ABSTRACT
The objective of this study was to evaluate the influence of collard green stalks as environmental enrichment on the behavior of cockatiels (Nymphicus hollandicus) maintained in captivity. Sixteen birds were housed individually in cages and fed a commercial diet for parrots and seed mix, with water ad libitum during 12 days in climatic chamber with temperature and relative humidity simulating tropical conditions. The animals (n = 8) were divided into two groups, control and enriched, and the experimental period was divided into two phases (during and after enrichment) of four days each. The collard green stalks were used as enrichment only during the first period of the experiment, daily changed and maintained only during the day. The stalks increased the feed intake and decreased the sleep activities. No effect was observed on the body surface temperature, locomotion, maintenance and other resting activities. Small undesirable activities were detected. It was concluded that collard green stalks can be used as environmental enrichment, becoming part of food preferences of cockatiels. However, its use does not significantly alter the behavior of birds kept in captivity
Keywords:
birds; animal welfare; captive animals; food enrichment; psittacine
RESUMO
O objetivo deste estudo foi avaliar a influência de talos de couve como enriquecimento ambiental sobre o comportamento de calopsitas (Nymphicus hollandicus) mantidas em cativeiro. Dezesseis aves foram alojadas individualmente em gaiolas e alimentadas com dieta comercial para psitacídeos e mistura de sementes, com água ad libitum durante 12 dias, em câmara climática com temperatura e umidade relativa do ar simulando condições tropicais. Os animais (n=8) foram divididos em dois grupos, controle e enriquecido, e o período experimental foi dividido em duas fases (durante e após o enriquecimento) de quatro dias cada. Os talos de couve foram usados como enriquecimento apenas durante o primeiro período do experimento, trocados diariamente e mantidos apenas durante o dia. Os talos aumentaram o consumo de ração e diminuíram o comportamento dormir. Nenhum efeito foi observado nas outras atividades de descanso, na temperatura da superfície corporal, na locomoção e manutenção. Foram detectadas pequenas atividades indesejáveis. Concluiu-se que os talos de couve podem ser usados como enriquecimento ambiental, formando parte das preferências alimentares de calopsitas. No entanto, seu uso não altera de maneira expressiva o comportamento das aves.
Palavras-chave:
pássaro; bem-estar animal; animais cativos; enriquecimento alimentar; psitacídeo
INTRODUCTION
Recently, the use of cockatiels (Nymphicus hollandicus) as pets is increasing around the world. However, studies with environmental enrichment and particularly on its influence on the behavior of this species are scarce. Due to an increase in the number of animals maintained in captivity, there is a great interest in the behavior of these birds when maintained with different substrates as environmental enrichment (Meehan and Mench, 2002MEEHAN, C.L.; MENCH, J.A. Environmental enrichment affects the fear and exploratory responses to novelty of young Amazon parrots. Appl. Anim. Behav. Sci., v.79, p.75-88, 2002. ; Meehan et al., 2004). This knowledge contributes with future studies in order to improve captivity conditions of the animals.
Environmental enrichment can be defined as modification of the environment of captive animals, in order to increase opportunities for expression of biological behavior and improve the health conditions of animals (Newberry, 1995NEWBERRY, R.C. Environmental enrichment: increasing the biological relevance of captive environments. Appl. Anim. Behav. Sci. , v.44, p.229-243, 1995.). However, to provide adequate environment, it is necessary to know the behavior of species maintained in different conditions. Cockatiels are mostly characterized as docile animals but different behavioral manifestation can be expressed when the birds are submitted to different environmental conditions (Seibert and Crowell-Davis, 2001SEIBERT, L.M.; CROWELL-DAVIS, S.L. Gender effects on aggression, dominance rank, and affiliative behaviors in a flock of captive adult cockatiels (Nymphicus hollandicus). Appl. Anim. Behav. Sci. , v.71, p.155-170, 2001. ).
