ABSTRACT
A ten-month-old male Yorkshire terrier dog was evaluated via CT four months after traumatic brain injury. The head CT scan showed dilated ventricles associated with a peripheral crescent-shaped collection of blood near the right parietal bone with a mineralized area. The visceral layer of the hematoma was hyperattenuating on the native scan and showed moderate contrast enhancement after administration of intravenous iodinated contrast medium. No fractures were identified in the calvarium. These findings were compatible with acute-on-chronic calcified subdural hematoma, which have been described with more detail in humans. This is the first report to include the description of the imaging findings related to this condition using computed tomography in a dog.
Keywords:
canine; head trauma; traumatic brain injury; computed tomography
RESUMO
Um cão Yorkshire Terrier, de 10 meses de idade, foi avaliado por tomografia computadorizada, quatro meses após lesão encefálica de origem traumática. A tomografia da cabeça demonstrou dilatação dos ventrículos associada com coleção de sangue periférico, em formato crescente, próxima ao osso parietal direito, com uma área mineralizada interna. A camada visceral do hematoma era hiperatenuante no exame pré-contraste e demonstrou moderado aumento de atenuação radiográfica após a administração de contraste iodado intravenoso. Fraturas no calvário não foram identificadas. Esses achados foram compatíveis com agudização de hematoma subdural crônico calcificado, condição que tem sido descrita com mais detalhes em humanos. Este é o primeiro relato que descreve os achados de imagem, por tomografia computadorizada, em um cão, relacionados a essa condição.
Palavras-chave:
canino; trauma cranioencefálico; tomografia computadorizada
INTRODUCTION
Chronic subdural hematoma (CSH) is a form of hematoma that occurs between the dura and arachnoid mater and is a usual complication of cranial trauma in humans (Iplikçioglu et al., 1991IPLIKÇIOGLU, A.C.; AKKAS, O.; SUNGUR, R. Ossified chronic subdural hematoma: case report. J. Trauma, v.31, p.272-275, 1991.). In small animals, to the authors’ knowledge, only subdural hematomas have been reported (Nykamp et al., 2001NYKAMP, S.; SCRIVANI, P.; DELAHUNTA, A. et al. Chronic subdural hematomas and hydrocephalus in a dog. Vet. Radiol. Ultrasound, v.42, p.511-514, 2001.; Kitagawa et al., 2008KITAGAWA, M.; OKADA, M.; KOIE, H. et al. Magnetic resonance imaging and computed tomography appearance of chronic subdural haematoma in a dog. Aust. Vet. J., v.86, p.100-101, 2008.; Hecht, 2011HECHT, S. Brain In: SCHWARZ, T.; SAUNDERS, J. (Eds). Veterinary computed tomography. United Kingdom: Wiley-Blackwell, 2011. p.185-196.). The CT appearance of subdural hematomas ranges between crescent and biconvex shape, depending on the curvature of the calvarium (Hecht, 2011).
Calcified CSH is considered rare in humans. When present, the extent of the calcification can range from small areas up to the entire surface of the brain hemisphere, which is then called an armored brain (Iplikçioglu et al., 1991IPLIKÇIOGLU, A.C.; AKKAS, O.; SUNGUR, R. Ossified chronic subdural hematoma: case report. J. Trauma, v.31, p.272-275, 1991.; Kanu et al., 2012KANU, O.O.; IGWILO, A.L.; DAINI, O. et al. Armoured brain: a case of bilateral calcified chronic subdural haematoma complicating infantile hydrocephalus. Rom. Neurosurg., n.1, 2012.). Pathogenesis for the calcification in humans is not clearly defined, however, vascular and metabolic causes may have a role in the development of this process (Moon et al., 2003MOON, H.G.; SHIN, H.S.; KIM, T.H. et al. Ossified chronic subdural haematoma. Yonsei Med. J., v.44, p.915-918, 2003.). In humans, calcification in CSH has not been reported earlier than three months after head trauma (Iplikçioglu et al., 1991). Chronic subdural hematoma with calcification has not been previously reported in dogs. In people there is a clear and clinically important distinction between the crescent shaped subdural and biconvex epidural hematomas (Parizel et al., 2001PARIZEL, P.M.; MAKKAT, S.; VAN MIERT, E. et al. Intracranial hemorrhage: principles of CT and MRI interpretation. Eur. Radiol., v.11, p.1770-1783, 2001.). In the limited reported cases of dogs and cats with intracranial hematoma, these were all subdural and had both biconvex and crescent shapes. This was also the case in this dog, where parts of the hematoma were crescent shaped and other parts biconvex, and this is probably related to the different skull shape compared to humans. Although there are anecdotal reports, to the authors’ knowledge, epidural cranial hematomas have not been reported in dogs.
