ABSTRACT
The aim of this study was to evaluate the effect of pST injections on metabolism, testicular size, and sperm characteristics in young boars. Sixty 22-day old piglets were divided into two groups: pST (n=30) and Control (n=30). The pST group was submitted to pST injections (90µg/kg body weight) every three days up to 330 days of age. Blood collections were performed weekly. Testicular weight was measures at 22, 82, 142, 202 and 365 days of age. Libido and fresh semen characteristics were evaluated between 150 and 210 days of age. Semen characteristics were also evaluated during a 72h storage period (15ºC). Testosterone, albumin, and phosphorus blood concentrations were higher in the pST group (P<0.05). The pST group had a higher IGF-I concentration in seminal plasma (P=0.05) and higher testicular weight (P<0.001) compared to the Control group. The pST group had higher ejaculate volume (P<0.001), total sperm count (P=0.047) and number of inseminating doses/ejaculate (P=0.047). During the 72h storage period, the pST group had a lower number of morphological alterations (P<0.001) compared to the Control group. In sum, pST injection in young boars increased testosterone concentration, testicular size, and sperm quality.
Keywords:
swine; growth hormone; pST; testicle; semen
RESUMO
O objetivo deste estudo foi determinar o efeito da administração de pST sobre o metabolismo, o tamanho testicular e a qualidade espermática de cachaços jovens. Foram usados leitões com 22 dias de idade, divididos em dois grupos: pST (n=30) e controle (n=30). O grupo pST foi submetido a injeções de pST (90µg/kg de peso vivo) a cada três dias até 330 dias de idade. Peso testicular foi avaliado aos 22, 82, 142, 202 e 365 dias de idade. Libido e qualidade do sêmen fresco foram avaliados entre 150 e 210 dias de idade. Qualidade espermática foi avaliada durante refrigeração (15ºC) por um período de 72 horas. Concentrações sanguíneas de testosterona, albumina e fósforo foram maiores no grupo pST (P<0,05). O grupo pST apresentou maior concentração de IGF-I no plasma seminal (P=0,05) e maior peso testicular, quando comparado ao grupo controle (P<0,001). O grupo pST apresentou maior volume espermático (P<0,001), concentração espermática (P=0,047) e número de doses espermáticas por ejaculado (P=0,047). Durante o período de 72 horas de refrigeração, o grupo pST teve menor número de patologias espermáticas (P<0,001). Assim, conclui-se que a administração de pST aumenta a concentração sanguínea de testosterona, o tamanho testicular e a qualidade espermática de cachaços jovens.
Palavras-chave:
suíno; hormônio do crescimento; pST; testículo; sêmen
INTRODUCTION
The sexual maturity of male pigs is influenced by different factors, such as breed, nutrition and environmental effects (Kumaresan et al., 2011KUMARESAN, A.; BUJARBARUAH, K.M.; KADIRVEL, G. et al. Early sexual maturity in local boars of Northeastern India: age-related changes in testicular growth, epididymal sperm characteristics and peripheral testosterone levels. Theriogenology, v.75, p.687-695, 2011.), and therefore can vary greatly between individuals. The gonadotropin-releasing hormone (GnRH) regulates the levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) and, as a consequence, testosterone production and Sertoli cell activity, respectively, regulating age at sexual maturity (Finnerty et al., 1998FINNERTY, M.; ENRIGHT, W.J.; ROCHE, J.F. Testosterone, LH and FSH episodic secretory patterns in GnRH-immunized bulls. J. Reprod. Fertil., v.114, p.85-94, 1998.). The increase of testosterone production plays a crucial role in the establishment of puberty and the onset of sperm production, as its serum levels are positively correlated to testicular development, puberty, sexual maturity and sperm production (Park and Yi, 2002PARK, C.S.; YI, Y.J. Comparison of semen characteristics, sperm freezability and testosterone concentration between Duroc and Yorkshire boars during seasons. Anim. Reprod. Sci., v.73, p.53-61, 2002.). Metabolic hormones, such as growth hormone (GH), may also influence sexual maturity. Exogenous GH supplementation is positively associated with improved testicular development, gametogenesis and steroidogenesis, and GH replacement therapy can accelerate puberty in young men with endogenous GH deficiency (Kamp et al., 2002KAMP, G.A.; WAELKENS, J.J.J.; DE MUINCK KEIZER-SCHRAMA, S.M.P.F. et al. High dose growth hormone treatment induces acceleration of skeletal maturation and an earlier onset of puberty in children with idiopathic short stature. Arch. Dis. Child., v.87, p.215-220, 2002.). However, the specific effects of GH supplementation in animals and humans with normal reproductive function are still unclear. Some studies indicate improvement of sperm quality in horses (Storer et al., 2005STORER, W.A.; THOMPSON JR, D.L.; CARTMILL, J.A. The effects of equine somatotropin on pituitary and testicular function in the stallion during the nonbreeding season. J. Equine Vet. Sci., v.25, p.106-112, 2005.) and bulls receiving exogenous GH (Hafez et al., 2005HAFEZ, Y.M.; FAWZY, S.A.; EL-HENAWY, M.A. et al. Effect of recombinant bovine somatotropin (rbST) on semen physical characteristics and some biochemical constituents in seminal plasma of Friesian bulls. Egypt. J. Anim. Prod., v.42, p.87-94, 2005.; Vieira et al., 2010VIEIRA, M.B.; BIANCHI, I.; MADEIRA, E.M. et al. Effect of recombinant bovine somatotropin on plasma concentrations of insulin-like growth factor I, insulin and membrane integrity of bull spermatozoa. Reprod. Domest. Anim., v.45, p.1110-1113, 2010.); while others have demonstrated adverse effects on testicular development in dogs (Sjogren et al., 1998SJOGREN, I.; JONSSON, M.; MADEJ, A. et al. Effects of very high doses of human growth hormone (hGH) on the male reproductive system in the dog. Andrologia, v.30, p.37-42, 1998.).
