Abstracts
This non-concurrent cohort study aims to identify predictors of tuberculosis mortality in a large population database in Brazil. Tuberculosis, death, and TB/HIV cases were validated respectively from the tuberculosis surveillance (SINAN/TB), mortality (SIM), and SINAN/AIDS databases for a five-year period. Analysis included proportional hazard models with relative risk estimates. Out of 5,451 individuals reported with tuberculosis, 320 (5.9%) died (incidence and mortality rates of 98.6 and 12.2/100 thousand inhabitants, respectively). After adjustment, relative risk of dying from tuberculosis was 9.8 for individuals > 50 years of age; 9.0 for TB/HIV co-infection; 3.0 for mixed TB clinical presentation; and 2.0 for treatment dropout. In the multivariate model, using cases with HIV/AIDS, all adjusted predictors lost significance except mixed clinical presentation (RR 1.9; 1.1-3.1). TB/HIV co-infection is an important predictor of TB mortality. However, among individuals without HIV/AIDS, mortality is still highly associated with older age, mixed clinical forms, and treatment dropout.
Tuberculosis; Survivorship; Mortality; Patient Dropouts
Este estudo de coorte não concorrente objetivou identificar fatores associados ao óbito por tuberculose em pacientes notificados ao sistema de vigilância de tuberculose (SINAN/TB), residentes em Recife, Pernambuco, Brasil, de 1996-2000. Óbito por tuberculose e co-infecção HIV/AIDS foram validados nos Sistemas de Mortalidade (SIM) e SINAN/AIDS. Análise de sobrevida modelos de riscos proporcionais de Cox foram utilizados. Dos 5.451 indivíduos diagnosticados (coeficiente de incidência de 98,6/100 mil habitantes), 320 (5,9%) foram ao óbito por tuberculose (coeficiente de mortalidade de 12,2/100 mil habitantes). O risco relativo ajustado de morrer foi de 9,8 para maiores de 50 anos, 9,0 para indivíduos com co-infecção HIV/AIDS, 3,0 para aqueles com apresentação mista e 2,0 para pacientes que abandonaram o tratamento. Quando se estratificou a população em dois grupos: pacientes com e sem co-infecção HIV/AIDS, os estimadores estudados (faixa etária, modo de entrada e apresentação clínica) mantiveram o poder de associação no grupo sem a co-infecção. Entretanto, na presença da co-infecção, apenas apresentação mista manteve-se associada ao óbito (RR = 1,9). Neste estudo, apesar da relevância do HIV/AIDS, idade acima de cinqüenta anos, apresentação mista e abandono de tratamento foram importantes preditores de óbito em pacientes com tuberculose.
Tuberculose; Sobrevida; Mortalidade; Desistência do Paciente
ARTIGO ARTICLE
Mortality, TB/HIV co-infection, and treatment dropout: predictors of tuberculosis prognosis in Recife, Pernambuco State, Brazil
Mortalidade, co-infecção por HIV/AIDS e abandono do tratamento como fatores prognósticos para tuberculose em Recife, Pernambuco, Brasil
Mirian Pereira DomingosI, II; Waleska Teixeira CaiaffaII; Enrico Antônio ColosimoIII
IGerência Geral de Vigilância à Saúde, Secretaria de Saúde do Estado de Pernambuco, Recife, Brasil
IIFaculdade de Medicina, Universidade Federal de Minas Gerais, Belo Horizonte, Brasil
IIIInstituto de Ciências Exatas, Universidade Federal de Minas Gerais, Belo Horizonte, Brasil
Correspondence Correspondence: M. P. Domingos Gerência Geral de Vigilância à Saúde Secretaria de Saúde do Estado de Pernambuco Rua 15 de Março 298, Recife, PE 50761-070, Brasil miriandomingos@medicina.ufmg.br
ABSTRACT
This non-concurrent cohort study aims to identify predictors of tuberculosis mortality in a large population database in Brazil. Tuberculosis, death, and TB/HIV cases were validated respectively from the tuberculosis surveillance (SINAN/TB), mortality (SIM), and SINAN/AIDS databases for a five-year period. Analysis included proportional hazard models with relative risk estimates. Out of 5,451 individuals reported with tuberculosis, 320 (5.9%) died (incidence and mortality rates of 98.6 and 12.2/100 thousand inhabitants, respectively). After adjustment, relative risk of dying from tuberculosis was 9.8 for individuals > 50 years of age; 9.0 for TB/HIV co-infection; 3.0 for mixed TB clinical presentation; and 2.0 for treatment dropout. In the multivariate model, using cases with HIV/AIDS, all adjusted predictors lost significance except mixed clinical presentation (RR 1.9; 1.1-3.1). TB/HIV co-infection is an important predictor of TB mortality. However, among individuals without HIV/AIDS, mortality is still highly associated with older age, mixed clinical forms, and treatment dropout.
