Abstracts
The objective of the present study was to determine the sperm enrichment with X-bearing spermatozoa, after one centrifugation in a Percoll or OptiPrep continuous density gradient, using quantitative real-time polymerase chain reaction (qPCR) of sperm DNA and resultant in vitro-produced bovine embryos by PCR. Frozen/thawed sperm was layered on density gradients and the tubes were centrifuged. Supernatants were gently aspirated and the sperm recovered from the bottom of the tubes. Cleavage and blastocyst rates were determined through in vitro production of embryos and PCR was performed to identify the embryos' genetic sex. A difference in blastocyst rate was found in the Percoll treatment compared to OptiPrep (P<0.05). The percentage of female embryos in the Percoll and OptiPrep groups was 62.0% and 47.1%, respectively. These results were confirmed by qPCR of spermatozoa DNA and underestimation was seen only in the Percoll group. It was possible to sexing sperm using simple approach.
bovine; sperm sexing; centrifugation; embryo sexing; qPCR; PCR
O objetivo do presente estudo foi determinar o enriquecimento de espermatozoides portadores do cromossomo X após a centrifugação em gradiente de densidade contínuo de Percoll ou OptiPrep, utilizando reação em cadeia da polimerase quantitativa em tempo real (qPCR) do DNA do espermatozoide e dos embriões bovinos produzidos in vitro resultantes pela PCR convencional. Espermatozoides descongelado foram depositados em gradientes de densidade e os tubos foram centrifugados. Os sobrenadantes foram gentilmente aspirados e os espermatozoides recuperados do fundo dos tubos. As taxas de clivagem e de blastocisto foram determinadas pela produção in vitro de embriões e a PCR foi realizada para a identificação genética do sexo dos embriões. Verificou-se diferença na taxa de blastocistos entre os grupos Percoll e OptiPrep (P<0,05). A porcentagem de embriões de fêmeas nos grupos Percoll e OptiPrep foi de 62,0% e 47,1%, respectivamente. Estes resultados foram confirmados pela qPCR do DNA de espermatozoides e uma subestimação foi observada no grupo do gradiente de densidade de Percoll. Foi possível a sexagem de espermatozoides utilizando uma metodologia simples.
bovino; sexagem espermatozoides; centrifugação; sexagem embriões; qPCR; PCR
VETERINARY MEDICINE
Comparative validation using quantitative real-time PCR (qPCR) and conventional PCR of bovine semen centrifuged in continuous density gradient
Validação comparativa utilizando PCR quantitativo em tempo real (qPCR) e PCR convencional de sêmen bovino centrifugado em gradiente de densidade contínuo
M.V. ResendeI; A.C. LucioII; A.P. PeriniI; L.Z. OliveiraI; A.O. AlmeidaI; B.C.A. AlvesIII; C.A. Moreira-FilhoIV; I.W. SantosV; V.F.M. Hossepian de LimaI
IDepartamento de Medicina Veterinária Preventiva e Reprodução Animal - FCAV - UNESP, Jaboticabal, SP
IIDepartamento de Zootecnia - FCAV - UNESP, Jaboticabal, SP
IIIInstituto Israelita de Ensino e Pesquisa Albert Einstein, São Paulo, SP
IVDepartamento de Pediatria, Faculdade de Medicina - USP, São Paulo, SP
VDepartamento de Medicina Veterinária, UFPR, Palotina, PR
ABSTRACT
The objective of the present study was to determine the sperm enrichment with X-bearing spermatozoa, after one centrifugation in a Percoll or OptiPrep continuous density gradient, using quantitative real-time polymerase chain reaction (qPCR) of sperm DNA and resultant in vitro-produced bovine embryos by PCR. Frozen/thawed sperm was layered on density gradients and the tubes were centrifuged. Supernatants were gently aspirated and the sperm recovered from the bottom of the tubes. Cleavage and blastocyst rates were determined through in vitro production of embryos and PCR was performed to identify the embryos' genetic sex. A difference in blastocyst rate was found in the Percoll treatment compared to OptiPrep (P<0.05). The percentage of female embryos in the Percoll and OptiPrep groups was 62.0% and 47.1%, respectively. These results were confirmed by qPCR of spermatozoa DNA and underestimation was seen only in the Percoll group. It was possible to sexing sperm using simple approach.
