Abstract:
This study sought to evaluate the occurrence of adverse events and their impacts on length of stay and mortality in an intensive care unit (ICU). This is a prospective study carried out in a teaching hospital in Rio de Janeiro, Brazil. The cohort included 355 patients over 18 years of age admitted to the ICU between August 1, 2011 and July 31, 2012. The process we used to identify adverse events was adapted from the method proposed by the Institute for Healthcare Improvement. We used a logistical regression to analyze the association between adverse event occurrence and death, adjusted by case severity. We confirmed 324 adverse events in 115 patients admitted over the year we followed. The incidence rate was 9.3 adverse events per 100 patients-day and adverse event occurrence impacted on an increase in length of stay (19 days) and in mortality (OR = 2.047; 95%CI: 1.172-3.570). This study highlights the serious problem of adverse events in intensive care and the risk factors associated with adverse event incidence.
Keywords:
Adverse Effects; Critical Care; Patient Safety; Mortality; Quality of Health Care
Resumen:
Este estudio tuvo como objetivo evaluar la ocurrencia de eventos adversos y el impacto de los mismos sobre el tiempo de permanencia y mortalidad en una unidad de cuidados intensivos (UCI). Se trata de un estudio prospectivo, desarrollado en un hospital universitario de Río de Janeiro, Brasil. La cohorte estaba formada por 355 pacientes mayores de 18 años admitidos en la UCI, durante el período del 1º agosto de 2011 al 31 de julio de 2012. El proceso de identificación de eventos adversos se basó en una adaptación del método propuesto por el Institute for Healthcare Improvement. La regresión logística fue utilizada para analizar la asociación entre la ocurrencia de evento adverso y el óbito, ajustado por la gravedad del paciente. Se confirmaron 324 eventos adversos en 115 pacientes internados a lo largo de un año de seguimiento. La tasa de incidencia fue de 9,3 eventos adversos por 100 pacientes-día y la ocurrencia de evento adverso impactó en el aumento del tiempo de internamiento (19 días) y en la mortalidad (OR = 2,047; IC95%: 1,172-3,570). Este estudio destaca el serio problema de los eventos adversos en la asistencia a la salud prestada en cuidados intensivos y los factores de riesgo asociados a la incidencia de eventos.
Palabras-clave:
Efectos Adversos; Cuidados Críticos; Seguridad del Paciente; Mortalidad; Calidad de la Atención de Salud
Resumo:
Este estudo teve como objetivo avaliar a ocorrência de eventos adversos e o impacto deles sobre o tempo de permanência e a mortalidade na unidade de terapia intensiva (UTI). Trata-se de um estudo prospectivo desenvolvido em um hospital de ensino do Rio de Janeiro, Brasil. A coorte foi formada por 355 pacientes maiores de 18 anos, admitidos na UTI, no período de 1º de agosto de 2011 a 31 de julho de 2012. O processo de identificação de eventos adversos baseou-se em uma adaptação do método proposto pelo Institute for Healthcare Improvement. A regressão logística foi utilizada para analisar a associação entre a ocorrência de evento adverso e o óbito, ajustado pela gravidade do paciente. Confirmados 324 eventos adversos em 115 pacientes internados ao longo de um ano de seguimento. A taxa de incidência foi de 9,3 eventos adversos por 100 pacientes-dia, e a ocorrência de evento adverso impactou no aumento do tempo de internação (19 dias) e na mortalidade (OR = 2,047; IC95%: 1,172-3,570). Este estudo destaca o sério problema dos eventos adversos na assistência à saúde prestada na terapia intensiva e os fatores de risco associados à incidência de eventos.
Palavras-chave:
Efeitos Adversos; Cuidados Críticos; Segurança do Paciente; Mortalidade; Qualidade da Assistência à Saúde
Introduction
Patients with severe conditions, who are hemodynamically unstable and subjected to several diagnostic-therapeutic interventions have a higher risk for adverse events. The complexity of care for critical patients requires advanced technology and specialized professionals able to immediately intervene in any change in their clinical condition 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.), (22. Resar RK, Rozich JD, Simmonds T, Haraden CR. A trigger tool to identify adverse events in the intensive care unit. Jt Comm J Qual Patient Saf 2006; 32:585-90.), (33. Merino P, Álvarez J, Cruz Martín M, Alonso A, Gutiérrez I; SYREC Study Investigators. Adverse events in Spanish intensive care units: the SYREC study. Int J Qual Health Care 2012; 24:105-13.), (44. Pagnamenta A, Rabito G, Arosio A, Perren A, Malacrida R, Barazzoni F, et al. Adverse event reporting in adult intensive care units and the impact of a multifaceted intervention on drug-related adverse events. Ann Intensive Care 2012; 2:47.. Intensive care units (ICU) concentrate patients with this profile, however, there is a high frequency of adverse events in hospital wards 55. Mendes W, Martins M, Rozenfeld S, Travassos C. The assessment of adverse events in hospitals in Brazil. Int J Qual Health Care 2009; 21:279-84.), (66. Sousa P, Uva AS, Serranheira F, Leite E, Nunes C. Segurança do doente: eventos adversos em hospitais portugueses: estudo piloto de incidência, impacte e evitabilidade. Lisboa: Escola Nacional de Saúde Pública, Universidade de Lisboa; 2011.. Standardization of processes and adoption of restrictive measures are not enough to explain this pattern. Studies that evaluate adverse events occurrence should therefore consider a complex set of factors that include the severity of the case, the quality of care and the institutional context.
Advanced age, comorbidities, compromised kidney and liver functions, lower levels of consciousness, polypharmacy use, inappropriate drug administration, invasive procedures and long hospital stays are risk factors associated with adverse events occurrence 77. Padilha KG. Ocorrências iatrogênicas em unidade de terapia intensiva (UTI): análise dos fatores relacionados. Rev Paul Enferm 2006; 25:18-23.), (88. Zhang M, Holman CDJ, Price SD, Sanfilippo FM, Preen DB, Bulsara MK. Comorbidity and repeat admission to hospital for adverse drug reactions in older adults: retrospective cohort study. BMJ 2009; 338:a2752.), (99. Naessens JM, Campbell CR, Shah N, Berg B, Lefante JJ, Williams AR, et al. Effect of illness severity and comorbidity on patient safety and adverse events. Am J Med Qual 2012; 27:48-57., defined as an incident that produces a damage or lesion on the patient that is attributed to care 1010. Runciman W, Hibbert P, Thomson R, Van Der Schaaf T, Sherman H, Lewalle P. Towards an international classification for patient safety: key concepts and terms. Int J Qual Health Care 2009; 21:18-26.. These events compromise the quality of care and may lead to death, disability, loss of confidence in and dissatisfaction with the service 1111. Brown C, Hofer T, Johal A, Thomson R, Nicholl J, Franklin BD, et al. An epistemology of patient safety research: a framework for study design and interpretation. Part 1. Conceptualising and developing interventions. Qual Saf Health Care 2008; 17:158-62..
