Abstracts
A parasitological study was undertaken to determine the health status of 15 mouse and 10 rat colonies bred in 18 Brazilian laboratory animal houses maintained under different sanitary barrier conditions which supply animals for teaching, research purposes and manufacture of biological products for medical or veterinary use. Parasitological methods were used for diagnosis of mites, lices, helminthes and protozoan parasites. A questionnaire was answered by institutions with the intention to obtain information about the existence of barriers against infections and of regular sanitary monitoring program of their colonies. The questionnaire data show that the majority of the animal houses investigated do not possess an efficient sanitary barrier system able to keep animals under controlled health sanitary conditions. Ecto and endoparasite infections are widespread in the colonies and multiple infections were common in animals from most facilities investigated. The prevalences of parasites detected among the mouse and rat colonies of the laboratory animal houses investigated were: Myocoptes musculinus (46.6%), Myobia musculi (26.6%), Radfordia ensifera (13.3%), Syphacia obvelata (86.6%), Aspiculuris tetraptera (60.0%), Hymenolepis nana (53.3%), Spironucleus muris (80.0%), Tritrichomonas muris (80.0%), Giardia muris (66.0%), Entamoeba muris (20.0%), Eimeria sp. (13.3%), Hexamastix muris (26.6%), Poliplax spinulosa (30.0%), Poliplax serrata (10.0%), Radfordia ensifera (30.0%), Syphacia muris (80.0%), Hymenolepis nana (40.0%), Trichosomoides crassicauda (55.5%), Spironucleus muris (90.0%), Tritrichomonas muris (80.0%), Giardia muris (60.0%), Entamoeba muris (80.0%), Eimeria sp. (60.0%) and Hexamastix muris (60.0%).
Rat; mouse; Brazilian laboratory animal houses; parasite; sanitary barrier
Um estudo parasitológico foi realizado para verificar as condições de saúde de 15 colônias de camundongos e 10 colônias de ratos produzidos em 18 biotérios de instituições brasileiras que fornecem animais para ensino, pesquisa e produção de imunobiológicos de uso médico ou veterinário. Métodos parasitológicos foram utilizados para diagnóstico de ácaros, piolhos, helmintos e protozoários parasitos. Um questionário foi respondido pelas instituições com o intuito de obter informações sobre a existência de barreiras contra infecções e programa de fiscalização sanitária de suas colônias. Os dados do questionário mostram que a maioria dos biotérios analisados não possui um sistema de barreiras sanitárias eficiente capaz de manter animais sob condições sanitárias controladas. Infecções por ecto e endoparasitos são generalizadas nas colônias e a associação de infecções múltiplas foi comum na maioria dos animais dos biotérios analisados. A prevalência dos parasitos detectados entre as colônias de camundongos e de ratos investigadas foi: Myocoptes musculinus (46,6%), Myobia musculi (26,6%), Radfordia ensifera (13,3%), Syphacia obvelata (86,6%), Aspiculuris tetraptera (60,0%), Hymenolepis nana (53,3%), Spironucleus muris (80,0%), Tritrichomonas muris (80,0%), Giardia muris (66,0%), Entamoeba muris (20,0%), Eimeria sp. (13,3%), Hexamastix muris (26,6%), Poliplax spinulosa (30,0%), Poliplax serrata (10,0%), Radfordia ensifera (30,0%), Syphacia muris (80,0%), Hymenolepis nana (40,0%), Trichosomoides crassicauda (55,5%), Spironucleus muris (90,0%), Tritrichomonas muris (80,0%), Giardia muris (60,0%), Entamoeba muris (80,0%), Eimeria sp. (60,0%) e Hexamastix muris (60,0%).
