ABSTRACT
Vomiting and diarrhea are two important clinical signs that can cause significant electrolytic and acid-base imbalances. The purposes of this study were to characterize hydric, electrolytic and acid-base disorders presented by puppies with hemorrhagic gastroenteritis and to compare the traditional and quantitative approaches to acid-base status interpretation. Sixty-one animals with a history of vomiting and/or diarrhea were used in this study and the following tests were performed: complete blood count, total plasma protein concentration and hemogasometry. Mean, standard deviation and Kappa values were calculated. The imbalances characterized by both approaches were: 42 (69%) animals without imbalance, 17 (28%) with metabolic alkalosis and 2 (3%) with metabolic acidosis by the traditional approach and 17 (28%) dogs without imbalance, 26 (43%) with metabolic alkalosis and 18 (29%) with metabolic acidosis by the quantitative approach. The agreement calculated between two approaches coincide in 28 cases, with a moderate Kappa value equivalent to 0.459. The most found imbalances were metabolic alkalosis, hypokalemia, and mild dehydration. Most of acid-base disturbances were not identified by the traditional approach, whereas by the quantitative approach, they were easily determined. Thus quantitative approach proved to be superior in identification of possible acid-base imbalances.
Keywords:
acid-base disorders; dehydration; strong ion model
RESUMO
Vômito e diarreia são dois sinais clínicos importantes, que podem causar desequilíbrios eletrolíticos e ácido-base importantes. Os objetivos deste trabalho foram caracterizar os desequilíbrios hídrico, eletrolítico e ácido-base apresentados por filhotes de cães com gastroenterite hemorrágica e comparar as abordagens de interpretação do equilíbrio ácido-base tradicional e quantitativa. Sessenta e um animais com histórico de vômito e/ou diarreia foram utilizados neste estudo, e os seguintes testes foram realizados: hemograma, concentração de proteína total plasmática e hemogasometria. Média, desvio-padrão e valor de Kappa foram calculados. Os desequilíbrios caracterizados pelas duas abordagens foram: 42 (69%) animais sem desequilíbrio, 17 (28%) com alcalose metabólica e dois (3%) com acidose metabólica, pela abordagem tradicional, e 17 (28%) cães sem desequilíbrio, 26 (43%) com alcalose metabólica e 18 (29%) com acidose metabólica, pela abordagem quantitativa. A concordância calculada entre as duas abordagens coincidiu em 28 casos, com um valor de Kappa moderado, equivalente a 0,459. Os desequilíbrios mais encontrados foram alcalose metabólica, hipocalemia e desidratação leve. A maioria dos distúrbios não foram identificados pela abordagem tradicional, enquanto, pela abordagem quantitativa, foram facilmente determinados. Portanto, a abordagem quantitativa provou-se superior na identificação de possíveis desequilíbrios ácido-base.
Palavras-chave:
desidratação; distúrbios ácido-base; modelo de íons fortes
INTRODUCTION
Hemorrhagic gastroenteritis in puppies can have several etiologies such as parasitic, bacterial and viral. Among viral diseases canine coronavirus (CCoV) and canine parvovirus (CPV) are the most common causes and usually affect dogs up to one year old, with high morbidity and mortality in puppies from three to six months of age and not vaccinated (Ghiggi et al., 2013GHIGGI, E.; PADILHA, V.S.; MORAES, A.N. et al. Reposição volêmica com hidroxietilamido ou solução de ringer lactato em cães com gastroenterite hemorrágica por parvovírus. Semin. Cienc. Agrar., v.34, p.1783-1791, 2013.; Duijvestijn et al., 2016DUIJVESTIJN, M.; MUGHINI-GRAS, L.; SCHUURMAN, N. et al. Enteropathogen infections in canine puppies: (co-)occurrence, clinical relevance and risk factors. Vet. Microbiol., v.195, p.115-122, 2016.). Clinical diagnosis is based on the history, clinical signs of vomiting and/or bloody diarrhea, and laboratory changes such as leukopenia due to neutropenia.
