Abstracts
Identification of species with allelopathic potential has been a target of researches aiming to use them to control crop weeds. Ipomoea cairica is considered a weed with allelopathic potential, which has already been reported. The goal of this study was to evaluate the allelopathic properties of leaf extracts from Ipomoea cairica on the germination and early development of four of the worst crop weeds in Brazil: Bidens pilosa L., Echinochloa crus-galli (L.) Beauv., Euphorbia heterophylla L. and Ipomoea grandifolia (Dammer) O´Donel. We tested the effects of leaf extracts, in four concentrations, on the germination and early development of these species. The extracts negatively affected the germination, early development and the morphology of all target species, and the phytotoxic effect was higher as the concentration of the extracts increased. The influence of the I. cairica extracts on germination, in addition to their effects on seedling development, made them more effective.
allelopathy; Bidens pilosa; Echinochloa crus-galli; Euphorbia heterophylla; Ipomoea grandifolia
A identificação de espécies com potencial alelopático tem sido alvo de estudos visando sua aplicação no controle de espécies infestantes de cultura. Ipomoea cairica é considerada uma espécie infestante com potencial alelopático já relatado. O objetivo desse trabalho foi avaliar as propriedades alelopáticas de extratos foliares aquosos de Ipomoea cairica sobre a germinação e o crescimento inicial de quatro das piores espécies infestantes de cultura no Brasil: Bidens pilosa L., Echinochloa crus-galli (L.) Beauv., Euphorbia heterophylla L. e Ipomoea grandifolia (Dammer) O´Donel. Testamos os efeitos dos extratos foliares em quatro concentrações sobre a germinação e o crescimento inicial dessas espécies. Os extratos afetaram negativamente a germinação, o crescimento inicial e a morfologia de todas as espécies alvo, sendo o efeito fitotóxico maior obtido com o aumento da concentração dos extratos testados. O conjunto de alterações ocasionadas pelo extrato de I. cairica sobre a germinação adicionadas aos seus efeitos sobre o crescimento das plântulas pode ser mais efetivo.
alelopatia; Bidens pilosa; Echinochloa crus-galli; Euphorbia heterophylla; Ipomoea grandifolia
ARTICLES ARTIGOS
Allelopathic effects of Ipomoea cairica (L.) Sweet on crop weeds1 1 Part of Master's dissertation of the first Author
Efeitos alelopáticos de Ipomoea cairica (L.) Sweet sobre espécies infestantes de cultura
Leandro Kenji Takao2 2 Author for correspondence: lktakao@gmail.com ; Jose Pedro Nepomuceno Ribeiro; Maria Inês Salgueiro Lima
Universidade Federal de São Carlos, Programa de Pós-Graduação em Ecologia e Recursos Naturais, Laboratório de Sistemática e Ecologia Química, São Carlos, SP, Brasil
ABSTRACT
Identification of species with allelopathic potential has been a target of researches aiming to use them to control crop weeds. Ipomoea cairica is considered a weed with allelopathic potential, which has already been reported. The goal of this study was to evaluate the allelopathic properties of leaf extracts from Ipomoea cairica on the germination and early development of four of the worst crop weeds in Brazil: Bidens pilosa L., Echinochloa crus-galli (L.) Beauv., Euphorbia heterophylla L. and Ipomoea grandifolia (Dammer) O´Donel. We tested the effects of leaf extracts, in four concentrations, on the germination and early development of these species. The extracts negatively affected the germination, early development and the morphology of all target species, and the phytotoxic effect was higher as the concentration of the extracts increased. The influence of the I. cairica extracts on germination, in addition to their effects on seedling development, made them more effective.