Adverse events incidence varies between 0.87 to 34.7 per 100 patients admitted to ICUs 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.), (22. Resar RK, Rozich JD, Simmonds T, Haraden CR. A trigger tool to identify adverse events in the intensive care unit. Jt Comm J Qual Patient Saf 2006; 32:585-90.), (44. Pagnamenta A, Rabito G, Arosio A, Perren A, Malacrida R, Barazzoni F, et al. Adverse event reporting in adult intensive care units and the impact of a multifaceted intervention on drug-related adverse events. Ann Intensive Care 2012; 2:47.), (1212. McLaughlin N, Leslie GD, Williams TA, Dobb GJ. Examining the occurrence of adverse events within 72 hours of discharge from the intensive care unit. Anaesth Intensive Care 2007; 35:486-93.), (1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.),(1414. Garrouste-Orgeas M, Timsit JF, Vesin A, Schwebel C, Arnodo P, Lefrant JY, et al. Selected medical errors in the intensive care unit. Am J Respir Crit Care Med 2010; 181:134-42.),(1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63.),(1616. Ilan R, Squires M, Panopoulos C, Day A. Increasing patient safety event reporting in 2 intensive care units: a prospective interventional study. J Crit Care 2011; 26:431.e11-431.e18.), (1717. Pottier V, Daubin C, Lerolle N, Gaillard C, Viquesnel G, Plaud B, et al. Overview of adverse events related to invasive procedures in the intensive care unit. Am J Infect Control 2012; 40:241-6.), (1818. Nilsson L, Pihl A, Tågsjö M, Ericsson E. Adverse events are common on the intensive care unit: results from a structured record review. Acta Anaesthesiol Scand 2012; 56:959-65.), (1919. Sommella L, de Waure C, Ferriero AM, Biasco A, Mainelli MT, Pinnarelli L, et al. The incidence of adverse events in an Italian acute care hospital: findings of a two-stage method in a retrospective cohort study. BMC Health Serv Res 2014; 14:358.), (2020. Ahmed AH, Thongprayoon C, Schenck LA, Malinchoc M, Konvalinová A, Keegan MT, et al. Adverse in-hospital events are associated with increased in-hospital mortality and length of stay in patients with or at risk of acute respiratory distress syndrome. Mayo Clin Proc 2015; 90:321-8.. The high variability observed in estimates can be partly explained by notification and information obtainment mechanisms, and processes for classifying the event and possible damage (who determines the classification and how). The most frequently used methods in investigating adverse events are voluntary notification and retrospective review of medical records, which have limitations in identifying associated risk factors and determining the chain of causality 1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63.), (2121. Beccaria LM, Pereira RAM, Contrin LM, Lobo SMA, Trajano DHL. Nursing care adverse events at an intensive care unit. Rev Bras Ter Intensiva 2009; 21:276-82.), (2222. Nascimento CCP, Toffoletto MC, Gonçalves LA, Freitas WG, Padilha KG. Indicators of healthcare results: analysis of adverse events during hospital stays. Rev Latinoam Enferm 2008; 16:746-51.), (2323. Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32..
Studies that used combined methods, such as reviewing medical records, direct observation and voluntary notification, favor the identification of a higher proportion of adverse events 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.), (1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.), (1717. Pottier V, Daubin C, Lerolle N, Gaillard C, Viquesnel G, Plaud B, et al. Overview of adverse events related to invasive procedures in the intensive care unit. Am J Infect Control 2012; 40:241-6.. Three cohort studies conducted by Rothschild et al. 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700. and Foster et al. 1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.), (1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63. used a combination of these methods and found, respectively, 20.2%, 19% and 24.6% of adverse events in hospitals. In these studies, the most frequent adverse events were infections associated with care, procedure complications and adverse drug events. In Brazil, the main studies that analyzed adverse events in intensive therapy used a sectional approach with medical record review or voluntary notification 2121. Beccaria LM, Pereira RAM, Contrin LM, Lobo SMA, Trajano DHL. Nursing care adverse events at an intensive care unit. Rev Bras Ter Intensiva 2009; 21:276-82.), (2222. Nascimento CCP, Toffoletto MC, Gonçalves LA, Freitas WG, Padilha KG. Indicators of healthcare results: analysis of adverse events during hospital stays. Rev Latinoam Enferm 2008; 16:746-51.), (2323. Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32.. In a cohort study on 195 patients admitted for clinical reasons in São Paulo, 81.7% had an adverse events. The most frequent events were related to procedures/clinical process and adverse drug events 2424. Zambon LS. Segurança do paciente em terapia intensiva: caracterização de eventos adversos em pacientes críticos, avaliação de sua relação com mortalidade e identificação de fatores de risco para sua ocorrência [Doctoral Dissertation]. São Paulo: Faculdade de Medicina, Universidade de São Paulo; 2014..
Given the lack of longitudinal studies seeking to investigate patient safety incident, particularly those resulting in damage, and the vulnerability to adverse events of patients with severe cases in intensive therapy, we believe it is crucial to evaluate patient profile and type of damage resulting from care. This study sought to evaluate adverse events occurrence and their impact on length of hospital stay and mortality in a teaching hospital ICU.
Methods
This is a concurrent cohort study of patients over 18 years of age admitted to the ICU of a teaching hospital located in Rio de Janeiro city, Brazil. We considered eligible patients admitted between August 2011 and July 2012. Of these, 23 were considered ineligible due to the following criteria: under 18 years of age (n = 12), length of ICU stay under six hours (n = 5), absence of a signed Informed Consent Form by a legal representative (n = 6) (Figure 1). Patients were followed from admission to the ICU until discharge, transfer to a hospital ward or death. There were no losses during follow up.
Flow chart representing the study population and adverse events detected, at an universitary hospital intesive care unit (ICU). Rio de Janeiro, Brazil, August 2011 to July 2012.
We analyzed harmful incident/adverse events, evaluating events associated with care which reached the patient and caused harm (lesion or damage) and disregarding those related to the underlying disease 1010. Runciman W, Hibbert P, Thomson R, Van Der Schaaf T, Sherman H, Lewalle P. Towards an international classification for patient safety: key concepts and terms. Int J Qual Health Care 2009; 21:18-26.. Incidents were identified based on tracking criteria adapted from the method proposed by the Institute for Healthcare Improvement (IHI) 2525. Institute for Healthcare Improvement. IHI global trigger tool for measuring adverse events. Cambridge: Institute for Healthcare Improvement; 2009. (IHI Innovation Series White Paper).. Of the trigger tool proposed by the IHI, we used one related to health care, four related to intensive care and twelve related to medication use.