Diarrhea due to infectious diseases can result in serious consequences to the health of puppies and it may lead to death in cases associated with severe dehydration (Duijvestijn et al., 2016DUIJVESTIJN, M.; MUGHINI-GRAS, L.; SCHUURMAN, N. et al. Enteropathogen infections in canine puppies: (co-)occurrence, clinical relevance and risk factors. Vet. Microbiol., v.195, p.115-122, 2016.) and metabolic acidosis is the expected acid-base imbalance in these cases (Tello and Perez-Freytes, 2017TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.), initially due to a higher loss of sodium ions (Na⁺) than chlorides (Cl-), with consequent decrease in the strong ions difference (SID) (Navarro et al., 2005NAVARRO, M.; MONREAL, L.; SEGURA, D. et al. A comparison of traditional and quantitative analysis of acid-base and electrolyte imbalances in horses with gastrointestinal disorders. J. Vet. Intern. Med., v.19, p.871-877, 2005.; Müller et al., 2012MÜLLER, K.R.; GENTILE, A.; KLEE, W.; CONSTABLE, P.D. Importance of the effective strong ion difference of an intravenous solution in the treatment of diarrheic calves with naturally acquired acidemia and strong ion (metabolic) acidosis. J. Vet. Intern. Med., v.26, p.674-683, 2012.). Simultaneously, bicarbonate (HCO3 -) present in pancreatic juice secretion in the duodenum is not reabsorbed in the intestine and is lost along with Na+ in feces (Brown e Otto, 2008BROWN, A.J.; OTTO, C.M. Fluid therapy in vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.653-675, 2008.; Trefz et al., 2012TREFZ, F.M.; LORCH, A.; FEIST, M. et al. Metabolic acidosis in neonatal calf diarrhea-clinical findings and theoretical assessment of a simple treatment protocol. J. Vet. Intern. Med., v.26, p.162-170, 2012.; Tello e Perez-Freytes, 2017).
To evaluate dogs with vomiting and/or diarrhea the degree of dehydration should be estimated as well as performing the hemogasometric and an electrolytic examination in order to establish adequate fluid therapy (Brown and Otto, 2008BROWN, A.J.; OTTO, C.M. Fluid therapy in vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.653-675, 2008.). By hemogasometry, one can access the acid-base status of the animal to know if it has metabolic acidosis/alkalosis.
The traditional approach is based on the Henderson-Hasselbach equation, it considers pH as a dependent variable while partial pressure of carbon dioxide (pCO2) and concentration of HCO3 - as independent variables. Therefore, it does not consider concentrations of electrolytes and proteins in maintaining acid-base status (Gomez and Kellum, 2015GOMEZ, H.; KELLUM, J.A. Understanding acid base disorders. Crit. Care Clin., v.31, p.849-860, 2015.). It is very important to take in consideration these last two variables since they are often altered in cases of gastrointestinal disorders, also the use of other methods with more variables have more accurate/specific results.
The quantitative approach, known as Stewart's Strong Ion Model, establishes that blood pH depends on pCO2, Strong Ion Difference (SID) and concentration of non-volatile weak acids (Atot) - determined by plasmatic proteins and phosphate concentrations - and this approach may explain changes in acid-base status that the traditional approach is unable to answer, for example: animals with altered electrolytes levels might have a normal pH and concomitant disorders may remain unnoticed by clinicians (Hopper et al., 2014aHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a., 2014b).
The purposes of this study were to characterize the hydric, electrolytic and acid-base disorders presented by dogs with hemorrhagic gastroenteritis and to compare results from traditional (Henderson-Hasselbach equation) and quantitative (Stewart's Strong Ion Model) approaches.
MATERIAL AND METHODS
Samples were collected from 61 dogs under one year old, males and females, presenting clinical signs and laboratory changes of hemorrhagic gastroenteritis at the time of admission to the Veterinary Hospital, in the period from March to December 2016, with a consent form signed by the guardians. This study was approved by the Institutional Ethical Committee in the Use of Animals (CEUA / UEL), under protocol number 21447,2015,64.
Inclusion criteria were: dogs with age under one year old, presenting clinical signs of vomiting and/or hemorrhagic diarrhea, dehydration and leukopenia (<6,000/mm3). Severe leukopenia was present when total leukocytes were <1,000/mm3, absolute neutrophil count less than 3,000/mm³ was considered as neutropenia and absolute lymphocyte count below 1,000/mm³ was considered as lymphopenia (Feldman et al., 2000FELDMAN, B.F.; ZINKL, J.G.; JAIN, N.C. Schalm’s veterinary hematology. 5.ed. Baltimore: Lippincott Williams & Wilkins, 2000. 1206p.). Exclusion criteria were the manifestation of coexisting respiratory compromising and animals over one-year-old age.
Blood samples were obtained by jugular vein puncture immediately before intravenous hydration and stored in EDTA commercial tubes and in pre-heparinized syringes, which were subsequently sealed in order to prevent equilibrium with atmospheric air and promptly sent to the laboratory for analysis.
A Complete Blood Cell count (CBC) was performed in automated analyzer, (poCH-100 iV Diff®; Sysmex) for global red and white cell counts, PCV, HGB, MCV, MCH, MCHC and a blood smear was stained with Diff-Quick stain for hematology in order to obtain leukocyte differential and manual platelet estimation; Total plasmatic protein (TPP) concentration was measured in an Attago® refractometer.