Key words: allelopathy, Bidens pilosa, Echinochloa crus-galli, Euphorbia heterophylla, Ipomoea grandifolia
RESUMO
A identificação de espécies com potencial alelopático tem sido alvo de estudos visando sua aplicação no controle de espécies infestantes de cultura. Ipomoea cairica é considerada uma espécie infestante com potencial alelopático já relatado. O objetivo desse trabalho foi avaliar as propriedades alelopáticas de extratos foliares aquosos de Ipomoea cairica sobre a germinação e o crescimento inicial de quatro das piores espécies infestantes de cultura no Brasil: Bidens pilosa L., Echinochloa crus-galli (L.) Beauv., Euphorbia heterophylla L. e Ipomoea grandifolia (Dammer) O´Donel. Testamos os efeitos dos extratos foliares em quatro concentrações sobre a germinação e o crescimento inicial dessas espécies. Os extratos afetaram negativamente a germinação, o crescimento inicial e a morfologia de todas as espécies alvo, sendo o efeito fitotóxico maior obtido com o aumento da concentração dos extratos testados. O conjunto de alterações ocasionadas pelo extrato de I. cairica sobre a germinação adicionadas aos seus efeitos sobre o crescimento das plântulas pode ser mais efetivo.
Palavras-chave: alelopatia, Bidens pilosa, Echinochloa crus-galli, Euphorbia heterophylla, Ipomoea grandifolia
Introduction
Studies about the allelopathic process have recently focused on its application in agriculture, with the goal of using allelochemicals as herbicides to develop more sustainable practices (Chon et al. 2003). Using natural products, allelochemicals and plant extracts cannot provide the same level of weed control when compared to synthetic herbicides, but they can be worthwhile when only a small quantity of herbicide can be used (Khanh et al. 2006). Excessive use of synthetic herbicides can cause pollution of soil/water and injuries to humans (Ahn et al. 2008), and natural compounds have some advantages over synthetic compounds, such as absence of halogen molecules and a shorter half-life (Duke et al. 2000). Moreover, most natural products with biological activity are partially water soluble and are bioactive at lower concentrations (Vyvyan 2002).
Annual global crop losses because of weed species amount to around 95 billion dollars in food production (FAO 2009), and weeds are among the main components of the agroecosystem that interfere in crop production (Kuva et al. 2008). They compete for resources, reducing production, and can deposit a high quantity of seeds in the soil, perpetuating the problem during future plantings (Vyvyan 2002). Bidens pilosa L. (hairy beggarticks), Echinochloa crus-galli (L.) Beauv. (barnyardgrass), Euphorbia heterophylla L. (wild poinsettia) and Ipomoea grandifolia (Dammer) O´Donel (morning glory) are among the worst weeds in several crops in Brazil and many other countries (Kissmann & Groth 1992; Norris et al. 2001).
Aquatic macrophytes have been the target of allelopathic studies showing a great number of promising species (Ahn et al. 2008). We previously verified the allelopathic potential of 25 aquatic macrophyte species and Ipomoea cairica (L.) Sweet was among those with the highest inhibition of lettuce germination. Allelopathy is pointed out as a common characteristic of weeds, which favors them in the environment (Wu et al. 2006). Ipomoea cairica is a perennial species of the Convolvulaceae that is widely distributed in tropical regions, considered noxious, invasive and becomes monodominant in invaded regions (Llamas 2003; Ma et al. 2009). The chemical nature of its secondary compounds was recently examined and two compounds, 3-3´-5-Trihidroxi-4´-7-dimethoxyflanove and 3-3´-5-Trihidroxi-4´-7-dimethoxyflavone-3-O-sulphate, were identified as being responsible for its allelopathic properties on radish (Raphanus sativus L.), cucumber (Cucumis sativus L.), Chinese cabbage (Brassica pekinensis (Lou.) Rupr.) and a weed (Ligularia virgaurea (Maxim.) Mattf), making it a possible candidate for the development of new herbicides based on natural products (Ma et al. 2009). However, its allelochemical effect on other crop weeds still needs to be elucidated.