Keywords: bovine, sperm sexing, centrifugation, embryo sexing, qPCR, PCR
RESUMO
O objetivo do presente estudo foi determinar o enriquecimento de espermatozoides portadores do cromossomo X após a centrifugação em gradiente de densidade contínuo de Percoll ou OptiPrep, utilizando reação em cadeia da polimerase quantitativa em tempo real (qPCR) do DNA do espermatozoide e dos embriões bovinos produzidos in vitro resultantes pela PCR convencional. Espermatozoides descongelado foram depositados em gradientes de densidade e os tubos foram centrifugados. Os sobrenadantes foram gentilmente aspirados e os espermatozoides recuperados do fundo dos tubos. As taxas de clivagem e de blastocisto foram determinadas pela produção in vitro de embriões e a PCR foi realizada para a identificação genética do sexo dos embriões. Verificou-se diferença na taxa de blastocistos entre os grupos Percoll e OptiPrep (P<0,05). A porcentagem de embriões de fêmeas nos grupos Percoll e OptiPrep foi de 62,0% e 47,1%, respectivamente. Estes resultados foram confirmados pela qPCR do DNA de espermatozoides e uma subestimação foi observada no grupo do gradiente de densidade de Percoll. Foi possível a sexagem de espermatozoides utilizando uma metodologia simples.
Palavras-chave: bovino, sexagem espermatozoides, centrifugação,sexagem embriões, qPCR, PCR
INTRODUCTION
Sperm sexing has aroused great interest due to the extensive application in animal production as well as medicine, and new separation techniques with both better accuracy and low cost are necessary. Density gradient centrifugation could be an approach to spermatozoa sexing because more DNA content of X- bearing spermatozoa can be related to more 0.06% density in those cells (Windsor et al., 1993).
For the separation of bovine X- or Y-chromosome bearing spermatozoa (X or Y sperm) satisfactory results have been obtained through recentrifugation of spermatozoa on two Percoll discontinuous density gradients. In cattle, Schwiderski et al. (1991) used two centrifugations in Percoll discontinuous gradients, consisting of 10 layers of 0.6mL of Percoll solution with densities ranging from 1.034 to 1.068g/mL. This method has provided an enrichment of over 75% and 65% of X or Y sperm fractions above and below, respectively, as verified by in situ hybridization. Blottner et al. (1993) using the same methodology used the sperm of both fractions for in vitro production of bovine embryos that had the sex confirmed by PCR, which demonstrated that the use of sperm from the top and bottom fraction resulted in 75% and 92% of the sex of embryos male and female, respectively. Lange et al. (1995) repeated both experiments and obtained 70% accuracy in the selection of X sperm in the sediment and 65% of Y sperm in the supernatant, after evaluation by in situ hybridization.
Hossepian de Lima et al. (2000) and Hossepian de Lima (2007) reported an accuracy of 70% using discontinuous Percoll density gradient in one or two centrifugations, but suggested many inconveniences that difficult commercial application, such as the impossibility of gradient storage, immediate use after semen collection or thawing, in addition to complex preparation (too many layers) that can increase variability and be time-consuming.
The objective of the present study was to determine the sperm enrichment with X sperm, after one centrifugation in a Percoll or OptiPrep continuous density gradient, using quantitative real-time polymerase chain reaction (qPCR) of sperm DNA and resultant in vitro-produced bovine embryos by PCR.