Hemogasometric exams were performed on blood gas analyzer (Omni C; Roche). Variables analyzed were: pH, partial carbon dioxide pressure (pCO2), base excess (BE), concentrations of bicarbonate (HCO3 -), sodium (Na+), potassium (K+) and chloride (Cl-), Anion Gap (AG) calculated by the formula (Na+ + K+) - (Cl- + HCO3 -), SID calculated by the formula (Na+ + K+ - Cl-) and Atot values were calculated according to the formula [Total Protein (g / L)] X 0.27 (Constable and Stämpfli, 2005CONSTABLE, P.D.; STÄMPFLI, H.R. Experimental determination of net protein charge and a(tot) and k(a) of nonvolatile buffers in canine plasma. J. Vet. Intern. Med., v.19, p.507-514, 2005.).
Total plasmatic protein values below 6.0g/dL were classified as hypoproteinemia (Feldman et al., 2000FELDMAN, B.F.; ZINKL, J.G.; JAIN, N.C. Schalm’s veterinary hematology. 5.ed. Baltimore: Lippincott Williams & Wilkins, 2000. 1206p.). Atot range reference value for dogs was determined and stipulated by Constable and Stämpfli (2005CONSTABLE, P.D.; STÄMPFLI, H.R. Experimental determination of net protein charge and a(tot) and k(a) of nonvolatile buffers in canine plasma. J. Vet. Intern. Med., v.19, p.507-514, 2005.) as 8.8 - 26mM. The Atot above reference interval indicates metabolic alkalosis and an increase in Atot suggests metabolic acidosis. The degree of dehydration was classified according to DiBartola (2012).
Regarding electrolyte concentrations, values above 155mmol/L Na+ were considered hypernatremia and below 140mmol/L, hyponatremia. For K+, concentrations below 3.7mmol/L were classified as hypokalemia, and Cl- concentrations above 120mmol/L hyperchloremia and less than 105mmol/L, hypochloremia (Kaneko et al., 2008KANEKO, J.; HARVEY, J.; BRUSS, M. Clinical biochemistry of domestic animals. 6.ed. San Diego: Academic Press, 2008. 928p.). As for the classification of acid-base disorders, values are presented in Table 1 for both approaches.
Statistical analysis of obtained data was performed using statistical software (RStudio© Version 1.0.136), and the mean and standard deviation were calculated. The Shapiro-Wilk normality test was performed and data distribution was considered normal when P< 0.05. Agreement between approaches was evaluated using the Kappa coefficient and a value between 0.4 and 0.6 was considered moderate. The α considered 0.05 was used to rule out random agreement.
RESULTS
Mean and standard deviation values for pCO2, pH, HCO3 -, BE ecf, Na+, K+, Cl-, SID, AG, TPP and Atot of dogs with hemorrhagic gastroenteritis according to the acid-base imbalance identified by both approaches are presented in Table 2. Regarding the classification of hydration levels evaluated, mild dehydration was identified in 24/61 (39.4%) dogs, 16/61 (26.2%) animals had moderate dehydration, 8/61 (13.1%) were severely dehydrated, and 13/61 (21.3%) dogs showed no signs of dehydration. Only 2/61 (3.2%) dogs had hypoproteinemia while the rest of the animals had protein values within the reference interval.
Electrolyte changes found were: hypokalemia 44/61 (72.1%), hypochloremia 19/61 (31.1%), hyperchloremia 1/61 (1.6%), hyponatremia 14/61 (22.9%) and hypernatremia 4/61 (6.5%).
The 44 dogs with hypokalemia either had no imbalances (66%) or had metabolic acidosis (4.5%) or alkalosis (29.5%) by the traditional approach. By the quantitative approach, the dogs had no imbalances (22.7%%) or were identified with metabolic acidosis (31.8%) or alkalosis (45.5%).
Regarding the 19 animals with hypochloremia, by the traditional approach it was not possible to identify disturbances (52.6%) or they were diagnosed with metabolic acidosis (10.5%) or alkalosis (39.9%). Using the quantitative approach, the dogs presented no imbalances (10.5%) or had metabolic acidosis (31.6%), but most of them had metabolic alkalosis (57.9%).
When comparing the two approaches, animals presenting normal acid-base status in the traditional approach were 42/61 (68.8%), whereas by the quantitative approach only 17/61 (27.8%) had no alterations. Two (2/61) (3.2%) dogs were diagnosed with metabolic acidosis by the traditional approach whereas quantitative approach revealed 18/61 (29.5%) animals. Metabolic alkalosis was the most identified acid-base disturbance, with 17/61 (27.8%) by traditional approach and 26/61 (42.6%) by quantitative approach.