There is evidence that birds can interact with several types of plants in the environment (Del Hoyo et al., 1997DEL HOYO, J.; ELLIOTT, A.; SARGATAL, J. Handbook of the birds of the world. Barcelona: Lynx Edicions, 1997. 696p.). In the literature, no study has reported on the use of collard green stalks (Brassica oleracea L.) as environmental enrichment on the behavior of captivity birds. In general, collards are considered to be a healthy food for humans (Chen et al., 2014CHEN, R.; ZHANG, P.; LI, L. et al. Analysis of nutrition of collard green grown in China. J. Food Proc. Technol., v.5, [p.1-5], 2014.), being a good source of vitamins (A, C, K and folic acid), calcium and soluble fiber, among several others nutrients like iron, copper, manganese, selenium and zinc (Miller-Cebert et al., 2009MILLER-CEBERT, R.L.; SISTANI, N.A.; CEBERT, E. Comparative mineral composition among canola cultivars and other cruciferous leafy greens. J. Food Compos. Anal., v.22, p.112-116, 2009.; Chen et al., 2014CHEN, R.; ZHANG, P.; LI, L. et al. Analysis of nutrition of collard green grown in China. J. Food Proc. Technol., v.5, [p.1-5], 2014.).
The knowledge of the effects of the use of collard green stalks as environmental enrichment for captivity cockatiels would assist in the discovery of new information that would improve the life quality of the birds. Thus, the aim of this study was to evaluate the use of collard green stalks as environmental enrichment for cockatiels (N. hollandicus) kept in captivity.
MATERIAL AND METHODS
The research was conducted at the Department of Animal Science of Federal University of Lavras (UFLA), in Lavras, Minas Gerais, Brazil. The experimental protocol was approved by the Ethics Committee on Animal Use (CEUA) of the same institution (protocol number 029/13).
Sixteen healthy cockatiels (N. hollandicus), eight males and females, aged approximately one year, derived from commercial breeding, were housed individually in galvanized wire cages (38(37(28cm), separated by 20cm from each other, at 1.0m of the floor, in a climatic chamber equipped with an automated system (MT-530 Super, Full Gauge Controls, Canoas, Brazil) that controlled the circulation of heated or cooled air in the room using infrared lamps, blowers and fans. All the equipment in a climatic chamber was connected to a central panel, allowing the automatic adjustment of the internal temperature of the room. The experimental conditions were adjusted to maintain the temperature near to 35°C from 6h00min to 18h00min and 25°C from 18h01min to 5h59min. The relative humidity was set to 70%. This change in day and night temperatures was determined based on the natural temperature variations that occur in the tropical zone (Verstappen et al., 2002VERSTAPPEN, F.A.L.M.; LUMEIJ, J.T.; BRONNEBERG, G.G. Plasma chemistry reference values in Ostriches. J. Wildlife. Dis., v.38, p.154-159, 2002.).
The air in the chamber was changed constantly and was regulated by blowers and fans attached to a pipe with small holes for air distribution. The system automatically stayed on for 15 minutes and off for two minutes. The temperature was monitored daily inside the rooms at 8h00min, 13h00min and 18h00min using maximum and minimum thermometers placed at half the height of the animals. The lighting program used was 24 hours of artificial light with infrared light 250w (Ecolume, Belo Horizonte, Brazil) located at 1.0m above the birds.
Each cage contained two plastic feeders, one for commercial diet (Complete Food for Parrots Birds CC Bio Tron, Rio Claro, São Paulo, Brazil) and other for seed mix (50% millet, 30% canary seed, 15% oat and 5% sunflower - Torloni, 1991TORLONI, C.E.E. Criação de calopsitas. [Guarulhos]: Lis Gráfica, 1991. 89p. ), and a drinking ceramic pot (10 cm diameter). Excreta were daily collected in an aluminum tray located under each cage.
The experiment was carried out in a completely randomized design with eight replicates in split-plot in time scheme (during and after the use of collard green stalks) with two treatments (with or without collard green stalks). The experimental unit was one bird evaluated during four days.
Initially, the animals were divided into two groups of eight birds each (four males and four females) that received or not the collard green stalks as environmental enrichment. The groups were organized so as to be the most homogeneous possible (size, weight and previous behavior such as aggression). The experimental period (8 days) was divided into two periods of four days (during and after the enrichment). The birds received water and diets ad libitum. Handlings (cage cleaning, feeding, water change, and evaluation of consumption) were made daily always around 6h00min. The feed and seed intake was determined based on the difference between the amount provided and the waste.
The environmental enrichment was made with collard green stalks during the first period of experiment (from 1st to 4th day). The stalks were heavy and then put in each cage of enriched group twice, at 6h00min and 13h00min. At 18h00min, the leftover stalks were removed and weighed again to evaluate consumption of the material. The body temperature was daily measured using the device FLIR E50 (Nashua, USA) around at 6h00min, 12h00 and at 18h:00min, and the daily mean calculated.