The effects of GH are mostly mediated by hepatic produced insulin-like growth factor-I (IGF-I), which is stimulated by GH (Sirotkin, 2005SIROTKIN, A.V. Control of reproductive processes by growth hormone: extra- and intracellular mechanisms. Vet. J., v.170, p.307-317, 2005.). IGF-I is a mitotic factor and can improve testicular development (Swanlund et al., 1995SWANLUND, D.J.; N’DIAYE, M.R.; LOSETH, K.J. et al. Diverse testicular responses to exogenous growth hormone and follicle-stimulating hormone in prepubertal boars. Biol. Reprod., v.53, p.749-757, 1995.). IGF-I can also stimulate the proliferation of Sertoli cells and the development of seminiferous tubules in prepubertal males (Swanlund et al., 1995). Besides, reduced IGF-I serum concentration is associated with a delay in testosterone production prior to puberty in undernourished steers (Brito et al., 2007BRITO, L.F.C.; BARTH, A.D.; RAWLINGS, N.C. et al. Effect of nutrition during calfhood and peripubertal period on serum metabolic hormones, gonadotropins and testosterone concentrations, and on sexual development in bulls. Domest. Anim. Endocrinol., v.33, p.1-18, 2007.). IGF-I has also been detected in seminal plasma (Henricks et al., 1998HENRICKS, D.M.; KOUBA, A.J.; LACKEY, B.R. et al. Identification of insulin-like growth factor-I in bovine seminal plasma and its receptor on spermatozoa: influence on sperm motility. Biol. Reprod., v.59, p.330-337, 1998.; Selvaraju et al., 2009SELVARAJU, S.; REDDY, I.J.; NANDI, S. et al. Influence of IGF-I on buffalo (Bubalus bubalis) spermatozoa motility, membrane integrity, lipid peroxidation and fructose uptake in vitro. Anim. Reprod. Sci., v.113, p.60-70, 2009.). Therefore, the effect of local IGF-I production should not be disregarded, since seminal plasma IGF-I is positively associated to sperm motility (Henricks et al., 1998). The administration of exogenous GH can also increase circulating concentrations of LH as well as the expression of its receptors in target tissues (Chatelain et al., 1991CHATELAIN, P.G.; SANCHEZ, P.; SAEZ, J.M. Growth hormone and insulin-like growth factor i treatment increase testicular luteinizing hormone receptors and steroidogenic responsiveness of growth hormone deficient dwarf mice. Endocrinology, v.128, p.1857-1862, 1991.; Sirotkin, 2005). Increased LH secretion in the postnatal period is related to maturation and differentiation of Leydig cells and an increased testosterone production (Bagu et al., 2006BAGU, E.T.; COOK, S.; GRATTON, C.L. et al. Postnatal changes in testicular gonadotropin receptors, serum gonadotropin, and testosterone concentrations and functional development of the testes in bulls. Reproduction, v.132, p.403-411, 2006.). Although it is known that GH is positively associated with circulating concentrations of gonadotropins in rats (Sirotkin, 2005), this is not true for ruminants (Folch et al., 2001FOLCH, J.; RAMON, J.P.; COCERO, M.J. et al. Exogenous growth hormone improves the number of transferable embryos in superovulated ewes. Theriogenology, v.55, p.777-785, 2001.), and is unknown for swine. Therefore, it can be hypothesized that the combined effects of GH on LH and IGF-I secretion can increase testicular cell proliferation and steroidogenesis, thus improving sperm production and quality.
Based on this, the aim of this study was to determine the effect of pST injections in young pigs on blood metabolites, testicular size, sperm characteristics and biochemical constituents of the seminal plasma.
MATERIALS AND METHODS
This research was approved by the Ethics Committee on Animal Experimentation of the Federal University of Pelotas (CEEA 6574).
Sixty 22-day old piglets (Landrace x Large White) were randomly assigned to one of two groups: pST (n=30) and Control (n=30). The pST group received pST injections (90µg/kg of body weight (BW) i.m.) (Reporcin, OzBioPharm Pty Ltd, Knoxfield, Vic., Australia) every three days (Rabassa et al., 2014RABASSA, V.R.; SCHWEGLER, E.; SCHNEIDER, A. et al. Effect of porcine somatotropin on metabolism and testicular characteristics of prepubertal pigs. Braz. J. Vet. Res. Anim. Sci., v.51, p.60-67, 2014.) up to 330 days of age. The Control group received placebo treatment (sodium chloride 0.9%, i.m.) at the same frequency. The pigs were weighed weekly up to 365 days of age.
Blood collections were performed weekly by venipuncture of the jugular during the period of hormonal treatments and continued for five weeks after the last injection (365 days of age). Serum was harvested after centrifugation at 3000g for 15min and frozen at - 80ºC for later analysis. Testosterone (Testosterone, NovaTec Immundiagnostica GmbH, Germany) and IGF-I (Uscn Sciences Co., Ltd., China) concentrations were evaluated by a commercial ELISA kit according to manufacturer’s instructions, and both had intra- and inter-assay coefficients of variation (CV) below 10%. IGF-I levels were measured after acid-ethanol extraction to remove IGF binding proteins and measure total IGF-I.