The aim of this study was to evaluate the allelopathic activity of aqueous leaf extracts, made from Ipomoea cairica, on the germination and early development of four crop weeds: B. pilosa, E. crus-galli, E. heterophylla and I. grandifolia. Aqueous extracts can present a great variety of active substances from the secondary metabolism. Thus, we expect I. cairica to present allelopathic potential on other species of agronomical interest.
Material and methods
Collection and extract preparation
We collected adult leaves with no signs of herbivory or diseases from flowering individuals of I. cairica in the Massaguaçu River estuary (23º37'20''S and 45º21'25''O), Caraguatatuba, Sao Paulo. We dried the leaves at 45ºC in a forced circulation chamber until mass stabilization, ground the material in a mill and stored it at -10ºC in plastic bags until use. We prepared a 10% (p/v) aqueous extract with the dry powdered leaves and distilled water, and stirred the extract with a magnetic stirrer for 5 minutes at room temperature (22ºC). After storing the extract at 6ºC for 12 hours we filtered it using a vacuum pump coupled to a Büchner funnel covered with filter paper (3 µm) (Ribeiro et al., 2009 - modified). We diluted the resulting solution (10%) in distilled water to obtain solutions at concentrations of 7.5; 5; 2.5 and 1.25%, and tested their effects on the germination of B. pilosa, E. crus-galli, E. heterophylla and I. grandifolia. We had previously scarified the seeds of I. grandifolia with pure sulphuric acid (H2SO4 PA) for 4 minutes to break dormancy, and then washed them in flowing water and dried them on filter paper.
Germination bioassay
We placed the seeds of thirty target species in Petri dishes (9 cm diameter) containing a double layer of filter paper (3 µm) that was moistened with 5 mL of solution, or distilled water for control. We sealed all of the Petri dishes with PVC film, closed the lid and placed them in a DBO incubator (28ºC and light-dark cycle of 12h-12h). We counted the seeds at 12h intervals and considered seeds to be germinated when there was a protrusion of one part of the embryo through the seed coat (Borghetti & Ferreira 2004). Germinated seeds were removed to avoid recount. After 10 days we calculated the percentage, average time and informational entropy of germination (Labouriau 1983).
Early development bioassay
We placed ten seedlings of the target species, previously germinated (2-4 mm radicle), in sterilized plastic boxes (8x13x5 cm) containing a double layer of filter paper (3 µm) moistened with 8 mL of solution or distilled water for control. We closed all plastic box lids and placed them in a DBO incubator (28ºC and 12h-12h light-dark cycle). After five days, we measured the length of radicles and aerial parts (hypocotyl+cotyledon) of each plant using a caliper and calculated the percentage of dead seedlings. We also analysed the morphology of the seedlings.
Osmotic potential and pH
We measured the osmotic potentials (Micro-Osmette, model: 5004 automatic osmometer) and pH of extracts and set a bioassay of germination and early development with the target species subjected to treatments of polyethylene glycol 6000 in concentrations of corresponding osmotic potentials.
Data analyses
Each target species received five treatments (witness, 2.5, 5, 7.5 and 10% solutions) in a completely randomized block design, with five replicas, for the germination and early development bioassays. After a normality test (Kolmogorov-Smirnov: p<0.05) we analysed the percentage of germination, informational entropy and percentage of dead seedlings using the Kruskall-Wallis test and Dunn's post test (p<0.05), and the average time of germination and early development by ANOVA and Tukey's post test (p<0.05).