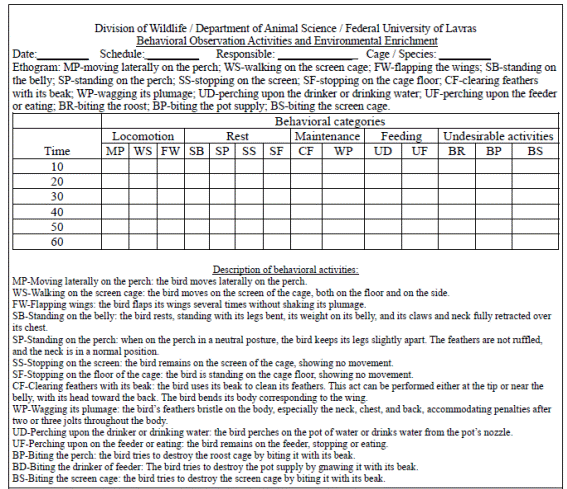
The behavior of the birds was assessed by filming during daylight with a camcorder (8.0 mm CCD Color Camera 1/3 Infrared ST1230, Campina Grande do Sul, Paraná, Brazil). The recorded images of each bird were analyzed at the same time by one evaluator. Every ten minutes, the activities of the animals were recorded following the focal sampling technique (Altmann, 1974ALTMANN, J. Observational study of behaviour: sampling methods. Behaviour, v.49, p.227-267, 1974.) in a pre-defined ethogram (Figure 1) elaborated for spectacled parrot (Amazona pretrei) of the order Psittaciformes in captivity (Prestes, 2000PRESTES, P.N. Description and quantitative analysis of etogram of Amazon petrei in captivity. Ararajuba , v.8, p.25-42, 2000.). With the total number of registered behaviors for each bird, the percentage of each behavior categories was calculated for each experimental unit.
Ethogram for evaluating the behavioral parameters of individual cockatiels (Nymphicus hollandicus).
The data were evaluated by ANCOVA after the normality test using Shapiro-Wilk test and the means were compared by F test. The values of each experimental unit obtained during the first period of experiment (1-4 days) were used as covariate. For the variables that not achieved the normality, Kruskal-Walis test was used. In all cases, α = 0.05. All statistical analysis was performed using the Action 2.3 statistical program.
RESULTS AND DISCUSSION
The room temperature was 35.0±2.0°C during the daylight (6h00min to 18h00min) and 25.0±2.0°C during the dark (18h01min to 5h59min). No interaction or effect of enriched environment or period of evaluation was observed (P>0.15) in the body surface temperature of cockatiels (Table 1). The birds, warm-blooded animals, control body temperature through behavioral changes or else physiological mechanisms, such as increase or decrease of the blood flow in the body surface which, in turn, facilitates or difficult the body heat dissipation to the environment when temperatures are outside the comfort zone (Macari et al., 1994MACARI, M.; FURLAN, R.L.; GONZALES, E. Fisiologia aviaria aplicada a frangos de corte. Jaboticabal: FUNEP / UNESP, 1994. 246p.). Thus, the temperature of the body surface of the birds can be used to evaluate the physiological response of the animal to an environment that is not conducive to their welfare (Nascimento et al., 2014NASCIMENTO, S.T.; SILVA, I.J.O.; MAIA, A.S.C. et al. Mean surface temperature prediction models for broiler chickens: a study of sensible heat flow. Int. J. Biometeorol., v.58, p.195-201, 2014.).
The enriched environment with collard green stalks increased (P<0.01) the feed intake of commercial diet but not (P>0.05) the seed mix after the use of enrichment. The approximate consumption of collard green stalks during the experimental period was 180 g. The intake of the stalks shows that the birds interacted with the material. It is known that foraging is an important feature for cockatiels, which, in their natural habitat, can take up to 70% of its activity time (Péron and Grosset, 2014PÉRON, F.; GROSSET, C. The diet of adult psittacids: veterinarian and ethological approaches. J. Anim. Physiol. Anim. Nutr., v.98, p.403-416, 2014. ). In this case, the use of substances that enhance this natural behavior of captive birds is important because it promotes not only animal welfare but also the captive breeding practice and possible reintroduction of the birds to the natural environment (Coulton et al., 1997COULTON, L.E.; WARAN, N.K.; YOUNG, R.J. Effects of foraging enrichment on the behaviour of parrots. Anim. Welf., v.6, p.357-364, 1997.).
