ABSTRACT
The interplay between plant-plant interactions and light heterogeneity in the understory of tropical forests has rarely been examined. We aimed to identify the relative importance of the understory light environment and terrestrial bromeliads in explaining the abundance and spatial organization of different plant life forms along a coastal forest gradient from seashore inland in southeastern Brazil. We estimated the abundance of various life forms (herbs, woody plants, bromeliads, climbers, and palms) and the degree of light availability using hemispherical photographs in 165 plots (1 m2) within a 1.75 ha site. We used ordination methods, partial redundancy analysis (pRDA), spatial filtering using Moran’s eigenvector mapping, and Moran’s I splines. Forest cover was highly heterogeneous, but did not explain variation in abundance of life forms. Spatially, bromeliads were negatively associated with woody saplings, herbs and climbing plants at scales between 5-20 m, while the distance to seashore was found to be unrelated to these patterns. Our findings revealed that terrestrial bromeliads play an important role in the spatial organization of various life forms near the forest floor. Overall, the presence of terrestrial bromeliads and the plant area index better explained the understory vegetation than forest cover and distance to seashore.
Keywords
community assembly; hemispherical photographs; herb layer; shrub layer; spatial patterns
Introduction
Light is commonly viewed as the most important resource determining establishment, reproduction and growth of plants within forests and, not surprisingly, it is also the most heterogeneous in time and space (Pearcy 2007Pearcy RW. 2007. Responses of Plants to Heterogeneous Light Environments. In: Pugnaire FI, Valladares F. (eds.) Functional Plant Ecology, 2nd. edn. Boca Raton, CRC Press. p. 724.). The understory light environment is both cause and result of forest dynamics (Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.). Variations in light availability are expected to affect structure and composition of herb layer vegetation within tropical forests (Lima & Gandolfi 2009Lima RAF, Gandolfi S. 2009. Structure of the herb stratum under different light regimes in the Submontane Atlantic Rain Forest. Brazilian Journal of Biology 69: 289-296.). Nevertheless, only a few studies ever quantified the patchiness of forest light conditions (Clark 1996Clark DB. 1996. Landscape-scale evaluation of understory light and canopy structure : methods and application in a neotropical lowland rain forest. Canadian Journal of Forest Research 26: 747-757.; Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.; Valladares & Guzmán 2006Valladares F, Guzmán B. 2006. Canopy structure and spatial heterogeneity of understory light in an abandoned Holm oak woodland. Annals of Forest Science 63: 749-761. ) or examined how spatial variation in light conditions relates to the spatial organization of herb layers in tropical forests (Montgomery 2004Montgomery RA. 2004. Effects of understory foliage on patterns of light attenuation near the forest floor. Biotropica 36: 33-39.), especially in coastal systems (Souza & Martins 2005Souza AF, Martins FR. 2005. Spatial variation and dynamics of flooding, canopy openness, and structure in a neotropical swamp forest. Plant Ecology 180: 161-173. ).
The light environment is mainly determined by canopy structure, which directly affects growth and survival of seedlings and saplings (Kabakoff & Chazdon 1996Kabakoff RP, Chazdon RL. 1996. Effects of canopy species dominance on understorey light availability in low-elevation secondary forest stands in Costa Rica. Journal of Tropical Ecology 12: 779-788.; Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.; Montgomery & Chazdon 2001Montgomery RA, Chazdon RL. 2001. Forest structure, canopy architecture, and light transmittance in tropical wet forests. Ecology 82: 2707-2718.; Melo et al. 2007Melo ACG, Miranda DLC, Durigan G. 2007. Cobertura de copas como indicador de desenvolvimento estrutural de reflorestamentos de restauração de matas ciliares no Médio Vale do Paranapanema, SP, Brasil. Árvore 31: 321-328.). This is because light availability has strong effects on the physiological processes of forest plants (Denslow et al. 1990Denslow JS, Schultz JC, Vitousek PM, Strain BR. 1990. Growth responses of tropical shrubs to treefall gap environments. Ecology 71: 165-179.). Spatial patchiness of light availability within forests is mainly caused by canopy gaps due to treefall (Denslow et al. 1990Denslow JS, Schultz JC, Vitousek PM, Strain BR. 1990. Growth responses of tropical shrubs to treefall gap environments. Ecology 71: 165-179.), selective logging (Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.; Beaudet & Messier 2002Beaudet M, Messier C. 2002. Variation in canopy openness and light transmission following selection cutting in northern hardwood stands: An assessment based on hemispherical photographs. Agricultural and Forest Meteorology 110: 217-228.) and crown architecture (Takeda & Kawamura 2002Takeda H, Kawamura K. 2002. Light environment and crown architecture of two temperate Vaccinium species: Inherent growth rules versus degree of plasticity in light response. Canadian Journal of Botany 80: 1063-1077. ; Kitajima et al. 2005Kitajima K, Mulkey SS, Wright SJ. 2005. Variation in crown light utilization characteristics among tropical canopy trees. Annals of Botany 95: 535-547. ; Pearcy et al. 2005Pearcy RW, Muraoka H, Valladares F. 2005. Crown architecture in sun and shade environments: assessing function and trade-offs with a three-dimensional simulation model. New Phytologist 166: 791-800. ). The role that canopy gaps play in forest regeneration is a central research topic (Pearcy 2007Pearcy RW. 2007. Responses of Plants to Heterogeneous Light Environments. In: Pugnaire FI, Valladares F. (eds.) Functional Plant Ecology, 2nd. edn. Boca Raton, CRC Press. p. 724.). Nevertheless, there is a multitude of other processes affecting the patterns of shrub layer vegetation within forests. These include plant-plant interactions (Ewel & Hiremath 2005Ewel JJ, Hiremath AJ. 2005. Plant-plant interactions in tropical forests. In: Burslem DFRP, Pinard MA, Hartley SE. (eds.) Biotic Interactions in the tropics: their role in the maintenance of species diversity. Cambridge, Cambridge University Press . p. 3-34.; Garbin et al. 2016Garbin ML, Guidoni-Martins KG, Hollunder RK, Mariotte P, Scarano FR, Carrijo TT. 2016. Spatial segregation of subordinate species is not controlled by the dominant species in a tropical coastal plant community. Perspectives in Plant Ecology Evolution and Systematics 18: 23-32.), deciduousness of trees (Souza et al. 2014Souza FFM, Gandolfi S, Rodrigues RRR. 2014. Deciduousness influences the understory community in a semideciduous tropical. Biotropica 46: 512-515.), soil heterogeneity (Tateno & Takeda 2003Tateno R, Takeda H. 2003. Forest structure and tree species distribution in relation to topography-mediated heterogeneity of soil nitrogen and light at the forest floor. Ecology Research 18: 559-571.; Townsend et al. 2008Townsend AR, Asner GP, Cleveland CC. 2008. The biogeochemical heterogeneity of tropical forests. Trends in Ecology & Evolution 23: 424-431.) and plant dispersal (Braz & Mattos 2010Braz MIG, Mattos EA. 2010. Seed dispersal phenology and germination characteristics of a drought-prone vegetation in southeastern Brazil. Biotropica 42: 327-335. ), all of which may interact with light heterogeneity. Thus, even though light can have a strong filtering effect on the organization of understory vegetation, the spatial organization of plant communities is a result of many different processes. Because these competing hypotheses are not mutually exclusive (Uriarte et al. 2005Uriarte M, Hubbell SP, John R, Condit R, Canham CD. 2005. Neighbourhood effects on sapling growth and survival in a neotropical forest and the ecological-equivalence hypothesis. In: Burslem DFRP, Pinard MA, Hartley SE. (eds.) Biotic Interactions in the tropics: their role in the maintenance of species diversity . Cambridge, Cambridge University Press . p. 89-106.), quantitative data are needed to understand the relative contribution of factors that determine spatial patterns of the shrub layer.