Computed tomography is very sensitive for the diagnosis of acute intracranial hemorrhage, which is evident as increased density due to attenuation of X-rays by the iron content of the hemoglobin portion of the blood (Garosi, 2010GAROSI, L.S. Cerebrovascular disease in dogs and cats. Vet. Clin. N. Am. Small Anim. Pract., v.40, p.65-79, 2010.). In such cases, CT is considered the most important radiological test because it not only provides a wealth of information on the intracranial status, but also helps to identify patients at risk for postoperative recurrence after surgical treatment and hematoma drainage (Stanisic et al., 2013STANISIC, M.; HALD, J.; RAUMUSSEN, I.A. et al. Volume and densities of chronic subdural haematoma obtained from CT imaging as predictors of postoperative recurrence: a prospective study of 107 operated patients. Acta Neurochir., v.155, p.323-333, 2013.). The attenuation gradually decreases until the hematoma is equal in density to the brain, approximately one month after onset (Garosi, 2010). In those cases, the CSH are frequently difficult to diagnose using non-contrast enhanced CT (Senturk et al., 2010SENTURK, S.; GUZEL, A.; BILICI, A. et al. CT and MR imaging of chronic subdural hematomas: a comparative study. Swiss Med. Wkly, v.140, p.335, 2010.). Computed tomography has some advantages for the diagnosis and evaluation of intracranial bleeding due to its greater availability and speed of the procedure (Senturk et al., 2010; Stanisic et al., 2013). However, magnetic resonance imaging (MRI) offers slightly better resolution in general and higher sensitivity, especially with minimum hemorrhage, because CT artifacts lead to decreased resolution of the brain adjacent to the calvarium. When the hematomas are small, without mass effect, and isoattenuating on CT, MRI offers the greatest advantage in detecting lesions (Nykamp et al., 2001NYKAMP, S.; SCRIVANI, P.; DELAHUNTA, A. et al. Chronic subdural hematomas and hydrocephalus in a dog. Vet. Radiol. Ultrasound, v.42, p.511-514, 2001.).
CASE REPORT
A ten-month-old male Yorkshire terrier dog was referred to a diagnostic imaging center for a computed tomography (CT) scan of the head. The dog had suffered a head trauma when it was six months old. Upon neurologic examination after the traumatic injury, the dog had ataxia and depression, and responded to visual and auditory stimuli. After initial treatment at a private clinic, the dog showed marked neurologic improvement and was sent home. After four months, neurologic signs were observed and the dog developed generalized seizures, ataxia, and visual deficits. Initially, some improvement occurred with the administration of oral prednisone. The dog was then referred for computed tomography imaging of the skull.
For the computed tomography scan, the dog was anesthetized with diazepam (0.5mg/kg, IV), propofol (0.4mg/kg, IV), and isoflurane. Computed tomography images were acquired using a helical scanner (GE High Speed FXi, Waukesha, Wisconsin, USA) with the dog in ventral recumbency. Transverse images were obtained using brain reconstruction kernels, in 2-mm-thick slices and pitch of 1. Images were acquired before and after administration of iodinated intravenous contrast (Optiray® 350). Images were reviewed by the authors, including a board certified veterinary radiologist, using dedicated DICOM viewer software (Horos, Purview, Annapolis MD, USA, version 1.1.7) on a computer workstation (Apple Mac Pro, Apple, USA) with a calibrated LCD flat screen monitor (Apple thunderbolt Display, 27 inch, Apple, USA). Density measurements in Hounsfield units (HU) were obtained using the region-of-interest tool of the viewing software.