Tuberculosis; Survivorship; Mortality; Patient Dropouts
RESUMO
Este estudo de coorte não concorrente objetivou identificar fatores associados ao óbito por tuberculose em pacientes notificados ao sistema de vigilância de tuberculose (SINAN/TB), residentes em Recife, Pernambuco, Brasil, de 1996-2000. Óbito por tuberculose e co-infecção HIV/AIDS foram validados nos Sistemas de Mortalidade (SIM) e SINAN/AIDS. Análise de sobrevida modelos de riscos proporcionais de Cox foram utilizados. Dos 5.451 indivíduos diagnosticados (coeficiente de incidência de 98,6/100 mil habitantes), 320 (5,9%) foram ao óbito por tuberculose (coeficiente de mortalidade de 12,2/100 mil habitantes). O risco relativo ajustado de morrer foi de 9,8 para maiores de 50 anos, 9,0 para indivíduos com co-infecção HIV/AIDS, 3,0 para aqueles com apresentação mista e 2,0 para pacientes que abandonaram o tratamento. Quando se estratificou a população em dois grupos: pacientes com e sem co-infecção HIV/AIDS, os estimadores estudados (faixa etária, modo de entrada e apresentação clínica) mantiveram o poder de associação no grupo sem a co-infecção. Entretanto, na presença da co-infecção, apenas apresentação mista manteve-se associada ao óbito (RR = 1,9). Neste estudo, apesar da relevância do HIV/AIDS, idade acima de cinqüenta anos, apresentação mista e abandono de tratamento foram importantes preditores de óbito em pacientes com tuberculose.
Tuberculose; Sobrevida; Mortalidade; Desistência do Paciente
Introduction
Tuberculosis is an important cause of death worldwide. Estimates indicate 8.8 million new cases of the disease and 1.6 million deaths per year 1,2. With 6% of all deaths worldwide attributed to TB, the disease is the fifth most common cause of death, next to cardiovascular diseases, respiratory infections, cancer, and diarrheal diseases 3,4.
Despite advances in knowledge on TB and available technology for its control, socioeconomic inequalities affecting vast sectors of the population, deterioration of the health infrastructure (aggravating problems with access to care), and the interface with the HIV epidemic suggest the need for a specific TB alert. This scenario is particularly severe in the developing countries 5,6,7.
Brazil occupies 14th place among the 23 countries that account for 80% of TB cases in the world. According to estimates, Brazil's incidence rate is 58 cases per 100 thousand inhabitants, with a total of 50 million infected individuals and 111 thousand new cases of the disease and six thousand deaths per year 1. In Recife, one of the country's ten largest State capitals, TB is one of the most serious public health problems. In addition to the high incidence rate (113.95/100 thousand), Recife also has one of the country's highest tuberculosis mortality rates (9.91/100 thousand) 8. The Brazilian government thus considers the city a priority for implementing TB control measures 8,9.
This non-concurrent cohort study aims to evaluate the impact of demographic and clinical/epidemiological factors on the survival of TB patients residing in Recife and reported to the official TB surveillance system.
Population and methods
Tuberculosis cases were identified in the Tuberculosis Reporting Data Base (SINAN/TB) under the Pernambuco State Health Department, which is the official system for mandatory reporting of diseases and health problems implemented in the State in 1993. TB reporting is mandatory nationwide for new cases, relapses, and reentries after treatment dropout 7.