Results and discussion
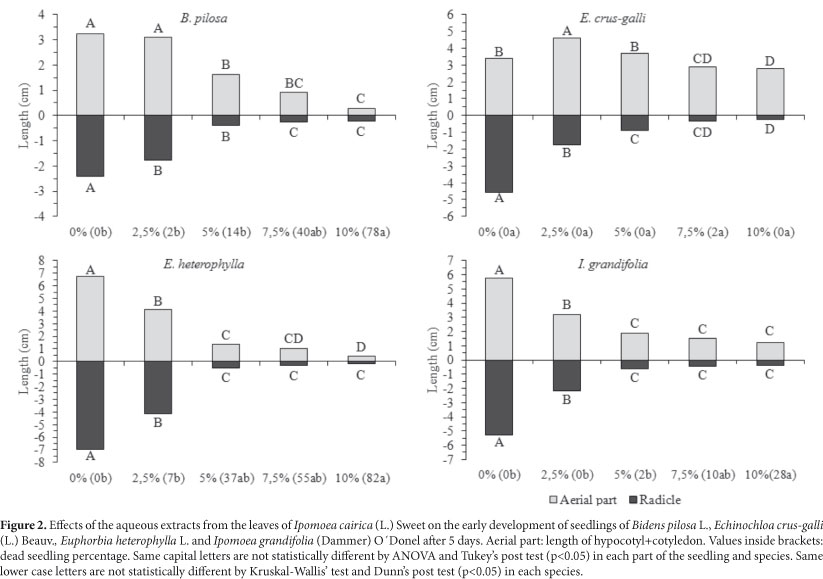
The extracts of I. cairica negatively affected the germination of all crop weed species (Tab. 1) and the effect was higher as the concentrations of the extracts increased. In general, the average time of germination was the most sensible parameter, followed by percentage and informational entropy of germination. The average time of germination for B. pilosa was significantly increased at all concentrations, and in E. crus-galli's, E. heterophylla's and I. grandifolia's for the 5% concentration. Regarding the percentage of germination, only the 10% extract significantly decreased values for I. grandifolia, E. heterophylla and E. crus-galli; and a concentration of 7.5% was toxic to B. pilosa. Besides this, the synchrony of the germination was also affected. Some informational entropy of germination values of I. grandifolia's seeds were increased. Informational entropy of germination values of B. pilosa, E. crus-galli and I. grandifolia seeds could not be calculated under the effect of higher concentration extracts due to the absence of germination in some replicas. However, germination curves (Fig. 1) emphasize the inhibitory effects of the extracts on synchrony and distribution at the time of germination. We observed erratic curves for B. pilosa, platikurtic (5, 7.5 and 10%) for E. heterophylla and E. crus-galli, and kurtosis for I. grandifolia, suggesting an extension of germination in time. Generally, higher extract concentrations were the same as lower percentage values and higher values of average time and informational entropy of germination.
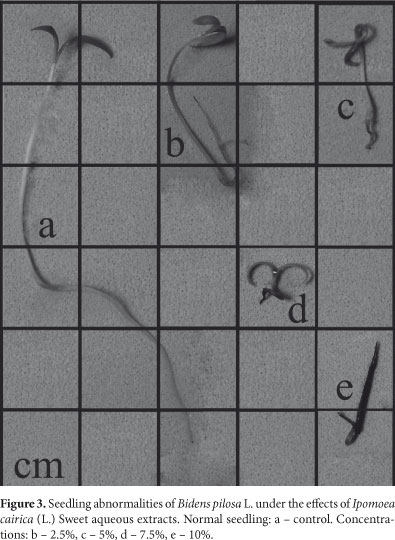
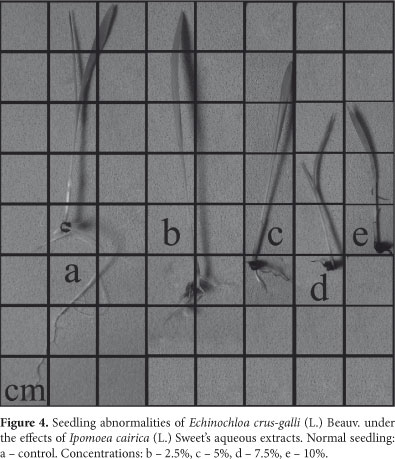
The extracts I. cairica also inhibited the early development of all target species (Fig. 2). There was an increasing inhibitory effect that concurred with and increase in extract concentration, and the radicle was the most sensitive organ. In addition, about 80% of the B. pilosa and E. heterophylla seedlings, and 30% of I. grandifolia seedlings, died as an effect of the 10% extract. In most of the cases the seedlings were rotten, impeding measurement. Generally, aerial parts are less sensitive to allelochemicals (Pires & Oliveira 2001) and only exhibit less growth. For example, the development of aerial parts of seedlings of E. crus-galli was stimulated by the 2.5% extract and inhibition increased at higher concentrations. Past studies have often related stimulation to low concentrations and inhibition to increasing concentrations (hormesis). Several species have presented a hormetic response because of allelopathic substances (Calabrese & Baldwin 2003) as well as many types of herbicides (Duke et al. 2006). Hormetic effects can influence the occurrence of primary/secondary succession in biological systems, and this concept is important in the development of herbicides based on natural products, where the focus is on toxic dose responses (Calabrese & Baldwin 2003). Qualitative evaluation of seedlings is another method used to analyze the effects of allelopathic substances (Ferreira & Aquila 2000). We verified abnormalities in the seedlings of at all target species and the radicular system was the most affected. It presented necrosis (Fig. 3c, 3d, 3e), was short and disproportionate compared to the aerial