Abstract
A citopatologia in vitro de uma cepa de rotavírus porcino adaptado em cultura de células foi comparada à estirpe-protótipo símia (SA-11). O efeito citopático (ECP) produzido pelos vírus foi semelhante embora a estirpe porcina tivesse apresentado algumas alterações diferentes, como o acentuado estreitamento do citoplasma, com grande perda do volume citoplasmático. O vírus porcino apresentou menor número de plaques de ECP porém com diâmetro maior em relação ao vírus símio, demonstrando maior capacidade de disseminação célula-célula, quase oito vezes mais, a julgar pelo diâmetro dos plaques de ECP. Os elementos do citoesqueleto das células infectadas revelaram uma reorganização semelhante para ambas as estirpes, não sendo possível observar nenhuma diferença, embora o ECP do vírus porcino tenha sido mais acentuado.
Rotavírus; citopatologia; cultura de células; plaques; citoesqueleto
Rotavírus; citopatologia; cultura de células; plaques; citoesqueleto
Rotavirus; cytopathology; cell culture; plaque; cytoskeleton
COMMUNICATION
[Comunicação]
The in vitro comparative cytopathology of a porcine rotavirus and the simian prototype (SA-11)
[Citopatologia comparativa in vitro entre uma estirpe de rotavírus porcino e o protótipo símio (SA-11)]
S.C.H. Lonien1, A.M. Bolognini2, R.E.C. Linhares1, C.M. Nozawa1* * Autor para correspondência E-mail: cnoz@uel.br
1Departamento de Microbiologia. CCB/Universidade Estadual de Londrina
Caixa Postal 6001
86051-970 - Londrina, PR
2Bolsista PIBIC/CNPq
Recebido para publicação em 17 de novembro de 2000
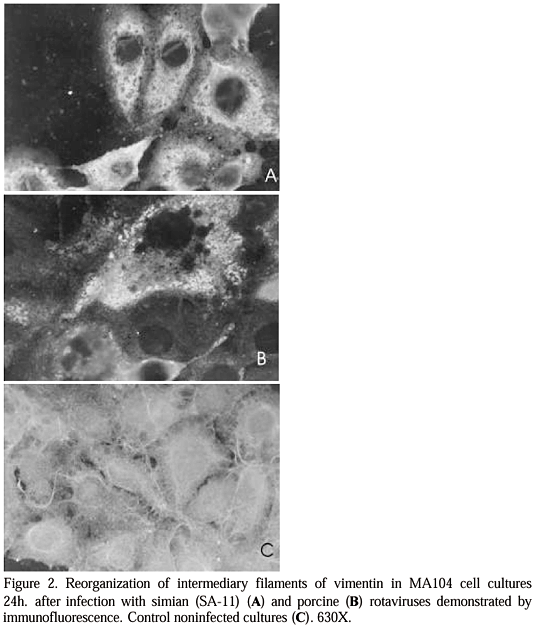
Rotaviruses are the causal agents of acute diarrhea and therefore play an important role in animal and human health. Virions present eleven segments of double-stranded RNA, triple-layered double-capsid and are nonenveloped (Estes, 1996). Replication occurs in the cytoplasm through morphogenesis carried out in a complex fashion. It involves RNA transcription already in the uncoating step, probably inside phagosomes and incomplete double-layered particles emerge in viroplasms, and bud into nearby endoplasmic reticulum. A transient pseudoenvelope is incorporated onto the particles and at the final stage of maturation it is lost and at the same time the viral surface polypeptides are rearranged (Patton, 1995; Estes, 1996). In general, the cytoskeleton is an extensive array of filaments that gives the cell its shape, the ability to move, and is responsible for the arrangements and internal motions of its organelles. Cytoskeleton components are also involved in cellular protein transport as well as RNA sorting (Ojakian & Schwimmor, 1988; Heskett & Pryme, 1991), and in several virus infections (Hua & Patton, 1994; Avalos et al., 1997; Favoreel et al., 1997). An outstanding role of the cytoskeleton was also attributed in the pathophysiology of rotavirus-induced diarrhea. Alteration of brush border-associated cytoskeleton by rotavirus can be the cause of blockage of sucrase/isomaltase transport to the apical surface of enterocytes, and actin-related changes were also associated with virus release (Jourdan et al., 1998). Although several studies were done in respect to the alterations of cytoskeleton elements by rotavirus infection, the actual meaning of these changes in the context of the cytopathology is poorly understood. The in vitro study of rotavirus/cell interaction may provide insights to the understanding of the rotavirus cytopathogenicity. This work is focused on the CPE, plaque size and changes in the cytoskeleton elements of a newly cell culture-adapted porcine rotavirus in comparison to a prototype simian strain (SA-11). Simian rotavirus strain SA-11 and porcine strain 1154 isolated from a pig farm and adapted to cell culture were used (Ramos et al., 2000). Seventy percent confluent cultures grown in DMEM (Gibco BRL, USA), supplemented with 5% fetal bovine serum (Cultilab, Brazil), added of 100IU penicillin/ml, 0.2 mg/ml streptomycin and 25 ug/ml fungizone were used throughout. Viruses were pretreated and maintained in culture, respectively, with 30ug/ml and 10ug/ml of crystalline trypsin1 throughout. Cells grown in Leighton tubes were washed and inoculated with viruses at m.o.i. of approximately 1. At time intervals of 24h, 48h and 72h postinfection (PI) coverslips were removed and submitted to Wright staining. For plaque assay, cells grown in 24-well polystyrene trays (Corning, USA) were washed, inoculated with ten-fold dilutions of the viruses and incubated at 37oC for 1h. Cultures were overlaid with 1.0ml of nutrient agarose at 1%, added of crystalline trypsin and 100ug/ml of DEAE-Dextran (Sigma Chem.Co., USA). Trays were incubated in 5% CO2 atmosphere and five days PI monolayers stained with 0.5% crystal violet added of 2.0% formalin. For immunofluorescence, coverslips were processed at time intervals of 24h and 48h PI. Cells were fixed and preincubated either with 5% normal goat or rabbit sera