Regardless of the importance of variation in light availability in explaining plant growth and distribution patterns, few studies made use of spatially explicit analytical tools to measure heterogeneity in forests (but see Valladares & Guzmán 2006Valladares F, Guzmán B. 2006. Canopy structure and spatial heterogeneity of understory light in an abandoned Holm oak woodland. Annals of Forest Science 63: 749-761. ). Most studies compared different vegetation types (e.g.Montgomery 2004Montgomery RA. 2004. Effects of understory foliage on patterns of light attenuation near the forest floor. Biotropica 36: 33-39.) or estimated the effects of gap creation on regeneration patterns (e.g.Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.). Many replicates are required to reliably estimate spatially explicit patterns of canopy openness or light availability, which may explain the lack of studies in this regard. Using spatially explicit mapping approaches would allow the quantification of patch size and magnitude (contrast) of the light environment in tropical forests. Patch size (spatial autocorrelation) varies between 1 and 20 meters (Clark et al. 1996Clark DB, Clark DA, Rich PM, Weiss S, Oberbauer SF. 1996. Landscape-scale evaluation of understory light and canopy structures: methods and application in a neotropical lowland rain forest. Canadian Journal of Forest Research 26: 747-757.; Nicotra et al. 1999Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.; Pearcy 2007Pearcy RW. 2007. Responses of Plants to Heterogeneous Light Environments. In: Pugnaire FI, Valladares F. (eds.) Functional Plant Ecology, 2nd. edn. Boca Raton, CRC Press. p. 724.) to about 50 meters (Yamada et al. 2014Yamada T, Yoshioka A, Hashim M, Liang N, Okuda T. 2014. Spatial and temporal variations in the light environment in a primary and selectively logged forest long after logging in Peninsular Malaysia. Trees - Structure and Function 28: 1355-1365.), or even 125 meters (Souza & Martins 2005Souza AF, Martins FR. 2005. Spatial variation and dynamics of flooding, canopy openness, and structure in a neotropical swamp forest. Plant Ecology 180: 161-173. ) when canopy openness is considered. However, the complex nature of the interplay between plant-plant interactions and spatial light heterogeneity in the understory of tropical forests has hardly been assessed.
Our study site, a coastal plain in Brazil (hereafter, restinga), has a marked zonation pattern in plant physiognomies from seashore to inland shaped by wind and sea level oscillations (Araujo & Pereira 2004Pereira MCA, Cordeiro SZ, Araujo DSD. 2004. Estrutura do estrato herbáceo na formação aberta de Clusia do Parque Nacional da Restinga de Jurubatiba, RJ, Brasil. Acta Botanica Brasilica 18: 677-687.). Coastal systems represent a great challenge to plants that are constantly stressed by wind, salt, flooding and the threat of losing habitat due to sea level variation (Crawford 2008Crawford RMM. 2008. Plants at the margin: Ecological limits and climate change. Cambridge, Cambridge University Press.), requiring a continuous adjustment to changing climate conditions (Church et al. 2013Church JA, Clark PU, Cazenave A, et al. 2013. Sea level change. In: Stocker TF, Qin D, Plattner G-K, et al. (eds.) Climate Change 2013: The physical science basis. contribution of working group i to the fifth assessment report of the intergovernmental panel on climate change. Cambridge, New York, Cambridge University Press. p. 1137-1216.). Although negative interactions can also occur among subordinate species (Garbin et al. 2016Garbin ML, Guidoni-Martins KG, Hollunder RK, Mariotte P, Scarano FR, Carrijo TT. 2016. Spatial segregation of subordinate species is not controlled by the dominant species in a tropical coastal plant community. Perspectives in Plant Ecology Evolution and Systematics 18: 23-32.), positive interactions are an important structuring mechanism in restingas (Scarano 2002Scarano FR. 2002. Structure, function and floristic relationships of plant communities in stressful habitats marginal to the Brazilian Atlantic rainforest. Annals of Botany 90: 517-524. ; Dias & Scarano 2007Dias ATC, Scarano FR. 2007. Clusia as nurse plant. In: Lüttge U. (ed.) Clusia: A woody neotropical genus of remarkable plasticity and diversity. Berlin, Heidelberg, Springer Berlin Heidelberg. p. 55-71.). Terrestrial bromeliads are an important component of these systems (Araujo 1992Araujo DSD. 1992. Vegetation types of sandy coastal plains of tropical Brazil: A first approximation. In: Seeliger U. (ed.) Coastal plant communities of Latin America. San Diego, Academic Press. p. 337-347.; Pereira et al. 2004Pereira MCA, Cordeiro SZ, Araujo DSD. 2004. Estrutura do estrato herbáceo na formação aberta de Clusia do Parque Nacional da Restinga de Jurubatiba, RJ, Brasil. Acta Botanica Brasilica 18: 677-687.), known for nursing the Clusia hilariana Schltdl. tree, the main nurse plant in the restingas (Correia et al. 2010Correia C, Dias A, Scarano F. 2010. Plant-plant associations and population structure of four woody plant species in a patchy coastal vegetation of Southeastern Brazil. Brazilian Journal of Botany 33: 607-613.) and for trapping palm seeds while negatively affecting palm seedlings (Brancalion et al. 2009Brancalion PHS, Gabriel VDA, Gómez JM. 2009. Do terrestrial tank bromeliads in Brazil create safe sites for palm establishment or act as natural traps for its dispersed seeds? Biotropica 41: 3-6. ).
These conflicting observations may be related to the strong effect of stress conditions on plant interactions (Bertness & Hacker 1994Bertness MD, Hacker SD. 1994. Physical stress and positive associations among marsh plants. The American Naturalist 144: 363.; Callaway et al. 2002Callaway RM, Brooker RW, Choler P, et al. 2002. Positive interactions among alpine plants increase with stress. Nature 417: 844-848. ; Michalet et al. 2006Michalet R, Brooker RW, Cavieres LA, et al. 2006. Do biotic interactions shape both sides of the humped-back model of species richness in plant communities? Ecology Letters 9: 767-773.; Brooker et al. 2007Brooker RW, Maestre FT, Callaway RM, et al. 2007. Facilitation in plant communities: the past, the present, and the future. Journal of Ecology 96: 18-34.). The benefit from nurse plants may not change with increasing distance from seashore (Castanho & Prado 2014Castanho CT, Prado PI. 2014. Benefit of shading by nurse plant does not change along a stress gradient in a coastal dune. PLoS One 9: e105082. doi: 10.1371/journal.pone.0105082.
https://doi.org/10.1371/journal.pone.010...
). Moreover, distance from seashore did not affect the outcome of associations between different life forms, such as trees, herbs, climbers or shrubs (Castanho et al. 2012Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. ). Brancalion et al. (2009Brancalion PHS, Gabriel VDA, Gómez JM. 2009. Do terrestrial tank bromeliads in Brazil create safe sites for palm establishment or act as natural traps for its dispersed seeds? Biotropica 41: 3-6. ) found that terrestrial bromeliads were mainly present in open canopy areas, where they negatively affected palm seedlings. Knowledge on how terrestrial bromeliads organize in space in response to canopy cover with increasing distance from seashore (a proxy for stress conditions, sensuCastanho et al. 2012Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. ) and how they associate with other life forms can provide a better understanding of the interplay between plant associations and light environment in restinga forests. This has not yet been quantitatively tested under a spatially explicit approach.
We aimed to identify the relative importance of the understory light environment and terrestrial bromeliads in explaining the abundance of different life forms along a coastal forest gradient from seashore towards inland in southeast Brazil. Our objectives were twofold. First, we aimed to identify patterns of association among different life-forms (bromeliads, palms, woody plants, climbing plants, and herbs), and the role of space (in the form of patches), forest cover and distance from seashore in explaining such patterns. We hypothesized that forest cover and distance from seashore are the main structuring factors that determine the abundance of the various life forms. Second, we sought to understand how canopy cover and bromeliads relate to other plant life forms by quantifying patterns of spatial covariation among these components, hypothesizing that terrestrial bromeliads, by preferentially occupying canopy gaps, negatively affect the abundance of other life forms in space due to their spatially aggregated growth patterns.