part (Fig. 4b, 4c), was atrophied (Fig. 4d, 4e and Fig. 5c, 5d, 5e) and defective (Fig. 5b and Fig. 6c, 6d, 6e). We also observed stimulation of the development of the lateral roots compared to the detriment of the primary root (Fig. 6b), suggesting hormonal disruption (Dayan et al. 2000). Many studies relate germination and inhibition of early development, and seedling abnormalities, to the effects of allelochemicals (Gatti et al. 2003; Maraschin-Silva & Aquila 2006; Ribeiro et al. 2009). However, changes in morphology and size of the roots and aerial parts can be the result of secondary effects and not because of the primary mechanism of action caused by a phytotoxic compound. For example, an evaluation of the effects of sorgoleone (a root exudate of Sorghum bicolor L.) on the early development of roots is not adequate because there is no direct relationship between this parameter and the mechanism of action of the allelochemical, which is the inhibition of the photosystem II (Dayan et al. 2000).
Generally it is assumed that the response of seeds and seedlings to plant extracts is due to allelopathy but osmotic potential can also have a negative effect on the target species (Astarita et al. 1996). The osmotic potential values of the leaf aqueous extracts of I. cairica varied from -0.13 to -0.43 MPa (Tab. 2). We observed that the effects of the 7.5 and 10% extracts on the percentage of germination of I. grandifolia and the average time of germination of E. crus-galli are partially related to osmotic potential. Osmotic potential is also partially responsible for the inhibition caused by the 5, 7.5 and 10% extracts on the aerial parts of the seedlings of E. heterophylla and I. grandifolia, and for the inhibition verified for the radicles of E. crus-galli for all concentration of the extracts. Germination and early development can also be negatively affected in extreme acid or alkaline conditions (Souza Filho et al. 1996). We observed low acidity in the extracts, and pH values varied from 5.74 to 5.92 (Tab. 2), which had no implications on germination and early development of the target species.
The effects of the I. cairica extracts on B. pilosa, E. crus-galli, E. heterophylla and I. grandifolia can benefit crops infested with them. In the control of weed species, reduction in the number of individuals in one stage (e.g., germination) increases the options that can be used to manage the following stages (e.g., development). The effectiveness of a treatment can be greater than indicated in studies that deal with the factor in an indirect way (Mohler 2001). For example, a weed population can have a few individuals capable of germinating under the effects of an allelochemical. This reduced number of individuals also germinates sparsely over time and suffers phytotoxic effects from the extracts during early development. The effects of the I. cairica extracts could be more effective if we considered them together, reducing competition for resources and space in the agricultural environment. Although our experiment is limited to the laboratory, the results show the sensitivity of another four important crop weeds in Brazil when exposed to aqueous extracts of Ipomoea cairica, and emphasizes the effects of the extracts on three different processes, germination, early development and seedling morphology.
