (Welz, 1999Welz, B. (1999). Atomic absorption spectrometry (3rd ed.). New York: Wiley-VCH.). In any case, it was possible to quantify the bioaccessible iron content in the samples as the concentrations measured were higher than the method LOQ. For Chicken Breast B, the total and bioaccessible iron levels were (58.4 ± 2.6) mg kg-1 and (13.5 ± 3.6) mg kg-1, respectively, while for liver these values were equal to (400 ± 10) mg kg-1 and (223 ± 18) mg kg-1, respectively.
The bioaccessible fractions of Fe obtained after an in vitro digestion of chicken breast and liver samples were equal to 23 and 56 %, respectively. The value found for chicken breast is compatible with results obtained in academic research done by Menezes (2010)Menezes, E. A. (2010). Determinação da disponibilidade de Ca, Cu, Fe, Mg e Zn em amostras de carnes bovinas, suínas e de frango in natura e processadas termicamente (Master’s thesis). Universidade Federal de São Carlos, São Carlos. for this kind of meat, approximately 20 %. For chicken liver, however, there are no studies which report determinations for this type of meat. Furthermore, the results obtained in this work indicate that iron found in the chicken liver sample is more available in the gastrointestinal tract than the amount present in chicken breast. In other words, these data indicate that chicken liver may be a much better source of this micronutrient, taking into account not only the total concentration but most importantly, its bioaccessible fraction. Figure 3 shows the comparison between total and bioaccessible fractions of Fe found in the samples analyzed in this study.
4 Conclusions
In this work, a procedure for the determination of iron bioaccessible fractions in chicken meat was developed and applied to two types of samples, breast and liver, which were cooked in an iron skillet as is customarily done in domestic cooking. Using a multivariate approach, it was established which variables affect, in vitro, the bioaccessible content of this element. These variables were sample mass in combination with pepsin concentrations. As a result, a robust and accurate method for the determination of iron concentrations was developed, which can be carried out directly on the chyme solutions. Moreover, through the proposed method, it was possible to optimize the mass/pepsin ratio that can be used for in vitro tests, resulting in an economy of the enzyme used to obtain a satisfactory analyte extraction. Comparing the results obtained for the different tissues, the chicken liver sample showed the highest level of total and bioaccessible iron compared to the chicken breast samples. This would confirm that liver is, in fact, a good source of iron.
Supplementary Material
Supplementary Material accompanies this paper.
1. 3² Factorial Design
2. Fe calibration curve
This material is available as part of the online article from http://www.scielo.br/cta
Acknowledgements
The authors would like to thank CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), FAPEMIG (Fundação de Amparo à Pesquisa do Estado de Minas Gerais), and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior). This work was also a collaboration research project of members from the Rede Mineira de Química (RQ-MG) supported by FAPEMIG (Project: CEX - RED-00010-14). The authors also thank Michelle Nascimento for assistance with the cooking procedure at the Dietetics laboratory and Dr. Richard Michael Grazul for his carefully language revision.
-
Practical Application: The method allows Fe bioaccessible determination in chicken with lower cost proper to routine.
References
- Associação Brasileira de Proteína Animal – ABPA. (2015). Relatório Anual. São Paulo: ABPA. Retrieved from http://abpa-br.com.br/files/publicacoes/c59411a243d6dab1da8e605be58348ac.pdf
» http://abpa-br.com.br/files/publicacoes/c59411a243d6dab1da8e605be58348ac.pdf - Azevedo, F. A., & Chasin, A. A. M. (2003). Metais: gerenciamento da toxicidade. São Paulo: Editora Atheneu.
- Barros, B. No, Scarmino, I. S., & Bruns, R. E. (2007). Como fazer experimentos (3. ed.). Campinas: Editora Unicamp.
- Brasil, Agência Nacional de Vigilância Sanitária. (2005, September 23). Resolução RDC n.º 269, de 22 de setembro de 2005. Aprova o “Regulamento Técnico sobre a Ingestão Diária Recomendada (IDR) de Proteína, Vitaminas e Minerais”. Diário Oficial [da] República Federativa do Brasil.
- Bressan, M. C., Vieira, J. O., Faria, P. B., Vicente, J. No, Andrade, P. L., & Figueiredo, E. E. S. (2004). História,aspectos econômicos, obtenção e ciência da carne. Lavras: Fundação de Apoio ao Ensino, Pesquisa e Extensão – FAEPE.