All patients reported from January 1, 1996, to December 31, 2000, and residing in Recife were selected for inclusion in the study. The information was located, retrieved, confirmed, or corrected in order to identify each individual, thereby eliminating multiple records. Patients with multiple entries due to dropout, hospitalization, or other factors were reorganized to comprise a single entry. Date of initial diagnosis was defined as the first report at which individuals received the TB diagnosis and specific treatment was initiated.
Data on death from tuberculosis and TB/HIV co-infection were confirmed and/or retrieved from databases in the national Mortality Information System (SIM) and SINAN/AIDS, through a systematic search, including previous and subsequent calendar years in relation to the study period (1993 to 2001 for HIV/AIDS and 1996 to 2001 for death from TB or AIDS). Death from TB was only considered when confirmed as primary cause of death in the mortality database, and TB/HIV co-infection when confirmed in the AIDS database. The censuring date for both events in the study was June 31, 2001.
The analysis excluded TB cases with reporting dates that coincided with or postdated death. The study also excluded cases with no information on follow-up and outcome in the SINAN/TB database and that were not found in the mortality database (SIM) or the SINAN/AIDS database after a systematic and exhaustive search, including the entire study period, i.e., until the censuring date.
The response variable was survival time, defined as time in months transpired from the date of initial diagnosis to death or, in the case of individuals who did not die, the last follow-up recorded by the health service. Individuals who died were considered failures and those who remained alive until the end of the study were considered censures.
The independent variables were: (a) socio-demographic: age, gender, and years of schooling; (b) clinical/epidemiological: TB clinical presentation, i.e., pulmonary, extrapulmonary, and mixed; TB/HIV co-infection; association with AIDS-defining disease; entry into the surveillance system, including new cases, relapses, and treatment dropouts; (c) temporal: calendar year (1996 to 2000); date of diagnosis; date of death from TB; closing date for reporting.
Descriptive analyses used Student's t-test and Pearson's chi-square test. The Kaplan-Meier method and log-rank test were used to estimate the probability of survival and compare the survival curves between the various categories in the same variable, respectively 10.
The independent effects of each covariable were measured using the Cox proportional risk model 11 and the hazard ratio was estimated as relative risk (RR). The importance of each covariable in the model was evaluated with the Wald test 10, and the hypotheses concerning the estimated parameters were tested according to the likelihood ratio statistic 11,12,13,14. Risk proportionality and goodness-of-fit were verified in the final models using graphic inspection and Martingale and Deviance residue analyses 11,13.
In order to verify whether the predictive covariables for TB survival were similar, two models were constructed according to the presence or absence of HIV co-infection. The first model considered all cases of TB and the second those associated with HIV/AIDS. In all the analyses, the significance level for rejecting the null hypothesis was p < 0.05.
To estimate the impact of predictive factors for death from tuberculosis in the population, the proportions of population attributable risk (PAR%) were calculated using the formula by Levin 15 (PAR% = p(RR-1)/p(RR-1)+1) for each variable that presented statistical significance in the final model.
The data were analyzed with Epi Info version 6.0 (Centers for Disease Control and Prevention, Atlanta, USA) and SPSS version 11.5 (SPSS Inc., Chicago, USA).
Access to the data was authorized by the Pernambuco State Health Department and approved by the Research Ethics Committee of the Universidade Federal de Minas Gerais [Federal University in Minas Gerais] (project 120/02), where the study was conducted.
Results
During the five years studied, 7,399 individuals residing in Recife were identified in the SINAN/TB database: 6,780 (91.6%) new cases, 280 (3.1%) relapses, 351 (4.7%) reentries due to prior treatment dropout, and 10 cases (0.1%) transferred from other municipalities or States. During the same period, 837 individuals (12.2%) died from TB. The mean incidence and mortality rates, respectively, were 98.6 and 12.2 per 100 thousand inhabitants, with a mean case-fatality rate of 12.3%.