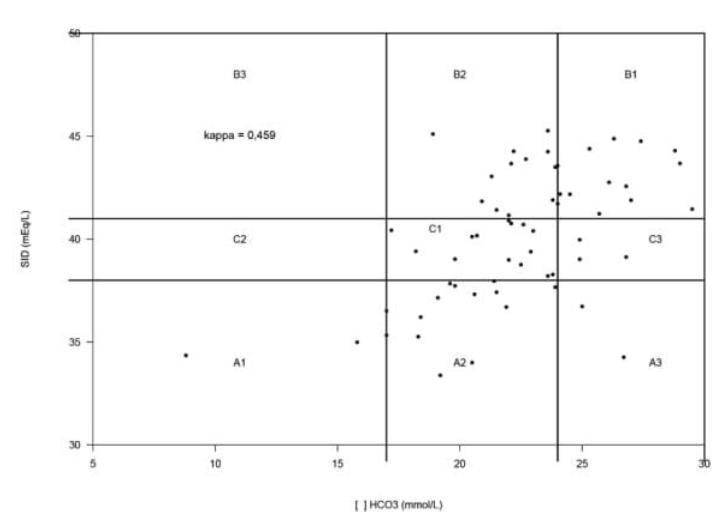
In total 19/61 (31%) imbalances were identified by the traditional approach against 44/61 (72%) disturbances detected through the quantitative approach. Kappa calculated between two approaches for these 61 animals was 0.459, meaning that in 28 cases (45.9%) the two approaches agreed in identifying the presence of imbalances and to classify them as normal acid-base status, metabolic acidosis or metabolic alkalosis as shown in Figure 1.
Comparison graphic of quantitative (SID) and traditional (HCO3 -) approach classifications of 61 dogs with hemorrhagic gastroenteritis attended at the Veterinary Hospital of Londrina State University, Londrina, PR, Brazil, 2016.
Respiratory acidosis was identified in 12/61 (19.6%) animals, in both approaches, as a compensatory mechanism to metabolic alkalosis, however, three dogs presented alkalemia. Only one animal had compensatory respiratory alkalosis as effective response from metabolic acidosis condition by both approaches, while seven dogs exhibited acidemia without compensation.
Of the studied animals, 14/61 (22.9%) died and severe leukopenia was present in these cases. Of these 14 dogs, using the traditional approach: one animal had metabolic acidosis, 5 dogs had metabolic alkalosis and 9 had no imbalances identified. Regarding the quantitative approach: 5 animals had metabolic acidosis, 7 had metabolic alkalosis and 2 had no changes in the acid-base status.
DISCUSSION
It is well known that the traditional approach has reliable results only when proteins, phosphate, and electrolyte concentrations are not altered. This does not occur in virtually all sick animals, highlighting the importance of knowing other methods to aid in the diagnosis of possible acid-base imbalances in these animals (Hopper and Haskins, 2008HOPPER, K.; HASKINS, S.C. A case-based review of a simplified quantitative approach to acid-base analysis. J. Vet. Emerg. Crit. Care, v.18, p.467-476, 2008.; Hopper et al., 2014a, 2014b; Torrente et al., 2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.).
A1: metabolic acidosis by both approaches; A2: metabolic acidosis by quantitative and no imbalances by traditional; A3: metabolic acidosis by quantitative and metabolic alkalosis by traditional; B1: metabolic alkalosis by both approaches; B2: metabolic alkalosis by quantitative and no imbalances by traditional; B3: metabolic alkalosis by quantitative and metabolic acidosis by traditional; C1: no imbalances by both approaches; C2: no imbalances by quantitative and metabolic acidosis by traditional; C3: no imbalances by quantitative and metabolic alkalosis by traditional;
It was found that metabolic alkalosis, hypokalemia, and mild dehydration were the most frequently observed imbalances in the analyzed dogs. The identification of disturbances by the quantitative approach was superior to the traditional approach, which does not take into account concentrations of total proteins and electrolytes for determination of acid-base status (Hopper; Epstein, 2012HOPPER, K.; EPSTEIN, S.E. Incidence, nature, and etiology of metabolic acidosis in dogs and cats. J. Vet. Intern. Med., v.26, p.1107-1114, 2012.; Hopper et al., 2014a; Gomez and Kellum, 2015GOMEZ, H.; KELLUM, J.A. Understanding acid base disorders. Crit. Care Clin., v.31, p.849-860, 2015.).
In terms of hydric imbalances, severity depends on duration, type, and intensity of vomiting and/or diarrhea (Boag et al., 2005BOAG, A.K.; COE, R.J.; MARTINEZ, T.A.; HUGHES, D. Acid-base and electrolyte abnormalities in dogs with gastrointestinal foreign bodies. J. Vet. Intern. Med., v.19, p.816-821, 2005.; Tello and Perez-Freytes, 2017TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.). Thirty-nine (64%) dogs had a history of vomiting and/or diarrhea for less than two days, hence mild dehydration is compatible with the period when animals showed clinical signs.
