ABSTRACT
A comprehensive census of a hectare of cerrado s.s. in southeastern Brazil, a small-tree and scrub woodland physiognomy, allowed the evaluation of whether pollination and dispersal modes are correlated with the stratification of the vegetation and if so, in what way. Generalist pollination, and pollination by small bees, as well as ornithophily and anemophily were more frequent in the lower layers (ground and scrub), while species pollinated by large bees and beetles are more or less equally distributed among the ground, scrub and tree layers. The three nocturnal pollination modes, phalenophily, sphingophily, and chiropterophily indicated a preference for the upper layers (tree and scrub). Zoochory predominated in the tree layer, but autochory increased towards the ground at the expense of anemochory and zoochory. We discuss possible reasons for the height distribution of pollination and seed dispersal modes and compare the situation in Cerrado with other Neotropical forests.
Keywords:
cerrado s.s. woodland; dispersal systems; Neotropical forests; pollination modes; vertical stratification
Introduction
The Cerrado is a species-rich vegetation, which originally covered an area of 1.5 to 2 million sq. km (or 20 % to 25 %) of the Brazilian territory. There are some small atypical areas of these plant formations also in northern Paraguay and southeastern Bolivia. A more precise estimate of the total Cerrado biome area reaches 1.910 million km2. Even if the ca. 20 %, made up by other vegetation types in the Cerrado biome, for instance, upland forests, gallery forests, limestone forests, veredas, marshy campos, hummock campos, campos rupestres, white-sand savanna patches (Eiten 1972Eiten G. 1972. The cerrado vegetation of Brazil. Botanical Review: 201-341.), are subtracted, the original area of Cerrado sensu lato (Cerrado in its wide sense, including all physiognomies) would be about 1,530 million km2 (Gottsberger & Silberbauer-Gottsberger 2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.). The cerrado sensu lato (cerrado s.l.), besides of being species-rich, has also a series of different vegetation forms or physiognomies, from forests, over woodlands, savannas to grasslands, which originally, before the arrival of the Europeans all occurred naturally. Most phytogeographers since Warming (1892Warming E. 1892. Lagoa Santa. Kjøbenhavn, Bianco Lunos. ) distinguish five main forms of Cerrado s.l., which are essentially defined by the woody layer or its absence (Fig. 1). The cerradão is a dense forest (total arboreal cover more than 60 %) or a woodland (40-60 % tree crown cover), with a usually more than 7 m tall tree canopy. The cerrado sensu stricto (or cerrado s.s.; cerrado in its narrow or restricted sense) is dominated by trees and shrubs (canopy generally less than 7 m tall). The term is applied to a low forest (arboreal cover more than 60 %), a low arboreal woodland or a tree and scrub woodland (woody-plant cover 40-60 %). The campo cerrado formation is still dominated by low trees and shrubs (woody-plant cover 10 to less than 40 %). This is the physiognomy that best corresponds to the form of a low-tree and scrub savanna with short-grass or tall-grass. The campo sujo (dirty field), also called campo sujo of cerrado, has no trees or a few very low trees, and has scattered shrubs over a grass layer (woody-layer cover less than 10 % but more than zero). By its physiognomy, it is also called a “scrub savanna” or a “shrub savanna”. The last physiognomy, campo limpo (clean field), is also called campo limpo of cerrado or cerrado grassfield. This is a pure grassland without trees or shrubs rising above the ground layer and thus the woody-layer cover is zero. Woody plants can exist but then they are low and do not exceed the ground layer in height. The definition of Cerrado can be found among others in Goodland (1971Goodland R. 1971. A physiognomic analysis of the “cerrado” vegetation of Central Brazil. Journal of Ecology 59: 411-419.), Eiten (1972Eiten G. 1972. The cerrado vegetation of Brazil. Botanical Review: 201-341.; 1978Eiten G. 1978. Delimitation of the cerrado concept. Vegetatio 36: 169-178.; 2001Eiten G. 2001. Vegetação natural do Distrito Federal. Brasília, Editora da Universidade de Brasília.), Goodland & Pollard (1973)Goodland R, Pollard R. 1973. The Brazilian cerrado vegetation: a fertility gradient. Journal of Ecology 61: 219-224., Coutinho (1978Coutinho LM. 1978. O conceito de cerrado. Revista Brasileira de Botânica 1: 17-23.), Goodland & Ferri (1979)Goodland R, Ferri G. 1979. Ecologia do Cerrado. Belo Horizonte/ São Paulo, Livraria Itatiaia Editora, Editora Universidade São Paulo., Sarmiento (1983Sarmiento G. 1983. The savannas of tropical America. In: Boulièr F. (ed.) Tropical savannas (Ecosystems of the world 13). Amsterdam, Elsevier. ), Ribeiro (1998Ribeiro JF. (ed.) 1998. Cerrado: matas de galeria. Planaltina, Embrapa-CPAC.) and Gottsberger & Silberbauer-Gottsberger (2006a)Gottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.. The most common original form of Cerrado in Central Brazil is a tree-scrub woodland (Eiten 1972Eiten G. 1972. The cerrado vegetation of Brazil. Botanical Review: 201-341.). When the whole range of its physiognomies is taken into consideration, then Cerrado is much more than a savanna. Thus, the use of many authors of the term savanna or Brazilian savanna as synonymous for Cerrado s.l. makes no sense and neglects the breadth and richness of this unique vegetation. The different forms of Cerrado have affinities in their floristic composition because all forms share some species in common, which do not occur in forests, woodlands, savannas and grasslands other than Cerrado sensu lato (Gottsberger & Silberbauer-Gottsberger 2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.).
Cerrado sensu lato and the continuum of its five basic forms, from cerradão, cerrado sensu stricto, campo cerrado, campo sujo to campo limpo. The tallest drawn cerradão tree is about 12 m in height. From Gottsberger & Silberbauer-Gottsberger (2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.), reproduced with permission of Reta Verlag, Ulm.