Glucose, cholesterol, urea, albumin, phosphorous, aspartate amino transferase (AST), gamma glutaryltransferase (GGT) (Labtest Diagnóstica S.A., Brazil) and non-esterified fatty acids (NEFA - Wako Diagnostics, USA) serum concentrations were determined by colorimetric methods following manufacturer’s instructions (Labtest Diagnóstica S.A., Brazil). CV was below 10% for all analyses.
At 22, 82, 142, 202 and 365 days of age, six animals from each group were submitted to orchiectomy surgery. Animals were sedated using 40% azaperone (Stresnil, Janssen Animal Healty, Belgium; i.m. 4mL/20kg BW). The scrotum was locally anesthetized with 2% lidocaine (Anestésico L Pearson - Laboratório Pearson Ltda, Brazil) and two scrotal incisions were performed for testicle removal. The left testicle was separated from the epididymis and weighed.
Libido evaluation began after the boars had become used to the semen collection management, which ranged from 150 days up to 210 days of age. Boars were separately taken to a semen collection room equipped with an artificial sow three times a week. Individual training sessions lasted a maximum of 10min. During each session, boar libido was classified on a 1 - 4 score scale, similar to that described by Kozink et al. (2002KOZINK, D.M.; ESTIENNE, M.J.; HARPER, A.F. et al. The effect of lutalyse on the training of sexually inexperienced boars for semen collection. Theriogenology, v.58, p.1039-1045, 2002.) where: 1 - boars showed no interest in the artificial sow; 2 - slight interest in the artificial sow but did not attempt to mount; 3 - mounted the artificial sow but did not allow semen collection; 4 - mounted the artificial sow and allowed semen collection. Duration of interest for the artificial sow was recorded in seconds. The number of attempts needed until first mount and first ejaculation were also evaluated.
Whole ejaculates were collected using the gloved-hand technique. Semen (without the gelatinous fraction) was evaluated three times a week as to appearance (samples with urine or blood were discarded), volume (mL), motility (%) and vigor (0-5 score, higher values indicates more vigorous sperm) in the period between 150 and 210 days of age (during semen collection training).
After 210 days of age, whole ejaculates (without the gelatinous fraction) were collected weekly using the gloved-hand technique up to 35 days after the last pST treatment (365 days of age) for all boars in the study. After an initial appearance evaluation (samples with urine or blood were discarded), volume (mL), pH, motility (%) and vigor (0-5 score) were evaluated. The semen was diluted 1:1 with BTS extender (Beltsville Thawing Solution, BTS; Minitüb, Tiefenbach, Germany). The sperm concentration was counted in a Neubauer chamber (x106/mL), total count (x109/ejaculate) and the semen volume needed to obtain a dose with three billion spermatozoa. The ejaculate was brought to a final volume of 100mL with addition of BTS. Semen doses were then maintained at room temperature in low light condition for 2 hours and stored at 15-18ºC in a refrigerator for 72 hours.
Semen doses were evaluated every 24 hours (0, 24, 48 and 72 hours) for motility, vigor and morphology (%) during the 72 h storage period. For this assessment, a mixture of semen and 3% formol-citrate was made and evaluated under a phase contrast microscope (1000x). Tail abnormalities, isolated head, presence of proximal and distal cytoplasmic drops and total number of morphologically normal cells were calculated. Plasma and acrosome membrane integrity and mitochondrial function were also evaluated by fluorescents probes. Sperm membrane integrity was evaluated using carboxyfluoresceindiacetate (CFDA) and propidium iodide (PI) markers as described by Harrison and Vickers (1990HARRISON, R.A.P.; VICKERS, S.E. Use of fluorescent probes to assess membrane integrity in mammalian spermatozoa. J. Reprod. Fertil., v.88, p.343-352, 1990.). To evaluate acrosomal integrity, Lectin from Arachishypogaea FITC Conjugate and PI were used as fluorescence markers. Mitochondrial function was evaluated by using the method described by Fraser et al. (2002FRASER, L.; LECEWICZ, M.; STRZEŻEK, J. Fluorometric assessments of viability and mitochondrial status of boar spermatozoa following liquid storage. Pol. J. Vet. Sci., v.5, p.85-92, 2002.) with PI and rhodamine 123 (R123). Evaluations were performed under an epifluorescent microscope (Olympus BX 51, America Inc., Sapporo, Japan) at 400x magnification (524 nm filter wave length). Two hundred sperms were counted on each slide and the membranes (acrosomal and plasmatic) were classified as intact or damaged. All evaluations were performed by the same technician.
Ejaculate aliquots were separated immediately after collection and centrifuged at 3000 x g for 15min to separate the seminal plasma, which was frozen at -80ºC for analysis. IGF-I concentration was evaluated by a commercial ELISA kit (Uscn Sciences Co., Ltd., China), after acid-ethanol extraction to remove IGF binding proteins as previously described for serum. Cholesterol, total protein (Labtest Diagnóstica S.A., Brazil) and fructose concentrations were evaluated by colorimetric methods. For fructose evaluation, 500µL seminal plasma was diluted in 950µL distilled water. The sample was deproteinized by adding 2% zinc sulphate and 0.4% sodium hydroxide and centrifuged at 2000g for 5min; the supernatant was removed, 1mL of 0.1% resorcinol and 3mL of 30% HCl were added and the solution was heated for 20min at 85ºC. The final solution was read at 450nm in a spectrophotometer. CV was below 10% for all analyses.