As proteins are weak acids, changes in their concentrations can directly affect the acid-base status and an excess of water can cause relative hypoproteinemia, whereas a water deficit can promote a relative hyperproteinemia. Hypoproteinemia promotes the development of metabolic alkalosis because the decrease in Atot determines a secondary increase in HCO3 - concentrations (Boag et al., 2005BOAG, A.K.; COE, R.J.; MARTINEZ, T.A.; HUGHES, D. Acid-base and electrolyte abnormalities in dogs with gastrointestinal foreign bodies. J. Vet. Intern. Med., v.19, p.816-821, 2005.; Meltesen and Bohn, 2012MELTESEN, H.S.; BOHN, A.A. Using corrected serum chloride and predicted bicarbonate concentrations to interpret acid-base status in dogs. Vet. Clin. Pathol., v.41, p.509-517, 2012.; Constable, 2014CONSTABLE, P.D. Acid-base assessment. Vet. Clin. N. Am. Food Anim. Pract., v.30, p.295-316, 2014.; Torrente et al., 2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.). The decrease of 1g/L in total protein concentration leads to a decrease of 2.5mEq/L in AG of dogs with plasma pH close to 7.4 (Constable and Stämpfli, 2005; Morais et al., 2008MORAIS, H.A.; BACH, J.F.; DIBARTOLA, S.P. Metabolic acid-base disorders in the critical care unit. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.559-574, 2008.).
Alkalosis due to hypoproteinemia can be expected, as well as acidosis due to loss of Na+ and HCO3 - ions (Morais, et al., 2008MORAIS, H.A.; BACH, J.F.; DIBARTOLA, S.P. Metabolic acid-base disorders in the critical care unit. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.559-574, 2008.; Torrente et al., 2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.), but in cases of vomiting and diarrhea the identification of acid-base imbalances is more complex and multifactorial (Burchell et al., 2014BURCHELL, R.K.; SCHOEMAN, J.P.; LEISEWITZ, A.L. The central role of chloride in the metabolic acid-base changes in canine parvoviral enteritis. Vet. J., v.200, p.152-156, 2014.; Hopper et al., 2014aHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a., 2014b).
The 2 animals with hypoproteinemia were in the group with normal acid-base status by traditional and quantitative approaches. They had vomiting and diarrhea, so the normal acid-base status can indicate that there was loss of Cl- and HCO3 - along with proteins, which were responsible for not causing apparent changes. Another possibility is that the hypoproteinemia is masked by the dehydration that this disease can cause, therefore, few hypoproteinemia cases were identified in this study.
In addition, water loss can cause alkalosis by increasing SID and, at the same time, dehydration may cause a relative increase in protein concentration (Atot) leading to acidosis. Or, instead, alkalosis may occur due to significant albumin losses in cases of diarrhea (Burchell et al., 2014BURCHELL, R.K.; SCHOEMAN, J.P.; LEISEWITZ, A.L. The central role of chloride in the metabolic acid-base changes in canine parvoviral enteritis. Vet. J., v.200, p.152-156, 2014.).
While the majority of the animals were classified with no disturbance in the traditional approach, the quantitative approach was able to identify a greater number of imbalances in the evaluated dogs. Electrolyte and acid-base imbalances are expected in both vomiting and diarrhea because they are fluids rich in electrolytes (Brown and Otto, 2008BROWN, A.J.; OTTO, C.M. Fluid therapy in vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.653-675, 2008.; Tello and Perez-Freytes, 2017TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.).
Chloride is one of the most important and useful electrolytes for interpreting acid-base status (Meltesen and Bohn, 2012MELTESEN, H.S.; BOHN, A.A. Using corrected serum chloride and predicted bicarbonate concentrations to interpret acid-base status in dogs. Vet. Clin. Pathol., v.41, p.509-517, 2012.), with hypochloremia being the second most detected alteration and present in animals with more severe clinical conditions, as well as in the study of Burchell et al. (2014BURCHELL, R.K.; SCHOEMAN, J.P.; LEISEWITZ, A.L. The central role of chloride in the metabolic acid-base changes in canine parvoviral enteritis. Vet. J., v.200, p.152-156, 2014.). Hyperchloremia presented by one dog in this study demonstrates that acidosis in this animal is due to higher loss of sodium ions than chloride, culminating with hyperchloremic acidosis (Navarro et al., 2005NAVARRO, M.; MONREAL, L.; SEGURA, D. et al. A comparison of traditional and quantitative analysis of acid-base and electrolyte imbalances in horses with gastrointestinal disorders. J. Vet. Intern. Med., v.19, p.871-877, 2005.; Müller et al., 2012MÜLLER, K.R.; GENTILE, A.; KLEE, W.; CONSTABLE, P.D. Importance of the effective strong ion difference of an intravenous solution in the treatment of diarrheic calves with naturally acquired acidemia and strong ion (metabolic) acidosis. J. Vet. Intern. Med., v.26, p.674-683, 2012.).
