ABSTRACT
A new species of Borreria (Rubiaceae) from Chapada do Apodi in northeastern Brazil is described and illustrated. A distribution map, illustrations, a conservation assessment, a key to morphologically similar taxa and images of the seeds and pollen grains are provided.
Keywords
Caatinga; northeastern Brazil; pollen grains; Rubioideae; seeds; semiarid; taxonomy
Introduction
Borreria (Rubiaceae) is the largest genus in tribe Spermacoceae sensu stricto, with about 100 species. It is distributed throughout the American continent from the southern United States to Uruguay and central Argentina. More than half of these species (69) occur in Brazil and 33 are endemic to it (Miguel & Cabral 2013Miguel LM, Cabral EL. 2013. Borreria krapocarmeniana, a new cryptic species recovered through taxonomic analyses of Borreria scabiosoides and Borreria linoides (Spermacoceae, Rubiaceae). Systematic Botany 38: 769-781.; Miguel et al. 2015Miguel LM, Souza EB, Cabral EL. 2015. Two new species of Borreria (Spermacoceae, Rubiaceae) from the states of Goiás and Minas Gerais, Brazil. Phytotaxa 201: 149-157.; Cabral & Salas 2015Cabral EL, Miguel LM, Salas RM. 2011. Dos especies nuevas de Borreria (Rubiaceae), sinopsis y clave de las especies para Bahia, Brasil. Acta Botanica Brasilica 25: 255-276.). For the Northeastern region, 27 species are recorded and 12 occur in Ceará state (Cabral & Salas 2015Cabral EL, Salas RM. 2015. Borreria in Lista de Espécies da Flora do Brasil. Rio de Janeiro: Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB20690. 7 Jun. 2015.
http://floradobrasil.jbrj.gov.br/jabot/f...
).
Bacigalupo & Cabral (1996Bacigalupo NM, Cabral EL. 1996. Infrageneric classification of Borreria (Rubiaceae-Spermacoceae) on the basis of American species. Opera Botanica Belgica 7: 297-308.), studying American species of Borreria, recognized two subgenera (Borreria and Dasycephala), two sections (Borreria and Pseudodiodia), two subsections (Borreria and Latifoliae) and three series (Borreria, Brachystemonoides and Laeves) based on morphological data. Borreria subsect. Borreria is defined by the septicidal dehiscence of the fruit into two dehiscent mericarps, flowers with exserted stamens and style, the stigma mainly bilobed and zonocolporate and pantoporate pollen grains. Within Borreria subsect. Borreria the series are defined only by seed morphology: Borreria ser. Borreria by reticulate seeds, Borreria ser. Brachystemonoides by seeds with an elaisome and Borreria ser. Laeves by ruminate seeds.
The delimitation of the genera Borreria and Spermacoce is confused and remains unclear. Some infrageneric groups of Bacigalupo & Cabral´s classification (1996Bacigalupo NM, Cabral EL. 1996. Infrageneric classification of Borreria (Rubiaceae-Spermacoceae) on the basis of American species. Opera Botanica Belgica 7: 297-308.) seem not to be monophyletic. This has been made evident on phylogenetic studies for the tribe Spermacoceae, however those include a small number of species of Borreria sensu Bacigalupo & Cabral (1996)Bacigalupo NM, Cabral EL. 1996. Infrageneric classification of Borreria (Rubiaceae-Spermacoceae) on the basis of American species. Opera Botanica Belgica 7: 297-308. representing the American flora (nine species in Dessein 2003Dessein S. 2003. Systematic studies in the Spermacoceae (Rubiaceae). PhD Thesis, Leuven, Belgium.; five in Kårehed et al. 2008Kårehed J, Groeninckx I, Dessein S, et al. 2008. The phylogenetic utility of chloroplast and nuclear DNA markers and the phylogeny of the Rubiaceae tribe Spermacoceae. Molecular Phylogenetics and Evolution 49: 843-866.; four in Groeninckx et al. 2009Groeninckx I, Dessein S, Ochoterena H, et al. 2009. Phylogeny of the herbaceous tribe Spermacoceae (Rubiaceae) based on plastid DNA data. Annals of the Missouri Botanical Garden 96: 109-132.; eleven in Salas et al. 2015Salas RM, Viana PL, Cabral, EL, et al. 2015. Carajasia (Rubiaceae), a new and endangered genus from Carajás mountain range, Pará, Brazil. Phytotaxa 206: 14-29.). Further molecular studies and a revision of the American species of Borreria are needed to assess whether these genera should be considered separate or merged into a wider, all-encompassing Spermacoce. But for the time being we follow the generic circumscription proposed by Bacigalupo & Cabral (1996)Bacigalupo NM, Cabral EL. 1996. Infrageneric classification of Borreria (Rubiaceae-Spermacoceae) on the basis of American species. Opera Botanica Belgica 7: 297-308..