Also in relation to nutritional balance, it is known that the cockatiel is a small granivorous bird, which can survive without drinking large amounts of water, metabolically producing enough water through the oxidation of carbohydrates and fats (MacMillen and Baudinette, 1993). In this case, collard green stalks can be used not only as environmental enrichment, but also as an indirect source of water for parrots, since vegetables and fresh vegetables have approximately 85% water in their composition (Koutsos et al., 2001KOUTSOS, E.A.; MATSON, K.D.; KLASING, K.C. Nutrition of birds in the order psittaciformes: a review. J. Avian. Med. Surg., v.15, p.257-275, 2001.).
During the use of collard green stalks as environmental enrichment, lower sleeping activities were registered (P < 0.05) in the enriched group (Table 2). Similarly, in blue-fronted parrot (Amazona aestiva, Psittacidae) it was observed that the rates of stationary display of inactive behavior (sleep) were significantly lower during the enrichment phase and significantly increased post enrichment (Andrade and Azevedo, 2011ANDRADE, A.A.; AZEVEDO, C.S. Efeitos do enriquecimento ambiental na diminuição de comportamentos anormais exibidos por papagaios-verdadeiros (Amazona aestiva, Psittacidae) cativos. Rev. Bras. Ornitol., v.19, p.56-62, 2011.). The result obtained in the present study suggests that collard green aroused the interest or curiosity of the birds without to affect significantly other specific behavioral characteristics. To reduce the ‘sleep’ activity indicates reduction of idleness which could contribute to reduce undesirable activities (Carlstead and Shepherdson, 2000CARLSTEAD, K.; SHEPHERDSON, D. Alleviating stress in zoo animals with environmental enrichment. MOBERG, G.P.; MENCH, J.A. (Eds). The biology of animal stress: basic principles and implications for animal welfare. Oregon: National Zoological Park, 2000. p.337-354.).
After removal of enrichment, there is a numerical tendency towards increase in bird undesirable activities such as biting the screen cage. In general, environmental enrichment involves several activities like searching for food, social interactions, training or intellectual stimulation and physical activities (Meehan et al., 2003MEEHAN, C.L.; GARNER, J.P.; MENCH, J.A. Isosexual pair housing improves the welfare of young Amazon parrots. Appl. Anim. Behav. Sci. , v.81, p.73-88, 2003.) which can substantially contribute to the psychological well-being of the birds (Jones, 2007JONES, M.P. Practical environmental enrichment in avian practice. In: North American Veterinary Conference/Small Animal and Exotics Section, 2007, Florida. Proceedings... Florida:NAVC, 2007. p.1487-1489.). This study shows that birds that once lived on favorable terms and are removed from their environments can manifest undesirable behavior. In such cases, the use of collard greens stalks can mitigate the problems arising from the marketing of cockatiels or seizure of wildlife birds kept in illegal captivity.
No effect of use of collard green stalks was observed (P>0.10) in the others behavior characteristics of the birds. These results are consistent with those obtained for superficial body temperature of birds. Behavior is generally considered to be the most effective form of body temperature regulation (Schlader et al., 2011SCHLADER, Z.J.; SIMMONS, S.E.; STANNARD, S.R.; MÜNDEL, T. Skin temperature as a thermal controller of exercise intensity. Eur. J. Appl. Physiol., v.111, p.1631-1639, 2011. ). Increase or decrease of body surface temperature in birds maintained in the same conditions could indicate higher or lower intensity of heaters movements (Refinetti et al., 1989REFINETTI, R. Contribution of locomotor activity to the generation of the daily rhythm of body temperature in golden hamsters. Physiol. Behav. , v.56, p.829-831, 1989.) or indicate presence of acute stress (Olivier et al., 2003OLIVIER, B.; ZETHOF, T.; PATTIJ, T. et al. Stress-induced hyperthermia and anxiety: pharmacological validation. Eur. J. Pharmacol., v.463, p.117-132, 2003.). Acute stress or stress-induced hyperthermia is mediated by the autonomic nervous system and occurs before and during exposure to anxiogenic stimuli or stress inducers such as noise, heat, handling, novelty or pain (Kleitman, 1945KLEITMAN, N. The effect of motion pictures on body temperature. Science, v.102, p.430-431, 1945.; Renbourn, 1960RENBOURN, E.T. Body temperature and pulse rate in boys and young men prior to sporting contests. A study of emotional hyperthermia: with a review of the literature. J. Psychosom. Res., v.4, p.149-175, 1960. ; Bermant et al., 1979BERMANT, R.I.; REEVES, D.L.; LEVINSON, D.M.; JUSTESEN, D.R. Classical conditioning of microwave‐induced hyperthermia in rats. Radio Sci., v.14, p.201-207, 1979.; Briese, 1995BRIESE, E. Emotional hyperthermia and performance in humans. Physiol. Behav., v.58, p.615-618, 1995.; Briese and DeQuijada, 1970BRIESE, E.; DEQUIJADA, M.G. Colonic temperature of rats during handling. Acta Physiol. Lat. Am., v.20, p.97-102, 1970.; Marazziti et al.,1992MARAZZITI, D.; DI MURO, A.; CASTROGIOVANNI, P. Psychological stress and body temperature changes in humans. Physiol. Behav. , v.52, p.393-395, 1992.). Thus, it is possible to presume that collard green stalks did not increase heating movements and consequently did not affect the bird's thermal status.