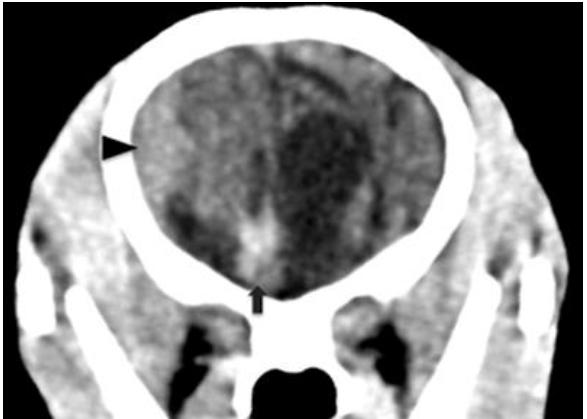
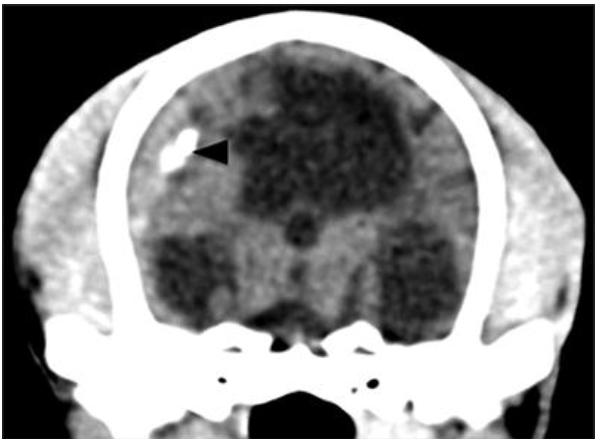
No skull fractures were observed and fontanelles and skull sutures were closed. There was a dilated ventricular system with irregular extensions into the brain tissue (Figure 1). There were relatively large intergyral spaces filled with subarachnoid cerebrospinal fluid (Figure 1A). In the right frontal lobe of the brain there was an 8mm lesion of hyperdense tissue (approximately 45 HU) compared to normal brain tissue (approximately 22 HU) (Figure 2). Adjacent to the right parietal bone there was a biconvex to crescent shaped area of material with a density similar to the hyperdense right frontal lobe lesion, causing mild compression of the right cerebrum and ventricular distortion (Figure 1A, 2). Within this mass lesion there was a 6mm lesion of very high density (approximately 250 HU) (Figure 3). After administration of intravenous contrast medium, there was moderate rim enhancement of the right parietal mass (Figure 1B). These imaging findings were interpreted as being consistent with focal parenchymal brain hemorrhage, subdural hematoma formation with central mineralization, and secondary cerebral and ventricular deviation and slight compression. The gyral atrophy and ventricular extensions into the brain tissue may be related to the reported trauma or independent porencephaly. A diagnosis of acute-on-chronic subdural hematoma was made based on the CT findings.
Brain computed tomographic images of an ataxic Yorkshire terrier. A) Transverse pre-contrast and B) post-contrast images at the level of the optic chiasm, C) Dorsal plane reconstructed pre-contrast image. There is marked ventricular distension (*) with irregular extensions into the brain tissue (white arrow), gyral atrophy with enlarged intergyral subarachnoid space (black arrow), a crescent to biconvex hyperdense mass lesion (black arrowheads) with rim-contrast enhancement (white arrowhead), consistent with an epi- or subdural hematoma.
Transverse pre-contrast CT image at the level of the frontal brain lobe, showing a hyperdense area in the right frontal lobe (arrow) and the hyperdense biconvex peripheral mass lesion (arrowhead).
Transverse pre-contrast CT image at the level of the thalamus, showing a hyperdense area (arrowhead) within the hyperdense biconvex peripheral mass lesion.