The stratification of a vegetation, especially of tropical and non-tropical forests, is a phenomenon apparently not settled yet. Authors disagree about the existence of strata, at least in tropical forests: “The idea that in tropical forests the crowns of the trees form several superposed strata or storeys (the words layer, tier and canopy are also used) has been current in the literature for a long time. The term stratification, as applied to rain forests has been variously interpreted and its meaning is often misunderstood. Sometimes it is stated categorically that there are three tree strata (according to a few authorities, more than three). There are also authors who state, or imply, that any grouping of the trees according to their height is arbitrary and that `strata´ have no objective reality.” (Richards 1996Richards PW. 1996. The tropical rainforest: an ecological study. 2nd. edn. Cambridge, Cambridge University Press.; see also Lowmann & Rinker 2004Lowmann MD, Rinker HB. 2004. Forest canopies. 2nd. edn. Burlington, Elsevier Academic Press.).
Notwithstanding the discussion about existence of stratification in tropical forests or not, Hallé et al. (1978Hallé F, Oldeman RAA, Tomlinson PB. 1978. Tropical trees and forests. Berlin/ Heidelberg/ New York, Springer.) find strata or layers, to be useful concepts for analyzing forest structure and functioning. In their opinion, whether strata exist or not is more or less irrelevant. “Our opinion is that there are no strata in the forest as subdivisions of the total population, but that in certain plots there are demonstrable horizontal “sets” composed of trees of the present. For this reason we have sparingly used the word “layer”, even then with some apprehension, but repeat that our layers do not correspond to the strata (real or imagined) of previous ecologists but refer to aggregations at certain levels of particularly built trees, not to the total tree population.” (Hallé et al. 1978Hallé F, Oldeman RAA, Tomlinson PB. 1978. Tropical trees and forests. Berlin/ Heidelberg/ New York, Springer.).
Even if stratified or layered tropical forests in reality do not exist, it is important to recognize that “stratification” of a vegetation results in gradients, which can be analyzed and thus can make the system easier to understand. For example, there are strong environmental gradients in a tropical forest. Light intensity is very high at the outer tree crowns, but becomes increasingly lower towards the ground of a forest. Air temperature during daytime is high at the upper canopy, but becomes cooler inside the forest and at lower levels. Air humidity is low at the outer canopy but increasingly higher towards the ground. These environmental gradients and further structural components of the vegetation are responsible for the distribution of life and growth forms of plants, being them trees of different heights, shrubs, herbs, lianas or epiphytes. Gradients are responsible also for the vertical and horizontal differences in density of plants and animals, and vertical and horizontal distribution of individual species of plants, animals, fungi and bacteria within a forest. All kinds of interactions in plants and animals, as well as also between plants and animals are shaped by gradients of a forest. For example, pollinating and seed dispersing animals have different flight or moving preferences, with the result that certain interactions occur more commonly in higher or lower levels of a forest (Bawa et al. 1985Bawa KS, Bullock SH, Perry DR, Coville RE, Grayum MH. 1985. Reproductive biology of tropical lowland rain forest trees. II. Pollination systems. American Journal of Botany 72: 346-356.).
There are few studies about spatial distribution of the vegetation-forming plants in the Cerrado, and there is no quantitative study yet of pollination and seed dispersal systems in Cerrado that relates to the stratification or layering of its vegetation; even partial indications about stratification of such systems are rare. Since we have worked for many years in a one hectare plot of cerrado s.s., a low-tree and scrub woodland, and did a whole census of pollination and seed dispersal (Gottsberger & Silberbauer-Gottsberger 1983Silberbauer-Gottsberger I, Eiten G. 1983. Fitossociologia de um hectare de cerrado. Brasil Florestal 54: 55-70.; 2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.; bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.; Silberbauer-Gottsberger & Gottsberger 1988Silberbauer-Gottsberger I, Gottsberger G. 1988. A polinização de plantas do cerrado. Revista Brasileira de Biologia 48: 651-663.), we are able to correlate the data with the layers of the vegetation occurring in the study plot.
For this, we posed the following questions: 1) Are there differences of smaller versus larger pollinating animals or diurnal versus nocturnal pollinators or seed dispersers in preferring certain vegetation layers? 2) Do zoochorous, anemochorous and autochorous dispersal units distribute equally or unequally in the height classes of the studied cerrado, and does this distribution correlate with the supposed dispersal modes or the seed dispersing animals?
In the present paper we present evidences of stratification in pollination and dispersal phenomena even in this relatively low and semi-open vegetation type. We further discuss the situation found in the studied cerrado s.s. and compare it with results from studies about pollination and dispersal in Neotropical forests elsewhere.
Materials and methods
The site of our long-term studies on floristics, vegetation, phenology and plant reproduction was a cerrado s.s. in the region of Botucatu (8 km distant from the city), São Paulo State, in a natural reserve of a Fazenda called “Treze de Maio” (22°45’S 48°25’W), at about 550 m altitude. At the time of our studies, the reserve was composed of several structurally different semideciduous cerrado s.l. physiognomies and gallery forests with a total extension of about 700 hectares1 1 Unfortunately, in 1982, after more than ten years of study, nearly the total area was cut by the owners of the sugarcane company São Manuel, and replanted mainly with sugarcane. . The climate of the Botucatu region is characterized by an average yearly precipitation of about 1,300 mm. The seasonal distribution of precipitation indicates that 36 % of the total annual rainfall occurs in spring, 44 % in summer, 10 % in autumn and only 9 % in winter, that is, a warm rainy season from October to March alternates with a cooler dry season from April to September. The yearly average temperature is about 21 °C.
A one hectare plot (100 m x 100 m) of cerrado s.s., a low-tree and scrub woodland with a total woody cover of 50 %, and tree canopy varying from 3 to 8 m height, was investigated intensively over several years. First, a quantitative floristic survey was conducted (Silberbauer-Gottsberger & Eiten 1983Silberbauer-Gottsberger I, Eiten G. 1983. Fitossociologia de um hectare de cerrado. Brasil Florestal 54: 55-70.; 1987Silberbauer-Gottsberger I, Eiten G. 1987. A hectare of cerrado. I. General aspects of trees and thick-stemmed shrubs. Phyton(Horn, Austria) 27: 55-91.), which eventually revealed the presence of 301 angiosperm “species” (some of them, however, representing infraspecific taxa) (Gottsberger & Silberbauer-Gottsberger 2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.; bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.). In the hectare, 4197 woody individuals were measured (≥10 cm circumference), of which 1180 were 3-8 m tall trees and 3017 thick-stemmed scrub elements (treelets, shrubs).