The new species here described was found as result of the study of Rubiaceae for the regional flora of Ceará. It is only known in the Chapada do Apodi, on the border between the Brazilian states of Ceará and Rio Grande do Norte. We present a morphological description, a distribution map, illustrations, a conservation assessment, a key to morphologically similar taxa and images of the seeds and pollen grains.
Materials and methods
This study is based on field observation and the study of collections from the herbaria CTES, EAC, HUEFS and HUVA (Thiers 2015Thiers, B. 2015. Index herbariorum. Part I: The herbaria of the world. New York, New York Botanical Garden. http://sweetgum.nybg.org/ih/. 15 Oct. 2015.
http://sweetgum.nybg.org/ih/...
).
Flower and seed samples were fixed in 70% alcohol, dried at critical-point, and sputter-coated with gold-palladium for scanning electron microscope (SEM) studies.
Pollen grains were obtained from anthers of one collection (Souza 3511). Samples for light microscopy (LM) were acetolyzed according to Erdtman (1966Erdtman, G. 1966. Pollen Morphology and Plant Taxonomy: Angiosperms. An introduction to Palynology-I. New York and London, Hafner Publishing Company. ) and mounted in glycerin jelly. Polar axis and equatorial diameter were measured on twenty five pollen grains using a Leica DM LB2 microscope. The terminology used to describe the pollen grains follows Punt et al. (2007Punt W, Blackmore S, Nilsson S, et al. 2007. Glossary of pollen and spore terminology. Review of Palaeobotany and Palynology 143: 1-81.).
Scanning Electron Microscope (SEM) images were taken from flowers, petals epidermis, stigma and acetolyzed pollen grains using a Jeol 5800 LV SEM at the Universidad Nacional del Nordeste, Corrientes, Argentina.
Characterization of the Chapada do Apodi
The Chapada do Apodi is located in the Northeast region of the Brazil, between the lower courses of the Jaguaribe and Piranhas-Açu rivers, along the border of the states of Ceará and Rio Grande do Norte (Prates et al. 1981Prates M, Gatto LCS, Costa MIP. 1981. Geomorfologia. In: Almeida ALS, Bittencourt-Netto O, Moreira HF. (eds.) Projeto Radambrasil. Vol. 23, Folha SB 24/25. Rio de Janeiro, Ministério das Minas e Energia. p. 301-348). It comprises a sedimentary relief formed by Cretaceous rocks of the Apodi Group, particularly calcareous from the Jandaíra Formation (Souza 1989Souza MJN. 1989. Geomorfologia. In: Martins MLR, Soares AML, Lucena GG, Souza GMB, Soares ZML. (eds.) Atlas do Ceará. Fortaleza, Iplance. p. 14-15) and partially covered by sandy and shallow soils (Fig. 1A). The altitude does not exceed 250 m and the predominant climate is semiarid with rainfall of 750-850 mm a year (Souza 1989). The vegetation in the Chapada do Apodi is Caatinga, composed of deciduous shrubs, subshrubs, small trees and annual herbs. The area was considered to be of "Extreme Importance" for the conservation of Caatinga flora by Tabarelli & Cardoso-Filho (2002Tabarelli M, Cardoso-Silva JM. 2002. Áreas e ações prioritárias para a conservação, utilização sustentável e repartição de benefícios da biodiversidade do bioma Caatinga. In: Araújo EL, Moura AN, Sampaio EVSB, Gestinari LMS, Carneiro JMT. (eds.) Biodiversidade, conservação e uso sustentável da flora do Brasil. Recife, Universidade Federal Rural de Pernambuco, Imprensa Universitária. p. 27-30). Typical Caatinga plants occur in the area, such as Poincianella pyramidalis (Tul.) L.P. Queiroz and Pityrocarpa moniliformis (Benth.) Luckow & R.W. Jobson (Leguminosae), Cordia trichotoma (Vell.) Arráb. ex Steud. (Boraginaceae), Jatropha spp. (Euphorbiaceae), Bromelia laciniosa Mart. ex Schult. & Schult.f. and Encholirium spectabile Martius ex Schult. f. (Bromeliaceae), Pilosocereus gounellei (A. Weber ex K. Schum.) Bly. ex Rowl. (Cactaceae) and Combretum leprosum Mart. (Combretaceae). Species endemic to the Caatinga are also a feature of the Chapada do Apodi landscape, such as Cordia oncocalyx Allemão and C. glazioviana (Taub.) Gottschling & J.S. Mill, as well as Hydrothrix gardneri Hook.f., a rare aquatic plant (Giulietti et al. 2004Giulietti AM, Bocage-Neta AL, Castro AAJF, et al. 2004. Diagnóstico da vegetação nativa do bioma Caatinga. In: Cardoso-Silva JM, Tabarelli M, Fonseca MT, Lins LV. (eds.) Biodiversidade da Caatinga: áreas e ações prioritárias para a conservação. Brasília, Ministério do Meio Ambiente. p. 47-90.).