Statistical analysis was performed using Statistical Analysis System 9 software (SAS Institute Inc. Cary, NC, USA). Analysis of variance for repeated measures was used to examine the effect of pST injections (effect of treatment, collection and treatment x collection interaction) on testicular size, metabolic profile (testosterone, IGF-I, glucose, cholesterol, urea, albumin, AST, GGT, NEFA and phosphorus), body weight gain, testicular weight, interest time for artificial sow, sperm characteristics (volume, motility, vigor, concentration, spermatic pathologies, acrosomal and plasma membrane integrity and mitochondrial function), and seminal plasma constituents (fructose, cholesterol, total protein and IGF-I) with a Tukey test adjustment. Semen analyses and seminal plasma constituents were grouped every two weeks. The number of attempts needed for first mount and number of attempts needed for first ejaculation were evaluated by one-way ANOVA. The libido score was evaluated by Chi-square test.
RESULTS
The pST group had increased body weight gain until 210 days of age (pST: 67.1±1.0kg; Control: 64.2±1.0kg; P=0.07). After 210 days of age, body weight was not different between groups (pST: 115.4±2.5kg; Control: 110.3±2.5kg; P>0.05). In addition, the pST group had higher testicular weight than Control group (P<0.001), with significant differences only observed at 365 days of age (Figure 1).
Testicular weight (mean ± standard error of mean) of the young pigs submitted to somatotropin (pST) injection from 22 to 330 days of age.
Testosterone concentration was higher in the pST group, being higher from 181 to 239 days of age in pST treated pigs (Figure 2). Serum IGF-I concentration was not different between groups throughout the period of study (pST: 15.4±2.4ng/mL; Control: 13.0±2.4ng/mL; P>0.05). Regarding the metabolic parameters, until 210 days of age concentrations of albumin (pST: 3.5±0.1g/dL; Control: 3.3±0.1g/dL), glucose (pST: 91.7±1.3mg/dL; Control: 90.2±1.2mg/dL), cholesterol (pST: 73.0±1.7mg/dL; Control: 74.1±2.1mg/dL), urea (pST: 30.8±0.7mg/dL; Control: 32.9±0.7mg/dL), AST (pST: 21.7±1.1UI/L; Control: 21.9±1.0UI/L) and GGT (pST: 115.2±7.5UI/L; Control: 126.9±8.8UI/L) were not different between groups (P>0.05). With reference to metabolic parameters after 210 days of age, only serum albumin (pST: 4.0±0.1g/dL; Control: 3.6±0.1g/dL; P<0.001) and phosphorus (pST: 7.5±0.1mg/dL; Control: 7.0±0.1mg/dL; P=0.06) were higher in the pST than Control group. Glucose (pST: 74.9±1.4mg/dL; Control: 74.4±1.4mg/dL), cholesterol (pST: 77.5±2.0mg/dL; Control: 74.5±2.0mg/dL), urea (pST: 30.1±1.2mg/dL; Control: 29.5±1.2mg/dL), AST (pST: 24.2±1.5UI/L; Control: 26.8±1.5UI/L) and GGT (pST: 95.7±14.0UI/L; Control: 87.6±14.0UI/L) concentrations were also not different between groups after 210 days of age (P>0.05).
Testosterone concentration (ng/mL; mean ± standard error of the mean) of male pigs submitted to administration of porcine somatotropin (pST) from 22 to 330 days of age and submitted to orchiectomy at 365 days of age.
Regarding the reproductive behavior, pST treated boars had a greater number of mounts with semen collection compared to the Control boars (P<0.01) (Figure 3). Average time of interest in the artificial sow was higher for pST treated boars (87.9±6.4 sec) than that for Control ones (63.0±3.9 sec) (P<0.001). The number of attempts needed for the first mount was 6.3±1.4 for the pST group and 6.2±1.0 for the Control group (P>0.05), and the number of attempts needed for first ejaculation was 7.9±0.8 for the pST Group and 6.4±1.3 for the Control Group (P>0.05).
Effect of porcine somatotropin (pST) on libido score of the young boars from 150 to 210 days of age. Libido score: 1 - boars showed no interest in the artificial sow; 2 - slight interest in the artificial sow but did not attempt to mount; 3 - mounted the artificial sow but did not allow semen collection; 4 - mounted the artificial sow and allowed semen collection.
Seminal plasma IGF-I concentrations were higher for the pST group (pST: 99.4±4.7ng/mL; Control: 82.7±7.2ng/mL; P=0.05), although no treatment by collection interaction was observed (P>0.05). Other biochemical constituents of the seminal plasma were not different between groups: total protein (pST: 3.4±0.6g/dL; Control: 3.4±1.0g/dL; P>0.05), cholesterol (pST: 8.8±1.5mg/dL; Control: 11.5±2.4mg/dL; P>0.05) and fructose (pST: 5.8±1.2mg/mL; Control: 6.5±1.8mg/mL; P>0.05).