Low concentrations of chloride are often associated with vomiting losses and variation in hypochloremia degree is determined by the severity of vomiting episodes and the period in which they occur (Boag et al., 2005BOAG, A.K.; COE, R.J.; MARTINEZ, T.A.; HUGHES, D. Acid-base and electrolyte abnormalities in dogs with gastrointestinal foreign bodies. J. Vet. Intern. Med., v.19, p.816-821, 2005.; Brown and Otto, 2008BROWN, A.J.; OTTO, C.M. Fluid therapy in vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.653-675, 2008.; Tello and Perez-Freytes, 2017TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.). Regarding the electrolyte disorders found, the hypochloremia was the second most frequent electrolyte abnormality, 58% of the animals with hypochloremia had concurrent hypochloremic metabolic alkalosis.
Decreases in potassium levels occur due to gastrointestinal losses and through movement from extracellular fluid to intracellular fluid in cases of metabolic alkalosis that can be caused by hypochloremia. In these cases, corrections with potassium chloride (KCl) are more efficient to revert the metabolic disturbance (Hopper and Epstein, 2012HOPPER, K.; EPSTEIN, S.E. Incidence, nature, and etiology of metabolic acidosis in dogs and cats. J. Vet. Intern. Med., v.26, p.1107-1114, 2012.; Tello and Perez-Freytes, 2017TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.).
By the traditional approach, despite having had hypokalemia, no acid-base disturbances were found in most of the animals. When clinicians tried to identify some acid-base disorder they had fewer probabilities to find it by this approach, which is a misleading diagnostic (Hopper et al., 2014aHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a.). The hypokalemia might be an alert for the clinicians that all is not well with the animal, and that they must investigate deeper the cause of this alteration.
In the traditional approach, the use of AG to aid in the identification of acid-base disorders is indicated. But in some cases the AG is not very accurate, because its value can suffer interferences from proteins and phosphate concentrations. The AG is important in cases of increased unmeasured anions as ketoacidosis, lactic acidosis and in cases of hyperchloremic acidosis (Artero, 2017ARTERO, C.T. A quick reference on anion gap and strong ion gap. Vet Clin N. Am Small Anim Pract., v.47, p.191-196, 2017.). In this study the AG did not exceed the reference interval and did not provide additional information to discover the cause of the disturbances, because the phosphate and protein concentrations were not altered in these animals.
Clinical sign manifestations are important because they determine what imbalance the animal will develop. Dogs with vomiting and diarrhea had either metabolic alkalosis (25), metabolic acidosis (16) or had no acid-base imbalances (12). In this last case, possibly due to annulment of antagonistic imbalances caused by vomiting and diarrhea (Morais et al., 2008MORAIS, H.A.; BACH, J.F.; DIBARTOLA, S.P. Metabolic acid-base disorders in the critical care unit. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.559-574, 2008.).
Three animals presented only vomiting as a clinical sign, without electrolyte or acid-base imbalances, a fact attributed to one-day time evolution and less obvious changes. Five dogs with diarrhea as only clinical sign had both metabolic acidosis (4) and metabolic alkalosis (1).
The traditional approach does not enable the identification of the cause of imbalances and also does not explain the interference of electrolytes and plasmatic proteins in the maintenance of pH. Consequently, changes in these parameters are frequently not identified in sick animals (Constable, 2000CONSTABLE, P.D. Clinical assessment of acid-base status: comparison of the henderson-hasselbalch and strong ion approaches. Vet. Clin. Pathol., v.29, p.115-128, 2000., 2014; Müller et al., 2012MÜLLER, K.R.; GENTILE, A.; KLEE, W.; CONSTABLE, P.D. Importance of the effective strong ion difference of an intravenous solution in the treatment of diarrheic calves with naturally acquired acidemia and strong ion (metabolic) acidosis. J. Vet. Intern. Med., v.26, p.674-683, 2012.; Hopper et al., 2014aHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a.; Torrente et al., 2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.; Gomez and Kellum, 2015GOMEZ, H.; KELLUM, J.A. Understanding acid base disorders. Crit. Care Clin., v.31, p.849-860, 2015.).
The quantitative approach provides a better characterization of acid-base imbalances in dogs with hemorrhagic gastroenteritis (Burchell et al., 2014BURCHELL, R.K.; SCHOEMAN, J.P.; LEISEWITZ, A.L. The central role of chloride in the metabolic acid-base changes in canine parvoviral enteritis. Vet. J., v.200, p.152-156, 2014.). In this study, the number of animals detected with some type of acid-base imbalance by this approach was superior to the traditional approach. As the quantitative approach takes into consideration the electrolyte concentrations as well as proteins, it is a more accurate method to identify any kind of disturbance and can provide a better idea of what is happening with the animal. Most of the animals studied had metabolic alkalosis by quantitative approach and presented vomiting and/or diarrhea, therefore the development of hypokalemia is compatible with the results in this study.