A-C. Habitat and morphology of Borreria apodiensis. A. Calcareous rocks in Chapada do Apodi; B. Detail of a terminal glomerule and elliptic bracts; C. Detail of four cyathiform corollas. (A. by E.B. Souza; B-C by E. Thomaz).
Results and discussion
Taxonomic treatment
Borreriaapodiensis E.L. Cabral, L.M. Miguel & E.B. Souza, sp. nov. Type: BRAZIL. CEARÁ. Chapada do Apodi. Limoeiro do Norte, 05°11'34''S 37°59'20''W, 86 m, 28/V/2015, fl., fr., E.B. Souza 3555, R.A.S. Pontes, E.C. Thomaz & L.C. Silvestre (Holotype: EAC; Isotypes: CTES, HUEFS, HUVA).
Borreria apodiensis is very close to Borreria brownii (Rusby) Standl., but differs in the inflorescence having terminal and with 2-5 axillary glomerules [vs. 1 terminal and 1 (rarely 2) subterminal glomerules], the calyx lobes narrowly ovoid, 2-2.5 mm long with pilose margin (vs. calyx lobes ovate 0.8-1 mm long with ciliate margin) and non-ruminate seeds (vs. ruminate seeds).
Herb to subshrub, erect, (5-)20-50(-100) cm tall; stem simple or with 3-4 opposite lateral branches, quadrangular to subquadrangular, glabrous to glabrescent, green-yellowish when dry, internodes (0.9)-2-8.5-(12) cm long. Leaves opposite, rarely pseudoverticillate, sessile, rarely pseudopetiolate, leaf blades (3.5-)5-7.5(-10) x 0.5-2 cm, narrowly elliptic to elliptic, apex acute and base attenuate, sometimes with a pseudopetiole of 5-7 mm long, membranaceous to chartaceous, glabrescent above, pilose on the nerves underneath, margins scabrous, 5-6 pairs of strongly marked secondary veins; stipular sheath 3-5 mm long, pubescent to puberulous, with (6-)8-10(-14) fimbriae; fimbriae 1-3(-5) mm long, glabrous or pubescent at the base and colleter-tipped. Inflorescence terminal and with 2-4(-5) axillary glomerules, (5-)10-20(-25) mm wide, hemispheric,; involucral bracts 4-8(-12), unequal, (0.7-)2-6.7(-9.2) x (0.3-)1-2.7 cm, the external pair twice as long as glomerule diameter. Flower subsessile, pedicel 0.7-1 mm long, hypanthium 1.2-2 mm long, pilose on the third upper part; calyx 4-lobed, lobes 2-2.5 mm long, narrowly triangular, pilose at the margin; corolla 5-6.5 mm long, cyathiform, white, tube 4-5 mm long, lobes 1-1.5 mm long, triangular, externally glabrescent with long hairs on the apex of lobes, internally with short and sparse hairs on the lobes and a ring of hairs in the throat; stamens exserted, filaments 1-1.3 mm long, anthers 0.7-0.9 mm long; style 5.3-6.8 mm long, stigma bilobed, papillose; nectariferous disk bilobed. Capsule (3.5-) 4-5 x 1-1.5 mm, oblong ellipsoid, pilose in the upper part. Seeds 2-2.5 ( 0.5-0.8 mm, ellipsoid, brown, ventral surface with a longitudinal groove covered by a strophiole with numerous raphides; testa reticulate-areolate.
A-H. Borreria apodiensis (Souza 3555). A. Habit; B. Stipular sheath; C. Flower; D. Open corolla; E. Hypanthium, calyx, style and stigma; F. Fruit; G-H. Seeds; G. dorsal view; H. ventral view. (Drawn by Laura Simón)
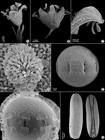
Pollen morphology: Pollen grains 8-10 zonocolpate, small (P= 25.7-(30)-33.2 µm and E= 26.4-(31.5)-35.1 µm), oblate spheroidal (P/E= 0.91-0.98), outline circular in polar view. Ectocolpus 5.8-7.6 µm long, slit-like. Endoaperture an endocingulum 2.78-5.76 µm wide which in LM analyses is seen as a lalongate pore. Exine 1.3-(1.7)-2.1 µm thick. Tectum perforate, uniformly nanospinulate, infratectum columellar. Perforations subcircular or irregular, small, 0.1-0.25 µm, nanospines 0.15-0.3 µm long (Fig. 3E-F).