Survival analysis excluded the following patients: 441 (6%) reported after death; 28 (0.4%) who presented change of diagnosis; 139 (1.9%) with diagnosis date on or later than the closing of reporting; 38 (0.5%) due to transfers; and 104 (1.4%) for having died of a cause unrelated to TB.
A total of 1,198 (16.2%) lacked any record of the last observation within the study period and were thus considered lost to follow-up. Individuals excluded or lost to follow-up did not show any statistically significant differences from those who remained in the study in relation to age, gender, HIV co-infection, form of entry, or calendar year (p > 0.05).
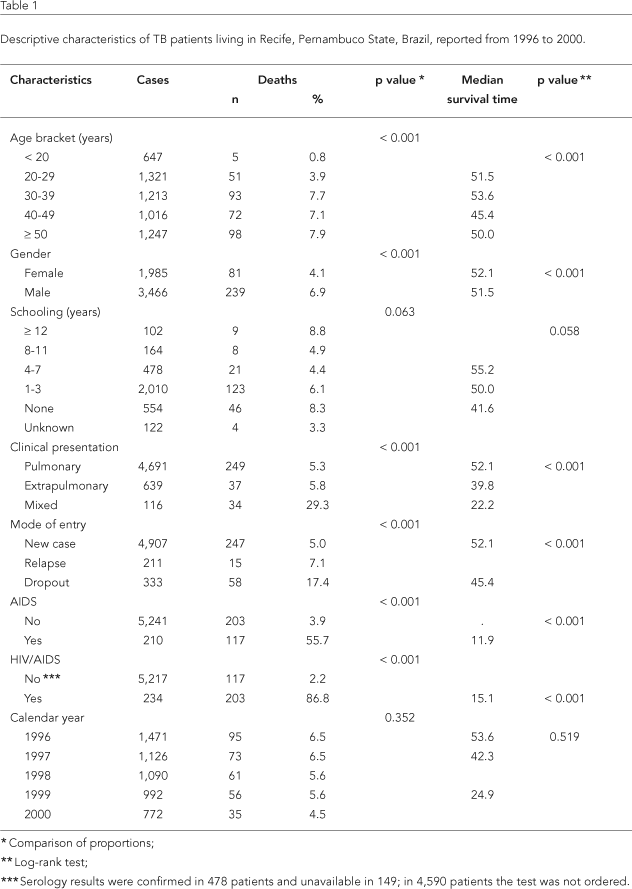
The survival study thus included 5,451 (73.7%) individuals whose characteristics are summarized in Table 1. Mean and median ages were 37.2 (± 18.3) and 36 years. The cohort was mostly male (3,466; 63.9%), and the male/female ratio was 1.7:1. Information on schooling was recorded in 3,466 cases (62.9%), of whom 58.6% had up to three years of schooling; 16.2% had no schooling; and only 3% had 12 or more years of schooling.
As for clinical presentation, the most frequent manifestation was the isolated pulmonary form, with 4,675 of cases (85.8%). Of the 639 patients (11.7%) with the isolated extrapulmonary clinical form and 137 (2.5%) with the combined clinical form, 25 (3.2%) had miliary tuberculosis and 36 (4.6%) had tuberculous meningitis.
A total of 861 patients (15.8%) had undergone HIV serological testing. Of these, 234 (27.2%) were recorded as HIV-positive, of whom 210 (89.7%) presented a diagnosis of AIDS-defining illness.
Death occurred in 320 cases (5.9%) in the cohort. Median survival for the entire cohort was 51.5 months (95%CI: 48.06-55.00), while the probability of surviving for 12, 36, and 50 or more months after TB diagnosis was 93%, 69%, and 37%, respectively.
All the variables were significantly associated with survival except for year of diagnosis, which was not associated, and schooling, which showed borderline association (Table 1).
Lower median survival times were observed in the 40-49-year age bracket and in males (45.4 and 51.5 months, respectively). Survival time was visibly reduced among patients with no schooling (41.6 months); extrapulmonary and mixed clinical presentation (39.6 and 22.2 months, respectively); treatment dropout (45.4 months); HIV+ serology (15.1 months); and AIDS co-morbidity (11.9 months), as shown in the penultimate column in Table 1.