Identification and confirmation of incidents involved three evaluation stages. The first, based on explicit criteria (trigger tool) and stimulated voluntary notification, was carried out by two nurses through daily, prospective review of medical records. This daily review involved 17 trigger tool in the following sections of the medical record: discharge summary, medication and nurse prescriptions, progress notes, medical procedures and laboratory exams. Information related to the interdisciplinary team's progress notes, to laboratory exams and to the discharge summary were directly collected from the electronic records. Data related to prescribed medication and capillary glycemia results were extracted from the printed prescriptions. In order to identify the trigger tool "excessive sedation/presence of hypotension", we required data from the daily evaluation of arterial pressure values, manually recorded in a form. The presence of at least one trigger tool guaranteed case selection for the second evaluation stage.
On the second stage, medical records considered eligible were re-evaluated by two experienced nurses in order to identify evidences of damage attributed to care. In cases in which information recorded on the medical records was insufficient for evaluation, complementary data were drawn from diverse sources (nursing records, change of shift reports, consultations with the medical, nursing and physiotherapy teams).
On the last stage, we confirmed incidents through expert consensus (one doctor, one pharmacist and two nurses). Medical records with possible adverse events were submitted to clinical evidence evaluation and adverse events confirmation by the experts. We used the Naranjo algorithm in judging and establishing a causal relationship for adverse events related to medication 2626. Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30:239-45.. We resolved divergences through consensus based on clinical judgment. We excluded events that took place outside the period when patients were in the ICU.
We classified adverse events into groups, following World Health Organization (WHO) guidelines 1010. Runciman W, Hibbert P, Thomson R, Van Der Schaaf T, Sherman H, Lewalle P. Towards an international classification for patient safety: key concepts and terms. Int J Qual Health Care 2009; 21:18-26.. Adverse events were sorted into these groups based on incident origin and shared characteristics. Groups were classified into five types: "clinical process/procedure" (pressure ulcer, failure to carry out a recommended procedure, complications from procedures such as orotracheal intubation, gastric catheterization, urinary catheterization, venous puncture, arterial puncture, problems in ventilation handling, and diagnostic error), "healthcare-associated infections" (bloodstream infection, respiratory tract infection, urinary tract infection), "intravenous medication/fluids" (dose error, dose omission, wrong scheduling, contraindications and medication past the expiration date), "medical device/equipment" (equipment failure/damage) and "infrastructure/buildings/fixtures" (water supply shortage). Since we were studying incidents that occurred in intensive care, we did not evaluate surgical infections resulting from procedures carried out in the operating room.
We classified pressure ulcers according to the European Pressure Ulcer Advisory Panel (EPUAP) and the National Pressure Ulcer Advisory Panel (NPUAP) 2727. National Pressure Ulcer Advisory Panel; European Pressure Ulcer Advisory Panel. Pressure ulcer treatment: technical report. Washington DC: National Pressure Ulcer Advisory Panel; 2009. in: stage I, stage II, stage III, stage IV and other stages.
In order to evaluate case severity, we used the Charlson comorbidity index (CCI), which quantifies a patient's risk of death. We calculated the CCI according to the algorithm developed by Quan et al. 2828. Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi J-C, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 2005; 43:1130-9., adapted for the International Classification of Diseases, 10th revision (ICD-10).
The covariates we analyzed were related to age, sex, educational level, severity profile, number of comorbidities, length of hospital ward stay, length of ICU stay, number of medications, vasoactive amine use and sedative use.
For average comparison, we used Student's t test for the independent samples. For the comparative analysis between categorical variables, we used the chi-squared test with Yate's correction and Fisher's exact test. Results were considered statistically significant for a p-value < 0.05.
We calculated the following frequency measures: proportion of patients with adverse events (number of patients with at least one adverse event/patient total), adverse event incidence rate (number of new adverse event cases/sum of time in which individuals were at risk for developing adverse event), crude mortality rate (number of deaths/sum of time in which individuals were at risk), proportion of patients with a pressure ulcer (number of patients with at least one pressure ulcer/patient total), pressure ulcer incidence rate (number of pressure ulcers/sum of time in which individuals were at risk for developing a pressure ulcer), proportion of patients with an adverse drug event (number of patients with at least one adverse drug event/patient total), adverse drug event incidence rate (number of adverse drug events/sum of time in which individuals were at risk for developing an adverse drug event) and proportion of patients with infection associated with care (number of patients with at least one infection/patient total).
We calculated survival considering the interval between when a patient entered the cohort (date of ICU admission) until the moment of death or censoring. In the exploratory analysis, in order to identify adverse events impact during hospital stay, we compared survival curves using the Kaplan-Meier method. The times until the outcome were compared through nonparametric tests, log-rank and peto, which enable us to verify if there is a significant difference between the groups in terms of survival.
We evaluated the association between adverse event occurrence and the outcome (death in the ICU) through multivariate logistical regression models that estimated the odds ratio. We used the following explanatory variables in the analysis: age (in years), sex (female, male), total CCI score, adverse event occurrence and reason for ICU admission (clinical, surgical). We included risk adjustment variables (sex, age and Charlson index) in all models. We used a significance level of 0.10. We verified the final model's fit with deviance, Hosmer-Lemeshow and Pearson tests.
For the statistical analyses, we used the open software R 3.1 (The R Foundation for Statistical Computing, Vienna, Austria; http://www.r-project.org).
This study was approved by the Ethical Review Board of the hospital we studied, process number 171/10.
Results
There were 355 patients in the cohort, with an average age of 60.2 years (standard deviation ‒ SD = 17.9). Of the patients, 25.2% were over 74 years of age, most had comorbidities related to the Charlson index (Figure 1; Table 1). Of the clinical conditions included in the index, the most frequent were malignant neoplasias (26.6%) and diabetes (17.9%). Comorbidities related to lung, liver and kidney diseases were reported in, respectively, 7.9%, 6.9% and 6.7% of admissions. Of the 355 admissions, 28.2% presented a Charlson index score equal to or higher than 3 and had, therefore, a high risk for death.
Admission characteristics of patients admitted to an universitary hospital intensive care unit (ICU), according to occurrence of adverse events. Rio de Janeiro, Brazil, August 2011 to July 2012.
We evaluated 4,129 trigger tool in 311 patients with potential adverse events through daily, prospective medical record review and stimulated voluntary notification; 155 cases were re-evaluated by experts in a consensus meeting. We confirmed 324 adverse events in 115 patients (Figure 1).
In the cohort, 32.4% of patients had adverse event (115 of 355; 95%CI: 27.7-37.4). The incidence rate was 9.4 adverse events per 100 patients-day (324 out of 3,448 patients-day) and there were, on average, 2.8 adverse events per patient (324 for 115).
Most patients were admitted for surgical reasons (74.9%) during admission to the ICU. Average hospital ward stay was 9.9 days (SD = 17.6) and average ICU stay was 8.9 days (SD = 11.7), varying between 1 to 248 days (Table 1). Patients with adverse event had the longest average ICU stay, when compared to those without adverse event (p-value ≤ 0.000).
There was no statistically significant relationship between adverse event occurrence and characteristics related to case severity (age, sex, Charlson index). However, there was a significant relationship regarding number of comorbidities (directly proportional). Variables related to the care process (specialty, drug therapy and length of stay) were statistically significant (Table 1).