Materials and methods
Study site
The study was conducted in the Paulo César Vinha State Park (1,574.85 ha) in the municipality of Guarapari (20°33’-20°38’S and 40° 26’-40° 23’W, sea level). The plant communities in the park are located on quaternary sandy coastal plains in southeast Brazil (Scarano 2002Scarano FR. 2002. Structure, function and floristic relationships of plant communities in stressful habitats marginal to the Brazilian Atlantic rainforest. Annals of Botany 90: 517-524. ; Araujo & Pereira 2004Pereira MCA, Cordeiro SZ, Araujo DSD. 2004. Estrutura do estrato herbáceo na formação aberta de Clusia do Parque Nacional da Restinga de Jurubatiba, RJ, Brasil. Acta Botanica Brasilica 18: 677-687.). There is a wide variety of vegetation types including forests, shrubs and herbaceous plant communities delimited by sharp boundaries as a result of the geomorphological diversity caused by changes in sea level (Araujo & Pereira 2004; Pimentel et al. 2007Pimentel MCP, Barros MJ, Cirne P, et al. 2007. Spatial variation in the structure and floristic composition of “restinga” vegetation in southeastern Brazil. Revista Brasileira Botânica 30: 543-551. ). The climate is characterized as Aw (Köppen), with an average annual temperature of 23.3 ºC, annual average rainfall of 1,307 mm and annual relative humidity of 80 % (Assis et al. 2004aAssis AM, Pereira OJ, Thomaz LD. 2004a. Fitossociologia de uma floresta de restinga no Parque Estadual Paulo César Vinha, Setiba, município de Guarapari (ES). Revista Brasileira de Botânica 27: 349-361. ). Our study was conducted in the non-flooded coastal plain forest formation (Assis et al. 2004aAssis AM, Pereira OJ, Thomaz LD. 2004a. Fitossociologia de uma floresta de restinga no Parque Estadual Paulo César Vinha, Setiba, município de Guarapari (ES). Revista Brasileira de Botânica 27: 349-361. ) on a spatial gradient (about 350 meters) between the inland limits of the beach vegetation and the open shrub C. hilariana Schltdl. vegetation (Araujo & Pereira 2004Araujo DSD, Pereira MCA. 2004. Sandy coastal vegetation. In: Encyclopedia of Life Support Systems. Oxford, UNESCO, Eolss Publishers.; Thomazi et al. 2013Thomazi R, Rocha R, Oliveira M, Bruno AS, Silva AG. 2013. Um panorama da vegetação das restingas do Espírito Santo no contexto do litoral brasileiro. Natureza On Line 11: 1-6.). This restinga forest has a continuous canopy of about 8 meters height and a herb layer vegetation that varies in composition and includes bromeliads, Arecaceae, Piperaceae, and Rubiaceae (Assis et al. 2004bAssis AM, Thomaz LD, Pereira OJ. 2004b. Florística de um trecho de floresta de restinga no município de Guarapari, Espírito Santo, Brasil. Acta Botanica Brasilica 18: 191-201. ). These forests are termed dry forests, sandy forests, non-flooded forests, or simply restinga forests (Assis et al. 2004aAssis AM, Pereira OJ, Thomaz LD. 2004a. Fitossociologia de uma floresta de restinga no Parque Estadual Paulo César Vinha, Setiba, município de Guarapari (ES). Revista Brasileira de Botânica 27: 349-361. ; Thomazi & Silva 2014Thomazi RD, Silva AG. 2014. Florística , diversidade e estrutura horizontal e vertical de uma área de vegetação arbstiva aberta numa planície arenosa costeira do Espírito Santo , sudeste do Brasil. Natureza On Line 12: 10-18.). The most abundant plant families in these forests are Myrtaceae, Bromeliaceae, Orchidaceae, Sapotaceae, Lauraceae, and Rubiaceae (Assis et al. 2004aAssis AM, Pereira OJ, Thomaz LD. 2004a. Fitossociologia de uma floresta de restinga no Parque Estadual Paulo César Vinha, Setiba, município de Guarapari (ES). Revista Brasileira de Botânica 27: 349-361. ). Details on the study location are available in Figure S1 in supplementary material.
Sampling and image acquisition
Field work was conducted between December 2015 and January 2016. We established 15 transects parallel to seashore (oriented in azimuth angle of 30 degrees). Each transect was 50 m long and the distance between them was 25 m. We installed 11 sampling units (1 m x 1 m plots) at every 5 m interval in each transect, resulting in 165 plots (15 transects x 11 plots) within 1.75 hectares (350 m x 50 m). Details on the study plot and sampling scheme are available in Figure S2 in supplementary material. In each sampling unit, we surveyed the vegetation and acquired hemispherical photographs. We sampled seven different life forms: woody plants (tree saplings), climbing plants (woody or herbaceous), cacti, herbaceous plants, palms, bromeliads, and pteridophytes. However, cacti and pteridophytes were removed from the analyses due to their very low abundance and frequency in the plots. Sampling was conducted using an adapted cover pin frame approach (Dias et al. 2005Dias ATC, Teixeira Zaluar HL, Ganade G, Scarano FR. 2005. Canopy composition influencing plant patch dynamics in a Brazilian sandy coastal plain. Journal of Tropical Ecology 21: 343-347. ; Garbin et al. 2012Garbin ML, Carrijo TT, Sansevero JBB, Sánchez-Tapia A, Scarano FR. 2012. Subordinate, not dominant, woody species promote the diversity of climbing plants. Perspectives in Plant Ecology Evolution and Systematics 14: 257-265.): at every 0.5 m interval of the plot, a thin stick (0.8 cm diameter) was positioned vertically, and the identity and the number of times each life form touched the stick was recorded up to a height of 1.5 m. The nine pin assessments were pooled to obtain a measure of life form abundance (number of times each life form touched the stick).
Light environment was assessed indirectly using hemispherical photographs (Jonckheere et al. 2004Jonckheere I, Fleck S, Nackaerts K, et al. 2004. Review of methods for in situ leaf area index determination Part I. Theories, sensors and hemispherical photography. Agricultural and Forest Meteorology 121: 19-35. ; Weiss et al. 2004Weiss M, Baret F, Smith GJ, Jonckheere I, Coppin P. 2004. Review of methods for in situ leaf area index (LAI) determination Part II. Estimation of LAI, errors and sampling. Agricultural and Forest Meteorology 121: 37-53. ; Chianucci & Cutini 2012Chianucci F, Cutini A. 2012. Digital hemispherical photography for estimating forest canopy properties: current controversies and opportunities. iForest - Biogeosciences and Forestry 5: 290-295. ). We used a digital camera model (Canon 1200D, resolution of 5184 x 3456, and 72 dpi), with a fish eye lens (180 degrees field of view, 4,5 mm; Sigma Corp. of America, Ronkonhoma, NY, EUA), resulting in circular images. The camera was mounted on a tripod of 1.7 m height. Images were obtained in summer (growing season) when leaves are at their maximum expansion (Pérez-Harguindeguy et al. 2013Pérez-Harguindeguy N, Díaz S, Garnier E, et al. 2013. New handbook for standardised measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167. ). All photographs were taken before sunrise, just after sunset or under an evenly overcast sky to ensure optimal backlight conditions (Jelaska 2004Jelaska SD. 2004. Analysis of canopy closure in the dinaric silver fir - Beech Forests (Omphalodo fagetum) in Croatia using hemispherical photography. Hacquetia 3: 43-49.).
Data analysis
We used ordination methods, partial redundancy analysis (pRDA), spatial filtering using Moran’s eigenvector mapping (MEM; see Dray et al. 2006Dray S, Legendre P, Peres-Neto PR. 2006. Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecological Modelling 196: 483-493. and Griffith & Peres-Neto 2006Griffith DA, Peres-Neto P. 2006. Spatial modeling in ecology: the flexibility of eigenfunction spatial analyses. Ecology 87: 2603-2613.), and Moran’s I splines. The objectives were: 1) to describe the main patterns of variation in life form abundance, 2) to partition this variation into spatial components (linear, gradient from seashore, and MEMs) and 3) to quantify univariate and bivariate spatial patterns for the different life forms and forest cover. All analyses were carried out in the R environment (R Development Core Team 2015R Development Core Team. 2015. A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL URL http: //www.R-project.org/
. 15 Jul. 2016.
http: //www.R-project.org/...
): simple and constrained ordinations were run in the vegan package (Oksanen et al. 2015Oksanen J, Blanchet FG, Kindt R, et al. 2015. Vegan: community ecology package. R package version 2.3-2. http: //CRAN.R-project.org/package=vegan. 15 Jul. 2016.
http: //CRAN.R-project.org/package=vegan...
), MEMs were run in the PCNM package (Legendre et al. 2013Legendre P, Borcard D, Blanchet FG, Dray S. 2013. PCNM: MEM spatial eigenfunction and principal coordinate analyses [R package version 2.1-2/r109]. ), Moran’s I spline univariate and cross-correlograms were run in the ncf package (Bjørnstad 2015Bjørnstad ON. 2015. ncf: Spatial Nonparametric Covariance Functions. R package version 1.1-6. https: //CRAN.R-project.org/package=ncf.
https: //CRAN.R-project.org/package=ncf...
).
Images were processed using CAN-EYE version 6.313 (2014, INRA, France, available at http://www6.paca.inra.fr/can-eye). The camera fish-eye system was calibrated using the method described in http://www.avignon.inra.fr/can_eye to compute the optical center and the projection function (Weiss & Baret 2014Weiss M, Baret F. 2014. CAN-EYE V6.313 User Manual. https: //www6.paca.inra.fr/can-eye/
https: //www6.paca.inra.fr/can-eye/...