To get an idea whether plant height and stratification were linked with pollination and seed dispersal modes, the individuals of the 301 species of the cerrado hectare were arranged according to height classes. The height classes used were “tree layer” (equal to or greater than 3 m), “scrub layer” (less than 3 m and equal to or greater than 1 m) and “ground layer” (less than 1 m). According to our scheme, whenever individuals of a tree species were shorter than 3 m but taller than 1 m, they were considered a scrub element, or even as a ground-layer element if they were flowering and fruiting when less than 1 m tall. Moreover, herbaceous plants between 1 and 3 m tall, such as a tall grass, were also categorized as scrub elements. Many species, therefore, occurred in different categories such as tree and scrub, or scrub and ground layers. Bearing in mind this potential overlap, the hectare of cerrado s.s. was analyzed with respect to height classes and pollination and dispersal modes. The arrangement of plant individuals in height classes, on one hand is necessary because the reproductive age of Cerrado plants varies strongly. On the other hand, this situation makes the measured and established height classes a kind of “snapshot”, which is valid only for a certain time scale. The Cerrado is a very dynamic and flexible vegetation, including the fact that Cerrado trees consist mostly of fast growing tree types (Gottsberger & Silberbauer-Gottsberger 2006aGottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.). Thus, the only way to obtain reliable data about vertical stratification of reproductive data in the Cerrado was to rely on height classes and not on growth or life forms of plants. The numbers of species in each height class or the numbers of species of a certain pollination or dispersal mode were absolute, while the percentages of the same species were rounded values. Studies of pollination were made between 1969 and 1981, and of seed dispersal mainly between 1970 and 1976.
With respect to pollination modes, there were a few non-generalist species, apparently pollinated effectively by more than one agent (e.g., Ananas ananassoides, Bromeliaceae; hummingbirds, large and small bees, butterflies); Palicourea rigida, Rubiaceae (the same pollinator groups as the previous species); Styrax ferruginea, Styracaceae (large bees and hummingbirds). However, for the presented estimates, those few species were included in their most important mode of pollination. The whole data set of primary and eventual secondary pollinators is available elsewhere (Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta. for the general data set). However, in the dispersal data set presented here (also in Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.), all species were included in their main dispersal mode only (or in this case by the main animal dispersers) and no secondary dispersers or generalist group were established.
Results
The distribution of flowering and fruiting species in the cerrado s.s. hectare varied among the three height classes. The tree layer had 61 flowering species, the scrub layer 123 and the ground layer 230 species. The sum of these species (414) was much greater than the actual species richness in the hectare (301) because, as mentioned above, flowering and fruiting individuals of the same species frequently fell into different height classes. The diagram shows (Fig. 2) that in reality in the cerrado hectare only 13 species (ca. 4 %) were strictly reproductive trees, 44 (15 %) trees and scrub, 4 (1 %) were in all three layers, 14 (5 %) strictly scrub, 61 (29 %) scrub and ground layer, and 165 (55 %) strictly ground layer. However, in the following paragraphs we will use the 61 trees, 123 scrub species and 230 ground-layer species as the basis for comparison.
Distribution of the 301 species among the three height classes tree, scrub and ground layer in a one hectare plot of cerrado s.s. in Botucatu, São Paulo State.
Pollination
With regard to pollination modes, the evaluation of the 301 angiosperms in the hectare revealed that the majority of plants were either bee-pollinated or generalist entomophilous (Fig. 3). The largest number of species was bee pollinated (melittophilous) which accounted for 38 % of pollination overall, and comprised pollination by small to medium-sized bees (body length less than 12 mm; 22 %) and large bees (body length 12 mm and greater; 16 %). Second in importance was generalist pollination (37 %). Within this category, we recognized the following forms of generalist insect pollination: lacking beetle participation (27 %) and having beetle participation (10 %). Bees overall were extremely important in the cerrado and even within the generalist pollination mode they may predominate. Next in importance, and directly related to the number of grasses and sedges, was anemophily (13 %). The remaining pollination modes totaled 12 %, of which cantharophily represented 3 % and the other pollination modes (ornithophily, chiropterophily, sphingophily, phalenophily, psychophily, myiophily) accounted for less than this. Of the 261 zoophilous species, only five were ornithophilous, three chiropterophilous, and the remaining 253 species were entomophilous.
Distribution of pollination modes among the 301 species in a one hectare plot of cerrado s.s. in Botucatu.
When considering pollination modes in the three height classes together (Fig. 4), generalist pollination numerically increased in number of species from tree (26 spp., 43 %) over scrub (58 spp., 46 %) to the ground layer (89 spp., 40 %), although the percentage of its representation in the three height classes was about the same. Pollination by large bees was numerically high in the scrub (29 spp., 24 %) and ground layer (29 spp., 13 %) and lower in the tree layer (15 spp., 25 %), although the percentage was quite higher in the tree and scrub layer than in the ground layer. Small bees apparently preferred the ground layer (54 spp., 23 %) where they visited, numerically and in percentage, substantially more flowers than in the other two layers (seven spp., 11 % in tree, and 12 spp., 10 % in scrub layer). Anemophily in cerrado apparently was also a phenomenon of the ground layer (40 spp., 17 %), since it was very low in the scrub layer (two spp., 2 %), and did not occur in trees. Bird pollination at the Botucatu site apparently was also restricted to the ground (five spp., 2 %) and scrub layers (four spp., 3 %), and was lacking in trees. Nocturnal beetle-pollinated species occurred in similar numbers in all three height classes (four spp., 7 % in tree, four spp., 3 % in scrub, and five spp., 2 % in ground layer), however with a somewhat higher percentage among trees (four spp., 7 %). Diurnal myiophily appeared to be more common also on the lower layers (one sp., 1 %, in scrub, and four spp., 2 % in ground layer). The three nocturnal pollination modes of phalenophily, sphingophily and chiropterophily were more common in the upper layers (trees and scrub) and were rare or non-existent at the ground. Further pollination modes had a very low representation and no height preference was detected.
Distribution of pollination modes among the three height classes in a one hectare plot of cerrado s.s. in Botucatu.
In general, the data indicate that species pollinated by small bees, generalist insect-pollinated species, hummingbird-, fly-, butterfly- and wind-pollinated species were more common in the lower layers of the vegetation. Large bee- and beetle-pollinated species were ± evenly distributed among the three vegetation layers, while sphingophilous, phalenophilous and chiropterophilous species occurred nearly exclusively in the taller scrub and tree layers.