- Cámara, F., Amaro, M. A., Barberá, R., & Clemente, G. (2005). Bioaccessibility of minerals in school meals: Comparison between dialysis and solubility methods. Food Chemistry, 92(3), 481-489. http://dx.doi.org/10.1016/j.foodchem.2004.08.009
» http://dx.doi.org/10.1016/j.foodchem.2004.08.009 - Franco, G. (1992). Tabela de composição química dos alimentos. (9. ed.). São Paulo: Editora Atheneu.
- Grotto, H. Z. W. (2008). Metabolismo do ferro: uma revisão sobre os principais mecanismos envolvidos em sua homeostase. Revista Brasileira de Hematologia e Hemoterapia, 30(5), 390-397. http://dx.doi.org/10.1590/S1516-84842008000500012
» http://dx.doi.org/10.1590/S1516-84842008000500012 - Harris, D. (2005). Análise química quantitativa (6. ed.). Rio de Janeiro: LTC.
- Hur, S. J., Lim, B. O., Decker, E. A., & McClements, D. J. (2011). In vitro human digestion models for food applications. Food Chemistry, 125(1), 1-12. https://doi.org/10.1016/j.foodchem.2010.08.036
» https://doi.org/10.1016/j.foodchem.2010.08.036 - Koch, I., Moriarty, M., House, K., Sui, J. & Cullen, W. R. Saper R. B, Reimer K. J. (2011). Bioaccessibility of lead and arsenic in traditional Indian medicines. Science of the total environment, 409(21), 4545-4552. http://doi.org/10.1016/j.scitotenv.2011.07.059 PMid:21864885
» http://doi.org/10.1016/j.scitotenv.2011.07.059 - Kulkarni, S. D., Acharya, R., Rajurkar, N. S., & Reddy, A. V. R. (2007). Evaluation of bioaccessibility of some essential elements from wheatgrass (Triticum aestivum L.) by in vitro digestion method. Food Chemistry, 103(2), 681-688. http://dx.doi.org/10.1016/j.foodchem.2006.07.057
» http://dx.doi.org/10.1016/j.foodchem.2006.07.057 - López, M. A. A., & Martos, F. C. (2004). Iron availability: an updated review. International Journal of Food Sciences and Nutrition, 55(8), 597-606. http://dx.doi.org/10.1080/09637480500085820
» http://dx.doi.org/10.1080/09637480500085820 - Luten, J., Crews, H., Flynn, A., Van Dael, P., Kastenmayer, P., Hurrell, R., Deelstra, H., Shen, L.-H., Fairweather-Tait, S., Hickson, K., Farré, R., Schlemmer, U., & Frøhlich, W. (1996). Interlaboratory trial on the determination of the in vitro iron dialysability from food. Journal of the Science of Food and Agriculture, 72(4), 415-424. http://dx.doi.org/10.1002/(SICI)1097-0010(199612)72:4<415::AID-JSFA675>3.0.CO;2-X
» http://dx.doi.org/10.1002/(SICI)1097-0010(199612)72:4<415::AID-JSFA675>3.0.CO;2-X - Maulvault, A. L., Machado, R., Afonso, C., Lourenço, H. M., Nunes, M. L., Coelho, I., Langerholc, T., & Marques, A. (2011). Bioaccessibility of Hg, Cd and As in cooked black scabbard fish and edible crab. Food and Chemical Toxicology, 49(11), 2808-2815. http://dx.doi.org/10.1016/j.fct.2011.07.059
» http://dx.doi.org/10.1016/j.fct.2011.07.059 - Menezes, E. A. (2010). Determinação da disponibilidade de Ca, Cu, Fe, Mg e Zn em amostras de carnes bovinas, suínas e de frango in natura e processadas termicamente (Master’s thesis). Universidade Federal de São Carlos, São Carlos.
- Miller, D. D., Schricker, B. R., Rasmussen, R. R., & Van Campen, D. (1981). An in vitro method for estimation of iron availability from meals. The American Journal of Clinical Nutrition, 34(10), 2248-2256.