Regarding semen characteristics sperm motility (pST: 58.4±5.8 %; Control: 48.0±8.1 %; P>0.05), vigor (pST: 2.1±0.2; Control: 2.0±0.3; P>0.05) and volume (pST: 25.6±9.6mL; Control: 10.5±12.6mL; P>0.05) were not different between groups in the period from 150 to 210 days of age. Nevertheless, fresh and stored semen characteristics were improved by pST treatment after 210 days of age. Sperm vigor was higher for fresh semen in the pST Group (Table 1), but no effect during storage was observed (Table 2). pST treatment had no effect on fresh semen motility (Table 1); however, when semen was stored for 72 hours, lower motility was observed in the Control group after 48 hours, while semen from the pST group presented stable motility during the 72-hour storage (Table 2). Additionally, during the 72-hour storage sperm from the pST group had fewer morphological alterations and reduced plasma membrane damage (P=0.07) (Table 2). The pST group also presented lower sperm concentration, but a higher ejaculate volume and increased total sperm output. Due to this alteration, the pST group had a higher number of inseminating doses per ejaculate (Table 1).
DISCUSSION
The current work has demonstrated that GH plays a pivotal role in the steroidogenesis and spermatogenesis of boars, as previously reported for other species in literature (Hafez et al., 2005HAFEZ, Y.M.; FAWZY, S.A.; EL-HENAWY, M.A. et al. Effect of recombinant bovine somatotropin (rbST) on semen physical characteristics and some biochemical constituents in seminal plasma of Friesian bulls. Egypt. J. Anim. Prod., v.42, p.87-94, 2005.; Storer et al., 2005STORER, W.A.; THOMPSON JR, D.L.; CARTMILL, J.A. The effects of equine somatotropin on pituitary and testicular function in the stallion during the nonbreeding season. J. Equine Vet. Sci., v.25, p.106-112, 2005.; Vieira et al., 2010VIEIRA, M.B.; BIANCHI, I.; MADEIRA, E.M. et al. Effect of recombinant bovine somatotropin on plasma concentrations of insulin-like growth factor I, insulin and membrane integrity of bull spermatozoa. Reprod. Domest. Anim., v.45, p.1110-1113, 2010.). Testicular steroidogenic activity is influenced by physiological development and endocrine status, and is associated to sexual maturity (Park and Yi, 2002PARK, C.S.; YI, Y.J. Comparison of semen characteristics, sperm freezability and testosterone concentration between Duroc and Yorkshire boars during seasons. Anim. Reprod. Sci., v.73, p.53-61, 2002.). Based on the serum testosterone concentration, the present study suggests that sexual maturity occurred at an earlier age in pST treated boars, as indicated by the time of the first testosterone peak (Kumaresan et al., 2011KUMARESAN, A.; BUJARBARUAH, K.M.; KADIRVEL, G. et al. Early sexual maturity in local boars of Northeastern India: age-related changes in testicular growth, epididymal sperm characteristics and peripheral testosterone levels. Theriogenology, v.75, p.687-695, 2011.). This earlier onset of sexual maturity is further supported by the sexual behavior of boars, since pST treated boars showed more interest in the artificial sow and mounts with ejaculation also at an earlier age. The effect of pST on testosterone concentration can be the result of an increased expression of LH receptors in Leydig cells as previously reported (Chatelain et al., 1991CHATELAIN, P.G.; SANCHEZ, P.; SAEZ, J.M. Growth hormone and insulin-like growth factor i treatment increase testicular luteinizing hormone receptors and steroidogenic responsiveness of growth hormone deficient dwarf mice. Endocrinology, v.128, p.1857-1862, 1991.). Furthermore, IGF-I can directly stimulate the expression of steroidogenic acute regulatory (StAR) protein and increase steroidogenesis in the testis (Yoon and Roser, 2011YOON, M.J.; ROSER, J.F. A synergistic effect of insulin-like growth factor (IGFI) on equine luteinizing hormone (eLH)-induced testosterone production from cultured Leydig cells of horses. Anim. Reprod. Sci., v.126, p.195-199, 2011.).
pST treatment had no effect on semen quality of boars from 150 to 210 days old. Nonetheless, El-Gohary et al. (2011) observed increased sperm motility and ejaculate volume in rams treated with rbST around the establishment of puberty. In addition, pST had no effect on testicular size during this period in our study. Nevertheless, pST treatment increased testicular size after puberty establishment, corroborating with the improved semen parameters observed in the post-pubertal period. Several studies indicate that testis size is positively correlated with sperm count per ejaculate (Borg et al., 1993BORG, K.E.; LUNSTRA, D.D.; CHRISTENSON, R.K. Semen characteristics, testicular size, and reproductive hormone concentrations in mature Duroc, Meishan, Fengjing, and Minzhu boars. Biol. Reprod., v.49, p.515-521, 1993.). This is in agreement with our current findings, since pST treatment concomitantly increased testicular weight, semen volume, total sperm count and number of semen doses per ejaculate. Our observations are also in agreement with findings from Hafez et al. (2005HAFEZ, Y.M.; FAWZY, S.A.; EL-HENAWY, M.A. et al. Effect of recombinant bovine somatotropin (rbST) on semen physical characteristics and some biochemical constituents in seminal plasma of Friesian bulls. Egypt. J. Anim. Prod., v.42, p.87-94, 2005.), who observed increased ejaculate volume, mass motility, total number of spermatozoa and a decreased percentage of abnormal sperm cells for bulls treated with rbST. No differences in testosterone concentration after puberty were observed by Hafez et al. (2005), so it is believed that pST did not improve sperm quality by this pathway. The pST effect on spermatogenesis is probably due to an increased local production of IGF-I, which is significantly correlated with the percentage of morphologically normal spermatozoa and sperm motility (Glander et al., 1996GLANDER, H.J.; KRATZSCH, J.; WEISBRICH, C. et al. Insulin-like growth factor-I and α2-macroglobulin in seminal plasma correlate with semen quality. Hum. Reprod., v.11, p.2454-2460, 1996.). Therefore, improved semen quality of boars in our study can be attributed to the increased levels of IGF-I in the seminal plasma of pST treated boars. Sertoli, Leydig and peritubular cells secrete IGF-I, which stimulates DNA synthesis in spermatogenic cells and cell proliferation (Söder et al., 1992SÖDER, L.; BANG, P.; WAHAB, A. et al. Insulin-like growth factors selectively stimulate spermatogonial, but not meiotic, deoxyribonucleicacid synthesis during rat spermatogenesis. Endocrinology, v.131, p.2344-2350, 1992.), increasing the number of spermatic cells in the ejaculate. Still, the improvement of sperm motility is attributed to increased energy metabolism (Selvaraju et al., 2009SELVARAJU, S.; REDDY, I.J.; NANDI, S. et al. Influence of IGF-I on buffalo (Bubalus bubalis) spermatozoa motility, membrane integrity, lipid peroxidation and fructose uptake in vitro. Anim. Reprod. Sci., v.113, p.60-70, 2009.). However, mitochondrial function in spermatozoa and energetic parameters in seminal plasma did not vary in our study. The positive pST effect on motility is probably due to direct actions of GH or IGF-I in the spermatozoa (Henricks et al., 1998HENRICKS, D.M.; KOUBA, A.J.; LACKEY, B.R. et al. Identification of insulin-like growth factor-I in bovine seminal plasma and its receptor on spermatozoa: influence on sperm motility. Biol. Reprod., v.59, p.330-337, 1998.).
