Abstract
O presente trabalho teve como objetivo avaliar a biocompatibilidade e a osseointegração de compósitos de hidroxiapatita (HA), policaprolactona (PCL) e alendronato (ALN) em defeitos ósseos produzidos no olécrano de coelhos. Trinta e seis coelhos foram distribuídos em quatro grupos, recebendo como tratamento: (1) compósito de HA (49,5%), PCL (49,5%) e ALN (1%); (2) HA (50%) e PCL (50%); (3) PCL (100%); e (4) solução salina 0,9%. As amostras para análise histológica foram coletadas de três animais de cada grupo aos oito, 45 e 90 dias de pós-operatório. No oitavo dia, as bordas do defeito ainda eram identificáveis em todos os grupos. Tecido ósseo novo era formado em contato com o biomaterial apenas nas formulações que incluíam HA. Essas características continuaram evidentes nos outros momentos analisados, embora o defeito estivesse preenchido pelo tecido regenerado. A presença dos biomateriais foi verificada nos três momentos em todos os grupos. Não houve evidências de reação indesejável ao biomaterial. As análises histológicas e histomorfométricas mostraram que os biomateriais são biocompatíveis e aqueles contendo a hidroxiapatita favoreceram a formação óssea no início do processo de regeneração, embora o alendronato não tenha apresentado qualquer efeito.
regeneração óssea; biocompatibilidade; biomaterial
bone regeneration; biocompatibility; biomaterial
regeneração óssea; biocompatibilidade; biomaterial
Bone diseases and lesions affect the world population with great economic and social impact and assume increasing importance because the prevalence of many of these conditions is related to aging, and can be addressed as a global public health problem (Woolf et al., 2012WOOLF, A.D.; ERWIN, J.; MARCH, L. The need to address the burden of musculoskeletal conditions. Best Pract. Res. Clin. Rheumatol., v.26, p.183-224, 2012. ). Thus, the development of biomaterials to support the bone regeneration is an expanding area. Hydroxyapatite (HA) is the major mineral component of bone and its synthetic form is used in bone fracture repair (Borges et al., 2000BORGES, A.P.B.; REZENDE, C.M.F.; RIBEIRO, M.F.B. et al. Hidroxiapatita sintética como substituto ósseo em defeito experimental provocado no terço proximal da tíbia em cão: aspectos à microscopia eletrônica de transmissão. Arq. Bras. Med. Vet. Zootec., v.52, p.616-620, 2000.). Association between HA and polycaprolactone (PCL) in the formulation of biomaterials is a promising initiative to enhance bone regeneration by compensating individual limitations such as fragility and hydrophobicity of the former and low bioactivity of the latter (Fabbri et al., 2010FABBRI, P.; BONDIOLI, F.; MESSORI, M. et al. Porous scaffolds of polycaprolactone reinforced with in situ generated hydroxyapatite for bone tissue engineering. J. Mater. Sci. Mater. Med., v.21, p.343-351, 2010.). Alendronate (ALN) is a bisphosphonate used in the treatment of various bone conditions because of their powerful inhibitory bone resorption activity (Pérez-López, 2004). The aim of this study was to evaluate the effect of the combination of these substances in the regeneration of bone defects experimentally induced in rabbits' olecranon.
Two composites were prepared: one consisting of 49.5% of synthetic hydroxyapatite (HAP-91(r) Plus, JHS Laboratório Químico Ltda., Brasil), 49.5% of polymer polycaprolactone (PCL) and 1% of alendronate (ALN) and was named composite HA+PCL+ALN; and the second was composed of 50% HA and 50% PCL and was designated PCL+HA composite.