MATERIAL AND METHODS
Chemicals and media were purchased from Sigma-Aldrich (St. Louis, MO), unless otherwise stated. The semen samples were obtained from cyopreserved bovine semen of two Gir breed bulls and two Jersey breed bulls from a commercial company. The percentages of total motile sperm and sperm vigor were determined subjectively on a drop of semen placed in a pre-warmed glass-slide covered with a coverslip, and examined under a bright field microscope at 400x magnification. Sperm concentration was determined in a hemocytometer at a 1:200 dilution.
The preparation of continuous density gradient was as follows: Percoll (Pharmacia Fine Chemicals, Uppsala, Sweden) gradients were made by mixing a stock solution of Percoll with DMEM, pH 7.4, 280 to 290mOsm/kg/H20 with 0.3% (wt/vol) of BSA (Calbiochem, Darmstadt, Germany) in order to obtain densities ranging from 1.110 to 1.123g/mL. OptiPrep was prepared by mixing different proportions in DMEM containing 0.3% (wt/vol) of BSA (Calbiochem, Darmstadt, Germany), pH 7.4, in order to obtain densities ranging from 1.110 to 1.123g/mL.
Discontinuous Percoll and OptiPrep density gradients were then consecutively layered in 15mL polystyrene centrifuge tubes. The tubes were maintained at 4ºC for 24h to convert a discontinuous into a continuous density gradient, according to instructions in the manual for the preparation of density gradients (Density..., 2007). Continuous gradients are characterized by a smooth change in density from the top to the bottom of the tube. Instead of the obvious interfaces present in the discontinuous gradient, a continuous gradient can be thought of as having an infinite number of interfaces (Cell..., 2007).
Forty million thawed sperm were overlaid on Percoll and OptiPrep continuous density gradients and one centrifugation was performed. Motility was evaluated subjectively using light microscopy, and the concentration was determined by counting spermatozoa in a hemocytometer. They were then centrifuged 500 X g for 15min at 22ºC. The supernatants were carefully aspirated and sperm located at the bottom fraction were collected from the tubes. Sperm concentration, motility, and recovery rates were determined and the sperm was used for quality control by in vitro production (assessment of cleavage and blastocyst rates) and qPCR (assessment of X and Y sperm proportion) procedures.
For IVF, bovine ovaries were transported from the abattoir to the laboratory in a saline solution at 30 to 35ºC. Antral follicles of 3 to 7mm were manually aspirated using a 19-gauge needle attached to a 20mL syringe. Oocytes with a compact cumulus and at least four layers of cells were selected for in vitro maturation (IVM). Groups of 15 to 20 oocytes were placed in 100µL droplets of IVM medium under mineral oil. IVM medium was TCM 199 (GIBCO BRL; Grand Island, USA) supplemented with 10% heat-inactivated (at 55ºC for 30min) FCS, FSH (FolltropinTM, Bioniche Animal Health, Belleville, Canada) (1.0µg/µL), hCG (Profasi HPTM; ProfasiTM, Serono, São Paulo, Brazil) (10U/mL), estradiol (1.0µg/mL), sodium pyruvate (0.2mM) and 16.67µg/µL amikacin (AminocinaTM, Instituto Biochimico, Rio de Janeiro, Brazil) and 5mM sodium bicarbonate. Oocytes were cultured for 22h at 38.5ºC under an atmosphere of 5% CO2 in the air.