Although environmental enrichment is a potentially promising strategy to reduce fear responses of captive parrots and similar birds, little is known about the elements necessary for effective environmental enrichment for these birds. Thus, it is essential to select carefully the environmental enrichment used and adjust the complexity of the environment to the behavioral characteristics and ability of each species to interact with the enrichment item.
Behavioral studies with cockatiels (N. hollandicus) kept in captivity are scarce in the literature. Most studies with psittacines were based on parrots. In the present study, the use of cabbage stalks as environmental enrichment was not sufficient to alter significantly the behavior of the birds; however, it cannot be said that the use of enrichment was not effective. We observed that all birds interacted with the enrichment item immediately so that we can affirm that this substrate can be used to enhance the ambience conditions of cockatiels kept in captivity.
CONCLUSION
Collard green stalks can be used as environmental enrichment, forming part of food preferences of cockatiels. However, its use does not expressively alter the behavior of birds kept in captivity.
ACKNOWLEDGEMENTS
The authors acknowledge the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES PNPD Institucional), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG - Programa Pesquisador Mineiro - PPM 00460-12), CNPq (Process number 305478/2015-0) and Programa de Pós-graduação em Ciências Veterinárias of Federal University of Lavras.
REFERENCES
- ALTMANN, J. Observational study of behaviour: sampling methods. Behaviour, v.49, p.227-267, 1974.
- ANDRADE, A.A.; AZEVEDO, C.S. Efeitos do enriquecimento ambiental na diminuição de comportamentos anormais exibidos por papagaios-verdadeiros (Amazona aestiva, Psittacidae) cativos. Rev. Bras. Ornitol., v.19, p.56-62, 2011.
- BERMANT, R.I.; REEVES, D.L.; LEVINSON, D.M.; JUSTESEN, D.R. Classical conditioning of microwave‐induced hyperthermia in rats. Radio Sci., v.14, p.201-207, 1979.
- BRIESE, E. Emotional hyperthermia and performance in humans. Physiol. Behav., v.58, p.615-618, 1995.
- BRIESE, E.; DEQUIJADA, M.G. Colonic temperature of rats during handling. Acta Physiol. Lat. Am., v.20, p.97-102, 1970.
- CARLSTEAD, K.; SHEPHERDSON, D. Alleviating stress in zoo animals with environmental enrichment. MOBERG, G.P.; MENCH, J.A. (Eds). The biology of animal stress: basic principles and implications for animal welfare. Oregon: National Zoological Park, 2000. p.337-354.
- CHEN, R.; ZHANG, P.; LI, L. et al. Analysis of nutrition of collard green grown in China. J. Food Proc. Technol., v.5, [p.1-5], 2014.
- COULTON, L.E.; WARAN, N.K.; YOUNG, R.J. Effects of foraging enrichment on the behaviour of parrots. Anim. Welf., v.6, p.357-364, 1997.
- DEL HOYO, J.; ELLIOTT, A.; SARGATAL, J. Handbook of the birds of the world. Barcelona: Lynx Edicions, 1997. 696p.