The Ethics Committee on Animal Use from the institution approved the procedures adopted in this study (protocol 309/2011). Thirty-six New Zealand rabbits were distributed into four groups. In each group, one treatment was employed: (1) HA+PCL+ALN; (2) HA+PCL; (3) PCL alone; or (4) 0.9% sterile saline solution, constituting the control group. All animals underwent surgical procedure for the production of the bone defect in the right olecranon. Before surgery, each animal received enrofloxacin (10mg/kg, intramuscular), then was sedated with acepromazine (0.25mg/kg, subcutaneous) and anesthetized with a combination of tiletamine-zolazepam (28mg/kg, intramuscular). Trichotomy was done in the right forelimb. For better analgesia, the brachial plexus block was performed with 1.5ml of 2% lidocaine. The surgical field was prepared with topic povidone and isolated with sterile surgical field cloths. After skin incision, muscle fascia divulsion and tricep bag incision, a bone defect was produced on the lateral cortex of the right olecranon with a manual trephine with 5mm in diameter. The defect was filled with a paste resulting from diluting 250mg of biomaterials, produced in powder form, in 1ml of 0.9% sterile saline solution. Each animal received one treatment according to its group as previously mentioned. The tissues were closed with nylon 4-0 using a simple continuous pattern. Postoperatively, the animals received two doses of morphine (1mg/kg, subcutaneous) with a 12 hour interval between applications.
At eight, 45 and 90 days after surgery, three animals from each group were euthanized for collection of the olecranon, which was dissected and separated from the ulna. The samples were fixed in 10% formalin and processed by routine histological techniques, with decalcification in a solution of formic acid and sodium citrate. 5mm thick sections were stained with hematoxylin and eosin. Three fields of each slide were photographed under a light microscope at 100x magnification, covering only the area of the cortical defect.
The images were analyzed on ImageJ software version 1.45s and biomaterial and bone percentage was determined by planimetric histomorphometry, in which a grid of 9 columns and 7 lines was superimposed on each image, counting 63 points of intersection per photo and 189 per animal. The points over biomaterial and bone were divided by the total points to obtain a percentage value. The histologic features seen in the defect region and the interaction between the tissue and the biomaterial were also described. "Bone", considering all groups, and "biomaterial", considering only those who received the treatment, obtained in the histomorphometric analysis were submitted to nonparametric Kruskal-Wallis test followed by the Student-Newman-Keuls, considering 5% level of significance.
After eight days, the defect edges were easily identifiable in the samples from the control group. Thin trabeculae emerging from the edges and arranged among a disorganized and vascularized connective tissue indicated the beginning of the repair process. In some samples it was still possible to view a clot mainly occupying the outer region of the defect. These features were also present in the groups that received the biomaterials, which could also be viewed in the samples, but they behaved differently between them. In the group that received only PCL, the polymer showed no contact with the newly formed bone tissue. In the groups that received HA+PCL+ALN or HA+PCL, besides having bone formation around the biomaterial in contact with it, there was also evident development of trabeculae in the medullary cavity of the olecranon (Fig. 1A). At 45 days in the control group, the entire cortical defect was already filled by bone tissue, containing, however, large and numerous pores filled with bone marrow, allowing the identification of the original defect edges. Newly formed bone deposition was still active. This process was similar in the other groups, however, those who received the HA in the formulation of treatment still had active formation of new trabeculae that continued throughout the biomaterial, suggesting osseointegration (Fig. 1B and C), indicating that the HA adds osseointegration property to the composite. Carlo et al. (2009)CARLO, E.D; BORGES, A.P.B; VARGAS, M.I.V. et al. Resposta tecidual ao compósito 50% hidroxiapatita: 50% poli-hidroxibutirato para substituição óssea em coelhos. Arq. Bras. Med. Vet. Zootec., v.61, p.844-852, 2009. , associating HA to polyhydroxybutyrate, also found that the bioactive activity of HA can promote osseointegration of a composite to which it is associated. These characteristics were extended until 90 days, when the presence of biomaterial was still detected and bone tissue continued to be deposited in contact with the biomaterial in the formulations that had HA (Fig. 1D). According to Lam et al. (2009)LAM, C.X.F.; HUTMACHER, D.W.; SCHANTZ, J.T. et al. Evaluation of polycaprolactone scaffold degradation for 6 months in vitro and in vivo. J. Biomed. Mater. Res. A, v.90, p.906-919, 2009., when PCL is used in the construction of three-dimensional structures to be employed as a biomaterial, it presents slow removal in vivo, since it is degraded by hydrolysis from the surface of the structure. Besides, Mitsak et al. (2011)MITSAK, A.G.; KEMPPAINEN, J.M.; HARRIS, M.T.; HOLLISTER, S.J. Effect of polycaprolactone scaffold permeability on bone regeneration in vivo. Tissue Eng. Part A, v.17, p.1831-1839, 2011. state that the increased permeability of PCL structures favors the bone growth in vivo.