IGF-I has another important role as a seminal plasma antioxidant (Selvaraju et al., 2009SELVARAJU, S.; REDDY, I.J.; NANDI, S. et al. Influence of IGF-I on buffalo (Bubalus bubalis) spermatozoa motility, membrane integrity, lipid peroxidation and fructose uptake in vitro. Anim. Reprod. Sci., v.113, p.60-70, 2009.), protecting spermatozoa against damage in the plasmatic membrane during storage. During cold storage of boar semen between 17 and 19ºC, the sperm cell ability to resist the refrigeration process is impaired by its inability to adjust membrane fluidity, which in turn is related to integrity and changes in the lipid composition of the plasma membrane (Cerolini et al., 2000CEROLINI, S.; ARRUDA, R.P.; DE ANDRADE, A.F.C. et al. Viability, susceptibility to peroxidation and fatty acid composition of boar semen during liquid storage. Anim. Reprod. Sci., v.58, p.99-111, 2000.). As demonstrated in this study, pST treatment increased the proportion of spermatozoa with intact membranes, which is probably due to the protective IGF-I effect on the sperm membrane, previously reported in bulls (Vieira et al., 2010VIEIRA, M.B.; BIANCHI, I.; MADEIRA, E.M. et al. Effect of recombinant bovine somatotropin on plasma concentrations of insulin-like growth factor I, insulin and membrane integrity of bull spermatozoa. Reprod. Domest. Anim., v.45, p.1110-1113, 2010.). As a result, sperm motility decreased less in pST treated boars at 48 hours of storage, which contributed to increased semen quality during conservation. pST treated boars also had lower sperm defects, which can be explained by the effect of IGF-I on spermatozoa maturation, decreasing the concentration of abnormal cells, as reported before (Glander et al., 1996GLANDER, H.J.; KRATZSCH, J.; WEISBRICH, C. et al. Insulin-like growth factor-I and α2-macroglobulin in seminal plasma correlate with semen quality. Hum. Reprod., v.11, p.2454-2460, 1996.). Additionally, the previous reports of IGF-I effects on the sperm membrane integrity can lead to reduced rates of sperm defects.
In the current study, GH also influenced protein and mineral metabolism. The effect of pST on albumin concentration is attributed to its influence on liver protein turnover, as demonstrated by the decrease in albumin levels in hypophysectomised rats and its partial restoration after GH supplementation (Feldhoff et al., 1977FELDHOFF, R.C.; TAYLOR, J.M.; JEFFERSON, L.S. Synthesis and secretion of rat albumin in vivo, in perfused liver, and in isolated hepatocytes - effects of hypophysectomy and growth hormone treatment. J. Biol. Chem., v.252, p.3611-3616, 1977.). The increase in phosphorus concentration can be explained by the antiphosphaturic effect of GH mediated by IGF-I, which increases renal phosphorus reabsorption (Kopple et al., 1995KOPPLE, J.D.; DING, H.; HIRSCHBERG, R. Effects of recombinant human insulin-like growth factor 1 on renal handling of phosphorus, calcium, and sodium in normal humans. Am. J. Kidney Dis., v.26, p.818-824, 1995.). This effect can contribute to GH influence on energy metabolism, through the retention of phosphorus for ATP production, and which could be related to an increased sperm motility during storage (Lamirande and Gagnon, 1992LAMIRANDE, E.; GAGNON, C. Reactive oxygen species and human spermatozoa. II. Depletion of adenosine triphosphate plays an important role in the inhibition of sperm motility. J. Androl., v.13, p.379-386, 1992.). pST administration did not improve IGF-I blood concentrations in this study, differing from the results found by Samaras et al. (1994SAMARAS, S.E.; HAGEN, D.R.; BRYAN, K.A. et al. Effects of growth hormone and gonadotropin on the insulin-like growth factor system in the porcine ovary. Biol. Reprod., v.50, p.178-186, 1994.), suggesting that the pST effect on spermatogenesis and steroidogenesis can be mediated by an increase in local IGF-I production in this study, and be independent of endocrine effects.