Acknowledgements
We are grateful to Dr. Sonia Cristina Juliano Gualtieri for the laboratorial assistance, and for financial support from CAPES ("Coordenação de Aperfeiçoamento de Pessoal de Nível Superior") and CNPq ("Conselho Nacional de Desenvolvimento Científico e Tecnológico") granted to the first and second authors, respectively.
Bibliographic references
Ahn, J.K.; Park, H.Y.; Hwang, S.J.; Kong, D.S.; Chun, S.C.; Khan, T.D. & Chung, I.M. 2008. Screening of aquatic plant extracts for herbicidal, fungicidal and insecticidal activity. Allelopathy Journal 21: 361-372.
Astarita, L.V.; Ferreira, A.G. & Bergonci, J.I. 1996. Mimosa bimucronata: Allelopathy and osmotic stress. Allelopathy Journal 3: 43-50.
Borghetti, F. & Ferreira, A.G. 2004. Interpretação de resultados de germinação. Pp. 323. In: Ferreira, A. G. & Borghetti, F. (ed.). Germinação: do básico ao aplicado. Porto Alegre, Artmed.
Calabrese, E.J. & Baldwin, L.A. 2003. Hormesis: the dose-response revolution. Annual review of pharmacology and toxicology 43: 175-1Chon, S.U.; Kim, Y.M. & Lee, J.C. 2003. Herbicidal potential and quantification of causative allelochemicals from several Compositae weeds. Weed Research 43: 444-450.
Dayan, F.E.; Romagni, J.G. & Duke, S.O. 2000. Investigating the mode of action of natural phytotoxins. Journal of Chemical Ecology 26: 2079-2094.
Duke, S.O.; Cedergreen, N.; Velini, E.D. & Belz, R.G. 2006. Hormesis: Is it an important factor in herbicide use and allelopathy? Outlooks on Pest Management 17: 29-33.
Duke, S.O.; Dayan, F.E.; Ramagnani, J.G. & Rimando, A.M. 2000. Natural products as sources of herbicides: current status and future trends. Weed Research 40: 99-111.
FAO. Food and Agriculture Organization of the United Nations. http://www.fao.org/news/story/en/item/29402/icode/ (Acesso em 03/11/2010).
Ferreira, A.G. & Aquila, M.E.A. 2000. Alelopatia: Uma área emergente da ecofisiologia. Revista Brasileira de Fisiologia Vegetal 12: 175-204.
Gatti, A.B.; Perez, S.C.J.G.A. & Lima, M.I.S. 2003. Atividade alelopática de extratos aquosos de Aristolochia esperanzae O. Kuntze na germinação e no crescimento de Lactuca sativa L. e Raphanus sativus L. Acta Botanica Brasilica 3: 459-472.
Khanh, T.D.; Chung, I.M.; Tawata, S. & Xuan, T.D. 2006. Weed supression by Passiflora edulis and its potential allelochemicals. European Weed Research Society 46: 296-303.
Kissmann, K.G. & Groth, D. 1992. Plantas Infestantes e nocivas. 2. ed. São Paulo, BASF.
Kuva, M.A.; Ferraudo, A.S.; Pitelli, R.A.; Alves, P.L.C.A. & Salgado, T.P. 2008. Weed plant infestation patterns in raw sugarcane agricultural systems. Planta Daninha 26: 549-557.
Labouriau, L.F.G. 1983. A germinação das sementes. Washington, Departamento de Assuntos Científicos e Tecnológicos da Secretaria Geral da Organização dos Estados Americanos.
Llamas, K.A. 2003. Tropical Flowering Plants: a guide to identification and cultivation 1. ed. Portland, Timber Press.