DISCUSSION
The authors present the imaging findings for a dog with acute-on-chronic subdural hematoma. In this case, there were signs of distortion and asymmetry of the ventricles due to the mass effect caused by the CSH. Ventriculomegaly and hydrocephalus are common conditions in Yorkshire terriers and, although the dog in this case had closed sutures and fontanelles, the possibility of these being present prior to the trauma cannot be excluded. In humans, extra parenchymal hemorrhage is a common complication of hydrocephalus, where atrophy and distortion of the brain leads to fragility of the lining of the ventricles, which becomes prone to tearing secondary to minor trauma or sudden increases in intracranial pressure. With hydrocephalus, there is a fragile outer gray matter layer and a lack of counter pressure provided by the cerebral hemispheres, which results in minor bleeding that is not promptly resolved (Nykamp et al., 2001NYKAMP, S.; SCRIVANI, P.; DELAHUNTA, A. et al. Chronic subdural hematomas and hydrocephalus in a dog. Vet. Radiol. Ultrasound, v.42, p.511-514, 2001.).
Another differential diagnosis to be considered for the “CSF-like expansion into the brain parenchyma” is porencephaly, which has been described as fluid cavities within brain parenchyma, uni or bilateral, usually connecting the ventricles to the subarachnoid space. The fluid within porencephaly lesions has the same characteristics as CSF (Schmidt et al., 2012SCHMIDT, M.J.; KLUMPP, S.; AMORT, K. et al. Porencephaly in dogs and cats: magnetic resonance imaging findings and clinical signs. Vet. Radiol. Ultrasound, v.53, p.142-149, 2012.). In the present case, there was no communication between the fluid cavity and the ventricles, and the radiodensity of the fluid within the lesion was different from the normal density of the CSF within the ventricles. The latter characteristic especially would suggest the lesion in the present paper is not porencephaly independent from the trauma.
The tomographic aspect of a subdural hematoma shows a gradual reduction in density over time (Hecht, 2011HECHT, S. Brain In: SCHWARZ, T.; SAUNDERS, J. (Eds). Veterinary computed tomography. United Kingdom: Wiley-Blackwell, 2011. p.185-196.). On CT, subdural hematomas show hyperdense areas in the acute stage, isodense areas in the sub-acute stage and hypodense areas in the chronic stage (Kitagawa et al., 2008KITAGAWA, M.; OKADA, M.; KOIE, H. et al. Magnetic resonance imaging and computed tomography appearance of chronic subdural haematoma in a dog. Aust. Vet. J., v.86, p.100-101, 2008.). In this report, CT images showed differences in the degree of radiographic attenuation of the hematoma, with the area adjacent to the cerebral parenchyma oscillating between 45 and 57 Hounsfield units, whereas the areas closer to the parietal bone had lower radiographic attenuation. The post-contrast images showed non-homogeneous contrast enhancement, which is compatible with a new hemorrhagic event, as well as a mild ring enhancement, which is consistent with a capillary-rich membrane encapsulating the chronic subdural hematoma (Parizel et al., 2001PARIZEL, P.M.; MAKKAT, S.; VAN MIERT, E. et al. Intracranial hemorrhage: principles of CT and MRI interpretation. Eur. Radiol., v.11, p.1770-1783, 2001.).
In humans, acute-on-chronic subdural hematomas are not unheard of. One study observed an incidence of 8% in a total of 107 patients which had been previously diagnosed with a CSH (Lee et al., 2011LEE, K.S.; SHIM, J.J.; YOON, S.M. et al. Acute-on-chronic subdural hematoma: not uncommon events. J. Korean Neurosurg. Soc., v.50, p.512-516, 2011.). Also in humans, loculation or septation have been reported as occurring between chronic hematomas and areas of new hemorrhage (Lee et al., 2011; Stanisic et al., 2013STANISIC, M.; HALD, J.; RAUMUSSEN, I.A. et al. Volume and densities of chronic subdural haematoma obtained from CT imaging as predictors of postoperative recurrence: a prospective study of 107 operated patients. Acta Neurochir., v.155, p.323-333, 2013.). In dogs, a case of chronic subdural hematoma with suspected new onset of hemorrhage was reported when CT and MRI images revealed different characteristics between the center and periphery of the hematoma (Kitagawa et al., 2008KITAGAWA, M.; OKADA, M.; KOIE, H. et al. Magnetic resonance imaging and computed tomography appearance of chronic subdural haematoma in a dog. Aust. Vet. J., v.86, p.100-101, 2008.).