The number of medications were directly related to adverse event occurrence. Use of five medications or more concentrated around 98.3% of adverse event. Vasoactive amine use and sedative use were associated with adverse event occurrence (p-value < 0.000) (Table 1).
During the ICU stay, 23.4% of patients died. The intensive care crude mortality rate was 2.4 per 100 patients in the period (83 out of 3,448 patients-day). Out of the 115 patients who had adverse event, 35.6% died.
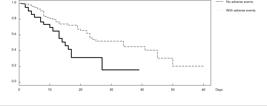
The distribution of the likelihood of survival in relation to length of ICU stay merits discussion. Since the observed differences were significant in the peto and log-rank tests (peto: Chisq = 15.2, p = 0.000; log-rank: Chisq = 15.2, p = 0.000), there are differences in the variation of the likelihood of dying over time. Median length of stay among patients with and without adverse event was, respectively, 34 and 15 days. The group with adverse event had a higher survival than the group without adverse event, a paradox apparently associated with case severity and reverse causality (Figure 2).
Survival curve (Kaplan-Meier) by adverse events occurrence in an universitary hospital intensive care unit (ICU). Rio de Janeiro, Brazil, August 2011 to July 2012.
The highest proportion of adverse event was related to what the WHO classifies as "clinical process/procedure" (59.3%). Of these adverse events, 156 were due to pressure ulcers (48.2%), 17 were due to damage from vascular catheter handling (5.3%), 15 were due to damage from ventilation handling (4.6%), two were due to damage from urinary catheter handling, one was due to damage from gastric catheter handling (0.6%) and one was due to hypoglycemia (0.3%) (Table 2). The proportion of patients who had at least one pressure ulcer was 19.7% (95%CI: 15.5-23.8). The incidence rate was 4.5 ulcers per 100 patients-day (156 out of 3,448 patients-day).
Among adverse events associated with intravenous medication/fluids, 34 were due to arterial hypotension (10.5%), 16 were due to hypoglycemia resulting from insulin and oral hypoglycemics use (5%) and five were due to nausea/vomiting (1.6%) (Table 2). The proportion of patients who had adverse drug events was 13.8% (95%CI: 10.6-17.8) and the incidence rate was 2.3 per 100 patients-day (79 out of 3,448 patients-day).
Thirty-two patients had 51 adverse events of healthcare-associated infection, a proportion of 9%. Primary bloodstream infection (6.1%), pneumonia (4.7%) and central vascular access infection (3.4%) were the most common types (Table 2).
Distribution of adverse events types identified in patients admitted to an universitary hospital intensive care unit (ICU). Rio de Janeiro, Brazil, August 2011 to July 2012.
Considering death as an outcome, we adjusted the three models in order to estimate the effect of the association between adverse event occurrence and death (Table 3). In the final model (model 3), we included the patient risk adjustment variables, adverse event occurrence and reason for admission to the unit. Variables age and adverse event had positive coefficients, that is, each one year increase in patient age (OR = 1.020; 95%CI: 1.005-1.037) and adverse event occurrence (OR = 2.047; 95%CI: 1.172-3.570) led to an increase in the odds of dying in intensive care. The variable surgical reason for ICU admission had a protective effect (OR = 0.166; 95%CI: 0.093-0.297).
Multivariate analysis (logistical model) of variables associated with death in patients admitted to an universitary hospital intensive care unit (ICU). Rio de Janeiro, Brazil.
Discussion
In this study, adverse event incidence was high, higher than estimates from international studies which also used a combination of methods in detecting events 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.), (1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.), (1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63.), (1919. Sommella L, de Waure C, Ferriero AM, Biasco A, Mainelli MT, Pinnarelli L, et al. The incidence of adverse events in an Italian acute care hospital: findings of a two-stage method in a retrospective cohort study. BMC Health Serv Res 2014; 14:358.), (2020. Ahmed AH, Thongprayoon C, Schenck LA, Malinchoc M, Konvalinová A, Keegan MT, et al. Adverse in-hospital events are associated with increased in-hospital mortality and length of stay in patients with or at risk of acute respiratory distress syndrome. Mayo Clin Proc 2015; 90:321-8.. This divergence is in part due to operational differences in adverse event and damage definitions present in the International Classification for Patient Safety (ICPS) 1010. Runciman W, Hibbert P, Thomson R, Van Der Schaaf T, Sherman H, Lewalle P. Towards an international classification for patient safety: key concepts and terms. Int J Qual Health Care 2009; 21:18-26. on which this study was based. Additionally, the trigger tool proposed by the IHI 2525. Institute for Healthcare Improvement. IHI global trigger tool for measuring adverse events. Cambridge: Institute for Healthcare Improvement; 2009. (IHI Innovation Series White Paper). are considered highly sensitive for identifying events 2929. Naessens JM, Campbell CR, Huddleston JM, Berg BP, Lefante JJ, Williams AR, et al. A comparison of hospital adverse events identified by three widely used detection methods. Int J Qual Health Care 2009; 21:301-7.. A study carried out in the United States that combined different strategies verified that 20.2% of patients had adverse events (3.62 adverse events per 100 patients-day) 11. Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.. In Canada, in 2008 and 2011, studies detected, respectively, 19% 1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259. and 24.6% of patients with adverse events (4.50 adverse events per 100 patients-day) 1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63.. In comparisons with studies that only used medical record review, the differences are even greater 22. Resar RK, Rozich JD, Simmonds T, Haraden CR. A trigger tool to identify adverse events in the intensive care unit. Jt Comm J Qual Patient Saf 2006; 32:585-90.), (1212. McLaughlin N, Leslie GD, Williams TA, Dobb GJ. Examining the occurrence of adverse events within 72 hours of discharge from the intensive care unit. Anaesth Intensive Care 2007; 35:486-93.), (1414. Garrouste-Orgeas M, Timsit JF, Vesin A, Schwebel C, Arnodo P, Lefrant JY, et al. Selected medical errors in the intensive care unit. Am J Respir Crit Care Med 2010; 181:134-42..
In Brazil, studies by Nascimento et al. 2222. Nascimento CCP, Toffoletto MC, Gonçalves LA, Freitas WG, Padilha KG. Indicators of healthcare results: analysis of adverse events during hospital stays. Rev Latinoam Enferm 2008; 16:746-51. and Beccaria et al. 2121. Beccaria LM, Pereira RAM, Contrin LM, Lobo SMA, Trajano DHL. Nursing care adverse events at an intensive care unit. Rev Bras Ter Intensiva 2009; 21:276-82., which evaluated events that could possible cause, or did cause, damage to patients in intensive care, respectively identified a total of 229 adverse events through notification and 550 adverse events through clinical monitoring. The method used in identifying incidents, the data extraction technique, the rationality used in selecting a possible incident and the incident confirmation definition and process may explain a large part of the variability of adverse event estimates.