Camundongo; rato; parasito; biotério brasileiro; barreira sanitária
Parasite survey in mouse and rat colonies of Brazilian laboratory animal houses kept under differents sanitary barrier conditions
(Estudo de parasitos em colônias de ratos e de camundongos em biotérios brasileiros mantidos sob diferentes condições de barreiras sanitárias)
R. Gilioli, L.A.G. Andrade, L.A.C. Passos, F.A. Silva, D.M. Rodrigues, A.M.A. Guaraldo
Universidade de Campinas - Unicamp
Caixa Postal 6095
13083-970 - Campinas, SP
Recebido para publicação, após modificação, em 20 de outubro de 1999.
E-mail: rovilson@obelix.unicamp.br
ABSTRACT
A parasitological study was undertaken to determine the health status of 15 mouse and 10 rat colonies bred in 18 Brazilian laboratory animal houses maintained under different sanitary barrier conditions which supply animals for teaching, research purposes and manufacture of biological products for medical or veterinary use. Parasitological methods were used for diagnosis of mites, lices, helminthes and protozoan parasites. A questionnaire was answered by institutions with the intention to obtain information about the existence of barriers against infections and of regular sanitary monitoring program of their colonies. The questionnaire data show that the majority of the animal houses investigated do not possess an efficient sanitary barrier system able to keep animals under controlled health sanitary conditions. Ecto and endoparasite infections are widespread in the colonies and multiple infections were common in animals from most facilities investigated. The prevalences of parasites detected among the mouse and rat colonies of the laboratory animal houses investigated were: Myocoptes musculinus (46.6%), Myobia musculi (26.6%), Radfordia ensifera (13.3%), Syphacia obvelata (86.6%), Aspiculuris tetraptera (60.0%), Hymenolepis nana (53.3%), Spironucleus muris (80.0%), Tritrichomonas muris (80.0%), Giardia muris (66.0%), Entamoeba muris (20.0%), Eimeria sp. (13.3%), Hexamastix muris (26.6%), Poliplax spinulosa (30.0%), Poliplax serrata (10.0%), Radfordia ensifera (30.0%), Syphacia muris (80.0%), Hymenolepis nana (40.0%), Trichosomoides crassicauda (55.5%), Spironucleus muris (90.0%), Tritrichomonas muris (80.0%), Giardia muris (60.0%), Entamoeba muris (80.0%), Eimeria sp. (60.0%) and Hexamastix muris (60.0%).
Keywords: Rat, mouse, Brazilian laboratory animal houses, parasite, sanitary barrier.
RESUMO
Um estudo parasitológico foi realizado para verificar as condições de saúde de 15 colônias de camundongos e 10 colônias de ratos produzidos em 18 biotérios de instituições brasileiras que fornecem animais para ensino, pesquisa e produção de imunobiológicos de uso médico ou veterinário. Métodos parasitológicos foram utilizados para diagnóstico de ácaros, piolhos, helmintos e protozoários parasitos. Um questionário foi respondido pelas instituições com o intuito de obter informações sobre a existência de barreiras contra infecções e programa de fiscalização sanitária de suas colônias. Os dados do questionário mostram que a maioria dos biotérios analisados não possui um sistema de barreiras sanitárias eficiente capaz de manter animais sob condições sanitárias controladas. Infecções por ecto e endoparasitos são generalizadas nas colônias e a associação de infecções múltiplas foi comum na maioria dos animais dos biotérios analisados. A prevalência dos parasitos detectados entre as colônias de camundongos e de ratos investigadas foi: Myocoptes musculinus (46,6%), Myobia musculi (26,6%), Radfordia ensifera (13,3%), Syphacia obvelata (86,6%), Aspiculuris tetraptera (60,0%), Hymenolepis nana (53,3%), Spironucleus muris (80,0%), Tritrichomonas muris (80,0%), Giardia muris (66,0%), Entamoeba muris (20,0%), Eimeria sp. (13,3%), Hexamastix muris (26,6%), Poliplax spinulosa (30,0%), Poliplax serrata (10,0%), Radfordia ensifera (30,0%), Syphacia muris (80,0%), Hymenolepis nana (40,0%), Trichosomoides crassicauda (55,5%), Spironucleus muris (90,0%), Tritrichomonas muris (80,0%), Giardia muris (60,0%), Entamoeba muris (80,0%), Eimeria sp. (60,0%) e Hexamastix muris (60,0%).