Photomicrograph of the defect region held in rabbit olecranon treated with different composites in order to evaluate bone regeneration. A: The HA+PCL+ALN group, 8 days after surgery. B: HA+PCL group, 45 days. C: HA+PCL+ALN 45 days. D: HA+PCL+ALN 90 days. Biomaterial (a), newly formed bone (b) temporary matrix (c) defect edge (d), bone formation in contact with the biomaterial (arrow). Hematoxylin and eosin.
At 90 days, the cortical bone defect filling the defect presented lower porosity than the previous observation. The samples from the group that received ALN in the formulation presented proliferation of periosteal bone tissue. In addition to this feature, no other difference was noted between the groups containing the bisphosphonate and those that did not contain it. Fernandes et al. (2004)FERNANDES, R.G.; FAIG-LEITE, H.; CARVALHO, Y.R. Estudo da reparação óssea na mandíbula de coelhos sob a ação do alendronato sódico e da sua associação com a hidroxiapatita. Rev. Odontol. UNESP, v.33, p.123-30, 2004. studied the use of ALN alone or in combination with HA in rabbit mandible defect and reported that the natural tissue regeneration occurred from the endo and periosteal surfaces, but there was no difference between the treated groups and the control group. In some reported cases, periosteal reaction was observed, but it was probably related to bone callus resulting from small fracture regeneration (Somford et al., 2009SOMFORD, M.P; GEURTS, G.F.A; TEULING, J.W.A.M. et al. Long-term alendronate use not without consequences? Int. J. Rheumatol., v.2009, p.1-6, 2009.). The local administration of ALN, however, can cause bone compaction that can be exploited in the prior preparation of areas that will receive the implant fixation (Jakobsen et al., 2007JAKOBSEN, T.; KOLD, S.; BECHTOLD, J.E. et al. Local alendronate increases fixation of implants inserted with bone compaction: 12-week canine study. J. Orthop. Res., v.25, p.432-441, 2007.). The mechanism and clinical significance of periosteal reaction seen in the context of this work needs to be further investigated. These were not fibrosis, giant cells or reactive signs of infection or inflammation at any time of observation, confirming the biocompatibility of the constituents of the groups used in this experiment.
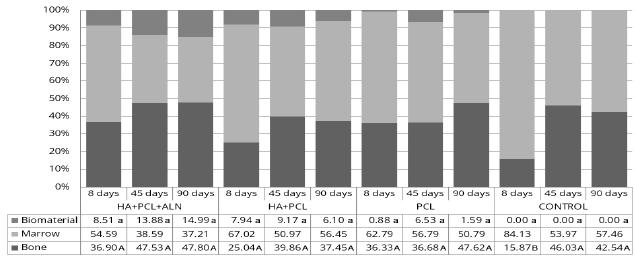
The histomorphometric analysis showed no statistical difference in the amount of biomaterial between the groups in the analyzed fields, although the p value obtained was 0.037, the post-hoc test was not able to detect the difference. However, the amount of bone was significantly lower in the control group at 8 days compared to all other groups and dates, with p = 0.001. This result indicates that the regeneration of a defect of this nature probably reaches a certain degree of stabilization before 90 days and, therefore, there was no difference between the values obtained for that time-point and 45 days, although the remodeling is still needed for the full restoration of the cortical bone (Fig. 2). Indeed, one must be careful in assessing the effect of biomaterials in non-critical defects, i.e., they may have the ability to regenerate in a short time without the need for therapeutic intervention (MacNeill et al., 1999MACNEILL, S.R.; COBB, C.M.; RAPLEY, J.W. et al. In vivo comparison of synthetic osseous graft materials. A preliminary study. J. Clin. Periodontol., v.26, p.239-245, 1999.). Vital et al. (2006)VITAL, C.; BORGES, A.P.B.; FONSECA, C.C. et al. Biocompatibilidade e comportamento de compósitos de hidroxiapatita em falha óssea na ulna de coelhos. Arq. Bras. Med. Vet. Zootec., v.58, p.175-183, 2006. employed HA composites in bone regeneration in ulna defects, which were produced in rabbits removing a fragment of 2cm in length and found that the regeneration process was earlier in the treated groups than in the control group up to 120 days.