Differences in adverse event identification strategies interfered with comparability between this study's estimates and those observed in the literature. We believe that using a combination of methods (medical record review, observation of care and notification) favored the detection of a higher proportion and spectrum of incident types. Nonetheless, use of these strategies in this study was not efficient in identifying near misses.
Adverse events were associated with an increase in average length of ICU stay. There was a statistically significant association between adverse event and mortality. The impact on increased length of stay and on mortality for patients who suffered adverse event in intensive care is a serious problem resulting from damages associated with care and shows the need for actions directed at the care process and at incident reduction. Length of stay may vary according to patient and hospital characteristics. Stays that are significantly shorter than expected may indicate the presence of efforts directed at reducing costs through premature patient discharge or greater case severity (high mortality in the first days/hours). On the other hand, stays that are significantly longer than expected may indicate low quality of care, since this pattern can result from complications arising from deficient care (or adverse event presence).
Adverse event occurrence was associated with an increase of 19 days in the average length of ICU stay, lower than results found by Forster at al. 1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259. (more than 31 days of hospital stay) and higher than results found by Ahmed et al. 2020. Ahmed AH, Thongprayoon C, Schenck LA, Malinchoc M, Konvalinová A, Keegan MT, et al. Adverse in-hospital events are associated with increased in-hospital mortality and length of stay in patients with or at risk of acute respiratory distress syndrome. Mayo Clin Proc 2015; 90:321-8. (2.4 days of ICU stay). The relationship between adverse event and length of ICU stay signals the possibility of reverse causality. The exposure (length of stay) may be influenced by the outcome's (adverse event) occurrence. Generally speaking, patients with less severe cases spend less time in intensive care and those who die within the first days do not have adverse events. However, patients with long stays often have more severe cases and suffer adverse events. In order to evaluate the impact of adverse events impact, the Ibero-American Study on Adverse Events (IBEAS) recommends considering length of stay attributed to adverse event, difference in length of stay with and without AE and additional procedures/treatments due to adverse event occurrence 3030. Ministerio de Sanidad y Política Social. Estudio IBEAS: prevalencia de efectos adversos en hospitales de Latinoamérica. Madrid: Ministerio de Sanidad y Política Social; 2010. (Informes, Estudios e Investigación)..
The proportion of deaths in intensive care in this study was similar to that of the Canadian study 1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259. (25%; 95%CI: 19-31) in that patients with adverse events had higher odds of dying in intensive care. A cohort study carried out in French ICUs identified a positive association between occurrence of two or more adverse events and death (OR = 3.09; 95%CI: 1.30-7.36) 1414. Garrouste-Orgeas M, Timsit JF, Vesin A, Schwebel C, Arnodo P, Lefrant JY, et al. Selected medical errors in the intensive care unit. Am J Respir Crit Care Med 2010; 181:134-42.. Brazilian studies evaluated the association between adverse events and hospital mortality and found a higher risk in their estimates than this study 3131. Daud-Gallotti R, Novaes HMD, Lorenzi MC, Eluf-Neto J, Okamura MN, Velasco IT. Adverse events and death in stroke patients admitted to the emergency department of a tertiary university hospital. Eur J Emerg Med 2005; 12:63-71.), (3232. Martins M, Travassos C, Mendes W, Pavão ALB. Hospital deaths and adverse events in Brazil. BMC Health Serv Res 2011; 11:223.), (3333. Dias MAE, Martins M, Navarro N. Adverse outcome screening in hospitalizations of the Brazilian Unified Health System. Rev Saúde Pública 2012; 46:719-29.. However, these studies 3131. Daud-Gallotti R, Novaes HMD, Lorenzi MC, Eluf-Neto J, Okamura MN, Velasco IT. Adverse events and death in stroke patients admitted to the emergency department of a tertiary university hospital. Eur J Emerg Med 2005; 12:63-71.), (3232. Martins M, Travassos C, Mendes W, Pavão ALB. Hospital deaths and adverse events in Brazil. BMC Health Serv Res 2011; 11:223.), (3333. Dias MAE, Martins M, Navarro N. Adverse outcome screening in hospitalizations of the Brazilian Unified Health System. Rev Saúde Pública 2012; 46:719-29. did not involve patients in intensive care and used different methods, which prevents us from establishing a parallel with their results.
Age was associated to risk of death. On the other hand, a surgical reason for admission was a protective factor. Studies have shown that advanced age, high Charlson score, emergency admission and adverse events occurrence are risk factors for mortality 1313. Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.), (3232. Martins M, Travassos C, Mendes W, Pavão ALB. Hospital deaths and adverse events in Brazil. BMC Health Serv Res 2011; 11:223.), (3333. Dias MAE, Martins M, Navarro N. Adverse outcome screening in hospitalizations of the Brazilian Unified Health System. Rev Saúde Pública 2012; 46:719-29..
The main types of events with damage that we identified were related to clinical processes/procedures, medication and infections associated with care. Among the adverse events that took place in intensive care, most were related to clinical processes/procedures. Of these, pressure ulcers were the most common, followed by damage from vascular catheter handling and damage from ventilation handling. In this study, the proportion of adverse events resulting from complications from procedures - damage from vascular catheter handling, damage from failure in ventilation handling, damage from urinary catheter handling, damage from gastric catheter handling (10.8%) - was lower than that found by Forster et al. 1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63., i.e., 23%. A possible explanation for this fact is that Forster et al. 1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63. use the taxonomy "avoidable and potential adverse event", including cases of errors which lead to a potential damage to the patient. A study by Zambon 2424. Zambon LS. Segurança do paciente em terapia intensiva: caracterização de eventos adversos em pacientes críticos, avaliação de sua relação com mortalidade e identificação de fatores de risco para sua ocorrência [Doctoral Dissertation]. São Paulo: Faculdade de Medicina, Universidade de São Paulo; 2014. detected 1,126 adverse events in an ICU, of which 54% were due to clinical processes/procedures, 25.8% were related to medication, 13.9% were related to nutrition and 5.5% were related to infections.
In the literature, the incidence of pressure ulcers in intensive care varies between 3.3% and 19.6% 3434. Shahin ESM, Dassen T, Halfens RJG. Incidence, prevention and treatment of pressure ulcers in intensive care patients: a longitudinal study. Int J Nurs Stud 2009; 46:413-21.), (3535. Manzano F, Navarro MJ, Roldán D, Moral MA, Leyva I, Guerrero C, et al. Pressure ulcer incidence and risk factors in ventilated intensive care patients. J Crit Care 2010; 25:469-76.), (3636. VanGilder C, Amlung S, Harrison P, Meyer S. Results of the 2008-2009 International Pressure Ulcer Prevalence Survey and a 3-year, acute care, unit-specific analysis. Ostomy Wound Manage 2009; 55:39-45.. Forster et al. 1515. Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63. did not identify this event in a Canadian ICU, something unusual in Brazil. In Brazilian ICUs, the incidence was much higher (31% to 62.5%) 3737. Rogensk NMB, Santos VLCG. Estudo sobre a incidência de úlcera por pressão em um hospital universitário. Rev Latinoam Enferm 2005; 13:474-80.), (3838. Fernandes LM, Caliri MHL. Using the Braden and Glasgow Scales to predict pressure ulcer risk in patients hospitalized at intensive care units. Rev Latinoam Enferm 2008; 16:973-8.), (3939. Gomes FSL, Bastos MAR, Matozinhos FP, Temponi HR, Velásquez-Meléndez G. Fatores associados à úlcera por pressão em pacientes internados nos Centros de Terapia Intensiva de Adultos. Rev Esc Enferm USP 2010; 44:1070-6.. In this study, 19.71% of patients developed pressure ulcers, an estimate that is comparable to international studies and inferior to national studies.