Phenology: All the specimens were collected from May to July with flowers and fruits.
Distribution and habitat: This species is known only in the Chapada do Apodi in Ceará and Rio Grande do Norte states. Field observations indicate that it is restricted to the Chapada do Apodi. This suggests a probably edaphic preference for calcareous substrates (Fig. 4). In contrast the related species Borreria brownii (Rusby) Standl. and B. remota (Lam.) Bacigalupo & E.L. Cabral are widely distributed in the Americas and Brazil and grow on a wide range of soils (Cabral et al. 2011).
A-G. Micromorphological features of Borreria apodiensis (Souza 3555). A. Complete flower; B. Opened cyathiform corolla; C. Apex of the corolla lobes showing internal and external papillae; D. Bilobed papillose stigma; E-F. Pollen grains; E. Equatorial view; F. Broken grain showed endocingulum; G. Dorsal and ventral view of seeds.
Remarks: Borreria apodiensis is similar to B. brownii (Rusby) Standl. and B. remota (Lam.) Bacigalupo & E.L. Cabral, because they share the annual herbaceous habit, elliptic leaves with strongly marked secondary veins and a cyathiform corolla. However, B. apodiensis has very narrowly triangular calyx lobes and seeds without transverse grooves, whereas B. brownii and B. remota have broadly triangular or ovate calyx lobes and seeds with 11-13 transverse grooves (ruminate seeds). Other features which distiguish B. apodiensis from the other two species with cyathiform corollas are presented in the key.
Etymology: The epithet 'apodiensis' refers to its distribution in Chapada do Apodi, where the species was collected.
Conservation status: The species is known from only five localities, within two adjacent Brazilian states. The extent of ocurrence (EOO) was calculated to be 864.346 km2. The area of occupancy (AOO) was estimated at 20 km2 and is being progressively reduced the impact of farms, mechanized fruitculture, charcoal production, fires, and limestone quarrying. Following the IUCN criteria (IUCN 2001IUCN. 2001. IUCN Red List Categories. Gland, Switzerland and Cambridge, Prepared by the IUCN Species Survival Comission. IUCN.), this species should be considered as Endangered: EN B1 a,b (iii) + B2 a,b (ii).
Representative specimens examined (paratypes): BRAZIL. Ceará: Limoeiro do Norte, 05°18'17''S 37°52'25''W, 144 m, 28/V/2015, fl., fr., E.B. Souza 3549, R.A.S. Pontes, E.C. Thomaz & L.C. Silvestre (EAC, HUVA, HUEFS); Quixeré, 3/VII/1996, fl., L.W. Lima-Verde et al. 269 (EAC). Rio Grande do Norte: Apodi, Lajedo da Soledade, 05°35'24''S 37°50'03''W, 110 m, 25/V/2015, fl., fr., E.B. Souza 3511, 3515, R.A.S. Pontes, E.C. Thomaz & L.C. Silvestre (HUVA, HUFS); Felipe Guerra, Cachoeira do Roncador, 05°34'29''S 37°40'47''W, 58 m, 27/V/2015, fl., fr., E.B. Souza 3538, 3539, R.A.S. Pontes, E.C. Thomaz & L.C. Silvestre (HUVA, HUEFS).
Key for Borreria with cyathiform corolla
1. Leaves narrowly elliptic or elliptic, sessile, rarely pseudopetiolate; calyx lobes very narrowly triangular; corolla 5-6.5 mm long.; pollen grains with endocingulum; seeds without transverse grooves .......................................................................... B. apodiensis
1ʼ. Leaves ovate or broadly elliptic, pseudopetiolate; calyx lobes broadly triangular or ovate; corolla 2-3.5 mm long; pollen grains without endocingulum; seeds with transverse grooves.
2. Inflorescence glomerules 1 terminal and 1 (rarely 2) subterminal, around the nodes; calyx lobes ovate ciliate; pollen grains with colpi 12-13 µm long; seeds oblongoid.......................................................................... B. brownii
2ʼ. Inflorescence glomerules 1 terminal and 7-9 subterminal, unilateral; calyx lobes broadly triangular with hirtulous margins; pollen grains with colpi 4-5 µm long; seeds ovoid .......................................................................... B. remota
Acknowledgments
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), through PPBio-Semiarid Project (Process 457427/2012-4) for their assistance in carrying out collections in Chapada do Apodi; Maria Iracema Loiola thanks the support from CNPq for the research grant. The first author also thanks the Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico (Funcap) for its financial support, the anonymous reviewers for their contributions and Laura Simón for preparing the illustrations.