). For each single image, the cover fraction (fCover) was computed. A set of 11 images of each transect was used to derive plant area index (PAI, at three different zenith angles, 38, 53, and 68 degrees, which correspond to the LAI2000 rings 3, 4, and 5, see Weiss et al. 2004Weiss M, Baret F, Smith GJ, Jonckheere I, Coppin P. 2004. Review of methods for in situ leaf area index (LAI) determination Part II. Estimation of LAI, errors and sampling. Agricultural and Forest Meteorology 121: 37-53. ), the fraction of absorbed photosynthetically active radiation (fAPAR, modeled and measured for white and black sky), gap fraction and modeled fCover. We adopted the term PAI, instead of LAI (leaf area index) because some leaves may be behind trunks, branches, and stems. Therefore, we followed Weiss & Baret (2014)Weiss M, Baret F. 2014. CAN-EYE V6.313 User Manual. https: //www6.paca.inra.fr/can-eye/
https: //www6.paca.inra.fr/can-eye/...
in that CAN-EYE provide estimates of PAI, but not LAI. Thus, nine light environment variables were used in the analyses.
Relative abundances of life forms were calculated for each sampling unit by dividing the total number of touches of each life form by the total number of touches of all life forms, with the resulting matrix (L) of sampling units per life form abundance. Matrix C comprised fCover per plot. A third matrix (X) was constructed using the local spatial coordinates (x and y) of each sampling unit. Another set of matrices was built considering each transect as a sampling unit. Raw abundance data (number of touches) for plots within a transect were pooled. Light environment data was composed by the nine variables generated in CAN-EYE (matrix P). Abundance matrices were normalized prior to the analyses (see Legendre & Gallagher 2001Legendre P, Gallagher E. 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271-280.). Environmental data (C and P) were standardized (McGarigal et al. 2000McGarigal K, Cushman S, Stafford S. 2000. Multivariate Statistics for wildlife and ecology research. New York, Springer .). Then, matrix X was expanded into Moran’s Eigenvector predictors with positive Moran´s I (calculated Moran´s I greater than expected Moran´s I) (Legendre & Legendre 2012Legendre P, Legendre L. 2012. Numerical Ecology, Third. Amsterdan, Elsevier.), resulting in matrix M. The Moran’s eigenvector mapping (MEM) approach (Griffith & Peres-Neto 2006Griffith DA, Peres-Neto P. 2006. Spatial modeling in ecology: the flexibility of eigenfunction spatial analyses. Ecology 87: 2603-2613.; Dray et al. 2012Dray S, Pélissier R, Couteron P, et al. 2012. Community ecology in the age of multivariate multiscale spatial analysis. Ecological Monographs 82: 257-275. ) allowed us to model the relationship between life forms and environment while ensuring independence among sampling units, and modeling of more complex spatial structures in a context of canonical methods (Borcard et al. 2011Borcard D, Gillet F, Legendre P. 2011. Numerical Ecology with R. New York, Springer. ; Legendre & Legendre 2012Legendre P, Legendre L. 2012. Numerical Ecology, Third. Amsterdan, Elsevier.). Using this approach, spatial dependence is filtered out by detrending, but the information from these spatial structures can be exploited as they may reveal the presence of unmeasured processes (McIntire & Fajardo 2009McIntire EJB, Fajardo A. 2009. Beyond description: the active and effective way to infer processes from spatial patterns. Ecology 90: 46-56.; Dray et al. 2012Dray S, Pélissier R, Couteron P, et al. 2012. Community ecology in the age of multivariate multiscale spatial analysis. Ecological Monographs 82: 257-275. ).
First, we ran a Principal Coordinates Analysis (Legendre & Legendre 2012Legendre P, Legendre L. 2012. Numerical Ecology, Third. Amsterdan, Elsevier.) on matrix L, using the Bray-Curtis dissimilarity on the raw relative abundances matrix, in order to reveal the main trends of variation of life form abundance in the forest understory. Second, a partial redundancy analysis (pRDA; Økland 2003Økland RH. 2003. Partitioning the variation in a plot-by-species data matrix that is related to n sets of explanatory variables. Journal of Vegetation Science 14: 693-700. ) was run using matrix L as response and matrices M, X, and C as explanatory variables. Thus, we considered the gradient from seashore to inland as an explanatory linear spatial structure, and the MEMs as the more complex (patchy) structures (see Borcard et al. 2011Borcard D, Gillet F, Legendre P. 2011. Numerical Ecology with R. New York, Springer. ). A forward selection of variables was run on the explanatory matrices using the ordistep function of the package vegan. Thus, partial constrained ordination (pRDA) was used to partition the variation of life form abundance into three sources: fCover, MEMs, and the linear gradient from seashore. Third, a second pRDA was run using the transects as sampling units. The response matrix was composed by the pooled life form abundance in each plot within a transect after standardization by total (relative abundance) and normalization (Legendre & Legendre 2012). Explanatory datasets were the linear distances from seashore of each transect, matrix P, and the abundance of bromeliads. These were extracted from the raw abundance matrix, which comprised the remaining life forms. This pRDA determined the bromeliads as an external cause over the other life forms within the forest. In all partitioning procedures, we used the RsquareAdj function in vegan to obtain unbiased estimates of fractions (Peres-Neto et al. 2006Peres-Neto PR, Legendre P, Dray S, Borcard D, et al. 2006. Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87: 2614-2625. ; Borcard et al. 2011Borcard D, Gillet F, Legendre P. 2011. Numerical Ecology with R. New York, Springer. ). Lastly, spatial univariate and cross-correlation functions were estimated between bromeliads, fCover and the other life forms using spline (cross) correlograms (Bjørnstad and Falck 2001Bjørnstad ON, Falck W. 2001. Nonparametric spatial covariance functions: Estimation and Testing. Environmental Ecology Stat 8: 53-70.). Correlograms were built at a distance of 115 m, roughly one third of the maximum amplitude. Uncertainty for spline (cross) correlograms was estimated by bootstrapping (10,000 iterations) and used to construct 95 % envelopes.
Results
Patterns of variation in life form abundance
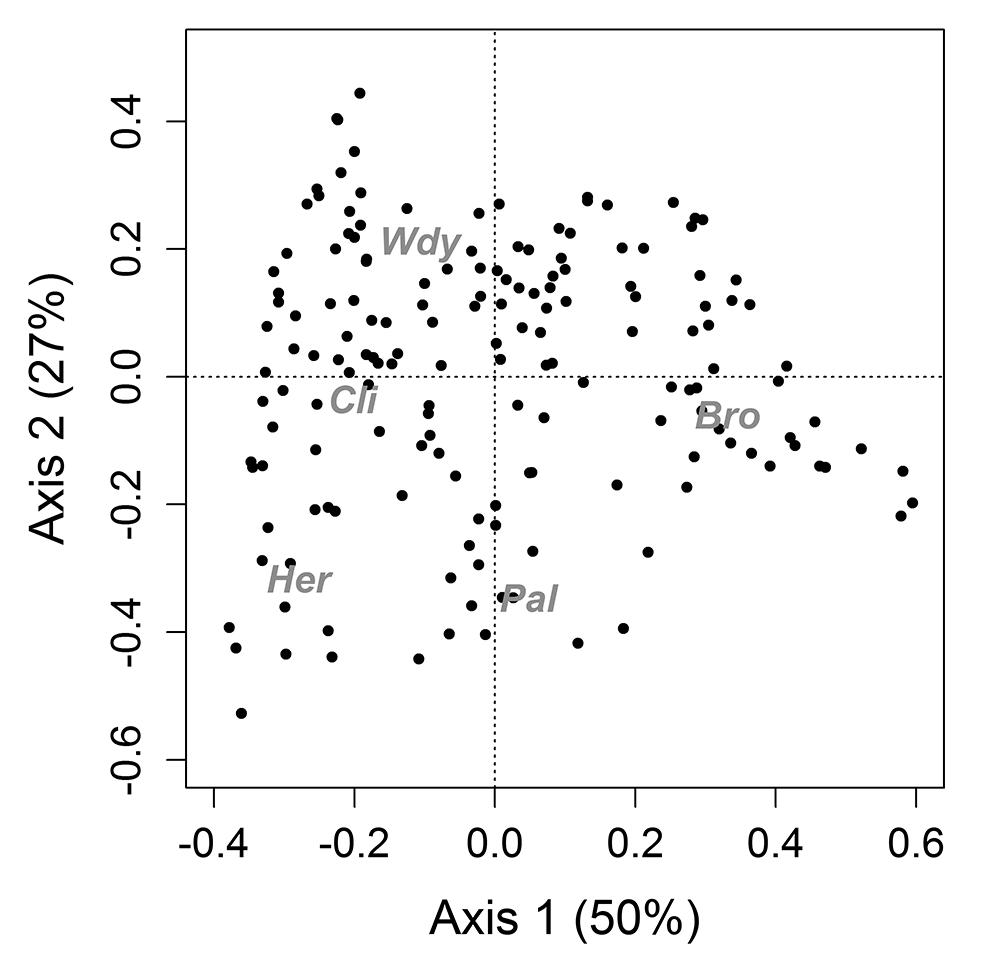
The first axis of the PCoA analysis showed that where bromeliads were abundant, other life forms were less present (Fig. 1). Two bromeliad species were dominant: Vrisea procera and Ananas sagenaria (arruda). The second axis distinguished the plots where herb species were more abundant than woody plants and bromeliads while abundance of climbers and palms was intermediate.