In a study carried out in France, 34.7% of patients admitted to an ICU had adverse events related to procedures 1717. Pottier V, Daubin C, Lerolle N, Gaillard C, Viquesnel G, Plaud B, et al. Overview of adverse events related to invasive procedures in the intensive care unit. Am J Infect Control 2012; 40:241-6.; hospital infections were considered a complication from procedures. In this study, infections were evaluated separately and affected 9% of patients. The most frequent types of infections were primary bloodstream infections, pneumonia and central vascular access infections. The literature shows that the occurrence of pneumonia associated with mechanical ventilation, surgical site infections and bloodstream infections associated with intravascular devices affect between 5% and 10% of hospitalized patients 4040. Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M. Adverse drug reactions in hospital in-patients: a prospective analysis of 3,695 patient-episodes. PLoS One 2009; 4:e4439.), (4141. Chittick P, Sherertz RJ. Recognition and prevention of nosocomial vascular device and related bloodstream infections in the intensive care unit. Crit Care Med 2010; 38(8 Suppl):S363-72.), (4242. Miller RS, Norris PR, Jenkins JM, Talbot TR, Starmer JM, Hutchison SA, et al. Systems initiatives reduce healthcare-associated infections: a study of 22,928 device days in a single trauma unit. J Trauma 2010; 68:23-31.. Infections associated with central venous catheters, in particular, represent an increase in mortality, length of stay and hospitalization costs. It is estimated that around 60% of nosocomial bacteremias are associated with some intravascular device 4141. Chittick P, Sherertz RJ. Recognition and prevention of nosocomial vascular device and related bloodstream infections in the intensive care unit. Crit Care Med 2010; 38(8 Suppl):S363-72.), (4242. Miller RS, Norris PR, Jenkins JM, Talbot TR, Starmer JM, Hutchison SA, et al. Systems initiatives reduce healthcare-associated infections: a study of 22,928 device days in a single trauma unit. J Trauma 2010; 68:23-31.), (4343. Casey AL, Elliott TS. Prevention of central venous catheter-related infection: update. Br J Nurs 2010; 19:78-87..
The proportion of patients who had adverse drug events was 13.8%. This was the second most frequent type of event. The incidence of adverse drug events in this study was similar to that found in other studies: Switzerland (15.4%), England (15.8%) and United States (16.4%) 44. Pagnamenta A, Rabito G, Arosio A, Perren A, Malacrida R, Barazzoni F, et al. Adverse event reporting in adult intensive care units and the impact of a multifaceted intervention on drug-related adverse events. Ann Intensive Care 2012; 2:47.), (4040. Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M. Adverse drug reactions in hospital in-patients: a prospective analysis of 3,695 patient-episodes. PLoS One 2009; 4:e4439.), (4444. Seynaeve S, Verbrugghe W, Claes B, Vandenplas D, Reyntiens D, Jorens PG. Adverse drug events in intensive care units: a cross-sectional study of prevalence and risk factors. Am J Crit Care 2011; 20:e131-40.. Brazilian studies conducted by Rozenfeld et al. 4545. Rozenfeld S, Chaves SMC, Reis LGC, Martins M, Travassos C, Mendes W, et al. Efeitos adversos a medicamentos em hospital público: estudo piloto. Rev Saúde Pública 2009; 43:887-90., Roque & Melo 4646. Roque KE, Melo ECP. Adaptação dos critérios de avaliação de eventos adversos a medicamentos para uso em um hospital público no Estado do Rio de Janeiro. Rev Bras Epidemiol 2010; 13:607-19., Giordani et al. 4747. Giordani F, Rozenfeld S, Oliveira DFM, Versa GLGS, Terencio JS, Caldeira LF, et al. Vigilância de eventos adversos a medicamentos em hospitais: aplicação e desempenho de rastreadores. Rev Bras Epidemiol 2012; 15:455-67. and Reis & Cassiani 2323. Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32. identified the occurrence of adverse drug events, with frequencies between 7% and 15.6%, similar to that of this study. These studies 2323. Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32.), (4545. Rozenfeld S, Chaves SMC, Reis LGC, Martins M, Travassos C, Mendes W, et al. Efeitos adversos a medicamentos em hospital público: estudo piloto. Rev Saúde Pública 2009; 43:887-90.), (4646. Roque KE, Melo ECP. Adaptação dos critérios de avaliação de eventos adversos a medicamentos para uso em um hospital público no Estado do Rio de Janeiro. Rev Bras Epidemiol 2010; 13:607-19.), (4747. Giordani F, Rozenfeld S, Oliveira DFM, Versa GLGS, Terencio JS, Caldeira LF, et al. Vigilância de eventos adversos a medicamentos em hospitais: aplicação e desempenho de rastreadores. Rev Bras Epidemiol 2012; 15:455-67. were based on the approach proposed by IHI 2525. Institute for Healthcare Improvement. IHI global trigger tool for measuring adverse events. Cambridge: Institute for Healthcare Improvement; 2009. (IHI Innovation Series White Paper)., using trigger tool, showing reproducibility in other contexts.
Arterial hypotension was the main adverse drug event identified in this study. Hypotension events were related to the use of antiarrhythmics, coronary vasodilators, antihypertensives, diuretics, general anesthetics, opioid analgesics and benzodiazepines. Among antihypertensives, sodium nitroprusside was associated with most hypotension episodes. Failures in monitoring hourly arterial pressure and in dose readjustment were the main factors that contributed to hypotension events.
We detected 16 hypoglycemia events associated with use of oral hypoglycemics, regular and NPH insulin, and one related to interruption of enteral nutrition. In this study, we identified all glycemias equal or inferior to 50mg/dL, however, only episodes related to medication use were considered adverse events. Hypoglycemia events that met the established cutoff point and were not related to medication use were not submitted to expert evaluation.