References
- Bacigalupo NM, Cabral EL. 1996. Infrageneric classification of Borreria (Rubiaceae-Spermacoceae) on the basis of American species. Opera Botanica Belgica 7: 297-308.
- Cabral EL, Miguel LM, Salas RM. 2011. Dos especies nuevas de Borreria (Rubiaceae), sinopsis y clave de las especies para Bahia, Brasil. Acta Botanica Brasilica 25: 255-276.
- Cabral EL, Salas RM. 2015. Borreria in Lista de Espécies da Flora do Brasil. Rio de Janeiro: Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB20690 7 Jun. 2015.
» http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB20690 - Dessein S. 2003. Systematic studies in the Spermacoceae (Rubiaceae). PhD Thesis, Leuven, Belgium.
- Erdtman, G. 1966. Pollen Morphology and Plant Taxonomy: Angiosperms. An introduction to Palynology-I. New York and London, Hafner Publishing Company.
- Giulietti AM, Bocage-Neta AL, Castro AAJF, et al. 2004. Diagnóstico da vegetação nativa do bioma Caatinga. In: Cardoso-Silva JM, Tabarelli M, Fonseca MT, Lins LV. (eds.) Biodiversidade da Caatinga: áreas e ações prioritárias para a conservação. Brasília, Ministério do Meio Ambiente. p. 47-90.
- Groeninckx I, Dessein S, Ochoterena H, et al. 2009. Phylogeny of the herbaceous tribe Spermacoceae (Rubiaceae) based on plastid DNA data. Annals of the Missouri Botanical Garden 96: 109-132.
- IUCN. 2001. IUCN Red List Categories. Gland, Switzerland and Cambridge, Prepared by the IUCN Species Survival Comission. IUCN.
- Kårehed J, Groeninckx I, Dessein S, et al. 2008. The phylogenetic utility of chloroplast and nuclear DNA markers and the phylogeny of the Rubiaceae tribe Spermacoceae. Molecular Phylogenetics and Evolution 49: 843-866.
- Miguel LM, Cabral EL. 2013. Borreria krapocarmeniana, a new cryptic species recovered through taxonomic analyses of Borreria scabiosoides and Borreria linoides (Spermacoceae, Rubiaceae). Systematic Botany 38: 769-781.
- Miguel LM, Souza EB, Cabral EL. 2015. Two new species of Borreria (Spermacoceae, Rubiaceae) from the states of Goiás and Minas Gerais, Brazil. Phytotaxa 201: 149-157.
- Prates M, Gatto LCS, Costa MIP. 1981. Geomorfologia. In: Almeida ALS, Bittencourt-Netto O, Moreira HF. (eds.) Projeto Radambrasil. Vol. 23, Folha SB 24/25. Rio de Janeiro, Ministério das Minas e Energia. p. 301-348
- Punt W, Blackmore S, Nilsson S, et al. 2007. Glossary of pollen and spore terminology. Review of Palaeobotany and Palynology 143: 1-81.
- Salas RM, Viana PL, Cabral, EL, et al. 2015. Carajasia (Rubiaceae), a new and endangered genus from Carajás mountain range, Pará, Brazil. Phytotaxa 206: 14-29.
- Souza MJN. 1989. Geomorfologia. In: Martins MLR, Soares AML, Lucena GG, Souza GMB, Soares ZML. (eds.) Atlas do Ceará. Fortaleza, Iplance. p. 14-15
- Tabarelli M, Cardoso-Silva JM. 2002. Áreas e ações prioritárias para a conservação, utilização sustentável e repartição de benefícios da biodiversidade do bioma Caatinga. In: Araújo EL, Moura AN, Sampaio EVSB, Gestinari LMS, Carneiro JMT. (eds.) Biodiversidade, conservação e uso sustentável da flora do Brasil. Recife, Universidade Federal Rural de Pernambuco, Imprensa Universitária. p. 27-30
- Thiers, B. 2015. Index herbariorum. Part I: The herbaria of the world. New York, New York Botanical Garden. http://sweetgum.nybg.org/ih/ 15 Oct. 2015.
» http://sweetgum.nybg.org/ih/
-
1.
Part of first author´s post-doctoral research
Publication Dates
-
Publication in this collection
03 May 2016 -
Date of issue
Apr-Jun 2016
History
-
Received
16 Jan 2016 -
Accepted
08 Apr 2016