There are only a few reports of chronic subdural hematoma in dogs (Nykamp et al., 2001NYKAMP, S.; SCRIVANI, P.; DELAHUNTA, A. et al. Chronic subdural hematomas and hydrocephalus in a dog. Vet. Radiol. Ultrasound, v.42, p.511-514, 2001.; Kitagawa et al., 2008KITAGAWA, M.; OKADA, M.; KOIE, H. et al. Magnetic resonance imaging and computed tomography appearance of chronic subdural haematoma in a dog. Aust. Vet. J., v.86, p.100-101, 2008.). As far as the authors are aware, this is the second report of a possible case of acute-on-chronic subdural hematoma, and the first report of calcification of the subdural hematoma in a dog. This may be an underdiagnosed condition, and it is important for clinicians and radiologists to entertain the possibility when neurologic signs recur some time after the initial trauma.
REFERENCES
- GAROSI, L.S. Cerebrovascular disease in dogs and cats. Vet. Clin. N. Am. Small Anim. Pract., v.40, p.65-79, 2010.
- HECHT, S. Brain In: SCHWARZ, T.; SAUNDERS, J. (Eds). Veterinary computed tomography. United Kingdom: Wiley-Blackwell, 2011. p.185-196.
- IPLIKÇIOGLU, A.C.; AKKAS, O.; SUNGUR, R. Ossified chronic subdural hematoma: case report. J. Trauma, v.31, p.272-275, 1991.
- KANU, O.O.; IGWILO, A.L.; DAINI, O. et al. Armoured brain: a case of bilateral calcified chronic subdural haematoma complicating infantile hydrocephalus. Rom. Neurosurg., n.1, 2012.
- KITAGAWA, M.; OKADA, M.; KOIE, H. et al. Magnetic resonance imaging and computed tomography appearance of chronic subdural haematoma in a dog. Aust. Vet. J., v.86, p.100-101, 2008.
- LEE, K.S.; SHIM, J.J.; YOON, S.M. et al. Acute-on-chronic subdural hematoma: not uncommon events. J. Korean Neurosurg. Soc., v.50, p.512-516, 2011.
- MOON, H.G.; SHIN, H.S.; KIM, T.H. et al. Ossified chronic subdural haematoma. Yonsei Med. J., v.44, p.915-918, 2003.
- NYKAMP, S.; SCRIVANI, P.; DELAHUNTA, A. et al. Chronic subdural hematomas and hydrocephalus in a dog. Vet. Radiol. Ultrasound, v.42, p.511-514, 2001.
- PARIZEL, P.M.; MAKKAT, S.; VAN MIERT, E. et al. Intracranial hemorrhage: principles of CT and MRI interpretation. Eur. Radiol., v.11, p.1770-1783, 2001.
- SCHMIDT, M.J.; KLUMPP, S.; AMORT, K. et al. Porencephaly in dogs and cats: magnetic resonance imaging findings and clinical signs. Vet. Radiol. Ultrasound, v.53, p.142-149, 2012.
- SENTURK, S.; GUZEL, A.; BILICI, A. et al. CT and MR imaging of chronic subdural hematomas: a comparative study. Swiss Med. Wkly, v.140, p.335, 2010.
- STANISIC, M.; HALD, J.; RAUMUSSEN, I.A. et al. Volume and densities of chronic subdural haematoma obtained from CT imaging as predictors of postoperative recurrence: a prospective study of 107 operated patients. Acta Neurochir., v.155, p.323-333, 2013.
Publication Dates
-
Publication in this collection
03 Apr 2020 -
Date of issue
Jan-Feb 2020
History
-
Received
16 Apr 2018 -
Accepted
05 July 2019