Seed dispersal
Of the total of 301 species in the hectare plot in Botucatu, 206 species (68 %) had the whole fruit as dispersal unit, and in 95 species (32 %) the dispersal unit was the seed. 135 species (45 %) were found to be zoochorous, 88 species (30 %) anemochorous (53 species had plumed dispersal units and 35 had winged dispersal units), and 78 species (25 %) were autochorous (Fig. 5).
Distribution of dispersal modes among the 301 plant taxa in a one hectare plot of cerrado s.s. in Botucatu.
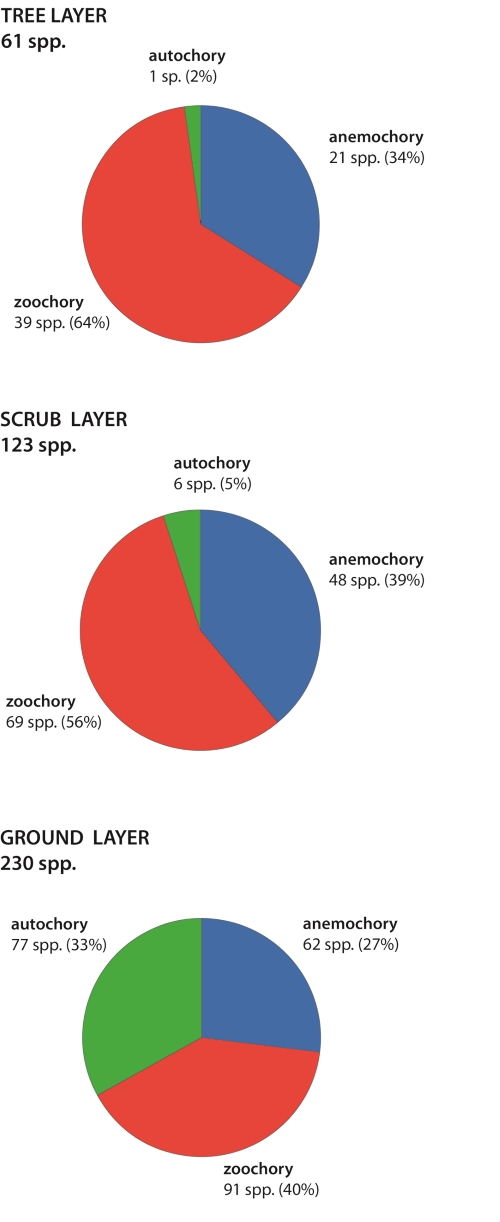
A comparison of the three height classes (Fig. 6) showed that the percentage of zoochorous species decreased with height, from the tree layer (64 %), to the scrub layer (56 %), and down to the ground layer (40 %). However, the total number of zoochorous species increased because of the much higher number of species at the ground layer. Likewise, the percentage of anemochorous species, contrary to their total number, decreased at the ground layer (27 %). Their highest percentage was in the scrub layer (39 %), with the tree layer occupying an intermediate position (34 %). The autochory increased considerably in number of species (from one to 77) and in percentage (from 2 % to 33 %) from the tree to the ground layer.
Distribution of dispersal modes among the three height classes in a one hectare plot of cerrado s.s. in Botucatu.
Thus, in the tree layer (61 species), zoochory predominated (39 species, 64 %) over anemochory (21 spp., 34 %), while autochory occurred in only one species (2 %). In the scrub layer (123 spp.) the situation was somewhat similar, with zoochory still dominating (69 spp, 56 %), but with anemochory (48 spp., 39 %) and autochory (six spp., 5 %) increasing in number and percentage. In the ground layer (230 spp.), autochory (77 spp., 33 %) was represented by a higher number, mainly at the expense of anemochory (62 spp., 27 %) and of zoochory (91 spp., 40 %).
As already mentioned, zoochory was the dispersal mode for 135 (45 %) species of the Botucatu hectare. Of these zoochorous species, 91 (67 %) were endozoochorous, 28 (21 %) epizoochorous, 12 (9 %) synzoochorous, and four species exhibited mixed endo- and synzoochory (Fig. 7). The highest number of species having endozoochory as a dispersal mode was found in the scrub layer (63 spp., 92 %), followed by the ground (49 spp., 54 %) and tree layers (37 spp., 94 %). Conversely, the highest percentage was in the tree layer (Fig. 8).
Distribution of dispersal modes among zoochorous species in a one hectare plot of cerrado s.s. in Botucatu.
Distribution of zoochorous dispersal modes among the three height classes in a one hectare plot of cerrado s.s. in Botucatu.
Synzoochory was best represented in the ground layer. Species with both endo- and synzoochory were more or less equally well represented in all three layers. Epizoochores were best represented among the ground layer (28 species, 31 %), only two did occur in the scrub layer (Tristachya leiostachya, Poaceae, and Lippia salviaefolia, Lamiaceae), and none among the trees.
Discussion
A satisfactory habit or growth form classification is very difficult in the Cerrado, because there is a complete intergradation between trees, treelets, shrubs, semishrubs and herbs (Eiten 1972Eiten G. 1972. The cerrado vegetation of Brazil. Botanical Review: 201-341.). As already mentioned, the reproductive age of Cerrado plants varies strongly. For this reason, there are cases where species produce flowers and fruits as a “tree”, but also as a “scrub” or even already as a “ground-layer” plant. Despite this overlap, the data provided here allow to envisage some trends on the distribution of reproductive features among height strata, which are discussed below.
Pollination
Some authors apparently did not detect any differences in the distribution of pollinators and pollination systems in Cerrado (Martins & Batalha 2005Martins FQ, Batalha MA. 2005. Vertical and horizontal distribution of pollination systems in ocerrado fragments of Central Brazil. In: The 2005 meeting of the association for tropical biology and conservation, frontiers in tropical biology and conservation. Abstracts.Universidade Federal de Uberlândia. p. 120. ), while further authors, such as Oliveira & Gibbs (2002Oliveira PE, Gibbs PE. 2002. Pollination and reproductive biology in cerrado plant communities. In: Oliveira PS, Marquis RJ. (eds.). The Cerrados of Brazil. Ecology and natural history of a Neotropical savanna. New York, Columbia University Press. p. 329-347. ) noted “Moths and bats are pollinators mostly of trees, while wind and hummingbird pollination appear mostly in the herbaceous layer.”