Principal coordinates analysis (PCoA) of different life forms (grey) in 165 plots in 1.75 hectares of a restinga forest understory. Bro, bromeliads; Pal, palms; Her, herbs; Cli, climbing plants; Wdy, woody saplings. The analysis revealed a pattern of segregation between bromeliads and other life forms.
Variation partitioning
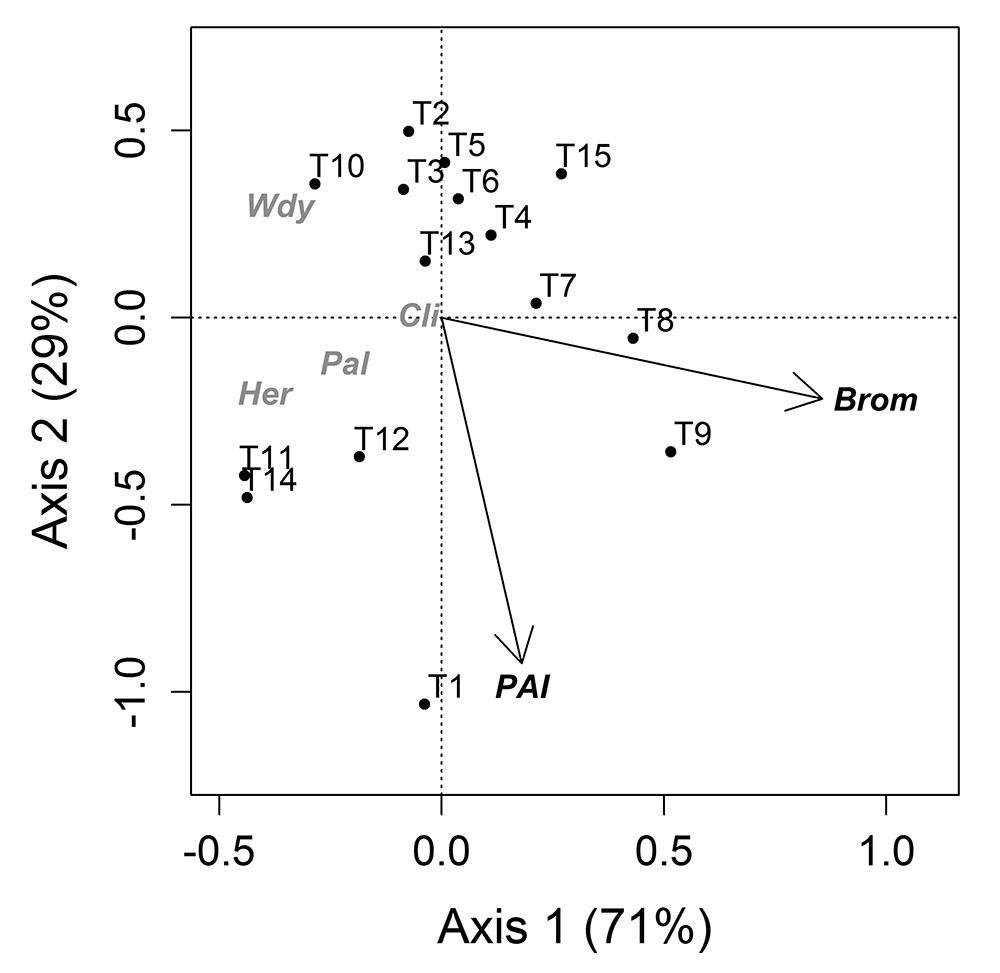
Canopy cover (fCover) was unrelated to the variation in life form abundance between plots (including bromeliads). The linear gradient from seashore explained only a small fraction (2 %) while MEMs explained the largest fraction (17 %) of variation in life form abundance (Fig. 2A). When transects were used as sampling units, bromeliads (32 %) and PAI (12 %) were the most important factors (Figs. 2B, 3) in explaining patterns of life form abundance. The linear gradient had no significant relationship with life form abundance (with: P=0.81, without: P=0.39, bromeliads) and was not considered in the second partitioning procedure.
A) Venn diagram for the partitioning of life form abundance related to the linear gradient (X), fCover (C) and MEMs (M) as explanatory data sets. Total variation explained (X∪C∪M) was 18 % (P = 0.005). B) Partition of the variation of life form abundance (after removing bromeliads) into two sources: bromeliads (B) and light environment (P). Total variation explained (B∪P) was of 41 % (P = 0.075). ∪, combined variation. ∩, shared variation. See Økland (2003Økland RH. 2003. Partitioning the variation in a plot-by-species data matrix that is related to n sets of explanatory variables. Journal of Vegetation Science 14: 693-700. ) for a detailed formulation of the variation partitioning procedures. Negative fractions should be interpreted as zeroes (Legendre 2008Legendre P. 2008. Studying beta diversity: ecological variation partitioning by multiple regression and canonical analysis. Journal of Plant Ecology 1: 3-8. ). See text for details.
Axis 1 and 2 of a constrained ordination (RDA) of life form abundance constrained by light environment (PAI, plant area index) and bromeliads (Brom). Total variation explained by the model was 41 %. See Fig. 2B for fractions. T, transect; numbering from 1 to 15 depicts the position of the transect in relation to seashore, the higher the number, the closer the transect is to seashore.
Univariate and bivariate spatial patterns
fCover (Fig. 4A), bromeliads (Fig. 4B) and herbs (Fig. 4C) were positively aggregated in space at scales between zero and 10 meters. Bromeliads were negatively associated in space with climbing plants (Fig. 4D), herbs (Fig. 4E) and woody plants (Fig. 4F). See Table S1 in supplementary material for light environment parameters of each transect derived using CAN-EYE.
Univariate spline correlograms of A) fCover, B) bromeliads, and C) herbs in 1.75 ha of a restinga forest. Spline cross-correlograms between bromeliads and: D) climbing plants, E) herbs, and F) woody plants. Upper and lower lines represent 95 % envelopes. Envelopes below or above zero in splines depict negative and positive significant spatial structures, respectively. In univariate splines (A, B and C), there was a positive association at scales between zero and 10 meters as envelope is above the zero line at these scales. For the cross-correlograms (D, E and F), there was a negative association at scales between zero and 10 meters as envelope is below the zero line at these scales.
Discussion
We found that bromeliads have a stronger impact on structuring the shrub layer vegetation than canopy cover or distance from seashore in this tropical coastal forest. Despite the expected predominant role of light and stress tolerance in filtering understory vegetation, bromeliads were able to occupy the shadier parts of the forest (with high PAI) and, due to their aggregated spatial organization, other life forms were excluded from these sites. Bromeliads have been shown before to affect the presence and density of other plant groups within forests (Brokaw 1983Brokaw N. 1983. Groundlayer dominance and apparent inhibition of tree regeneration by Aechmea magdalenae (Bromeliaceae) in a tropical forest. Tropical Ecology 24: 194-200.; Wada 1993Wada N. 1993. Dwarf bamboos affect the regeneration of zoochorous trees by providing habitats to acorn-feeding rodents. Oecologia 94: 403-407.; Barberis et al. 2002Barberis IM, Batista WB, Pire EF, Lewis JP, León RJC. 2002. Woody population distribution and environmental heterogeneity in a Chaco forest, Argentina. Journal of Vegetation Science 13: 607-614. ; Barberis & Lewis 2005Barberis IM, Lewis JP. 2005. Heterogeneity of terrestrial bromeliad colonies and regeneration of Acacia praecox (Fabaceae) in a humid-subtropical-Chaco forest, Argentina. Revista de Biologia Tropical 53: 377-385.). Nevertheless, in these studies, bromeliads had different effects on understory vegetation by either promoting regeneration or inhibiting other plant groups. In restingas and swamp forests, bromeliads are especially known to nurse tree species (Scarano et al. 1998Scarano FR, Rios RI, Esteves FA. 1998. Tree species richness, diversity and flooding regime: case studies of recuperation after anthropic impact in Brazilian flood-prone forests. International Journal of Ecology and Environmental Sciences 24: 223-235.; Scarano 2006Scarano FR. 2006. Plant community structure and function in a swamp forest within the Atlantic rain forest complex: a synthesis. Rodriguésia 57: 491-502.; Scarano 2009Scarano FR. 2009. Plant communities at the periphery of the Atlantic rain forest: Rare-species bias and its risks for conservation. Biology Conservation 142: 1201-1208. ; Correia et al. 2010Correia C, Dias A, Scarano F. 2010. Plant-plant associations and population structure of four woody plant species in a patchy coastal vegetation of Southeastern Brazil. Brazilian Journal of Botany 33: 607-613.). When acting as a trap for seeds, they can competitively exclude palm seedlings (Brancalion et al. 2009Brancalion PHS, Gabriel VDA, Gómez JM. 2009. Do terrestrial tank bromeliads in Brazil create safe sites for palm establishment or act as natural traps for its dispersed seeds? Biotropica 41: 3-6. ). These authors found that bromeliads were negatively associated with shaded conditions and that conditions for palms were better where bromeliads were absent. In summary, our results suggest that bromeliads adjust to shaded conditions, establish dense colonies, and then inhibit woody, herbaceous and climbing plants, regardless of distance from seashore.