Histomorphometric distribution of the amount of bone, biomaterial and medullary space in each group and date of collection in rabbit olecranon treated with different composites in order to evaluate bone regeneration. Different letters in the line indicate statistical difference (Kruskal-Wallis, SNK; α= 5%).
It is concluded that the biomaterials consisting of polycaprolactone associated hydroxyapatite are biocompatible and favored the formation of bone in olecranon defects in rabbits in the initial period of repair. Hydroxyapatite gave the property of osseointegration to the composite, while the addition of alendronate showed no effect on the process.
ACKNOWLEDGEMENT
The authors thank the agencies FAPEMIG, CNPq and CAPES for financial support and JHS Laboratório Químico Ltda. for providing the material for research.
- BORGES, A.P.B.; REZENDE, C.M.F.; RIBEIRO, M.F.B. et al. Hidroxiapatita sintética como substituto ósseo em defeito experimental provocado no terço proximal da tíbia em cão: aspectos à microscopia eletrônica de transmissão. Arq. Bras. Med. Vet. Zootec., v.52, p.616-620, 2000.
- CARLO, E.D; BORGES, A.P.B; VARGAS, M.I.V. et al. Resposta tecidual ao compósito 50% hidroxiapatita: 50% poli-hidroxibutirato para substituição óssea em coelhos. Arq. Bras. Med. Vet. Zootec., v.61, p.844-852, 2009.
- FERNANDES, R.G.; FAIG-LEITE, H.; CARVALHO, Y.R. Estudo da reparação óssea na mandíbula de coelhos sob a ação do alendronato sódico e da sua associação com a hidroxiapatita. Rev. Odontol. UNESP, v.33, p.123-30, 2004.
- FABBRI, P.; BONDIOLI, F.; MESSORI, M. et al. Porous scaffolds of polycaprolactone reinforced with in situ generated hydroxyapatite for bone tissue engineering. J. Mater. Sci. Mater. Med., v.21, p.343-351, 2010.
- JAKOBSEN, T.; KOLD, S.; BECHTOLD, J.E. et al. Local alendronate increases fixation of implants inserted with bone compaction: 12-week canine study. J. Orthop. Res., v.25, p.432-441, 2007.
- LAM, C.X.F.; HUTMACHER, D.W.; SCHANTZ, J.T. et al. Evaluation of polycaprolactone scaffold degradation for 6 months in vitro and in vivo. J. Biomed. Mater. Res. A, v.90, p.906-919, 2009.
- MACNEILL, S.R.; COBB, C.M.; RAPLEY, J.W. et al. In vivo comparison of synthetic osseous graft materials. A preliminary study. J. Clin. Periodontol., v.26, p.239-245, 1999.
- MITSAK, A.G.; KEMPPAINEN, J.M.; HARRIS, M.T.; HOLLISTER, S.J. Effect of polycaprolactone scaffold permeability on bone regeneration in vivo. Tissue Eng. Part A, v.17, p.1831-1839, 2011.
- PÉREZ-LÓPEZ, F.R. Postmenopausal osteoporosis and alendronate. Maturitas, v.48, p.179-192, 2004.
- SOMFORD, M.P; GEURTS, G.F.A; TEULING, J.W.A.M. et al. Long-term alendronate use not without consequences? Int. J. Rheumatol., v.2009, p.1-6, 2009.
- VITAL, C.; BORGES, A.P.B.; FONSECA, C.C. et al. Biocompatibilidade e comportamento de compósitos de hidroxiapatita em falha óssea na ulna de coelhos. Arq. Bras. Med. Vet. Zootec., v.58, p.175-183, 2006.
- WOOLF, A.D.; ERWIN, J.; MARCH, L. The need to address the burden of musculoskeletal conditions. Best Pract. Res. Clin. Rheumatol., v.26, p.183-224, 2012.
Publication Dates
-
Publication in this collection
Mar-Apr 2016
History
-
Received
05 Apr 2015 -
Accepted
15 Nov 2015