Although the Cerrado is a relatively low vegetation, a vertical variation of pollination systems, can be expected even in the lowest and most open physiognomies of cerrado s.l., namely in campo cerrado, campo sujo and even campo limpo. For example, there is an instructive case showing that even slightly different heights of inflorescences can affect the behavior of insects. Gumbert & Kunze (1999Gumbert A, Kunze J. 1999. Inflorescence height affects visitation behavior of bees - a case study of an aquatic plant community in Bolivia. Biotropica 31: 466-477.) studied aquatic plants visited by bees, in an inundated savanna region in Bolivia, and compared plants with inflorescence heights shorter than the grass height with other plants that had inflorescences projecting out of the surrounding vegetation. Larger bees of the genera Apis, Melipona, Bombus and Xylocopa visited only flowers above the surrounding vegetation. Smaller, mainly solitary bees (e.g., Augochlorella, Ancyloscelis) visited flowers in the dense vegetation near the water surface. The existence of foraging strata, in this case, may be explained by a preference for distinct flight heights. Additionally, a longer detection distance may allow faster and directional flights between inflorescences. Especially large and social bees which fly rapidly and which are especially common at species of the higher layers, are known to be highly competitive. Competitively inferior smaller bee species, many of which solitary, may be displaced to more hidden and less easily exploitable flowers (Gumbert & Kunze (1999Gumbert A, Kunze J. 1999. Inflorescence height affects visitation behavior of bees - a case study of an aquatic plant community in Bolivia. Biotropica 31: 466-477.). Wind also can force a vertical distribution of bees. In strong wind-exposed Brazilian dune vegetation, large and strong bees, such as Xylocopa frontalis, always flew relatively high and fully exposed to the wind, while smaller species had a much lower flight height, and very small bees took advantage of every depression in the dunes (Gottsberger et al. 1988Gottsberger G, Camargo JMF, Silberbauer-Gottsberger I. 1988. A bee-pollinated tropical community: the beach dune vegetation of Ilha de São Luís, Maranhão, Brazil. Botanische Jahrbücher für Systematik 109: 469-500.).
Indeed, pollination in general as well as by bees is very heterogeneous (summarized in Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.). There are not only differences in size of the bees, being small to medium-sized (smaller than Apis mellifera or less than 12 mm) or large ones (above this size), but they are also solitary or social, and their behavior often is not restricted to simple collection of pollen and nectar. There are very specialized behaviors in bee pollination. For example, there is the phenomenon of pollen transfer by smaller-sized bees that visit certain Lamiaceae (Eriope and Hyptis) which have an explosive release of pollen, or there is another specialization in Dalechampia (Euphorbiaceae), with species providing resin for certain small female bees for use in nest construction. Several large and some small bees are involved in the complex buzz pollination when flowers have poricidal anthers that have to be vibrated (e.g. Solanum, Ouratea, Cochlospermum, Miconia, Senna, Chamaecrista), while other plant groups (e.g. Malpighiaceae, Iridaceae) offer oil instead of pollen, which is used by large female Anthophorinae (Apidae) bees to feed their larvae. Certain Jacaranda (Bignoniaceae) species produce perfume and nectar in the flowers. Males of large euglossine bees mainly collect perfume, while small bees, also being active pollinators, feed on nectar and collect pollen. As mentioned above, the smaller-sized bees found their adequate flowers more commonly at the ground and scrub layer than in the tree layer. The strong and fast flying large bees (e.g. Xylocopa, Bombus, Euglossa, Epicharis, Centris) and their flowers were more evenly distributed in all three layers.
Other plant species pollinated jointly by insects of several orders, such as bees, flies, butterflies and beetles, presented a generalized pollination mode. Examples from the cerrado are species of Acosmium, Sclerolobium, Anadenanthera, Stryphnodendron, Connarus, Serjania, Aspilia and further Asteraceae, Copaifera, Anacardium, Schefflera, Licania, and many others. Main characteristics of generalist flowers in the Cerrado are that they have commonly diurnal anthesis and a relatively small size (from a few mm to 1 cm in diameter, rarely more). Flowers are open, either disc-shaped or cup-like, or more or less shortly tubular. They are often born in relative lax or dense inflorescences, and their floral symmetry is more commonly actinomorphic and only sometimes zygomorphic. The relatively large number of cerrado plants with generalist, polyphylic flowers is visited by fluctuating pollinator groups, according to local and regional distribution of available animals.
In the hectare, besides grasses and sedges, usually anemophilous, were also a few species of the genera Cissampelos or Myrsine that exhibited anemophily. Pollination by small beetles occurred in the small palm, Acrocomia hassleri, as well as in the Annonaceae genera Xylopia and Duguetia. Although Xylopia aromatica, in reality is pollinated by thrips (Thysanoptera) and beetles, we accounted this species here in the cantharophilous group since beetles are also prominent visitors. Several species of Annona exhibited the nocturnal phenomenon of pollination by large nocturnal dynastid-scarab beetles. Pure fly pollination (myiophily) was rare in the cerrado and occurred in Aristolochia species. Pollination by diurnal Lepidoptera (psychophily) was also rare in cerrado, but Lippia and Lantana (Verbenaceae) species, which have a suite of butterfly-associated characters, were visited by butterflies only occasionally in the cerrado, but were pollinated mainly by bees. More or less exclusive settling moth (nocturnal Lepidoptera) pollination (phalenophily) occurred in species of Pouteria (Sapotaceae), Roupala (Proteaceae) and Diospyros (Ebenaceae). Pollination by hovering moths or hawkmoths (sphingophily) occurred in Couepia (Chrysobalanaceae), Bauhinia (Fabaceae), Qualea grandiflora (Vochysiaceae), Aspidosperma, Hancornia (Apocynaceae) and Tocoyena (Rubiaceae).
Considering pollination by vertebrates, the pollination by bats, for instance in the genera Bauhinia (Fabaceae) and Caryocar (Caryocaraceae), was common in the hectare plot, mostly at the scrub and tree layer. In contrast, pollination by hummingbirds was important in the scrub and ground layer and lacking at the tree layer. Even epiphytic Bromeliaceae, such as Aechmea species were found growing relatively low in the vegetation, namely at the first ramification of trees and thus occurred in the scrub, and Ananas anassoides grew at the ground layer only.