The role of forest cover and distance from seashore
In coastal systems, distance from seashore is a good proxy for abiotic stress (Castanho et al. 2012Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. ). In our study, the distance from seashore to the nearest transect was about 170 meters. It seems that the restinga forest and the beach vegetation (Araujo & Pereira 2004Pereira MCA, Cordeiro SZ, Araujo DSD. 2004. Estrutura do estrato herbáceo na formação aberta de Clusia do Parque Nacional da Restinga de Jurubatiba, RJ, Brasil. Acta Botanica Brasilica 18: 677-687.) are capable to buffer the effects of wind and salt spray. Distance from seashore did not affect spatial associations and nurse effects in restingas (Castanho et al. 2012Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. ; Castanho & Prado 2014Castanho CT, Prado PI. 2014. Benefit of shading by nurse plant does not change along a stress gradient in a coastal dune. PLoS One 9: e105082. doi: 10.1371/journal.pone.0105082.
https://doi.org/10.1371/journal.pone.010...
). Moreover, in open shrub formations of the restinga, distance from seashore was not associated with floristic composition or structural parameters of the vegetation. In contrast, vegetation differed among plots parallel to the sea (Pimentel et al. 2007Pimentel MCP, Barros MJ, Cirne P, et al. 2007. Spatial variation in the structure and floristic composition of “restinga” vegetation in southeastern Brazil. Revista Brasileira Botânica 30: 543-551. ). Overall, our data suggest a limited role of abiotic stress associated with increasing proximity to seashore. We did not observe discernable effects of stress gradients on the organization of forest understory communities in this coastal system. Likewise, Castanho et al. (2012)Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. found no evidence that spatial life form associations change with increasing distance from seashore. Our study showed that the findings for sand dunes (Castanho et al. 2012Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961. ) and shrub vegetation (Pimentel et al. 2007Pimentel MCP, Barros MJ, Cirne P, et al. 2007. Spatial variation in the structure and floristic composition of “restinga” vegetation in southeastern Brazil. Revista Brasileira Botânica 30: 543-551. ) also apply to the forest formations in the restinga. Canopy cover, PAI, fAPAR and gap fraction were not related to distance from seashore, and the same was true for life form abundance of the shrub layer vegetation. Thus, it seems that the understory vegetation of these forests is buffered against the potentially harmful effects caused by the proximity to the seashore.
Understory plant species are affected by competition with canopy trees and lianas, and by low levels of light reaching the forest floor (Wright 2002Wright SJ. 2002. Plant diversity in tropical forests: a review of mechanisms of species coexistence. Oecologia 130: 1-14. ). Low levels (less than 2-5 % of the above canopy) and temporal heterogeneity of light reaching the floor (e.g.Canham et al. 1990Canham CD, Denslow JS, Platt WJ, Runkle JR, Spies TA, White PS. 1990. Light regimes beneath closed canopies and tree-fall gaps in temperate and tropical forests. Canadian Journal of Forest Research 20: 620-631. ) drive a wide variation in morphology and physiological plasticity of the understory species, directly affecting the observed patterns of diversity and species coexistence (Théry 2001Théry M. 2001. Forest light and its influence on habitat selection. Plant Ecology 153: 251-261.; Valladares 2003Valladares F. 2003. Light heterogeneity and plants: from ecophysiology to species coexistence and biodiversity. In: Esser K, Lüttge U, Beyschlag W, Hellwig F. (eds.) Progress in botany: genetics physiology systematics ecology. Berlin, Heidelberg, Springer. p. 439-471.). The vertical leaf orientation of bromeliads that would impair their capacity to harvest light, may be compensated by their high photosynthetic capacity and biomass allocation to leaf tissue (Valladares et al. 2002Valladares F, Skillman JB, Pearcy RW. 2002. Convergence in light capture efficiencies among tropical forest understory plants with contrasting crown architectures: A case of morphological compensation. American Journal of Botany 89: 1275-1284. ). Restinga bromeliads acclimate poorly to high levels of light under exposed conditions, while they seem to perform better and acclimate to more shaded conditions in forests (Duarte et al. 2002Duarte HM, Rocas G, Barreto SMB, et al. 2002. Acclimation or stress symptom? An integrated study of intraspecific variation in the clonal plant Aechmea bromeliifolia, a widespread CAM tank-bromeliad. Botanical Journal of the Linnean Society 140: 391-401. ). In our study site, bromeliads appear to cope with high shade conditions (indicated by PAI) creating dense patches (as indicated by the large space fraction explained by MEMs), but the physiological mechanisms behind these patterns require further study. Moreover, the large fraction explained by MEMs indicated a highly-aggregated pattern of bromeliads unrelated to canopy cover. The patchy pattern of bromeliads and their dissociation in space with other plant life forms was evident in the spline and cross-correlogram outputs.
Spatial patterns
Terrestrial bromeliads seem to partition space with other groups of plant life forms. It is noteworthy that canopy cover and distance from seashore had no effect on the abundance of the different life forms studied. Understory light conditions are expected to affect shrub layer vegetation (Montgomery 2004Montgomery RA. 2004. Effects of understory foliage on patterns of light attenuation near the forest floor. Biotropica 36: 33-39.). The patterns of spatial segregation between bromeliads and the other life forms, and the limited role of canopy cover and gradient stress in determining these patterns, point to an important role of competition for space and of dispersal limitation (see Barberis et al. 2014Barberis IM, Torres PS, Batista WB, Magra G, Galetti L, Lewis JP. 2014. Two bromeliad species with contrasting functional traits partition the understory space in a South American xerophytic forest: Correlative evidence of environmental control and limited dispersal. Plant Ecology 215: 143-153. ) in the spatial organization of the shrub layer of the restinga forest. Our results indicate that terrestrial bromeliads may have a strong influence on structure and spatial organization of an understory plant community. Future studies should evaluate the physiological plasticity of bromeliads allowing them to cope with the different light environments of the restinga forest, and to measure patterns of tree seed germination and abundance of tree seedlings in sites with and without bromeliad colonies.
Conclusions
We showed that the assembly of the herb layer vegetation in this restinga forest is independent of canopy cover and distance from seashore. Importantly, terrestrial bromeliads tend to occupy the shadiest portions of the gradient (as indicated by PAI), and they seem to play a crucial role in determining the spatial organization of different life forms in this forest. Thus, these internal filters (sensuViolle et al. 2012Violle C, Enquist BJ, McGill BJ, et al. 2012. The return of the variance: Intraspecific variability in community ecology. Trends in Ecology & Evolution 27: 244-252.) of the community (the local environmental conditions driven by PAI) and the density-dependent processes are more important than the external filters, such as distance from seashore (as a proxy for the stress gradient). The nonrandom co-occurrence patterns between terrestrial bromeliads and other life forms detected here (the negative cross-correlation spatial structure) provides evidence that biotic internal filters can be more important than external filters in the assembly of the herb layer vegetation in these forests. Future studies should assess the plasticity of bromeliads in coping with changes in light conditions, and address the question if different bromeliad species have different effects on other plant life forms, especially on seedlings and saplings of tree species regenerants.