Agreement found between approaches in this study was slightly superior (45.9%) to Torrente et al. (2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.) in which imbalances identified coincided in 32/105 (30.5%) cases. That is because all animals analyzed in the study performed by Torrente et al. (2014) had hypoproteinemia as inclusion criteria, whereas in this study only 2/61 (3.2%) had low protein concentrations. Although few dogs had hypoproteinemia, the electrolyte changes have a greater impact on the acid-base balance in cases of gastrointestinal disorders of this study.
While there is a greater agreement between approaches in this study compared to Torrente et al. (2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.), the Kappa value found in this study is moderate and shows the lower ability of the traditional approach to find acid-base and electrolyte disturbances, while the quantitative approach was able to provide that information in most of the cases. This reinforces the importance of using the quantitative approach, even when there is no protein concentration change because electrolytic changes are also important in maintaining the acid-base status.
As stated, the traditional approach does not identify many changes, demonstrating its low capacity in observation of imbalances involved in cases of gastroenteritis (Hopper et al., 2014aHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a.). Yet in this study, the quantitative approach made possible to identify metabolic alkalosis in the hypoproteinemic animals.
Even if the etiological diagnosis has not been made, the results of this study show the importance of electrolytic and acid-base status evaluation in the choice of adequate fluid therapy to treat consequences that vomiting and diarrhea can induce.
As previously mentioned, the traditional approach does not take into account electrolyte concentrations in the classification of acid-base imbalances. Therefore it fails to detect changes in the acid-base status of sick animals, demonstrating its poor ability to observe imbalances in cases of gastroenteritis, which have underlying mechanisms causing abnormal acid-base disturbances.
The quantitative method allows the clinicians to identify more complex disturbances because it characterizes the electrolytes and protein changes and enables a more adequate treatment that might reduce or optimize the hospitalization time.
In this study, most of the dogs that died had an alteration in SID and that might be connected with the death of these animals, but a correlation analysis was not performed. It would be interesting to do this analysis in future studies, to determine if the SID can be a predictive factor in mortality, once SID is more efficient in identifying acid-base disturbances than the traditional approach.
Lactate, phosphate, and albumin concentrations were not measured and were considered as limitations for this study. Since they interfere in AG, it would be interesting to measure this metabolite and make adjustments for this variable to have a more accurate AG value as was done in previous studies (Constable and Stämpfli, 2005CONSTABLE, P.D.; STÄMPFLI, H.R. Experimental determination of net protein charge and a(tot) and k(a) of nonvolatile buffers in canine plasma. J. Vet. Intern. Med., v.19, p.507-514, 2005.; Meltesen and Bohn, 2012MELTESEN, H.S.; BOHN, A.A. Using corrected serum chloride and predicted bicarbonate concentrations to interpret acid-base status in dogs. Vet. Clin. Pathol., v.41, p.509-517, 2012.; Hopper et al., 2014bHOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 2: comparison of anion gap, strong ion gap, and semiquantitative analysis. J. Vet. Emerg. Crit. Care, v.24, p.502-508, 2014b., 2014a; Torrente et al., 2014TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.). Although lactate has not been measured the SID is not affected by it, since in this study the SID was calculated only with Na+, K+ and Cl- values.
CONCLUSION
The most common imbalances in this study were identified as: metabolic alkalosis, hypokalemia, and mild dehydration. Intensity and duration of clinical signs of vomiting and diarrhea must be taken into consideration since the severity of the disturbances varies in function of them. Animals presenting vomiting and diarrhea should be evaluated according to the quantitative approach since it enables the identification of a greater number of disturbances and makes it possible to clarify the cause of small alterations in acid-base status because it considers the concentrations of proteins and electrolytes.
REFERENCES
- ARTERO, C.T. A quick reference on anion gap and strong ion gap. Vet Clin N. Am Small Anim Pract., v.47, p.191-196, 2017.
- BOAG, A.K.; COE, R.J.; MARTINEZ, T.A.; HUGHES, D. Acid-base and electrolyte abnormalities in dogs with gastrointestinal foreign bodies. J. Vet. Intern. Med., v.19, p.816-821, 2005.
- BROWN, A.J.; OTTO, C.M. Fluid therapy in vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.653-675, 2008.
- BURCHELL, R.K.; SCHOEMAN, J.P.; LEISEWITZ, A.L. The central role of chloride in the metabolic acid-base changes in canine parvoviral enteritis. Vet. J., v.200, p.152-156, 2014.
- CAMPBELL, A.; CHAPMAN, M. Handbook of poisoning in dogs and cats. Wiley-Blackwell, 2000. 272p.