In general, the species-rich vegetation of cerrado s.l. exhibits nearly all pollination modes expected in the Neotropics (Oliveira & Gibbs 2000Oliveira PE, Gibbs PE. 2000. Reproductive biology of woody plants in a cerrado community of Central Brazil. Flora 195: 311-329.). In Cerrado, nearly all possible animal groups occur that usually serve as pollinators, from bees, wasps, diurnal and nocturnal butterflies and moths, flies, beetles, thrips, hummingbirds, bats to non-flying mammals. In terms of pollination types, the Cerrado is certainly as rich as any mesophytic rainforest. However, there should be differences in the distribution of pollination modes in the different strata of cerrado s.s. and rainforests. The cerrado s.s. is a relatively low, open and sunny plant formation, which during the day can become very hot in all three layers and down to the ground. It is perhaps notable that smaller insects, or not so strong flyers, prefer lower levels of the vegetation, such as small bees, diurnal butterflies and flies; additionally, also hummingbirds move or find their flowers at lower strata. However, ornithophily is a special case. Hummingbirds find a more continuous nectar supply in the adjacent gallery forests than in cerrado. It is notable that in cerrado, hummingbirds display opportunistic feeding behavior, that is, they often look for nectar in non-ornithophilous flowers ((Oliveira & Gibbs 2000Oliveira PE, Gibbs PE. 2000. Reproductive biology of woody plants in a cerrado community of Central Brazil. Flora 195: 311-329.; Maruyama et al. 2013Maruyama PK, Oliveira GM, Ferreira C, Dalsgaard B, Oliveira PE. 2013. Pollination syndromes ignored: importance of non-ornithophilous flowers to Neotropical savanna hummingbirds. Naturwissenschaften 100:1061-1068. ). However, in the present discussion we do not consider the visits of hummingbirds to non-ornithophilous flowers.
Large bees as well as beetles (the last being often nocturnal) do not show strong preference for one of the three layers, while the other nocturnal animals, such as nocturnal settling moths, the sphingids and bats appear to prefer the upper layers in the hectare. It might be a possibility that small insects that are not strong flyers might prefer the lower levels of the vegetation also to escape predation. For the large dynastid-scarab beetles, which visit Annona and Philodendron flowers during the evening or night, the darkness certainly is a protection against predatory lizards and birds. Also during daytime, these beetles are protected inside the closed pollination chambers and kettles of their flowers and inflorescences.
Ramirez (2004aRamirez N. 2004a. Pollination specialization and time of pollination on a tropical Venezuelan plain: variations in time and space. Botanical Journal of the Linnean Society 145: 1-16.; bRamirez N. 2004b. Ecology of pollination in a tropical Venezuelan savanna. Plant Ecology 173: 171-189.) studied a Venezuelan savanna region, which consisted of 1) a forest vegetation, 2) of an ecotone or forest-savanna transition vegetation, 3) a savanna vegetation, and 4) a secondary vegetation on disturbed areas. Similar to the cerrado hectare studied here, frequency distribution of nine pollination modes has showed, at the community level, bee pollination (38.6 %) as dominant. However. the occurrence of the other pollination types was different. Butterfly (13.9 %), fly (12.7 %), and wasp (10.8 %) pollination came a close second. Moth (6.2 %) and wind (10.4 %) pollination occurred with similar frequency, and the least common were bird (3.1 %), beetle (2.3 %) and bat (1.9 %) pollination. Pollination modes associated with growth forms has showed completely different results, as in the cerrado hectare. Bee pollination was dominant in all growth forms, but wasp, butterfly and fly pollination were secondary for trees, shrubs and lianas, while butterfly and wind pollination were secondary for herbaceous species.
We aimed to compare pollination systems in Cerrado and Neotropical rainforests. However, since the methods in the studies frequently differ, a direct comparison is complicated. Such a comparison is somewhat possible with the study of pollination in lowland rainforest of La Selva. The Biological Station La Selva in Costa Rica has a long tradition of research on plant reproductive biology, including community level distribution of pollination systems as related to vertical stratification of the forest. Kress & Beach (1994Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.) assembled and summarized the available information on floral biology of trees, shrubs and herbs at La Selva, supposedly using methods similar to the ones used for the cerrado hectare. Depending on their vertical position at reproductive maturity, plants are classified as to belong to the canopy, subcanopy and understory strata. Herbs, shrubs and small trees less than 5 m high make up the understory. Trees more than 5 m tall but of less than canopy stature at maturity, belong to subcanopy. The canopy includes trees flowering at the top of the forest and emergent species. Also in this forest, a classification of some large shrubs and small trees provides a gradual intergradation of the lower and upper strata that complicates interpretation of vertical stratification. One serious caveat in comparing the cerrado s.s. and the La Selva studies is that only part of the species are considered at La Selva. They focused their study on common and accessible species belonging to major taxonomic families. At that time, the canopy and subcanopy at La Selva comprised at least 320 species, of which only 125 species were analyzed with regard to their pollination. The estimate of the number of taxa in the understory was 365 species (excluding epiphytes) and only 151 species were studied. On the other hand, the cerrado study in Botucatu was based on a whole census of only one hectare, while the La Selva study is based on the whole area of about 1,536 ha of the entire Biological Station.
Since floristic composition differs substantially between second-growth and old-growth forest, not only in Costa Rica (Guariguata et al. 1997Guariguata MR, Chazdon RL, Denslow JS, Dupuy JM, Anderson L. 1997. Structure and floristics of secondary and old-growth forest stands in lowland Costa Rica. Plant Ecology 132: 107-120.; Chazdon et al. 2003Chazdon RL, Careaga S, Webb C, Vargas O. 2003. Community and phylogenetic structure of reproductive traits of woody species in wet tropical forests. Ecological Monographs 73: 331-348.), but probably everywhere (Girão et al. 2007Girão LC, Lopes AV, Tabarelli M, Bruna EM. 2007. Changes in tree reproductive traits reduce functional diversity in a fragmented Atlantic forest landscape. PLOS ONE 9(8): e908. doi: 10.1371/journal.pone.0000908
https://doi.org/10.1371/journal.pone.000...