Acknowledgements
We thank FAPES, CNPq and Capes (Espírito Santo State Research Agency/Brazilian Research Agencies), which granted fellowships to M.L. Garbin, F. Misaki, C. Lopes, and K. Santos. We thank FAPES for funding (grant n°69329524/2015). We are also grateful to IEMA (Espírito Santo Environmental Agency) for collection licenses.
References
- Araujo DSD. 1992. Vegetation types of sandy coastal plains of tropical Brazil: A first approximation. In: Seeliger U. (ed.) Coastal plant communities of Latin America. San Diego, Academic Press. p. 337-347.
- Araujo DSD, Pereira MCA. 2004. Sandy coastal vegetation. In: Encyclopedia of Life Support Systems. Oxford, UNESCO, Eolss Publishers.
- Assis AM, Pereira OJ, Thomaz LD. 2004a. Fitossociologia de uma floresta de restinga no Parque Estadual Paulo César Vinha, Setiba, município de Guarapari (ES). Revista Brasileira de Botânica 27: 349-361.
- Assis AM, Thomaz LD, Pereira OJ. 2004b. Florística de um trecho de floresta de restinga no município de Guarapari, Espírito Santo, Brasil. Acta Botanica Brasilica 18: 191-201.
- Barberis IM, Batista WB, Pire EF, Lewis JP, León RJC. 2002. Woody population distribution and environmental heterogeneity in a Chaco forest, Argentina. Journal of Vegetation Science 13: 607-614.
- Barberis IM, Lewis JP. 2005. Heterogeneity of terrestrial bromeliad colonies and regeneration of Acacia praecox (Fabaceae) in a humid-subtropical-Chaco forest, Argentina. Revista de Biologia Tropical 53: 377-385.
- Barberis IM, Torres PS, Batista WB, Magra G, Galetti L, Lewis JP. 2014. Two bromeliad species with contrasting functional traits partition the understory space in a South American xerophytic forest: Correlative evidence of environmental control and limited dispersal. Plant Ecology 215: 143-153.
- Beaudet M, Messier C. 2002. Variation in canopy openness and light transmission following selection cutting in northern hardwood stands: An assessment based on hemispherical photographs. Agricultural and Forest Meteorology 110: 217-228.
- Bertness MD, Hacker SD. 1994. Physical stress and positive associations among marsh plants. The American Naturalist 144: 363.
- Bjørnstad ON. 2015. ncf: Spatial Nonparametric Covariance Functions. R package version 1.1-6. https: //CRAN.R-project.org/package=ncf
» https: //CRAN.R-project.org/package=ncf - Bjørnstad ON, Falck W. 2001. Nonparametric spatial covariance functions: Estimation and Testing. Environmental Ecology Stat 8: 53-70.
- Borcard D, Gillet F, Legendre P. 2011. Numerical Ecology with R. New York, Springer.
- Brancalion PHS, Gabriel VDA, Gómez JM. 2009. Do terrestrial tank bromeliads in Brazil create safe sites for palm establishment or act as natural traps for its dispersed seeds? Biotropica 41: 3-6.
- Braz MIG, Mattos EA. 2010. Seed dispersal phenology and germination characteristics of a drought-prone vegetation in southeastern Brazil. Biotropica 42: 327-335.
- Brokaw N. 1983. Groundlayer dominance and apparent inhibition of tree regeneration by Aechmea magdalenae (Bromeliaceae) in a tropical forest. Tropical Ecology 24: 194-200.
- Brooker RW, Maestre FT, Callaway RM, et al. 2007. Facilitation in plant communities: the past, the present, and the future. Journal of Ecology 96: 18-34.
- Callaway RM, Brooker RW, Choler P, et al. 2002. Positive interactions among alpine plants increase with stress. Nature 417: 844-848.
- Canham CD, Denslow JS, Platt WJ, Runkle JR, Spies TA, White PS. 1990. Light regimes beneath closed canopies and tree-fall gaps in temperate and tropical forests. Canadian Journal of Forest Research 20: 620-631.
- Castanho CT, Oliveira AA, Prado PI. 2012. The importance of plant life form on spatial associations along a subtropical coastal dune gradient. Journal of Vegetation Science 23: 952-961.
- Castanho CT, Prado PI. 2014. Benefit of shading by nurse plant does not change along a stress gradient in a coastal dune. PLoS One 9: e105082. doi: 10.1371/journal.pone.0105082.
» https://doi.org/10.1371/journal.pone.0105082 - Chianucci F, Cutini A. 2012. Digital hemispherical photography for estimating forest canopy properties: current controversies and opportunities. iForest - Biogeosciences and Forestry 5: 290-295.
- Church JA, Clark PU, Cazenave A, et al. 2013. Sea level change. In: Stocker TF, Qin D, Plattner G-K, et al. (eds.) Climate Change 2013: The physical science basis. contribution of working group i to the fifth assessment report of the intergovernmental panel on climate change. Cambridge, New York, Cambridge University Press. p. 1137-1216.
- Clark DB. 1996. Landscape-scale evaluation of understory light and canopy structure : methods and application in a neotropical lowland rain forest. Canadian Journal of Forest Research 26: 747-757.
- Clark DB, Clark DA, Rich PM, Weiss S, Oberbauer SF. 1996. Landscape-scale evaluation of understory light and canopy structures: methods and application in a neotropical lowland rain forest. Canadian Journal of Forest Research 26: 747-757.
- Correia C, Dias A, Scarano F. 2010. Plant-plant associations and population structure of four woody plant species in a patchy coastal vegetation of Southeastern Brazil. Brazilian Journal of Botany 33: 607-613.
- Crawford RMM. 2008. Plants at the margin: Ecological limits and climate change. Cambridge, Cambridge University Press.
- Denslow JS, Schultz JC, Vitousek PM, Strain BR. 1990. Growth responses of tropical shrubs to treefall gap environments. Ecology 71: 165-179.
- Dias ATC, Scarano FR. 2007. Clusia as nurse plant. In: Lüttge U. (ed.) Clusia: A woody neotropical genus of remarkable plasticity and diversity. Berlin, Heidelberg, Springer Berlin Heidelberg. p. 55-71.
- Dias ATC, Teixeira Zaluar HL, Ganade G, Scarano FR. 2005. Canopy composition influencing plant patch dynamics in a Brazilian sandy coastal plain. Journal of Tropical Ecology 21: 343-347.
- Dray S, Legendre P, Peres-Neto PR. 2006. Spatial modelling: a comprehensive framework for principal coordinate analysis of neighbour matrices (PCNM). Ecological Modelling 196: 483-493.
- Dray S, Pélissier R, Couteron P, et al. 2012. Community ecology in the age of multivariate multiscale spatial analysis. Ecological Monographs 82: 257-275.
- Duarte HM, Rocas G, Barreto SMB, et al. 2002. Acclimation or stress symptom? An integrated study of intraspecific variation in the clonal plant Aechmea bromeliifolia, a widespread CAM tank-bromeliad. Botanical Journal of the Linnean Society 140: 391-401.
- Ewel JJ, Hiremath AJ. 2005. Plant-plant interactions in tropical forests. In: Burslem DFRP, Pinard MA, Hartley SE. (eds.) Biotic Interactions in the tropics: their role in the maintenance of species diversity. Cambridge, Cambridge University Press . p. 3-34.
- Garbin ML, Carrijo TT, Sansevero JBB, Sánchez-Tapia A, Scarano FR. 2012. Subordinate, not dominant, woody species promote the diversity of climbing plants. Perspectives in Plant Ecology Evolution and Systematics 14: 257-265.
- Garbin ML, Guidoni-Martins KG, Hollunder RK, Mariotte P, Scarano FR, Carrijo TT. 2016. Spatial segregation of subordinate species is not controlled by the dominant species in a tropical coastal plant community. Perspectives in Plant Ecology Evolution and Systematics 18: 23-32.
- Griffith DA, Peres-Neto P. 2006. Spatial modeling in ecology: the flexibility of eigenfunction spatial analyses. Ecology 87: 2603-2613.
- Jelaska SD. 2004. Analysis of canopy closure in the dinaric silver fir - Beech Forests (Omphalodo fagetum) in Croatia using hemispherical photography. Hacquetia 3: 43-49.
- Jonckheere I, Fleck S, Nackaerts K, et al. 2004. Review of methods for in situ leaf area index determination Part I. Theories, sensors and hemispherical photography. Agricultural and Forest Meteorology 121: 19-35.
- Kabakoff RP, Chazdon RL. 1996. Effects of canopy species dominance on understorey light availability in low-elevation secondary forest stands in Costa Rica. Journal of Tropical Ecology 12: 779-788.