CONCLUSIONS
The pST treatment of young boars increased the testosterone concentration and libido, and had a positive effect on metabolic balance. In the testicles, pST treatment increased the testicular weight and sperm quality of both fresh (vigor, volume, and total sperm concentration) and stored semen (motility, number of morphological alterations and integrity of plasmatic membrane).
ACKNOWLEDGEMENTS
This work was supported by CNPq (grant n°478385/2009-9) and FAPERGS (grant n°0904231).
REFERENCES
- BAGU, E.T.; COOK, S.; GRATTON, C.L. et al. Postnatal changes in testicular gonadotropin receptors, serum gonadotropin, and testosterone concentrations and functional development of the testes in bulls. Reproduction, v.132, p.403-411, 2006.
- BORG, K.E.; LUNSTRA, D.D.; CHRISTENSON, R.K. Semen characteristics, testicular size, and reproductive hormone concentrations in mature Duroc, Meishan, Fengjing, and Minzhu boars. Biol. Reprod., v.49, p.515-521, 1993.
- BRITO, L.F.C.; BARTH, A.D.; RAWLINGS, N.C. et al. Effect of nutrition during calfhood and peripubertal period on serum metabolic hormones, gonadotropins and testosterone concentrations, and on sexual development in bulls. Domest. Anim. Endocrinol., v.33, p.1-18, 2007.
- CEROLINI, S.; ARRUDA, R.P.; DE ANDRADE, A.F.C. et al. Viability, susceptibility to peroxidation and fatty acid composition of boar semen during liquid storage. Anim. Reprod. Sci., v.58, p.99-111, 2000.
- CHATELAIN, P.G.; SANCHEZ, P.; SAEZ, J.M. Growth hormone and insulin-like growth factor i treatment increase testicular luteinizing hormone receptors and steroidogenic responsiveness of growth hormone deficient dwarf mice. Endocrinology, v.128, p.1857-1862, 1991.
- EL-GOHARY, E.S.; ABDEL-KHALEK, E.A.; ASHMAWY, T.A.M. et al. Effect of recombinant bovine somatotropin (rbST) on growth performance and puberty incidence of male and female lambs born from rbST treated ewes. Egypt. J. Sheep Goat Sci., v.6, p.47-55, 2011.
- FELDHOFF, R.C.; TAYLOR, J.M.; JEFFERSON, L.S. Synthesis and secretion of rat albumin in vivo, in perfused liver, and in isolated hepatocytes - effects of hypophysectomy and growth hormone treatment. J. Biol. Chem., v.252, p.3611-3616, 1977.
- FINNERTY, M.; ENRIGHT, W.J.; ROCHE, J.F. Testosterone, LH and FSH episodic secretory patterns in GnRH-immunized bulls. J. Reprod. Fertil., v.114, p.85-94, 1998.
- FOLCH, J.; RAMON, J.P.; COCERO, M.J. et al. Exogenous growth hormone improves the number of transferable embryos in superovulated ewes. Theriogenology, v.55, p.777-785, 2001.
- FRASER, L.; LECEWICZ, M.; STRZEŻEK, J. Fluorometric assessments of viability and mitochondrial status of boar spermatozoa following liquid storage. Pol. J. Vet. Sci., v.5, p.85-92, 2002.
- GLANDER, H.J.; KRATZSCH, J.; WEISBRICH, C. et al. Insulin-like growth factor-I and α2-macroglobulin in seminal plasma correlate with semen quality. Hum. Reprod., v.11, p.2454-2460, 1996.
- HAFEZ, Y.M.; FAWZY, S.A.; EL-HENAWY, M.A. et al. Effect of recombinant bovine somatotropin (rbST) on semen physical characteristics and some biochemical constituents in seminal plasma of Friesian bulls. Egypt. J. Anim. Prod., v.42, p.87-94, 2005.
- HARRISON, R.A.P.; VICKERS, S.E. Use of fluorescent probes to assess membrane integrity in mammalian spermatozoa. J. Reprod. Fertil., v.88, p.343-352, 1990.
- HENRICKS, D.M.; KOUBA, A.J.; LACKEY, B.R. et al. Identification of insulin-like growth factor-I in bovine seminal plasma and its receptor on spermatozoa: influence on sperm motility. Biol. Reprod., v.59, p.330-337, 1998.
- KAMP, G.A.; WAELKENS, J.J.J.; DE MUINCK KEIZER-SCHRAMA, S.M.P.F. et al. High dose growth hormone treatment induces acceleration of skeletal maturation and an earlier onset of puberty in children with idiopathic short stature. Arch. Dis. Child., v.87, p.215-220, 2002.
- KOPPLE, J.D.; DING, H.; HIRSCHBERG, R. Effects of recombinant human insulin-like growth factor 1 on renal handling of phosphorus, calcium, and sodium in normal humans. Am. J. Kidney Dis., v.26, p.818-824, 1995.
- KOZINK, D.M.; ESTIENNE, M.J.; HARPER, A.F. et al. The effect of lutalyse on the training of sexually inexperienced boars for semen collection. Theriogenology, v.58, p.1039-1045, 2002.
- KUMARESAN, A.; BUJARBARUAH, K.M.; KADIRVEL, G. et al. Early sexual maturity in local boars of Northeastern India: age-related changes in testicular growth, epididymal sperm characteristics and peripheral testosterone levels. Theriogenology, v.75, p.687-695, 2011.