), we concentrate our discussion on old-growth vegetation types which are closer to the original situation of a “pristine forest”.
A tall and structured lowland rainforest, contrary to the cerrado, has a very hot environment only at the outer canopy level and becomes progressively cooler, more shady and humid towards the ground. Additionally, there are lianas growing up to the canopy and especially a number of epiphytes usually occur at the subcanopy, but also at the canopy and even at the ground layer. The distribution of pollination systems at la Selva, therefore, should be different as compared to the cerrado s.s., at least in some parts. The compilation by Kress & Beach (1994Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.) (Tab. 1) showed that fly, butterfly, beetle, and small bee pollination were most common in the understory. Pollination by medium to large bees was also most common in the understory, but occurred also in a lower number, in the subcanopy and canopy. Hummingbird flowers were considerably more common in the understory (although epiphytic, probably bird-pollinated Bromeliaceae in the canopy apparently were not considered in the study). Bat pollination was most common in the subcanopy, but occurred also in the understory and the canopy. Pollination by small diverse insects (the generalists) was more common in the canopy and less in the subcanopy and understory (Kress & Beach (1994Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.).
Pollination modes of the 276 studied flowering plant species at La Selva, Costa Rica, and their distribution among the three height classes. Adapted from Kress & Beach (1994Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.).
Thus, there are different trends in the distribution of pollination systems in the cerrado in Botucatu and the lowland rainforest at La Selva, Costa Rica (compare Tabs. 1 and 2), especially with regard to the distribution of pollination by varied generalist insects, larger and smaller bees, and beetles. The reasons for the different pollination systems distribution are largely open to speculation. Since the discussed compilation (Kress & Beach 1994Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.) is largely based on previous heterogeneous studies (Bawa et al. 1985Bawa KS, Bullock SH, Perry DR, Coville RE, Grayum MH. 1985. Reproductive biology of tropical lowland rain forest trees. II. Pollination systems. American Journal of Botany 72: 346-356.; Frankie 1975Frankie GW. 1975. Tropical forest phenology and pollinator plant coevolution. In: Gilbert LE, Raven PH. (eds.) Coevolution of animals and plants. Austin/ London, University of Texas Press. p. 192-209.), and, as already mentioned, there is also a large number of species in these forests, which were not evaluated at all, a more complete comparison would require a more complete study and may provide even a different picture.
Distribution of pollination modes among the three height classes in a one hectare plot of cerrado s.s. in Botucatu, São Paulo State, Brazil.
Further studies in forests considered stratum preferences of different pollinators. Bees differed between canopy and understory of a semideciduous lowland forest at Panama City (Roubik 1993Roubik DW. 1993. Tropical pollinators in the canopy and understory : Field data and theory for stratum "preferences". Journal of Insect Behavior 6: 659-673.). Other studies in dry forests of the Guanacaste Province, Costa Rica, found the large bee pollination system to be prominent primarily among trees and lianas (Frankie et al. 1983Frankie GW, Haber WA, Opler PA, Bawa KS. 1983. Characteristics and organization of the large bee pollination system in the Costa Rican dry forest. In: Jones CE, Little RJ. (eds.) Handbook of experimental pollination biology . New York, Van Nostrand Reinhold . p. 411-447. ), and among mass-flowering trees (Frankie & Haber 1983Frankie GW, Haber WA. 1983. Why bees move among mass-flowering neotropical trees. In: Jones CE, Little RJ. (eds.) Handbook of experimental pollination biology. New York, Van Nostrand Reinhold. p. 361-372. ). Again, in the Guanacaste Province dry forest, 65 hawkmoth species visit 31 native plant species, ca. 10 % of the trees, but also various species of shrubs, herbs, lianas, and epiphytes (Haber & Frankie 1989Haber WA, Frankie GW. 1989. A tropical hawkmoth community: Costa Rican dry forest Sphingidae. Biotropica 21: 155-172.).
There are more recent studies in Neotropical forests communities showing pollination stratification trends. Dulmen (2001Dulmen A. 2001. Pollination and phenology of flowers in the canopy of two contrasting rain forest types in Amazonia, Colombia. Plant Ecology 153: 73-85.), for example, compared the pollination systems in the canopy of an inundated forest with that of a terra firme forest in the Colombian Amazon. In both forests, most canopy trees and lianas, were pollinated by small bees, large bees, butterflies or by small, relatively unspecialized insects. Other pollinators, like hummingbirds, bats, moths and beetles were less common. Kanstrup & Olesen (2000Kanstrup J, Olesen JM. 2000. Plant-flower visitor interactions in a Neotropical rain forest canopy: community structure and generalisation level. Det Norske Videnskaps-Akademi. I. Mat.-Naturv. Klasse Skrifter, Ny Serie 39: 33-41.) studied the community of nine species of epiphytes and climbers in the canopy of a rainforest in Ecuador, and found 46 insect species (mostly beetles) and one bird species as flower visitors. Yamamoto et al. (2007Yamamoto LF, Kinoshita LS, Martins FR. 2007. Síndromes de polinização e de dispersão em fragmentos da Floresta Estacional Semidecídua Montana, SP, Brasil. Acta Botanica Brasilica 21: 553-573.) investigated semideciduous montane forest fragments in São Paulo state, considering trees and shrubs with dbh ≥ 3 cm in three strata. Melittophily dominated in the upper stratum.
Stratification trends are present in all plant formations studied to date. Supposedly, stratification of pollination modes in tropical forests is more accentuated than in the cerrado s.s. woodland. However, considering the present state of knowledge, such differences are hard to test.
Seed dispersal
The preponderance of the anemochorous and autochorous dispersal modes (55 %) over zoochory (45 %) in the cerrado sensu stricto hectare, shows that many species are able to disperse their diaspores without animals, and that for a little less than half of the species, animals are important dispersers. It is also possible to show that there are trends between woody and herbaceous growth forms of cerrado s.s. plants and dispersal modes. Woodiness is positively related with endozoochory, and endo- and synzoochory, as well as with anemochory of plants with winged dispersal units. With respect to the “herbaceous” plants (we are here considering the superficial appearance of the above ground portion of a plant), epizoochory and autochory are strongly related to this habit; and among anemochorous “herbs”, plumed dispersal units are quite common.