- Kitajima K, Mulkey SS, Wright SJ. 2005. Variation in crown light utilization characteristics among tropical canopy trees. Annals of Botany 95: 535-547.
- Legendre P. 2008. Studying beta diversity: ecological variation partitioning by multiple regression and canonical analysis. Journal of Plant Ecology 1: 3-8.
- Legendre P, Borcard D, Blanchet FG, Dray S. 2013. PCNM: MEM spatial eigenfunction and principal coordinate analyses [R package version 2.1-2/r109].
- Legendre P, Gallagher E. 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271-280.
- Legendre P, Legendre L. 2012. Numerical Ecology, Third. Amsterdan, Elsevier.
- Lima RAF, Gandolfi S. 2009. Structure of the herb stratum under different light regimes in the Submontane Atlantic Rain Forest. Brazilian Journal of Biology 69: 289-296.
- McGarigal K, Cushman S, Stafford S. 2000. Multivariate Statistics for wildlife and ecology research. New York, Springer .
- McIntire EJB, Fajardo A. 2009. Beyond description: the active and effective way to infer processes from spatial patterns. Ecology 90: 46-56.
- Melo ACG, Miranda DLC, Durigan G. 2007. Cobertura de copas como indicador de desenvolvimento estrutural de reflorestamentos de restauração de matas ciliares no Médio Vale do Paranapanema, SP, Brasil. Árvore 31: 321-328.
- Michalet R, Brooker RW, Cavieres LA, et al. 2006. Do biotic interactions shape both sides of the humped-back model of species richness in plant communities? Ecology Letters 9: 767-773.
- Montgomery RA. 2004. Effects of understory foliage on patterns of light attenuation near the forest floor. Biotropica 36: 33-39.
- Montgomery RA, Chazdon RL. 2001. Forest structure, canopy architecture, and light transmittance in tropical wet forests. Ecology 82: 2707-2718.
- Nicotra AB, Chazdon RL, Iriarte SVB. 1999. Spatial Heterogeneity of Light and Woody Seedling Regeneration in Tropical Wet Forests. Ecology 80: 1908-1926.
- Økland RH. 2003. Partitioning the variation in a plot-by-species data matrix that is related to n sets of explanatory variables. Journal of Vegetation Science 14: 693-700.
- Oksanen J, Blanchet FG, Kindt R, et al. 2015. Vegan: community ecology package. R package version 2.3-2. http: //CRAN.R-project.org/package=vegan 15 Jul. 2016.
» http: //CRAN.R-project.org/package=vegan - Pearcy RW. 2007. Responses of Plants to Heterogeneous Light Environments. In: Pugnaire FI, Valladares F. (eds.) Functional Plant Ecology, 2nd. edn. Boca Raton, CRC Press. p. 724.
- Pearcy RW, Muraoka H, Valladares F. 2005. Crown architecture in sun and shade environments: assessing function and trade-offs with a three-dimensional simulation model. New Phytologist 166: 791-800.
- Pereira MCA, Cordeiro SZ, Araujo DSD. 2004. Estrutura do estrato herbáceo na formação aberta de Clusia do Parque Nacional da Restinga de Jurubatiba, RJ, Brasil. Acta Botanica Brasilica 18: 677-687.
- Peres-Neto PR, Legendre P, Dray S, Borcard D, et al. 2006. Variation partitioning of species data matrices: estimation and comparison of fractions. Ecology 87: 2614-2625.
- Pérez-Harguindeguy N, Díaz S, Garnier E, et al. 2013. New handbook for standardised measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167.
- Pimentel MCP, Barros MJ, Cirne P, et al. 2007. Spatial variation in the structure and floristic composition of “restinga” vegetation in southeastern Brazil. Revista Brasileira Botânica 30: 543-551.
- R Development Core Team. 2015. A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL URL http: //www.R-project.org/ 15 Jul. 2016.
» http: //www.R-project.org/ - Scarano FR. 2002. Structure, function and floristic relationships of plant communities in stressful habitats marginal to the Brazilian Atlantic rainforest. Annals of Botany 90: 517-524.
- Scarano FR. 2006. Plant community structure and function in a swamp forest within the Atlantic rain forest complex: a synthesis. Rodriguésia 57: 491-502.
- Scarano FR. 2009. Plant communities at the periphery of the Atlantic rain forest: Rare-species bias and its risks for conservation. Biology Conservation 142: 1201-1208.
- Scarano FR, Rios RI, Esteves FA. 1998. Tree species richness, diversity and flooding regime: case studies of recuperation after anthropic impact in Brazilian flood-prone forests. International Journal of Ecology and Environmental Sciences 24: 223-235.
- Souza AF, Martins FR. 2005. Spatial variation and dynamics of flooding, canopy openness, and structure in a neotropical swamp forest. Plant Ecology 180: 161-173.
- Souza FFM, Gandolfi S, Rodrigues RRR. 2014. Deciduousness influences the understory community in a semideciduous tropical. Biotropica 46: 512-515.
- Takeda H, Kawamura K. 2002. Light environment and crown architecture of two temperate Vaccinium species: Inherent growth rules versus degree of plasticity in light response. Canadian Journal of Botany 80: 1063-1077.
- Tateno R, Takeda H. 2003. Forest structure and tree species distribution in relation to topography-mediated heterogeneity of soil nitrogen and light at the forest floor. Ecology Research 18: 559-571.
- Théry M. 2001. Forest light and its influence on habitat selection. Plant Ecology 153: 251-261.
- Thomazi R, Rocha R, Oliveira M, Bruno AS, Silva AG. 2013. Um panorama da vegetação das restingas do Espírito Santo no contexto do litoral brasileiro. Natureza On Line 11: 1-6.
- Thomazi RD, Silva AG. 2014. Florística , diversidade e estrutura horizontal e vertical de uma área de vegetação arbstiva aberta numa planície arenosa costeira do Espírito Santo , sudeste do Brasil. Natureza On Line 12: 10-18.
- Townsend AR, Asner GP, Cleveland CC. 2008. The biogeochemical heterogeneity of tropical forests. Trends in Ecology & Evolution 23: 424-431.
- Uriarte M, Hubbell SP, John R, Condit R, Canham CD. 2005. Neighbourhood effects on sapling growth and survival in a neotropical forest and the ecological-equivalence hypothesis. In: Burslem DFRP, Pinard MA, Hartley SE. (eds.) Biotic Interactions in the tropics: their role in the maintenance of species diversity . Cambridge, Cambridge University Press . p. 89-106.
- Valladares F. 2003. Light heterogeneity and plants: from ecophysiology to species coexistence and biodiversity. In: Esser K, Lüttge U, Beyschlag W, Hellwig F. (eds.) Progress in botany: genetics physiology systematics ecology. Berlin, Heidelberg, Springer. p. 439-471.
- Valladares F, Guzmán B. 2006. Canopy structure and spatial heterogeneity of understory light in an abandoned Holm oak woodland. Annals of Forest Science 63: 749-761.
- Valladares F, Skillman JB, Pearcy RW. 2002. Convergence in light capture efficiencies among tropical forest understory plants with contrasting crown architectures: A case of morphological compensation. American Journal of Botany 89: 1275-1284.
- Violle C, Enquist BJ, McGill BJ, et al. 2012. The return of the variance: Intraspecific variability in community ecology. Trends in Ecology & Evolution 27: 244-252.
- Wada N. 1993. Dwarf bamboos affect the regeneration of zoochorous trees by providing habitats to acorn-feeding rodents. Oecologia 94: 403-407.
- Weiss M, Baret F. 2014. CAN-EYE V6.313 User Manual. https: //www6.paca.inra.fr/can-eye/
» https: //www6.paca.inra.fr/can-eye/ - Weiss M, Baret F, Smith GJ, Jonckheere I, Coppin P. 2004. Review of methods for in situ leaf area index (LAI) determination Part II. Estimation of LAI, errors and sampling. Agricultural and Forest Meteorology 121: 37-53.
- Wright SJ. 2002. Plant diversity in tropical forests: a review of mechanisms of species coexistence. Oecologia 130: 1-14.
- Yamada T, Yoshioka A, Hashim M, Liang N, Okuda T. 2014. Spatial and temporal variations in the light environment in a primary and selectively logged forest long after logging in Peninsular Malaysia. Trees - Structure and Function 28: 1355-1365.
Publication Dates
-
Publication in this collection
13 Mar 2017 -
Date of issue
Jan-Mar 2017
History
-
Received
13 Dec 2016 -
Accepted
23 Jan 2017