The sperm centrifuged in Percoll and OptiPrep density gradients was counted, diluted IVF medium (100 x 103 cells for a 90µL IVF droplet) and incubated for 60 to 90min for capacitation before IVF. The oocytes were washed three times in TCM-1993 medium supplemented with 25mM of HEPES, 100mM of sodium pyruvate, BSA (BSA fraction V, fatty acid free, Inlab, São Paulo, Brazil) (10mg/mL) and once in IVF medium. The oocytes and sperm were incubated for 20h in 5% CO2 in humidified air at 38.5ºC. Presumptive zygotes were denuded of cumulus cells by repeated pipetting, and washed three times in modified synthetic oviduct fluid (SOF) medium, and they were then transferred to 500µL SOF medium in four well dishes. Embryo culture was carried out under mineral oil in a humidified atmosphere of 5% CO2, 5% O2 and 90% N2 at 38.5ºC and cleavage was assessed at 46 to 48 h and development to the blastocyst stage at day 7 to 8. Fetal calf serum (FCS) was not added during in vitro culture, as previously described (Gutiérrez-Adán et al., 2001). The blastocysts were frozen in liquid nitrogen individually in microtubes with 10µL of Milli-Q water and stored at -20ºC until PCR analysis for sex determination. In order to obtain embryonic cell DNA for sex determination by PCR, 221 and 239 embryos from Percoll and OptiPrep groups, respectively.
Proteinase K (Invitrogen, Cleveland, USA) was added at a final concentration of 5µg per embryo. The micro tubes were incubated at 37ºC for 60min (for enzyme activity) and at 98ºC for 10min (for enzyme inactivation). The sample of each micro tube was separated in two samples. Two pairs of primers of Y-specific sequences were split into two distinct samples. The first pair was 5'-CCT CCC CTT GTT CAA ACG CCC GGA ATC ATT-3' and 5'-TGC TTG ACT GCA GGG ACC GAG AGG TTT GGG-3' (Bondioli et al., 1989), and the second pair was 5'-ATC AGT GCA GGG ACC GAG ATG- 3' and 5'-AAG CAG CCG ATA AAC ACT CCT T-3' (Schwerin et al., 1991; Luz et al., 2000). The first pair detected a sequence of 210 bp specific to the bovine Y-chromosome and the second one detected a sequence of 196bp specific to Y-chromosome. A third pair of primers detected an autosomal sequence of 280bp, indicating the presence of bovine genomic DNA. The third pair was 5'-AGG TCG CGA GAT TGG TCG CTA GGT CAT GCA-3' and 5'-AAG ACC TCG AGA GAC CCT CTT CAA CAC GT-3' (Ellis et al., 1988). Multiplex PCR was carried out in the same tube with first (210bp) and third (280bp) primers and the PCR of the second pair of primers (196bp) was carried out in a second tube. Amplifications were performed in a thermocycler (MJ Research PTC 100 Thermal Cycler, GMI Inc., Ramsey, MN) as follows: a) for first and third primers: an initial step at 94ºC for 10sec, 40 denaturation cycles at 94ºC for 1 min, annealing at 58ºC for 1min and synthesis at 72ºC for 1min. A time extension of 7min at 72ºC was added at the end of the final cycle; b) for the second primer: an initial step at 94ºC for 10sec, 38 denaturation cycles at 94ºC for 1min, annealing at 58ºC for 1min and synthesis at 72ºC for 1min. A time extension of 7min at 72ºC was added at the end of the final cycle as well.
Before DNA extraction from spermatozoa, there were two washes with PBS by centrifugation at 900x g for 5min to remove as much residues of the density gradient as possible. DNA extraction from sperm samples was performed using the DNeasy Blood and Tissue Kit (Qiagen, GmbH, Germany). DNA concentration and purity were determined using a biophotometer (Eppendorf, Hamburg, Germany). The concentration of samples was around 100 ng. DNA samples were subjected to electrophoresis in 2% agarose gel for visualization of genomic DNA. The qPCR procedure (Parati et al., 2006) was used to determine the enrichment of samples with X sperm, after centrifugation in density gradients, without the need to perform validation by means of in vitro production of embryos and subsequent sexing by conventional PCR. For internal control a negative control with no DNA added was used, a control with DNA extracted from the blood of a male bovine and with DNA extracted from spermatozoa not centrifuged in density gradients.