In constructing the models, the variables HIV+ serology and AIDS comorbidity were combined in a single covariable (HIV/AIDS co-infection), due to the overlapping of information for positive and unknown cases, with a 89% concordance between these categories.
Compared to the references, univariate analysis (Table 2) showed that the RR of dying from TB increased significantly with age in a dose-response gradient (from 4.7 to 10.5); male gender (1.6); mixed clinical form (6.2); HIV/AIDS co-infection (10.2); and reentry into the information system after a report of previous treatment dropout (2.6). Higher schooling showed a positive but non-significant association. Entry as relapse and calendar year were also not significantly associated with mortality.
In the final multivariate model, considering the entire group, only the covariables age, gender, clinical presentation, mode of entry into the system, and HIV/AIDS co-infection remained statistically significant (Table 2). HIV/AIDS co-infection was one of the most important predictive factors for mortality in this cohort, accounting for 203 (63.4%) of the 320 deaths. Accumulated mortality for this group of patients was 86.8% (203/234) with an RR of 9.0 (95%CI: 7.0-11.8) as compared to patients without TB/HIV/AIDS co-infection. However, death from TB in patients who reentered the surveillance system after known previous treatment dropout also remained significant (RR = 2.0; 95%CI: 1.5-2.7).
Separate analysis of the models for patient groups with and without TB/HIV/AIDS (Table 3) showed a profound change in the estimators. In the model without HIV/AIDS co-infection, with the exception of male gender, which lost statistical significance, all the factors that previously predicted death remained significant. However, important changes in the magnitude of risks were observed for age, history of treatment dropout, and mixed clinical presentation.
In the model with TB/HIV/AIDS co-infection, mixed clinical presentation was the only factor that remained statistically associated with death, with a relative risk of death of 1.9 (95%CI: 1.1-3.1) when compared with pulmonary presentation alone, after adjusting for age, gender, and mode of entry.
Residual analysis of all the multivariate models showed good adequacy and fit for the final models 13,15.
For the entire cohort, PAR% for previous treatment dropout was 5.7%, independently of age, gender, clinical form, and TB/HIV/AIDS co-infection (Table 4). However, for the group without TB/HIV/AIDS co-infection, PAR% for dropout increased, meaning that independently of other factors, in patients without HIV/AIDS, if dropout were eliminated, death from TB would be reduced by 11.5%.
Discussion
This study showed high TB incidence and mortality rates in patients residing in a State capital in Northeast Brazil. Age greater than 20 years, male gender, mixed clinical presentation, TB/HIV/AIDS co-infection, and treatment dropout were the significant and independent predictive factors associated with shorter survival time.
Various studies have indicated that the risk of dying from TB increases with the patient's age. However, mortality from TB in older individuals can be attributed not only to age per se and the presence of co-morbidities, but also to difficulties in accessing diagnosis in this group of patients, thereby delaying the onset of treatment 16. In our study, in both the univariate and multivariate analysis, the relative risk of dying from TB increased in a dose-response gradient with age, i.e., the older the patient, the greater the risk of dying. Similar results have been reported in other studies 17,18,19. The correlation between age and TB mortality reinforces the importance of early diagnosis in elderly patients, since treatment efficacy is the same as in younger individuals 20.
The fact that the majority of deaths were in males (239/320) is also consistent with the literature 21,22. However, although the relative risk for males was significant in the final adjusted model, it lost significance in the model stratified by TB/HIV/AIDS co-infection. This finding is corroborated by reports of patients with TB and HIV/AIDS in New York (USA) 17, Tanzania 19, and Spain 23, in which the observed effect of male gender on mortality was not significant.
Some studies confirm that persons with isolated pulmonary TB survive longer than those with the extrapulmonary and mixed forms 16,23,24,25. In our study, patients with other forms combined with the pulmonary presentation showed half the median survival time and a threefold risk of death from TB as compared to cases with the isolated pulmonary form. Of the deaths in patients with mixed forms, 19.7% had HIV co-infection or AIDS. In fact, in the model for the group classified as TB/HIV/AIDS co-infection, the mixed pulmonary/extrapulmonary form was the only factor independently associated with death, while in the group without TB/HIV/AIDS diagnosis, age at diagnosis, mixed pulmonary/extrapulmonary form, and previous treatment dropout remained significantly associated with risk of death from TB.