Palavras-chave: Camundongo, rato, parasito, biotério brasileiro, barreira sanitária.
INTRODUCTION
Biomedical research still depends on the use of living laboratory animal models. Mice and rats are the most common laboratory animals used in research and testing. Experimental results from research performed with living laboratory animals may be affected by environmental conditions provided to breeding and maintaining of these animals and by infectious agents (Clough, 1982; Pakes et al., 1984; Homberger & Thomann, 1994). Despite the presently intensive use of isolated cells and molecules and in vitro methods such as tissue cell culture, recombinant DNA and monoclonal antibodies, biomedical research still depends to a large extent on data obtained from experiments performed with living laboratory animal models. As a consequence, sensitivity and accuracy of results may be compromised when infectious agents are present in the colonies. Thus, infected animals are not suitable for cancer and disease research or helminthes biology experiments (Baird et al., 1982; Ito, 1982). The use of laboratory animals bred under barrier-maintained system, controlled environment conditions and submitted to periodical genetic and sanitary monitoring is of utmost importance (Vessel et al., 1976; Melby & Balk, 1983).
Besides the high mortality in young animals caused by some infections, parasites can complicate research by inducing physiological and immunological alterations in the hosts, increasing or diminishing host susceptibility to experimental stress, inducing tissue damages, stimulating abnormal tissue growth, competing with the host for nutrients, decreasing the volume of hosts blood and body fluids and by mechanical interference (Hsu, 1980). This survey was performed to add new data on the knowledge about parasite infection of mice and rats used in research, bred in several Brazilian laboratory animal houses kept under different sanitary barrier conditions.
MATERIAL AND METHODS
A questionnaire was made to obtain information about the existence of barriers against infections and a regular sanitary monitoring program of mouse and rat colonies bred in different Brazilian institutions located in the states of São Paulo, Rio de Janeiro, Minas Gerais, Paraná, Mato Grosso do Sul, Goiás and Rio Grande do Sul.
Young and adult mice and rats of various inbred and outbred strains, ranging in age from 4 to 32 weeks, were randomly chosen from each breeding area facilities to make up groups of animals. The sample sizes were variable according to the sizes of the colonies and availability of animals to be scrutinized. The animals were examined within 24h after their obtaining and they were submitted to ether anesthesia, bled by cardiac puncture and sacrificed by cervical dislocation before necropsy. Sera obtained were inactivated at 56°C for 30 min and kept at 20°C until their use in serological assays to study possible Toxoplasma gondii and latent bacteria and virus infections.
Mites and lice were diagnosed by direct examination of animal carcasses under dissecting microscope and identified by light microscopy after clarification with Hoyer liquid according to Krantz to (1978).
For helminthes and protozoan diagnostic, slices of the small and large intestine were collected and the lumen contents submitted to direct light microscopy and the parasitological methods described by Willis (1921), Sheather (1923), Hoffman et al. (1934) and Faust et al. (1938) were applied. Feces suspensions were prepared in physiological saline solution and kept warm on a heating plate for preservation of adult worm and protozoan trophozoite motility. When necessary, protozoan cysts were stained by iodine solution to facilitate identification.
The investigation of Toxoplasma gondii infection was made by indirect immunofluorescence assay which determines serum antibody levels.
The rat parasite Trichosomoides crassicauda was investigated in the urinary bladder by egg and worm identification with direct microscopy.
RESULTS AND DISCUSSION
The data concerning sanitary conditions obtained with the questionnaire are shown in Table 1. They showed that most animal facilities maintain colonies under conventional conditions and are installed in buildings which were not planned to support Specific Pathogen Free (SPF) animals, sometimes, with different species in the same room. Although a meaningful number of them use disinfected materials, few animal houses have a regular sanitary monitoring program. Only two animal houses had a complete set of sanitary barriers and one of them had periodical sanitary program to monitor parasite, virus and bacteria infections.