The results of amplification (Ct values - cycle threshold) were analyzed in 7500 Sequence Detection System Software (Applied Biosystems, Foster City, USA) using a relative quantification (ΔΔCt) study assay. Figure 1 shows an example of amplification curves. The quality control of the centrifuged sperm evaluation was made by the proportion of X- and Y-sperm by qPCR. The calculation for predicting the proportion of X and Y sperm was performed based on the 2-ΔΔCt method described by (Livak and Schmittgen, 2001). The calculation of the proportion of relative quantification of X and Y sperm in the samples also took into account the efficiency of each amplification, where 2.0 is 100%, 1.9 is 90%, etc.
Five trials were performed for each bull in the Percoll and OptiPrep groups, totaling forty centrifugation procedure trials. Cleavage and blastocyst rates were analyzed through a chi-square test using an SAS program (SAS Inst. Inc., Cary, USA). The chi-square test was used to analyze the embryo PCR results, comparing the percentages of male and female embryos of sexing groups (Percoll and OptiPrep) with the expected 1:1 ratio. Means of female embryos obtained in the Percoll and OptiPrep groups were compared with means for DNA amplification of X sperm by qPCR, and analyzed by analysis of variance (PROC GLM of SAS program - SAS Inst. Inc., Cary, USA) followed by the Tukey's test. A 5% level of significance was used in all statistical analysis.
RESULTS
The mean motility before density gradient centrifugation was 64% and after centrifugation it was 78% and 30% for the Percoll and OptiPrep groups, respectively. The sperm recovery rate after centrifugation was 5.7% and 19.4% for the Percoll and OptiPrep groups, respectively.
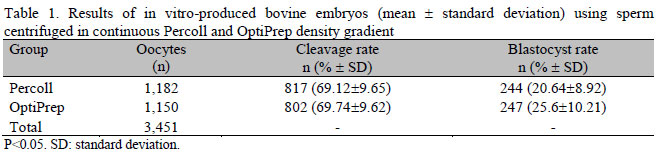
The findings of in vitro embryo production (cleavage and blastocyst rates) are shown in Table 1. A difference in blastocyst rate between the Percoll group and OptiPrep group was observed (P<0.05), and no differences (P>0.05) in cleavage and blastocyst rates after grouping the results of Gir and Jersey breeds were found between groups.
A total of 460 embryos were sexed by conventional PCR analysis (genetic sex identification). The results are summarized in Table 2. Data from sexing groups were compared to that of the expected 1:1 ratio. Female sex ratio deviation was found in the Percoll group (62.0%) (P<0.05) but not in the OptiPrep group (47.3%) (P>0.05).
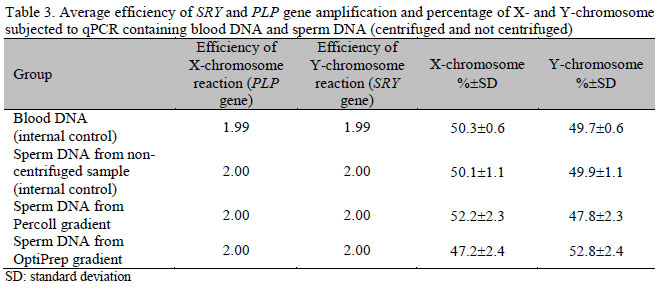
Data from qPCR was grouped in Table 3. The relative quantification of X- and Y-chromosome from blood DNA samples (internal control) was 50.3% for each chromosome. In the samples containing sperm DNA (not centrifuged - internal control), it was 50.1% for each chromosome, showing that qPCR was able to correctly identify X and Y sperm, as the expected proportion in males is 1:1.
In the samples centrifuged in Percoll or OptiPrep density gradient, the percentage of X-chromosome was 52.2 and 47.2%, respectively. In the Percoll gradient, the percentage of X- chromosome was statistically lower than in PCR of embryos (62% of females) (P<0.05). However, in the OptiPrep gradient, the percentage obtained in qPCR was not different (P>0.05) from that found in conventional PCR of embryos (47.3% of female embryos) (Table 4).