- CONSTABLE, P.D. Acid-base assessment. Vet. Clin. N. Am. Food Anim. Pract., v.30, p.295-316, 2014.
- CONSTABLE, P.D. Clinical assessment of acid-base status: comparison of the henderson-hasselbalch and strong ion approaches. Vet. Clin. Pathol., v.29, p.115-128, 2000.
- CONSTABLE, P.D.; STÄMPFLI, H.R. Experimental determination of net protein charge and a(tot) and k(a) of nonvolatile buffers in canine plasma. J. Vet. Intern. Med., v.19, p.507-514, 2005.
- DIBARTOLA, S.P. Fluid, electrolyte and acid-base disorders in small animal practice. 4.ed. St. Louis: Saunders, 2012. 744p.
- DUIJVESTIJN, M.; MUGHINI-GRAS, L.; SCHUURMAN, N. et al. Enteropathogen infections in canine puppies: (co-)occurrence, clinical relevance and risk factors. Vet. Microbiol., v.195, p.115-122, 2016.
- FELDMAN, B.F.; ZINKL, J.G.; JAIN, N.C. Schalm’s veterinary hematology. 5.ed. Baltimore: Lippincott Williams & Wilkins, 2000. 1206p.
- GHIGGI, E.; PADILHA, V.S.; MORAES, A.N. et al. Reposição volêmica com hidroxietilamido ou solução de ringer lactato em cães com gastroenterite hemorrágica por parvovírus. Semin. Cienc. Agrar., v.34, p.1783-1791, 2013.
- GOMEZ, H.; KELLUM, J.A. Understanding acid base disorders. Crit. Care Clin., v.31, p.849-860, 2015.
- HOPPER, K.; EPSTEIN, S.E. Incidence, nature, and etiology of metabolic acidosis in dogs and cats. J. Vet. Intern. Med., v.26, p.1107-1114, 2012.
- HOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 1: comparison of three methods of acid-base analysis. J. Vet. Emerg. Crit. Care, v.24, p.493-501, 2014a.
- HOPPER, K.; EPSTEIN, S.E.; KASS, P.H.; MELLEMA, M.S. Evaluation of acid-base disorders in dogs and cats presenting to an emergency room. part 2: comparison of anion gap, strong ion gap, and semiquantitative analysis. J. Vet. Emerg. Crit. Care, v.24, p.502-508, 2014b.
- HOPPER, K.; HASKINS, S.C. A case-based review of a simplified quantitative approach to acid-base analysis. J. Vet. Emerg. Crit. Care, v.18, p.467-476, 2008.
- KANEKO, J.; HARVEY, J.; BRUSS, M. Clinical biochemistry of domestic animals. 6.ed. San Diego: Academic Press, 2008. 928p.
- MELTESEN, H.S.; BOHN, A.A. Using corrected serum chloride and predicted bicarbonate concentrations to interpret acid-base status in dogs. Vet. Clin. Pathol., v.41, p.509-517, 2012.
- MORAIS, H.A.; BACH, J.F.; DIBARTOLA, S.P. Metabolic acid-base disorders in the critical care unit. Vet. Clin. N. Am. Small Anim. Pract., v.38, p.559-574, 2008.
- MÜLLER, K.R.; GENTILE, A.; KLEE, W.; CONSTABLE, P.D. Importance of the effective strong ion difference of an intravenous solution in the treatment of diarrheic calves with naturally acquired acidemia and strong ion (metabolic) acidosis. J. Vet. Intern. Med., v.26, p.674-683, 2012.
- NAVARRO, M.; MONREAL, L.; SEGURA, D. et al. A comparison of traditional and quantitative analysis of acid-base and electrolyte imbalances in horses with gastrointestinal disorders. J. Vet. Intern. Med., v.19, p.871-877, 2005.
- TELLO, L.; PEREZ-FREYTES, R. Fluid and electrolyte therapy during vomiting and diarrhea. Vet. Clin. N. Am. Small Anim. Pract., v.47, p.505-519, 2017.
- TORRENTE, C.; MANZANILLA, E.G.; GOPEGUI, R.R. A comparison of traditional and quantitative analysis of acid-base imbalances in hypoalbuminemic dogs. J. Vet. Emerg. Crit. Care, v.24, p.509-518, 2014.
- TREFZ, F.M.; LORCH, A.; FEIST, M. et al. Metabolic acidosis in neonatal calf diarrhea-clinical findings and theoretical assessment of a simple treatment protocol. J. Vet. Intern. Med., v.26, p.162-170, 2012.
Publication Dates
-
Publication in this collection
03 Apr 2020 -
Date of issue
Jan-Feb 2020
History
-
Received
31 Aug 2018 -
Accepted
24 July 2019