The IHI 2525. Institute for Healthcare Improvement. IHI global trigger tool for measuring adverse events. Cambridge: Institute for Healthcare Improvement; 2009. (IHI Innovation Series White Paper). does not recommend using decision algorithms in evaluating adverse drug events. However, just as in other studies 2323. Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32.), (4747. Giordani F, Rozenfeld S, Oliveira DFM, Versa GLGS, Terencio JS, Caldeira LF, et al. Vigilância de eventos adversos a medicamentos em hospitais: aplicação e desempenho de rastreadores. Rev Bras Epidemiol 2012; 15:455-67., in this study, we used the Naranjo et al. 2626. Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30:239-45. algorithm in order to evaluate the causality of adverse drug events. The use of this algorithm reduced judgment subjectivity in the consensus meetings. Nonetheless, all divergences were resolved through clinical judgment.
Electronic record use facilitated the data collection process. Information related to the interdisciplinary team's progress notes, laboratory exams and discharge summary were wholly consulted in the computer system of the hospital we studied. Nonetheless, we had to manipulate printed prescriptions and glycemia recording forms, increasing time spent on data collection. Electronic records and printed prescriptions avoided illegibility and favored chronological analysis of events. Access to electronic records and to the data collection form regarding medication prescription enabled us to quickly check data and favored the analysis of timing in identifying and confirming a potential event in the expert consensus stage.
Longitudinal studies produce better clinical evidences, favoring the identification and analysis of adverse events, in describing risk factors associated with adverse event occurrence and determining the chain of events. This approach guarantees the maintenance of timing when evaluating causal relationships, a criterion that cannot be guaranteed in cross-sectional studies 4848. Szklo M, Javier Nieto F. Epidemiology: beyond the basics. Burlington: Jones & Bartlett Learning; 2007.. The characteristics of the problem impose challenges that may not always be solved in the early stages of exploratory analyses of repeated measures. These are different events that may occur more than once for a single patient, may result from the same risk factor and take on different configurations. For this reason, it is crucial that we advance on the appropriate modeling for repeated measures, considering observation correlation and dependence.
One limitation of this study is the occurrence of non-diferential classification errors, which result from the degree of imperfection in which information is obtained for the study group. This type of error is not related to the exposure/outcome level, and is considered an inherent problem of data collection methods 4848. Szklo M, Javier Nieto F. Epidemiology: beyond the basics. Burlington: Jones & Bartlett Learning; 2007..
Conclusions
The occurrence of adverse events represents a serious problem for intensive care and impacts length of stay and mortality, increasing both. Determining adverse event magnitude, patient profile and factors associated with occurrence of damage resulting from care is a crucial issue in improving quality and patient safety. We emphasize the need to strengthen safety culture in order to intervene in the care process and establish a commitment to safety at all levels of health organization.
In Brazil, there are few longitudinal studies seeking to identify risk factors associated with event incidence. New studies must not only describe the temporal tendency, but also take into account the correlation between successive measures and estimate the rate of change over time.
The used of a combination of adverse event tracking methods enables us not only to obtain information through medical records, but also evaluate and discuss events with the professionals involved in the case. However, the method requires time and trained observers, and is costly to institutions.
Acknowledgments
We thank Capes (process n. BEX 15930/12-2) and FAPERJ (process n. E-26/103.052/2012) for funding this study. We also thank Professor Ricardo de Amorim Garcia of the Federal University of Rio de Janeiro.
References
-
1Rothschild JM, Landrigan CP, Cronin JW, Kaushal R, Lockley SW, Burdick E, et al. The Critical Care Safety Study: the incidence and nature of adverse events and serious medical errors in intensive care. Crit Care Med 2005; 33:1694-700.
-
2Resar RK, Rozich JD, Simmonds T, Haraden CR. A trigger tool to identify adverse events in the intensive care unit. Jt Comm J Qual Patient Saf 2006; 32:585-90.
-
3Merino P, Álvarez J, Cruz Martín M, Alonso A, Gutiérrez I; SYREC Study Investigators. Adverse events in Spanish intensive care units: the SYREC study. Int J Qual Health Care 2012; 24:105-13.
-
4Pagnamenta A, Rabito G, Arosio A, Perren A, Malacrida R, Barazzoni F, et al. Adverse event reporting in adult intensive care units and the impact of a multifaceted intervention on drug-related adverse events. Ann Intensive Care 2012; 2:47.
-
5Mendes W, Martins M, Rozenfeld S, Travassos C. The assessment of adverse events in hospitals in Brazil. Int J Qual Health Care 2009; 21:279-84.
-
6Sousa P, Uva AS, Serranheira F, Leite E, Nunes C. Segurança do doente: eventos adversos em hospitais portugueses: estudo piloto de incidência, impacte e evitabilidade. Lisboa: Escola Nacional de Saúde Pública, Universidade de Lisboa; 2011.
-
7Padilha KG. Ocorrências iatrogênicas em unidade de terapia intensiva (UTI): análise dos fatores relacionados. Rev Paul Enferm 2006; 25:18-23.
-
8Zhang M, Holman CDJ, Price SD, Sanfilippo FM, Preen DB, Bulsara MK. Comorbidity and repeat admission to hospital for adverse drug reactions in older adults: retrospective cohort study. BMJ 2009; 338:a2752.
-
9Naessens JM, Campbell CR, Shah N, Berg B, Lefante JJ, Williams AR, et al. Effect of illness severity and comorbidity on patient safety and adverse events. Am J Med Qual 2012; 27:48-57.
-
10Runciman W, Hibbert P, Thomson R, Van Der Schaaf T, Sherman H, Lewalle P. Towards an international classification for patient safety: key concepts and terms. Int J Qual Health Care 2009; 21:18-26.
-
11Brown C, Hofer T, Johal A, Thomson R, Nicholl J, Franklin BD, et al. An epistemology of patient safety research: a framework for study design and interpretation. Part 1. Conceptualising and developing interventions. Qual Saf Health Care 2008; 17:158-62.
-
12McLaughlin N, Leslie GD, Williams TA, Dobb GJ. Examining the occurrence of adverse events within 72 hours of discharge from the intensive care unit. Anaesth Intensive Care 2007; 35:486-93.
-
13Forster AJ, Kyeremanteng K, Hooper J, Shojania KG, van Walraven C. The impact of adverse events in the intensive care unit on hospital mortality and length of stay. BMC Health Serv Res 2008; 8:259.
-
14Garrouste-Orgeas M, Timsit JF, Vesin A, Schwebel C, Arnodo P, Lefrant JY, et al. Selected medical errors in the intensive care unit. Am J Respir Crit Care Med 2010; 181:134-42.
-
15Forster AJ, Worthington JR, Hawken S, Bourke M, Rubens F, Shojania K, et al. Using prospective clinical surveillance to identify adverse events in hospital. BMJ Qual Saf 2011; 20:756-63.
-
16Ilan R, Squires M, Panopoulos C, Day A. Increasing patient safety event reporting in 2 intensive care units: a prospective interventional study. J Crit Care 2011; 26:431.e11-431.e18.
-
17Pottier V, Daubin C, Lerolle N, Gaillard C, Viquesnel G, Plaud B, et al. Overview of adverse events related to invasive procedures in the intensive care unit. Am J Infect Control 2012; 40:241-6.