The analysis of male bovine blood DNA amplification (internal control) and samples of sperm DNA from Percoll, OptiPrep and sperm DNA from non-centrifuged sample groups showed that the average efficiency was close to 100% for the two genes used (PLP and SRY), with a correlation (r2) average of 0.996.
Table 4 shows a data comparison between conventional PCR of embryos and qPCR from sperm DNA subjected to centrifugation.
DISCUSSION
The objective in the present study was to determine the sperm enrichment with X-bearing spermatozoa, after one centrifugation in a Percoll or OptiPrep continuous density gradient, using quantitative real-time polymerase chain reaction (qPCR) of sperm DNA and resultant in vitro-produced bovine embryos by PCR. Also, results of qPCR of spermatozoa DNA centrifuged and sex ratio of in vitro-produced embryos by conventional PCR were compared, and how they affected the in vitro production of bovine embryos was studied.
The results of cleavage rate did not differ between the centrifuged groups that ranged from 68 to 70%, and 70 to 73%, respectively, indicating that this parameter was not affected by centrifugation. These results were similar to those obtained using two centrifugations in Percoll discontinuous density gradient (Schwiderski et al., 1991; Blottner et al., 1993; Lange et al., 1995; Hossepian de Lima, 2007). The only difference showing lower rate of blastocysts was observed in the Percoll gradient in Jersey breed. This was due to one of the bulls used in the study, which had a blastocyst rate of 11% compared to an average of 23% in the other three bulls (data not shown).
Despite the sex ratio deviation in the Percoll gradient (62% of females, P<0.05), the sexing results demonstrated that there was sex ratio towards females after PCR analysis. In bovine, one centrifugation on 12-layer discontinuous Percoll gradient produced a female sex ratio deviation that ranged from 55.7 to 74.3% (Hossepian de Lima et al., 2000; Kobayashi et al., 2004). In the present study, the discontinuous density gradient with only 3 layers was converted into continuous density gradient and could be stored for 24 h at 4ºC, which may simplify routine procedures of in vitro production in specialized companies.
OptiPrep has been used in medicine to separate motile and viable sperm as a substitute of Percoll, but since 1996 its clinical human use has been withdrawn due to potential endotoxin contamination (McCann and Chantler, 2000). Mousset-Simeon et al. (2004) reported that OptiPrep was less effective than Percoll to separate sperm with regular motility and morphology. In contrast, the present study obtained similar results in the comparison of motility between Percoll and OptiPrep, which was consistent with other reports in humans (Andersen and Byskov, 1997; Harrison, 1997; Claassens et al., 1998). However, OptiPrep was not effective to separate X sperm. The qPCR has recently been used as a reliable technique for the quantification of X and Y sperm cells in semen samples, especially to validate techniques for sexing sperm (Joerg et al., 2004; Parati et al., 2006; Puglisi et al., 2006).
For the first time the use of qPCR to validate sexing bovine sperm submitted to density gradient centrifugation was demonstrated. Furthermore, it was possible to quantify the proportion of X and Y sperm in semen samples before centrifugation without any adjustments by using linear regression, as described in other studies (Parati et al., 2006; Puglisi et al., 2006).
The results of predicting the proportion of X and Y sperm through qPCR confirmed the findings of genetic identification of sex in in vitro-produced bovine embryos with the use of sexed sperm in OptiPrep groups. In Percoll density gradient, the percentages of X-chromosomes found in the samples by qPCR (52%) were underestimated compared to the sex ratio of embryos obtained in conventional PCR (62%). This can be explained by the effect of some components found in Percoll, such as silica and PVP, which may have interfered with the DNA extraction process or amplification by qPCR, despite successive washes. There have been reports that Percoll at 80% concentration produced great variability during sexing sperm by flow cytometry (Stap et al., 1998; Puglisi et al., 2006). In the present study, centrifuged sperm were recovered in a fraction containing 90% Percoll, which theoretically could have affected qPCR results, as seen in sexing by flow cytometry. However, the interference of Percoll in the DNA extraction process and qPCR has not been described in literature.