- Minekus, M., Alminger, M., Alvito, P., Ballance, S., Bohn, T., Bourlieu, C., Carrière, F., Boutrou, R., Corredig, M., Dupont, D., Dufour, C., Egger, L., Golding, M., Karakaya, S., Kirkhus, B., Le Feunteun, S., Lesmes, U., Macierzanka, A., Mackie, A., Marze, S., McClements, D. J., Ménard, O., Recio, I., Santos, C. N., Singh, R. P., Vegarud, G. E., Wickham, M. S. J., Weitschies, W., & Brodkorb, A. (2014). A standardised static in vitro digestion method suitable for food - an international consensus. Food & Function, 5(6), 1113-1124. http://dx.doi.org/10.1039/c3fo60702j
» http://dx.doi.org/10.1039/c3fo60702j - Nascimento, A. N., Naozuka, J., & Oliveira, P. V. (2010). In vitro evaluation of Cu and Fe bioavailability in cashew nuts by off-line coupled SEC–UV and SIMAAS. Microchemical Journal, 96(1), 58-63. http://dx.doi.org/10.1016/j.microc.2010.01.016
» http://dx.doi.org/10.1016/j.microc.2010.01.016 - Olivio, N. (2008). Mercado Mundial de Carnes. Criciúma: Editora Varela.
- Peixoto, R. R. A., Mazon, E. A. M., & Cadore, S. (2013). Estimation of the bioaccessibility of metallic elements in chocolate drink powder using an in vitro digestion method and spectrometric techniques. Journal of the Brazilian Chemical Society, 24, 884-890. http://dx.doi.org/10.5935/0103-5053.20130111
» http://dx.doi.org/10.5935/0103-5053.20130111 - Pereira, C. C., Silva, E. N., Souza, A. O., Vieira, M. A., Ribeiro, A. S., & Cadore, S. (2016). Evaluation of the bioaccessibility of minerals from blackberries, raspberries, blueberries and strawberries. Journal of Food Composition and Analysis. In press. http://dx.doi.org/10.1016/j.jfca.2016.12.001
» http://dx.doi.org/10.1016/j.jfca.2016.12.001 - Purchas, R. W., Busboom, J. R., & Wilkinson, B. H. P. (2006). Changes in the forms of iron and in concentrations of taurine, carnosine, coenzyme Q(10), and creatine in beef longissimus muscle with cooking and simulated stomach and duodenal digestion. Meat Science, 74(3), 443-449. http://dx.doi.org/10.1016/j.meatsci.2006.03.015
» http://dx.doi.org/10.1016/j.meatsci.2006.03.015 - Ramos, A., Cabrera, M. C., & Saadoun, A. (2012). Bioaccessibility of Se, Cu, Zn, Mn and Fe, and heme iron content in unaged and aged meat of Hereford and Braford steers fed pasture. Meat Science, 91(2), 116-124. http://dx.doi.org/10.1016/j.meatsci.2012.01.001
» http://dx.doi.org/10.1016/j.meatsci.2012.01.001 - Silva, E. N., Heerdt, G., Cidade, M., Pereira, C. D., Morgon, N. H., & Cadore, S. (2015). Use of in vitro digestion method and theoretical calculations to evaluate the bioaccessibility of Al, Cd, Fe and Zn in lettuce and cole by inductively coupled plasma mass spectrometry. Microchemical Journal, 119, 152-158. http://dx.doi.org/10.1016/j.microc.2014.12.002
» http://dx.doi.org/10.1016/j.microc.2014.12.002 - Souza, S. N. P., Nascentes, C. C., & Costa, L. M. (2013). Validation of a microwave-assisted digestion procedure of pate samples using diluted HNO3 for Fe and Zn determination by FS FAAS. Analytical Methods, 5(22), 6411-6415. http://dx.doi.org/10.1039/c3ay41343h
» http://dx.doi.org/10.1039/c3ay41343h - Stelmach, E., Pohl, P., & Szymczycha-Madeja, A. (2014). Evaluation of the bioaccessability of Ca, Fe, Mg and Mn in ground coffee infusions by in vitro gastrointestinal digestion. Journal of the Brazilian Chemical Society, 25, 1993-1999. http://dx.doi.org/10.5935/0103-5053.20140183
» http://dx.doi.org/10.5935/0103-5053.20140183 - Welz, B. (1999). Atomic absorption spectrometry (3rd ed.). New York: Wiley-VCH.
Publication Dates
-
Publication in this collection
12 June 2017 -
Date of issue
Jan-Mar 2018
History
-
Received
27 Jan 2017 -
Accepted
30 Apr 2017