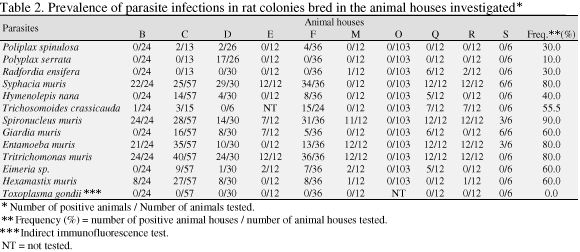
The prevalence and distribution of parasite infections in rat and mouse colonies from every animal house and the parasite frequencies among the animal facilities investigated are shown in Tables 2 and 3, respectively. Parasite infections were present in most of the animal houses investigated and despite the fact that some samples were limited, a high proportion of animals infected with four or more parasite species was observed in a single group of six animals, indicating that the colonies were heavily infected. No positive result was found in the serological tests for Toxoplasma gondii infection, but virus infections were also detected in the majority of these colonies investigated in the past (Gilioli et al., 1996).
As expected, the prevalences of parasite infections were due to the mixing of several species from different breeders in the same room under conventional conditions, the absence of a quarantine program and effective sanitary barrier systems able to keep animals under controlled sanitary conditions (Boot et al., 1996; Rehg & Toth, 1998). The results of the present survey were not too different from those obtained in the past in other countries (Weisbroth & Scher, 1971; Tanaka et al., 1974; Casebolt et al., 1988).
Parasites infect laboratory mice and rats worldwide at high prevalenceand usually highly contagious infection proceeds without symptoms unless heavy infection occurs (Flynn, 1973; Tanaka et al., 1974; Baker, 1998). However, these infections have several adverse effects on biological research, specially altering immune responses, blood parameters, nutritional and radiation studies making some experimental results invalid. Thus, animals infected with parasites are not suitable for cancer and disease research or helminthes biology experiments (Zubaid & Majeed, 1981; Baird, 1982; Ito, 1982).
Some parasites as Hymenolepis nana, frequently found in mouse and rat colonies, are potentially zoonotic agents and the health hazards for technicians can not be discharded (Fox et al., 1984).
Survey of several Brazilian laboratory animal houses in the past showed that no animal facility was able to produce SPF animals (Rosenkranz et al., 1978). Nowadays, at least one animal facility can produce rats and mice free of parasites and viruses (Gilioli et al., 1996). However, it could be concluded that in the present moment most of Brazilian laboratory animal houses are unable to produce or maintain animals under controlled sanitary conditions.
The fact that most of the colonies were found to be infected suggests the need of a continuous and sustained effort to improve the quality of laboratory animals bred in Brazil.
ACKNOWLEDGMENTS
The present work was sponsored by FAPESP, CNPq and KFA. The authors thank Dr. Regina Maura Bueno Franco for the manuscript overhaul.
- BAIRD, S.M., BEATTIE, g.m., LANNOM, R.A. et al. Induction of lymphoma in antigenically stimulated athymic mice. Cancer Res., v.42, p.198-206, 1982.
- BAKER, D.G. Natural pathogens of laboratory mice, rats, and rabbits and their effects on research. Clin. Microbiol. Rev., v.11, p.231-266, 1998.
- BOOT, R., VAN HERCK, H., VAN DER LOGT, J. Mutual viral and bacterial infections after housing rats of various breeders within an experimental unit. Lab. Anim, v.30, p.42-45, 1996.
- CASEBOLT, D.B., Lindsey, J.R., Casell, G.H. Prevalence rates of infectious agents among commercial breeding populations of rats and mice. Lab. Anim. Sci, v.38, p.327-329, 1988.
- CLOUGH, G. Environmental effects on animals used in biomedical research. Biol. Res, v.57, p.487-523, 1982.