In a complementary study samples enriched with X sperm separated by flow cytometry were used. The percentage of X sperm using qPCR was 89%, which was within the expected 85% to 95% obtained by flow cytometry (Cran, 2007; Hamano, 2007; Seidel Jr., 2009).
These results in estimating the percentage of X and Y sperm in semen samples with sexed sperm through flow cytometry were higher than those reported by Joerg et al. (2004). In this study, the percentages of both X and Y sperm were underestimated, and a maximum of 80% and 70% were obtained, respectively, when the expected percentage was 85% to 95% (Cran, 2007; Seidel Jr., 2009).
CONCLUSIONS
The centrifugation procedure did not interfere with cleavage rates during in vitro embryo production, but a lower rate of blastocysts was seen in the Percoll gradient. There was a sex ratio deviation in the Percoll gradient, confirmed by conventional PCR of in vitro-produced bovine embryos. These results were confirmed by the qPCR of spermatozoa DNA, and an underestimation was seen only in the Percoll group. It was possible to sexing sperm using a simple approach.
ACKNOWLEDGMENTS
This study was supported by FAPESP grants # 04/06044-0 and 05/59357-9.
Recebido em 10 de março de 2010
Aceito em 6 de abril de 2011
E-mail: mvresende@yahoo.com.br
- ANDERSEN, C.Y.; BYSKOV, A.G. Enhanced separation of X and Y bearing sperm cells by a combined density gradient centrifugation evaluated by fluorescence in situ hybridization of the Y-chromosome. Acta Obstet. Gynecol. Scand., v.76, p.131-134, 1997.
- BLOTTNER, S.; SCHWERIN, M.; BÖTTCHER, M. et al Selective enrichment of bovine X- and Y-spermatozoa by Percoll density gradient. Arch. Tierz, v. 36, p.153-162, 1993.
- BONDIOLI, K.R.; ELLIS, S.B.; PRYOR, J.H. et al The use of male-specific chromosomal DNA fragments to determine the sex of bovine preimplantation embryos. Theriogenology, v.31, p. 95-104, 1989.
- CELL SEPARATION MEDIA. Methodology and applications. Sweden: GE Healthcare, 2007. p.20.
- CLAASSENS, O.E.; MENKVELD, R.; HARRISON, K.L. Evaluation of three substitutes for Percoll in sperm isolation by density gradient centrifugation. Hum. Reprod., v.13, p.3139-3143, 1998.
- CRAN, D.G. XY sperm separation and use in artificial insemination and other ARTs. Soc. Reprod. Fertil., v.65, suppl., p.475-91, 2007
- DENSITY GRADIENT MEDIA: Applications and products. 5.ed. Norway: Axis-Shield, 2005. p.15-18.
- ELLIS, S.B.; BONDIOLI, K.R.; WILLIAMS, M.E. et al Sex determination of bovine embryos using male-specific DNA probes. Theriogenology, v.29, p.242, 1988.
- GUTIÉRREZ-ADÁN, A.; LONERGAN, P.; RIZOS, D. et al Effect of the in vitro culture system on the kinetics of blastocyst development and sex ratio of bovine embryos. Theriogenology, v.55, p.1117-1126, 2001.
- HAMANO, K. Sex preselection in bovine by separation of X- and Y-chromosome bearing spermatozoa. J. Reprod. Dev., v.53, p.27-38, 2007.
- HARRISON, K.L. Iodixanol as a density gradient medium for the isolation of motile spermatozoa. J. Assist. Reprod. Genet., v.14, p.385-387, 1997.
- HOSSEPIAN DE LIMA, V.F.M.; RAMALHO, M.D.T.; RODRIGUES, L.H. et al Separation of X- and Y-Bearing Bovine Spermatozoa by Percoll density gradient centrifugation. Theriogenology, v.53, p.280, 2000.