added of 0.3% Triton X-1004, for 15min at room temperature (25oC). This was followed by the incubation with goat anti-vimentin, rabbit anti-actin and anti-tubulin4 sera for 30 min at 37oC. After washings overlaid with FITC-conjugate anti-goat or anti-rabbit IgG4 for 30 min at 37oC and cells were observed under UV-light. Mock-infected cultures were used as control. The degenerative process observed in cell cultures infected with porcine and simian viruses was similar except that the CPE evolution of the former was faster than that of the latter. At 24h PI acidophilic inclusion bodies were observed generally round in shape and varied sizes, randomly scattered in a vacuolated cytoplasm. The cellular shape was round with a tendency for clumping. This was followed by the increase of cellular morphological changes progressively towards the loss of the cytoplasmic volume. The cytoplasm itself was restricted to narrow and long spindle-like structures, in particular for the porcine virus. Although CPE for both strains was similar, the plaque assay demonstrated that the simian strain produced mostly small plaques (average diameter 0.03mm, actual size in trays) in comparison to that of the porcine strain that presented larger plaques (average diameter 0.23mm, actual size in trays) (Fig.1). The intermediary filaments of vimentin were similarly altered as demonstrated 24h PI with porcine and simian rotaviruses (Fig.2). The well-organized network of vimentin represented by filaments irradiating from around cell nucleus which resembled a spider web was no longer observed but instead, a condensed amorphous mass with intense fluorescence took place by 24h up to 48h occupying two thirds of the cytoplasm. The microfilaments of actin were demonstrated by a fluorescent mass surrounding the nucleus. Twenty-four hours PI this reasonably organized mass was also substituted by an amorphous mass of fluorescence distributed throughout the cytoplasm. Tubulin also lost its initial disposition, 24h PI, demonstrated by long fibers throughout the cytoplasm (not shown). As for intermediary filaments, alterations for microfilaments of actin and tubulin were similar for both strains.
We compaired the cytopathology of a field adapted porcine rotavirus and the prototype simian virus. We found morphological and time-dependent differences in respect to the development of CPE. Cell rounding and clumping and the development of inclusion bodies of varied sizes were common for both strains. Simian virus produced smaller plaques albeit in larger number and a few larger plaques were also detected that may represent mutants. On the other hand, porcine virus developed larger plaques in smaller number but small plaques were also observed that could also represent mutants. The finding that porcine virus produced larger plaques is suggestive of a higher ability to spread from cell to cell and higher cytolycity/pathogenicity, as also suggested by Burke & Desselberger (1996). This was also in corroboration of faster evolution of porcine CPE in comparison to that of SA-11. Different plaque sizes were also observed elsewhere with feline and bovine rotaviruses (Hoshino et al., 1982; Archambault et al., 1984) and represent population heterogeneity (Taniguchi et al., 1994). As far as cytoskeleton is concerned it was demonstrated that both viruses caused depolimerization of vimentin, actin and tubulin. The reorganization of intermediate filaments, microfilaments and microtubules was represented by a condensed mass of these structures in the cytoplasm. This effect could cooperate with rounding observed during the development of CPE. However, in spite of porcine virus has produced a faster and generalized evolution of CPE, no difference in the aspect of cytoskeleton reorganization was observed, as expected. Several explanations have been put forward to justify cytoskeleton reorganization. Cervera et al. (1981) demonstrated that cellular and vesicular stomatitis virus mRNAs attach to vimentin filaments to be translated. According to Hua & Patton (1994) and Patton (1995), two nonstructural rotavirus proteins, NSP 1 and NSP 3, were found to be in association with cytoskeleton and as Weclewicz et al. (1994) demonstrated, vimentin filaments component were involved. It seems that cytoskeleton components act as an important network that regulates virus morphogenesis and/or release. This is possibly attained at expenses of the reorganization of these components that are associated in several virus infections with cytoplasmic transportation, maturation or release of virions, and also with CPE (Cudmore et al., 1995; Avalos et al., 1997; Jourdan et al., 1998). In conclusion, the comparison of simian and porcine rotaviruses cytopathology demonstrated that although the evolution of the CPE of the latter was faster and chronologically different on the morphological ground, no remarkable difference was found, apart from the number and plaque size.