-
18Nilsson L, Pihl A, Tågsjö M, Ericsson E. Adverse events are common on the intensive care unit: results from a structured record review. Acta Anaesthesiol Scand 2012; 56:959-65.
-
19Sommella L, de Waure C, Ferriero AM, Biasco A, Mainelli MT, Pinnarelli L, et al. The incidence of adverse events in an Italian acute care hospital: findings of a two-stage method in a retrospective cohort study. BMC Health Serv Res 2014; 14:358.
-
20Ahmed AH, Thongprayoon C, Schenck LA, Malinchoc M, Konvalinová A, Keegan MT, et al. Adverse in-hospital events are associated with increased in-hospital mortality and length of stay in patients with or at risk of acute respiratory distress syndrome. Mayo Clin Proc 2015; 90:321-8.
-
21Beccaria LM, Pereira RAM, Contrin LM, Lobo SMA, Trajano DHL. Nursing care adverse events at an intensive care unit. Rev Bras Ter Intensiva 2009; 21:276-82.
-
22Nascimento CCP, Toffoletto MC, Gonçalves LA, Freitas WG, Padilha KG. Indicators of healthcare results: analysis of adverse events during hospital stays. Rev Latinoam Enferm 2008; 16:746-51.
-
23Reis AMM, Cassiani SHDB. Adverse drug events in an intensive care unit of a university hospital. Eur J Clin Pharmacol 2011; 67:625-32.
-
24Zambon LS. Segurança do paciente em terapia intensiva: caracterização de eventos adversos em pacientes críticos, avaliação de sua relação com mortalidade e identificação de fatores de risco para sua ocorrência [Doctoral Dissertation]. São Paulo: Faculdade de Medicina, Universidade de São Paulo; 2014.
-
25Institute for Healthcare Improvement. IHI global trigger tool for measuring adverse events. Cambridge: Institute for Healthcare Improvement; 2009. (IHI Innovation Series White Paper).
-
26Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30:239-45.
-
27National Pressure Ulcer Advisory Panel; European Pressure Ulcer Advisory Panel. Pressure ulcer treatment: technical report. Washington DC: National Pressure Ulcer Advisory Panel; 2009.
-
28Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi J-C, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 2005; 43:1130-9.
-
29Naessens JM, Campbell CR, Huddleston JM, Berg BP, Lefante JJ, Williams AR, et al. A comparison of hospital adverse events identified by three widely used detection methods. Int J Qual Health Care 2009; 21:301-7.
-
30Ministerio de Sanidad y Política Social. Estudio IBEAS: prevalencia de efectos adversos en hospitales de Latinoamérica. Madrid: Ministerio de Sanidad y Política Social; 2010. (Informes, Estudios e Investigación).
-
31Daud-Gallotti R, Novaes HMD, Lorenzi MC, Eluf-Neto J, Okamura MN, Velasco IT. Adverse events and death in stroke patients admitted to the emergency department of a tertiary university hospital. Eur J Emerg Med 2005; 12:63-71.
-
32Martins M, Travassos C, Mendes W, Pavão ALB. Hospital deaths and adverse events in Brazil. BMC Health Serv Res 2011; 11:223.
-
33Dias MAE, Martins M, Navarro N. Adverse outcome screening in hospitalizations of the Brazilian Unified Health System. Rev Saúde Pública 2012; 46:719-29.
-
34Shahin ESM, Dassen T, Halfens RJG. Incidence, prevention and treatment of pressure ulcers in intensive care patients: a longitudinal study. Int J Nurs Stud 2009; 46:413-21.
-
35Manzano F, Navarro MJ, Roldán D, Moral MA, Leyva I, Guerrero C, et al. Pressure ulcer incidence and risk factors in ventilated intensive care patients. J Crit Care 2010; 25:469-76.
-
36VanGilder C, Amlung S, Harrison P, Meyer S. Results of the 2008-2009 International Pressure Ulcer Prevalence Survey and a 3-year, acute care, unit-specific analysis. Ostomy Wound Manage 2009; 55:39-45.
-
37Rogensk NMB, Santos VLCG. Estudo sobre a incidência de úlcera por pressão em um hospital universitário. Rev Latinoam Enferm 2005; 13:474-80.
-
38Fernandes LM, Caliri MHL. Using the Braden and Glasgow Scales to predict pressure ulcer risk in patients hospitalized at intensive care units. Rev Latinoam Enferm 2008; 16:973-8.
-
39Gomes FSL, Bastos MAR, Matozinhos FP, Temponi HR, Velásquez-Meléndez G. Fatores associados à úlcera por pressão em pacientes internados nos Centros de Terapia Intensiva de Adultos. Rev Esc Enferm USP 2010; 44:1070-6.
-
40Davies EC, Green CF, Taylor S, Williamson PR, Mottram DR, Pirmohamed M. Adverse drug reactions in hospital in-patients: a prospective analysis of 3,695 patient-episodes. PLoS One 2009; 4:e4439.
-
41Chittick P, Sherertz RJ. Recognition and prevention of nosocomial vascular device and related bloodstream infections in the intensive care unit. Crit Care Med 2010; 38(8 Suppl):S363-72.
-
42Miller RS, Norris PR, Jenkins JM, Talbot TR, Starmer JM, Hutchison SA, et al. Systems initiatives reduce healthcare-associated infections: a study of 22,928 device days in a single trauma unit. J Trauma 2010; 68:23-31.
-
43Casey AL, Elliott TS. Prevention of central venous catheter-related infection: update. Br J Nurs 2010; 19:78-87.
-
44Seynaeve S, Verbrugghe W, Claes B, Vandenplas D, Reyntiens D, Jorens PG. Adverse drug events in intensive care units: a cross-sectional study of prevalence and risk factors. Am J Crit Care 2011; 20:e131-40.
-
45Rozenfeld S, Chaves SMC, Reis LGC, Martins M, Travassos C, Mendes W, et al. Efeitos adversos a medicamentos em hospital público: estudo piloto. Rev Saúde Pública 2009; 43:887-90.
-
46Roque KE, Melo ECP. Adaptação dos critérios de avaliação de eventos adversos a medicamentos para uso em um hospital público no Estado do Rio de Janeiro. Rev Bras Epidemiol 2010; 13:607-19.
-
47Giordani F, Rozenfeld S, Oliveira DFM, Versa GLGS, Terencio JS, Caldeira LF, et al. Vigilância de eventos adversos a medicamentos em hospitais: aplicação e desempenho de rastreadores. Rev Bras Epidemiol 2012; 15:455-67.
-
48Szklo M, Javier Nieto F. Epidemiology: beyond the basics. Burlington: Jones & Bartlett Learning; 2007.
Publication Dates
-
Publication in this collection
Oct 2016
History
-
Received
22 May 2015 -
Reviewed
23 Aug 2015 -
Accepted
22 Feb 2016