- HOSSEPIAN DE LIMA, V.F.M. Aspectos metodológicos na seleção do sexo de espermatozóides bovinos para utilização no melhoramento genético e na produção animal. Rev. Bras. Zootec., v.36, p.219-228, 2007
- JOERG, H.; ASA, M.; GRAPHODATSKAYA, D. et al Validating bovine sexed semen samples using quantitative PCR. J. Anim. Breed. Genet.,v.121, p.209-215, 2004.
- KOBAYASHI, J.; OGURO, H.; UCHIDA, H. et al Assessment of bovine X- and Y-bearing spermatozoa in fractions by discontinuous percoll gradients with rapid fluorescence in situ hybridization. J. Reprod. Dev., v.50, p.463-469, 2004.
- LANGE, W.; PEMSEL, H.; BLOTTNER, S. et al Über de einfluβ des bullen auf die selective anreicherung der X-und Y- spermien mit der Percoll-dichtegradienten-zentrifugation. Arch. Tierz.,v.38, p.155-161, 1995.
- LIVAK, K.J.; SCHMITTGEN, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods, v.25, p.402-408, 2001.
- LUZ, M.R.; WATANABE, Y.F.; FERRO, J.A. et al Sexing in vitro fertilized bovine embryos by multiplex PCR. Braz. J. Vet. Res. Anim. Sci., v.37, p.453-456, 2000.
- McCANN, C.T.; CHANTLER, E. Properties of sperm separated using Percoll and IxaPrep density gradients. A comparison made using CASA, longevity, morphology and the acrosome reaction. Int. J. Androl., v. 23, p.205-209, 2000.
- MOUSSET-SIMÉON, N.; RIVES, N.; MASSE, L. et al Comparison of six density gradient media for selection of cryopreserved donor spermatozoa. J. Androl, v.25, p.1-4, 2004.
- PARATI, K.; BONGIONI, G.; ALEANDRI. R. et al Sex ratio determination in bovine semen: A new approach by quantitative real time PCR. Theriogenology, v.66, p.2202-2209, 2006.
- PUGLISI, R.; VANNI, R.; GALLI, A. et al. In vitro fertilisation with frozen-thawed bovine sperm sexed by flow cytometry and validated for accuracy by real-time PCR. Reproduction, v.132, p.519-526, 2006
- SCHWERIN, M.; BLOTTNER, S.; THOMSEN, P.D. et al Quantification of Y chromosome bearing spermatozoa of cattle using in situ hybridization. Mol. Reprod. Dev, v.30, p.39-43, 1991.
- SCHWIDERSKI, H.; BLOTTNER, S.; SCHWERIN, M. Mikroinjektion und in vitro befruchtung mit spermien aus versuchen zur anreicherung der geschlechtss pezifischen spermientypen beim rind. Reprod. Dom. Anim, v.26, p.143-144, 1991.
- SEIDEL Jr., G.E. Sperm sexing technology - The transition to commercial application An introduction to the symposium ''Update on sexing mammalian sperm''. Theriogenology, v.71, p.1-3, 2009
- STAP, J.; HOEBE, R.A.; MERTON, J.S. et al Improving the resolution of cryopreserved X- and Y-sperm during DNA flow cytometric analysis with the addition of Percoll to quench the fluorescence of dead sperm. J. Anim. Sci., v.76, p.1896-1902, 1998.
- WINDSOR, D.P.; EVANS, G.; WHITE, I.G. Sex predetermination by separation of X and Y chromosome-bearing sperm: a review. Mol. Reprod. Dev., v.5, p.155-171, 1993.
Publication Dates
-
Publication in this collection
08 Aug 2011 -
Date of issue
June 2011
History
-
Accepted
06 Apr 2011 -
Received
10 Mar 2010