The results found here are consistent with the various reports in the literature 25,26,27,28 that the HIV epidemic has changed the prognosis for TB patients, and that HIV has a stronger impact on TB mortality than on incidence of the disease. TB/HIV/AIDS co-infection had a striking effect on risk of death, especially given the magnitude of risk of dying from TB, some 9.0 for patients with TB/HIV/AIDS co-infection as compared to those without. These results were quite similar to those of other authors 17,18, suggesting the external validity of our study.
Incomplete treatment has been reported as another important risk factor for death from TB 17,19,29,30. In fact, among the patients who died in this study, 18.2% had a history of previous treatment dropout. In this group of patients, in the absence of TB/HIV/AIDS co-infection, the relative risk of dying was 3.2 times greater than for patients who entered as new cases. However, in the presence of TB/HIV/AIDS co-infection, previous treatment dropout did not remain as a significant variable for risk of death.
It is possible that incomplete treatment (evaluated here by proxy with the variable "treatment dropout") may merely be a marker for other confounding variables, like worse health status, adverse drug effects, or difficult access to health 16.
In this study, PAR% for treatment dropout in patients without HIV/AIDS was less than that reported by Garcia-Garcia et al. 30. However, since treatment dropout is the only factor associated with death that can be controlled directly by health services, our results corroborate the findings of other studies 19,31 and strengthen the position that complete treatment increases patient survival. It is important to emphasize the high efficacy of the directly observed treatment strategy (DOTS), even in patients with TB/HIV/AIDS co-infection 30,31.
Since the database used in this study aims at epidemiological surveillance, the results are limited by the absence of important variables described in the literature 23,24,30, generating possibilities for residual confounding. Particularly important are those related to TB/HIV/AIDS co-infection, like exposure categories, T-lymphocyte count, and viral load; those related to treatment dropout like social characteristics, alcohol abuse, housing conditions, occupation, illicit drug use, and associated diseases; and those related to health services access.
Other relevant limitations that should be mentioned include the incompleteness of some information, especially on schooling; some decisions made in case classification, mainly for TB/HIV/AIDS co-infection; and the exclusion of cases from the analysis. The loss of some 30% of information on schooling certainly reflects a deficiency in the capacity to evaluate the real significance of this variable, considered lacking, and thus not included in some analyses. Although this variable may admittedly be an important marker for socioeconomic status, and although the lack of this information for a large share of cases limits its interpretation, we chose to emphasize this deficiency in order to encourage discussion on the need to improve the quality of these databases. National studies in Brazil 32 that also used secondary databases similar to this and other events have reported the same problems, although they do not rule out other types of analysis.
In relation to the categorization of the presence or absence of TB/HIV/AIDS co-infection, the decision was based on the TB treatment protocol for health services in Brazil in the absence of HIV serology. In other words, given the absence of a routine protocol for ordering HIV serology for all patients with a TB diagnosis, added to the difficulties with laboratory confirmation and the fact that ordering HIV serology is based on clinical suspicion, health services consider patients without HIV serology as not presenting TB/HIV co-infection, adopting the standard regimen and treatment schedule prevailing at the time 33. Although we cannot rule out classification errors, i.e., including patients with HIV/AIDS in our group without co-infection, such errors might lead to underestimation rather than overestimation of our parameters.
As for internal validity, two questions can be discussed pertaining to cases excluded from the analyses. The first involves cases whose closing and outcome information was not found, even after an exhaustive search in the SINAN/TB database for possible reentries, followed by searches in the SINAN/AIDS and SIM databases for other outcomes. The confirmation and retrieval of information from other databases (SIM and AIDS) allowed improvement of the data, but did not ensure total quality of information.
Other cases died from causes unrelated to TB. In both situations, the excluded cases did not differ significantly from those that remained in the study, in relation to the principal target variables.