Keywords: Rotavirus, cytopathology, cell culture, plaque, cytoskeleton
ACKNOWLEDGEMENTS
The authors wish to thank to CAPES, CNPq, CPG/UEL and Prouni/Londrina for partial financial support. This paper is part of CSH Lonien M.Sc. manuscript who was in receipt of scholarship also granted by CAPES/DS.
RESUMO
A citopatologia in vitro de uma cepa de rotavírus porcino adaptado em cultura de células foi comparada à estirpe-protótipo símia (SA-11). O efeito citopático (ECP) produzido pelos vírus foi semelhante embora a estirpe porcina tivesse apresentado algumas alterações diferentes, como o acentuado estreitamento do citoplasma, com grande perda do volume citoplasmático. O vírus porcino apresentou menor número de plaques de ECP porém com diâmetro maior em relação ao vírus símio, demonstrando maior capacidade de disseminação célula-célula, quase oito vezes mais, a julgar pelo diâmetro dos plaques de ECP. Os elementos do citoesqueleto das células infectadas revelaram uma reorganização semelhante para ambas as estirpes, não sendo possível observar nenhuma diferença, embora o ECP do vírus porcino tenha sido mais acentuado.
Palavras-chave: Rotavírus, citopatologia, cultura de células, plaques, citoesqueleto
REFERENCES
- ARCHAMBAULT, D., ROY, R.S., DEA, S. et al. Comparative study of bovine rotavirus isolates by plaque assay. Can. J. Comp. Med, v.48, p.286-289, 1984.
- AVALOS, R.T., YU, Z., NAYAK, D.P. Association of influenza virus NP and M1 proteins with cellular cytoskeletal elements in influenza virus-infected cells. J. Virol, v.71, p.2947-2958, 1997.
- BURKE, B., DESSELBERGER, U. Rotavirus pathogenicity. Virology, v.218, p.299-305, 1996.
- CERVERA, M., DREYFUSS, G., PENMAN, S. Messenger RNA is translated when associated with the cytoskeletal framework in normal and VSV-infected HeLa cells. Cell, v.23, p.113-120, 1981.
- CUDMORE, S., COSSART, P., GRIFFITHS, G. et al. Actin-based motility of vaccinia virus. Nature, n.378, p.636-638, 1995.
- ESTES, M.K. Rotaviruses and their replication. In: FIELDS, B.N., KNIPE, P.M., HOWLEY, P.N. et al. (Ed). Virology, 3.ed. Philadelphia: Lippincott-Raven, 1996. p.1625-1655.
- FAVOREEL, H.W., NAUWYNCK, H.J., OOSTVELDT, P.V. et al. Antibody-induced and cytoskeleton-mediated redistribution and shedding of viral glycoproteins, expressed on pseudorabies virus-infected cells. J. Virol., v.71, p.8254-8261, 1997.
- HESKETT, J.E., PRYME, I.F. Interaction between mRNA ribosomes and the cytoskeleton. J. Biochem, v.277, p. 1-10, 1991.
- HOSHINO, Y., WYATT, R.G., SCOTT, F.W. et al. Isolation and characterization of a canine rotavirus. Arch. Virol, v.72, p.113-125, 1982.
- HUA, J., PATTON, J.T. The carboxyl-half of the rotavirus nonstructural protein NS53 (NSP1) is not required for virus replication. Virology, v.196, p.567-576, 1994.
- JOURDAN, N., BRUNET, J.P., SAPIN, C. et al. Rotavirus infection reduces sucrase-isomaltase expression in human intestinal epithelial cells by perturbing protein targeting and organization of microvillar cytoskeleton. J. Virol., v.72, p.7228-7236, 1998.
- OJAKIAN, G.K., SCHWIMMOR, R. The polarized distribution of an apical cell surface glycoprotein is maintained by interactions with the cytoskeleton of Madin-Darby canine kidney cells. J. Cell Biol., v.107, p.2377-2397, 1988.
- PATTON, J. T. Structure and function of the rotavirus RNA-binding proteins. J. Gen. Virol., v.76, p.2633-2644, 1995.
- RAMOS, A.P.D., STEFANELLI, C.C., LINHARES, R.E.C. et al. The stability of porcine rotavirus in feces. Vet. Microbiol, v.71, p.1-8, 2000.
- TANIGUCHI, K., NISHIKAWA, K., NOBUMICHI, K. et al. Differences in plaque size and VP4 sequence found in SA11 virus clones having simian authentic VP4. Virology, v.198, p.325-330, 1994.
- WECLEWICZ, K., KRISTENSSON, K., SVENSSON. L. Rotavirus causes selective vimentin reorganization in monkey kidney CV-1 cells. J. Gen. Virol., v.75, p.3267-3271, 1994.
Publication Dates
-
Publication in this collection
20 June 2002 -
Date of issue
Aug 2001
History
-
Received
17 Nov 2000