- LAMIRANDE, E.; GAGNON, C. Reactive oxygen species and human spermatozoa. II. Depletion of adenosine triphosphate plays an important role in the inhibition of sperm motility. J. Androl., v.13, p.379-386, 1992.
- PARK, C.S.; YI, Y.J. Comparison of semen characteristics, sperm freezability and testosterone concentration between Duroc and Yorkshire boars during seasons. Anim. Reprod. Sci., v.73, p.53-61, 2002.
- RABASSA, V.R.; SCHWEGLER, E.; SCHNEIDER, A. et al. Effect of porcine somatotropin on metabolism and testicular characteristics of prepubertal pigs. Braz. J. Vet. Res. Anim. Sci., v.51, p.60-67, 2014.
- SAMARAS, S.E.; HAGEN, D.R.; BRYAN, K.A. et al. Effects of growth hormone and gonadotropin on the insulin-like growth factor system in the porcine ovary. Biol. Reprod., v.50, p.178-186, 1994.
- SELVARAJU, S.; REDDY, I.J.; NANDI, S. et al. Influence of IGF-I on buffalo (Bubalus bubalis) spermatozoa motility, membrane integrity, lipid peroxidation and fructose uptake in vitro. Anim. Reprod. Sci., v.113, p.60-70, 2009.
- SIROTKIN, A.V. Control of reproductive processes by growth hormone: extra- and intracellular mechanisms. Vet. J., v.170, p.307-317, 2005.
- SJOGREN, I.; JONSSON, M.; MADEJ, A. et al. Effects of very high doses of human growth hormone (hGH) on the male reproductive system in the dog. Andrologia, v.30, p.37-42, 1998.
- SÖDER, L.; BANG, P.; WAHAB, A. et al. Insulin-like growth factors selectively stimulate spermatogonial, but not meiotic, deoxyribonucleicacid synthesis during rat spermatogenesis. Endocrinology, v.131, p.2344-2350, 1992.
- STORER, W.A.; THOMPSON JR, D.L.; CARTMILL, J.A. The effects of equine somatotropin on pituitary and testicular function in the stallion during the nonbreeding season. J. Equine Vet. Sci., v.25, p.106-112, 2005.
- SWANLUND, D.J.; N’DIAYE, M.R.; LOSETH, K.J. et al. Diverse testicular responses to exogenous growth hormone and follicle-stimulating hormone in prepubertal boars. Biol. Reprod., v.53, p.749-757, 1995.
- VIEIRA, M.B.; BIANCHI, I.; MADEIRA, E.M. et al. Effect of recombinant bovine somatotropin on plasma concentrations of insulin-like growth factor I, insulin and membrane integrity of bull spermatozoa. Reprod. Domest. Anim., v.45, p.1110-1113, 2010.
- YOON, M.J.; ROSER, J.F. A synergistic effect of insulin-like growth factor (IGFI) on equine luteinizing hormone (eLH)-induced testosterone production from cultured Leydig cells of horses. Anim. Reprod. Sci., v.126, p.195-199, 2011.
Publication Dates
-
Publication in this collection
Jan-Feb 2018
History
-
Received
30 Sept 2016 -
Accepted
18 Apr 2017


 pST Group (n=30): treated with pST every three days up to 330 days of age; Control Group (n=30): received placebo treatment at the same frequency; Collection: time of orchiectomy; Group x Collection: analyze the interaction between group and collection. a,b P<0.05.
pST Group (n=30): treated with pST every three days up to 330 days of age; Control Group (n=30): received placebo treatment at the same frequency; Collection: time of orchiectomy; Group x Collection: analyze the interaction between group and collection. a,b P<0.05.
 pST Group (n=6): treated with pST every three days up to 330 days of age; Control Group (n=6): received placebo treatment at the same frequency; Collection: blood collection used to determination of testosterone levels; Group x Collection: analyze the interaction between group and collection. * Groups P<0.05.
pST Group (n=6): treated with pST every three days up to 330 days of age; Control Group (n=6): received placebo treatment at the same frequency; Collection: blood collection used to determination of testosterone levels; Group x Collection: analyze the interaction between group and collection. * Groups P<0.05.
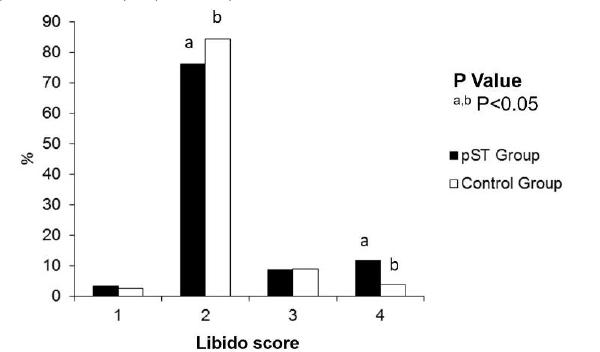 pST Group (n=12): treated with pST every three days up to 330 days of age; Control Group (n=12): received placebo treatment at the same frequency. pST group libido score (%): 1=3.25; 2=76.22; 3=8.79; 4=11.72. Control group libido score (%): 1=2.57; 2=84.56; 3=9.00; 4=3.85. a,b P<0.05.
pST Group (n=12): treated with pST every three days up to 330 days of age; Control Group (n=12): received placebo treatment at the same frequency. pST group libido score (%): 1=3.25; 2=76.22; 3=8.79; 4=11.72. Control group libido score (%): 1=2.57; 2=84.56; 3=9.00; 4=3.85. a,b P<0.05.