- JONES, M.P. Practical environmental enrichment in avian practice. In: North American Veterinary Conference/Small Animal and Exotics Section, 2007, Florida. Proceedings... Florida:NAVC, 2007. p.1487-1489.
- KLEITMAN, N. The effect of motion pictures on body temperature. Science, v.102, p.430-431, 1945.
- KOUTSOS, E.A.; MATSON, K.D.; KLASING, K.C. Nutrition of birds in the order psittaciformes: a review. J. Avian. Med. Surg., v.15, p.257-275, 2001.
- MACARI, M.; FURLAN, R.L.; GONZALES, E. Fisiologia aviaria aplicada a frangos de corte. Jaboticabal: FUNEP / UNESP, 1994. 246p.
- MACMILLEN, R.E.; BAUDINETTE, R.V. Water economy of granivorous birds: Australian parrots. Funct. Ecol., v.7, p.704-712, 1993.
- MARAZZITI, D.; DI MURO, A.; CASTROGIOVANNI, P. Psychological stress and body temperature changes in humans. Physiol. Behav. , v.52, p.393-395, 1992.
- MEEHAN, C.L.; MENCH, J.A. Environmental enrichment affects the fear and exploratory responses to novelty of young Amazon parrots. Appl. Anim. Behav. Sci., v.79, p.75-88, 2002.
- MEEHAN, C.L.; GARNER, J.P.; MENCH, J.A. Isosexual pair housing improves the welfare of young Amazon parrots. Appl. Anim. Behav. Sci. , v.81, p.73-88, 2003.
- MEEHAN, C.L.; GARNER, J.P.; MENCH, J.A. Environmental enrichment and development of cage stereotypy in orange-winged Amazon parrots (Amazona amazonica). Dev. Psychobiol. v.44, p.209-218, 2004.
- MILLER-CEBERT, R.L.; SISTANI, N.A.; CEBERT, E. Comparative mineral composition among canola cultivars and other cruciferous leafy greens. J. Food Compos. Anal., v.22, p.112-116, 2009.
- NASCIMENTO, S.T.; SILVA, I.J.O.; MAIA, A.S.C. et al. Mean surface temperature prediction models for broiler chickens: a study of sensible heat flow. Int. J. Biometeorol., v.58, p.195-201, 2014.
- NEWBERRY, R.C. Environmental enrichment: increasing the biological relevance of captive environments. Appl. Anim. Behav. Sci. , v.44, p.229-243, 1995.
- OLIVIER, B.; ZETHOF, T.; PATTIJ, T. et al. Stress-induced hyperthermia and anxiety: pharmacological validation. Eur. J. Pharmacol., v.463, p.117-132, 2003.
- PÉRON, F.; GROSSET, C. The diet of adult psittacids: veterinarian and ethological approaches. J. Anim. Physiol. Anim. Nutr., v.98, p.403-416, 2014.
- PRESTES, P.N. Description and quantitative analysis of etogram of Amazon petrei in captivity. Ararajuba , v.8, p.25-42, 2000.
- REFINETTI, R. Contribution of locomotor activity to the generation of the daily rhythm of body temperature in golden hamsters. Physiol. Behav. , v.56, p.829-831, 1989.
- RENBOURN, E.T. Body temperature and pulse rate in boys and young men prior to sporting contests. A study of emotional hyperthermia: with a review of the literature. J. Psychosom. Res., v.4, p.149-175, 1960.
- SCHLADER, Z.J.; SIMMONS, S.E.; STANNARD, S.R.; MÜNDEL, T. Skin temperature as a thermal controller of exercise intensity. Eur. J. Appl. Physiol., v.111, p.1631-1639, 2011.
- SEIBERT, L.M.; CROWELL-DAVIS, S.L. Gender effects on aggression, dominance rank, and affiliative behaviors in a flock of captive adult cockatiels (Nymphicus hollandicus). Appl. Anim. Behav. Sci. , v.71, p.155-170, 2001.
- TORLONI, C.E.E. Criação de calopsitas. [Guarulhos]: Lis Gráfica, 1991. 89p.
- VERSTAPPEN, F.A.L.M.; LUMEIJ, J.T.; BRONNEBERG, G.G. Plasma chemistry reference values in Ostriches. J. Wildlife. Dis., v.38, p.154-159, 2002.
Publication Dates
-
Publication in this collection
June 2017
History
-
Received
09 Aug 2016 -
Accepted
10 Oct 2016