Despite the above-mentioned limitations, the study highlights the importance of TB and related death in a city where the disease is highly endemic. It also points to the need for improved data quality, heightened surveillance, and better diagnosis in patients older than 50 years, with a relevant risk of dying. Equally urgent is the need for specific strategies to reduce treatment dropout.
Since Brazil practices important public policies for free distribution of antiretroviral drugs to all HIV+ patients, more detailed studies are needed to elucidate the real impact of the HIV epidemic on TB mortality, including research on the contribution of ordering HIV serology in patients with TB diagnosis.
Contributors
M. P. Domingos was in charge of elaborating the article (analyses, drafting, and review). W. T. Caiaffa participated actively in the elaboration of the article, from its conception to the final review. E. A. Colosimo contributed with the statistical analyses and discussion.
Submitted on 17/Jul/2006
Final version resubmitted on 26/Jun/2007
Approved on 13/Jul/2007
- 1. World Health Organization. Global tuberculosis control: surveillance, planning, financing. Geneva: World Health Organization; 2006.
- 2. Werf MJV, Borgdorff MW. Targets for tuberculosis control: how confident can we be about the data? Bull World Health Organ 2007; 85:371-7.
- 3. Raviglione MC, Snider DE, Kochi A. Global epidemiology of tuberculosis: morbidity and mortality of worldwide epidemic. JAMA 1995; 273:220-6.
- 4. Dye C, Schelele S, Dolin P, Pathania V, Raviglione MC. Global burden of tuberculosis: estimated incidence, prevalence and mortality by country. JAMA 1999; 282:677-86.
- 5. Castelo Filho A, Kritski AL, Barreto AW, Lemos ACM, Ruffino-Netto A, Guimarães CA, et al. II Consenso Brasileiro de Tuberculose. Diretrizes brasileiras para tuberculose 2004. J Bras Pneumol 2004; 30 Suppl 1:2-56.
- 6. World Health Organization. Global tuberculosis control. Geneva: World Health Organization; 1998.
- 7. Ministério da Saúde. Guia de vigilância epidemiológica. Brasília: Ministério da Saúde; 2005.
- 8. Sassaki CM, Vilela MJB, Gonzáles RIC, Villa TCS, Ruffino-Netto A. A experiência da implantação do DOTS na cidade do Recife, 2005. In: Ruffino-Netto A, Villa TCS, organizadores. Tuberculose. Implantação do DOTS em algumas regiões do Brasil: histórico e peculiaridades regionais. Riberão Preto: Rede TB; 2006. p. 167-79.
-
9Ministério da Saúde. Plano nacional de controle da tuberculose: normas técnicas, estrutura e operacionalização. 5ª Ed. Brasília: Fundação Nacional de Saúde; 2000.
- 10. Kaplan E, Meier P. Non-parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53:457-81.
- 11. Harris EK, Albert A. Survivorship analysis for clinical studies. New York: Dekker; 1991.
- 12. Bustamante-Teixeira MT, Faerstein E, Latorre MR. Técnicas de análise de sobrevida. Cad Saúde Pública 2002; 18:579-94.
- 13. Carvalho MS, Andreozzi VL, Codeço CT, Barbosa MTS, Shimakura SE. Análise de sobrevida: teoria e aplicações em saúde. Rio de Janeiro: Editora Fiocruz; 2005.
- 14. Bustamante-Montes LP, Escobar-Mesa A, Borja-Aburto VH, Gómez-Muños A, Becerra-Posada, F. Predictors of death from pulmonary tuberculosis: the case of Veracruz, Mexico. Int J Tuberc Lung Dis 2000; 4:208-15.
- 15. Schlesselman JJ. Case-control study. Oxford: Oxford University Press; 1982.
- 16. Chaimowics F. Age transition of tuberculosis incidence and mortality in Brazil. Rev Saúde Pública 2001; 35:81-7.
- 17. Pablos-Méndez A, Sterling TR, Frieden TR. The relationship between delayed or incomplete treatment and all-cause mortality in patients with tuberculosis. JAMA 1996; 276:1223-8.