Ma, R.J.; Wang, N.L.; Zhu, H.; Guo, S.J. & Chen, D.S. 2009. Isolation and identification of allelochemicals from invasive plant Ipomoea cairica. Allelopathy Journal 24: 77-84.
Maraschin-Silva, F. & Aquila, M.E.A. 2006. Potencial alelopático de espécies nativas na germinação e crescimento inicial de Lactuca sativa L. (Asteraceae). Acta Botanica Brasilica 20: 61-69.
Mohler, C.L. 2001. Weed life history: identifying vulnerabilities. Pp. 40-98. In: Liebman, M., Mohler, C. L. & Staver, C. P. (ed.). Ecological Management of Agricultural Weeds. Cambridge, Cambridge University Press.
Norris, R.F.; Elmore, C.L.; Rejmánek, M. & Akey, W.C. 2001. Spatial arrangement, density, and competition between barnyardgrass and tomato: I. Crop growth and yield. Weed Science 49: 61-68.
Pires, N.M. & Oliveira, V.R. 2001. Alelopatia. Pp. 145-185. In: Oliveira Jr., R.S. & Constantin, J. (Coord.). Plantas daninhas e seu manejo. Guaíba, Livraria e Editora Agropecuária.
Ribeiro, J.P.N.; Matsumoto, R.S.; Takao, L.K.; Voltarelli, V.M. & Lima, M.I.S. 2009. Efeitos alelopáticos de extratos aquosos de Crinum americanum L. Revista Brasileira de Botânica 32: 183-188.
Souza Filho, A.P.S.; Rodrigues, L.R.A. & Rodrigues, T.J.D. 1996. Efeitos de extratos aquosos de assa-peixe sobre a germinação de três espécies de braquiária. Planta Daninha 14: 93-101.
Vyvyan, J.A. 2002. Allelochemicals as leads for new herbicides and agrochemicals. Tetrahedron 58: 1631-1646.
Wu, J.R.; Chen, Z.Q. & Peng, S.L. 2006. Allelopathic potential of Alternanthera philoxeroides, Ipomoea cairica and Spartina alterniflora. Allelopathy Journal 18: 279-285.
Recebido em 29/03/2011
Aceito em 27/09/2011
- Ahn, J.K.; Park, H.Y.; Hwang, S.J.; Kong, D.S.; Chun, S.C.; Khan, T.D. & Chung, I.M. 2008. Screening of aquatic plant extracts for herbicidal, fungicidal and insecticidal activity. Allelopathy Journal 21: 361-372.
- Astarita, L.V.; Ferreira, A.G. & Bergonci, J.I. 1996. Mimosa bimucronata: Allelopathy and osmotic stress. Allelopathy Journal 3: 43-50.
- Borghetti, F. & Ferreira, A.G. 2004. Interpretação de resultados de germinação. Pp. 323. In: Ferreira, A. G. & Borghetti, F. (ed.). Germinação: do básico ao aplicado Porto Alegre, Artmed.
- Calabrese, E.J. & Baldwin, L.A. 2003. Hormesis: the dose-response revolution. Annual review of pharmacology and toxicology 43: 175-1Chon,
- S.U.; Kim, Y.M. & Lee, J.C. 2003. Herbicidal potential and quantification of causative allelochemicals from several Compositae weeds. Weed Research 43: 444-450.
- Dayan, F.E.; Romagni, J.G. & Duke, S.O. 2000. Investigating the mode of action of natural phytotoxins. Journal of Chemical Ecology 26: 2079-2094.
- Duke, S.O.; Cedergreen, N.; Velini, E.D. & Belz, R.G. 2006. Hormesis: Is it an important factor in herbicide use and allelopathy? Outlooks on Pest Management 17: 29-33.
- Duke, S.O.; Dayan, F.E.; Ramagnani, J.G. & Rimando, A.M. 2000. Natural products as sources of herbicides: current status and future trends. Weed Research 40: 99-111.