- FAUST, E.C., D'ANTONI, J.S., ODOM, V. et al. A critical study of clinical laboratory techinics for the diagnosis of protozoan cysts and helminth eggs in feces. I-Preliminary communication. Am. J. Trop. Med., v.18, p.169-183, 1938.
- FLYNN, R.J. Parasites of laboratory animals Ames: Iowa State University, 1973. 884 p.
- FOX, J.G., NEWCOMER, C.E., ROZMIAREK, H. Selected zoonoses and other health hazards. In: FOX, J.G., COHEN, B.J., LOEW, F.M. (Eds.), Laboratory animal medicine. New York: Academic, 1984. p.613-648.
- GILIOLI, R., SAKURADA, J.K., ANDRADE, L.A.G. et al. Virus infection in rat and mouse colonies reared in Brazilian animal facilities. Lab. Anim. Sci, v.46, p.582-584, 1996.
- HOFFMAN, W.A., PONS, J.A., JANER, J.L. The sedimentation-concentration method in schistosomiasis mansoni. Puerto Rico J. Publ. Health, n.9, p.281-298, 1934.
- HOMBERGER, F.R., THOMANN, P.E. Transmission of murine viruses and mycoplasma in laboratory mouse colonies with respect to housing conditions. Lab. Anim., v.2, p.113-120, 1994.
- HSU, C.K. Parasitic diseases: how to monitor them & their effects on research. Lab. Anim, v.14, p.48-53, 1980.
- ITO, A. Hymenolepis nana: immunogenicity of a lumen phase of the direct cycle and failure to auto-infection in BALB/c mice. Exp. Parasitol, v.54, p.113-120, 1982.
- KRANTZ, G.W. Examination and mounting techniques. In: MANUAL of acarology. 2.ed. Cervallis: Oregon State University, 1978.
- MELBY Jr., E.C., BALK, M.W. (Eds.) The importance of laboratory animal genetics, health and the environment in biomedical research New York: Academic Press, 1983.
- PAKES, S.P., LU, Y.S., MEUNIER, P.C. Factors that complicate animal research. In: Laboratory animal medicine FOX, J.G., COHEN, B.J., LOEW, F.M. (Eds.), New York: Academic, 1984. p.649-665.
- REHG, J.E., TOTH, L.A. Rodent quarantine programs: purpose, principles and practice. Lab. Anim. Sci, v.48, p.438-447, 1998.
- ROSENKRANZ, A., JURKIEWCZ, A., CORRADO, A. Situaçăo dos biotérios brasileiros: fator limitante de estudos farmacodinâmicos e toxicológicos de produtos naturais. Cięnc. Cult., v.32, p.156-163, 1978.
- SHEATHER, L. The detection of intestinal protozoa and mange parasites by flotation technique. J. Comp. Pathol., v.36, p.266-275, 1923.
- TANAKA, H., OHSHIMA, S., FUJINAMI, F. A survey of parasites in commercially available small laboratory mammals. Exp. Anim., v.23, p.15-30, 1974.
- VESSEL, E.S., LANG, C.M., WHITE, W.J. et al. Environmental and genetics factors affecting the response of laboratory animals to drugs. Fed. Proc, v.35, p.1125-1132, 1976.
- WEISBROTH, S.H., SCHER, S. Trichosomoides crassicauda infection of a commercial rat breeding colony. Observations on the life cycle and propagation. Lab. Anim. Sci, v.21, p.54-61, 1971.
- WILLIS, H.H. A simple levitation method for the detection of hookworm ova. Med. J. Aust., v.11, p.375-376, 1921.
- ZUBAID, A.J., MAJEED, S.K. Pathology of the nematode Trichosomoides crassicauda in the urinary bladder of laboratory rats. Lab. Anim. Sci, v.15, p.381-384, 1981.
Publication Dates
-
Publication in this collection
14 Aug 2000 -
Date of issue
Feb 2000
History
-
Received
20 Oct 1999