- 18. Conoully C, Davies GR, Wilkinson D. Impact of the immunodeficiency virus epidemic on mortality among adults with tuberculosis in rural South Africa, 1991 1995. Int J Tuberc Lung Dis 1998; 2:919-25.
- 19. van den Broek J, Mfinanga S, Moshiro C, O'Brien R, Mugomela A, Lefi M. Impact of human immunodeficiency virus infection on the outcome of treatment and survival of tuberculosis patients in Mwanza, Tanzania. Int J Tuberc Lung Dis 1998; 2:547-52.
- 20. Dutt AK, Stead WW. Tuberculosis. Clin Geriatr Med 1992; 8:761-77.
- 21. Braun MM, Cote TR, Rebkin CS. Trends in death with tuberculosis during AIDS era. JAMA 1993; 269:2865-8.
- 22. Deriemer K, Ruddoy I, Scheter GF, Hopewell PC, Delay L. The epidemiology of tuberculosis diagnosed after death in San Francisco, 1986-1995. Int J Tuberc Lung Dis 1999; 3:488-93.
- 23. Casanovas MF, Langohr K, Melis GG, Rizo PGO, Del Vallado JMJL, Buqueras JAC. Supervivencia en pacientes con tuberculosis infectados por HIV. Estudio de los fallecimientos en los primeros nueve meses de tratamiento. Rev Esp Salud Pública 1999; 73:549-62.
- 24. Whalen C, Okwera A, Johnson J, Vjecha M, Hom D, Wallis R, et al. Predictors of survival in human immunodeficiency virus-infected patients with pulmonary tuberculosis. Am J Respir Crit Care Med 1996; 153:1977-81.
- 25. Shafer RW, Bloch AB, Larkin C, Vasudavan V, Seligman S, Dehovitz JD, et al. Predictors of survival in HIV-infected tuberculosis patients. AIDS 1996; 10:269-72.
- 26. Stiblo K. The impact of HIV infection on the global epidemiology of tuberculosis. Bull Int Union Tuberc Lung Dis 1991; 66:27-32.
- 27. Glynn JR, Warnddorff DK, Fine PEM, Msiaka GK, Munthali MM, Ponnighaus JM. The impact of HIV on morbidity and mortality in sub-Saharan Africa: a study in rural Malawi and review of literature. Health Transit Rev 1997; 7 Suppl 2:75-87.
- 28. Gustafson P, Gomes VF, Vieira CS, Rabna P, Seng R, Johansson P, et al. Tuberculosis in Bissau: incidence and risk factors in an urban community in sub-Saharan Africa. Int J Epidemiol 2004; 33:163-72.
- 29. Albuquerque MFM, Sá-Leitão CC, Campelo AR, Souza WV, Salustiano A. Fatores prognósticos para o desfecho do tratamento da tuberculose pulmonar em Recife, Pernambuco, Brasil. Rev Panam Salud Pública 2001; 9:368-74.
- 30. Garcia-Garcia ML, Ponce-de-León A, Garcia-Sancho MC, Ferreira-Reyes L, Palacios-Martínez M, Fuentes J, et al. Tuberculosis-related deaths with a well-functioning DOTS control program. Emerg Infect Dis 2002; 8:1327-32.
- 31. Nunn P, Brindle R, Carpenter L, Odhiambo J, Wasunna K, Newnham R, et al. Cohort study of human immunodeficiency virus infection in patients with tuberculosis in Nairobi, Kenya. Analysis of early (6-month) mortality. Am Rev Respir Dis 1992; 146:849-54.
- 32. Fonseca MG, Bastos FI, Derriço M, Andrade CLT, Travassos C, Szwarcwald CL. AIDS e grau de escolaridade no Brasil: evolução temporal de 1986 a 1996. Cad Saúde Pública 2000; 16 Suppl 1:S77-87.
- 33. Ministério da Saúde. Guia de vigilância epidemiológica. 5Ş Ed. Brasília: Fundação Nacional de Saúde; 1998.
Publication Dates
-
Publication in this collection
26 Mar 2008 -
Date of issue
Apr 2008
History
-
Accepted
13 July 2007 -
Reviewed
26 June 2007 -
Received
17 July 2007