- FAO. Food and Agriculture Organization of the United Nations. http://www.fao.org/news/story/en/item/29402/icode/ (Acesso em 03/11/2010).
- Ferreira, A.G. & Aquila, M.E.A. 2000. Alelopatia: Uma área emergente da ecofisiologia. Revista Brasileira de Fisiologia Vegetal 12: 175-204.
- Gatti, A.B.; Perez, S.C.J.G.A. & Lima, M.I.S. 2003. Atividade alelopática de extratos aquosos de Aristolochia esperanzae O. Kuntze na germinação e no crescimento de Lactuca sativa L. e Raphanus sativus L. Acta Botanica Brasilica 3: 459-472.
- Khanh, T.D.; Chung, I.M.; Tawata, S. & Xuan, T.D. 2006. Weed supression by Passiflora edulis and its potential allelochemicals. European Weed Research Society 46: 296-303.
- Kissmann, K.G. & Groth, D. 1992. Plantas Infestantes e nocivas 2. ed. São Paulo, BASF.
- Kuva, M.A.; Ferraudo, A.S.; Pitelli, R.A.; Alves, P.L.C.A. & Salgado, T.P. 2008. Weed plant infestation patterns in raw sugarcane agricultural systems. Planta Daninha 26: 549-557.
- Labouriau, L.F.G. 1983. A germinação das sementes Washington, Departamento de Assuntos Científicos e Tecnológicos da Secretaria Geral da Organização dos Estados Americanos.
- Llamas, K.A. 2003. Tropical Flowering Plants: a guide to identification and cultivation 1. ed. Portland, Timber Press.
- Ma, R.J.; Wang, N.L.; Zhu, H.; Guo, S.J. & Chen, D.S. 2009. Isolation and identification of allelochemicals from invasive plant Ipomoea cairica Allelopathy Journal 24: 77-84.
- Maraschin-Silva, F. & Aquila, M.E.A. 2006. Potencial alelopático de espécies nativas na germinação e crescimento inicial de Lactuca sativa L. (Asteraceae). Acta Botanica Brasilica 20: 61-69.
- Mohler, C.L. 2001. Weed life history: identifying vulnerabilities. Pp. 40-98. In: Liebman, M., Mohler, C. L. & Staver, C. P. (ed.). Ecological Management of Agricultural Weeds Cambridge, Cambridge University Press.
- Norris, R.F.; Elmore, C.L.; Rejmánek, M. & Akey, W.C. 2001. Spatial arrangement, density, and competition between barnyardgrass and tomato: I. Crop growth and yield. Weed Science 49: 61-68.
- Pires, N.M. & Oliveira, V.R. 2001. Alelopatia. Pp. 145-185. In: Oliveira Jr., R.S. & Constantin, J. (Coord.). Plantas daninhas e seu manejo Guaíba, Livraria e Editora Agropecuária.
- Ribeiro, J.P.N.; Matsumoto, R.S.; Takao, L.K.; Voltarelli, V.M. & Lima, M.I.S. 2009. Efeitos alelopáticos de extratos aquosos de Crinum americanum L. Revista Brasileira de Botânica 32: 183-188.
- Souza Filho, A.P.S.; Rodrigues, L.R.A. & Rodrigues, T.J.D. 1996. Efeitos de extratos aquosos de assa-peixe sobre a germinação de três espécies de braquiária. Planta Daninha 14: 93-101.
- Vyvyan, J.A. 2002. Allelochemicals as leads for new herbicides and agrochemicals. Tetrahedron 58: 1631-1646.
- Wu, J.R.; Chen, Z.Q. & Peng, S.L. 2006. Allelopathic potential of Alternanthera philoxeroides, Ipomoea cairica and Spartina alterniflora Allelopathy Journal 18: 279-285.
Publication Dates
-
Publication in this collection
24 Jan 2012 -
Date of issue
Dec 2011
History
-
Received
29 Mar 2011 -
Accepted
27 Sept 2011