A comparison with a nearby one hectare plot of cerradão (height 6-10 m, tree crown cover close to 100 %) in Botucatu showed that the importance of zoochory in the forest-like plot was considerably higher than in the cerrado s.s. hectare plot. Zoochory made up 69 %, anemochory only 25 %, and autochory was as low as 6 % (Gottsberger & Silberbauer-Gottsberger 1983Gottsberger G, Silberbauer-Gottsberger I. 1983. Dispersal and distribution in the cerrado vegetation of Brazil. Sonderband naturwissenschaftlicher Verein für Hamburg 7: 315-352. ; 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.; Silberbauer-Gottsberger & Gottsberger 1984Silberbauer-Gottsberger I, Gottsberger G. 1984. Cerrado - Cerradão. A comparison with respect to number of species and growth forms. Phytocoenologia 12: 293-303.). Thus the tall forest-like physiognomy, the cerradão, is approaching the dispersal characteristics of humid tropical forests (Gottsberger & Silberbauer-Gottsberger 1983Gottsberger G, Silberbauer-Gottsberger I. 1983. Dispersal and distribution in the cerrado vegetation of Brazil. Sonderband naturwissenschaftlicher Verein für Hamburg 7: 315-352. ).
With respect to the major dispersal modes, the cerrado s.s. appears to occupy an intermediate position between Neotropical dry forests and moist rainforests. While in dry forests woody plants are predominantly wind dispersed and/or have a lower representation of zoochory than in the cerrado s.s., in rainforests the woody species are predominantly zoochorous, with more than 90 % of trees and about 75 % of lianas exhibiting this dispersal mode (Levey et al. 1984Levey DJ, Moermond TC, Denslow JS. 1984. Fruit choice in Neotropical birds: the effect of distance between fruits on preference patterns. Ecology 65: 844-850.; Gentry 1995Gentry AH. 1995. Diversity and floristic composition of neotropical dry forests. In: Bullock SH, Mooney HA, Medina E. (eds.) Seasonally dry tropical forests. Cambridge/ New York/ Melbourne, Cambridge University Press. p. 146-194.; Chazdon et al. 2003Chazdon RL, Careaga S, Webb C, Vargas O. 2003. Community and phylogenetic structure of reproductive traits of woody species in wet tropical forests. Ecological Monographs 73: 331-348.). Similar trends have been described for Brazilian Amazon and Atlantic forests (Frisch 1988Frisch RO. 1988. Fruchtbiologische Untersuchungen an einem amazonischen Regenwald der Terra Firme. Doctoral Thesis, University of Vienna, Vienna.; Morellato & Leitão-Filho 1992Morellato LPC, Leitão-Filho HF. 1992. Padrões de frutificacão e dispersão na Serra do Japi. In: Morellato LPC. (ed.) Histório natural da Serra do Japi: Ecologia e preservação de uma área florestal no sudeste do Brasil. Campinas, Editora UNICAMP/FAPESP. p. 112-140. ; Griz & Machado 2001Griz LMS, Machado ICS. 2001. Fruiting phenology and seed dispersal syndromes in caatinga, a tropical dry forest in the Northeast of Brazil. Journal of Tropical Ecology 17: 303-321.), for caatinga and Atlantic forest (Vicente et al. 2003Vicente A, Santos AMM, Tabarelli M. 2003. Variação no modo de dispersão de espécies lenhosas em um gradiente de precipitação entre floresta seca e úmida no nordeste do Brasil. In: Leal IR, Tabarelli M, Silva JMC. (eds.) Ecologia e conservação da caatinga. Recife, Universidade Federal de Pernambuco. p. 565-592. ). Further studies of seed dispersal in Brazilian Cerrado vegetation also confirm the described trends (Mantovani 1983Mantovani W. 1983. Composição e similaridade florística, fenologia e espectro biológico do cerrado da Reserva Biológica de Mogi Guaçu, Estado São Paulo. MSc thesis, Universidade de Campinas, Campinas.; Mantovani & Martins 1988Mantovani W, Martins FR. 1988. Variações fenológicas das espécies do cerrado da Reserva Biológica de Moji Guaçu, Estado de São Paulo. Revista Brasileira de Botânica 11: 101-112.; Varassin & Silva 1999Varassin IG, Silva WR. 1999. Padrões estacionais de frutificação e germinação de sementes em cerrado, Minas Gerais. Boletim do Museu de Biologia Mello Leitão 10: 13-28.; Batalha & Mantovani 2000Batalha MA, Mantovani W. 2000. Reproductive phenological patterns of cerrado plant species at the Pe-de-Gigante reserve (Santa Rita do Passa Quatro, SP, Brazil): a comparison between the herbaceous and woody floras. Revista Brasileira de Biologia 60: 129-145.; Vieira et al. 2002Vieira DLM, Aquino FG, Brito MA, Fernandes-Bulhão C, Henriques RPB. 2002. Síndromes de dispersão de espécies arbustivo-arbóreas em cerrado strictu sensu do Brasil Central e savanas amazônicas. Revista Brasileira de Botânica 25: 215-220.; Batalha & Martins 2004Batalha MA, Martins FR. 2004. Reproductive phenology of the cerrado plant community in Emas National Park (Central Brazil). Australian Journal of Botany 52: 149-161.; Yamamoto et al. 2007Yamamoto LF, Kinoshita LS, Martins FR. 2007. Síndromes de polinização e de dispersão em fragmentos da Floresta Estacional Semidecídua Montana, SP, Brasil. Acta Botanica Brasilica 21: 553-573.).
Animals responsible in the cerrado s.s. hectare for endo-, syn- or epizoochory are several large reptiles, such as the teiú (Tupinambis teguixim), or birds, such as the ema (Rhea americana) along with a large number of the further 900 bird species occurring in the Cerrado region (Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.). Many mammals eat or transport fruits and seeds, as for instance, bats, opossums, agoutis, cervids, the maned wolf, Chrysocyon brachyurus (fond of the wolf-fruit, Solanum lycocarpum, as well as of many other fruits), the tapirs, some primates, omnivorous canids and even some dung beetles or ants (for animals as potential seed dispersers in cerrado in former times and at present see Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.).
The zoochorous scrub species of the cerrado s.s. hectare are visited mostly by flying or climbing animals (birds, bats, monkeys, spiny rats, etc.), and their fruits are likewise in reach of almost all larger ground animals, such as wolves, tapirs, forest dogs, iguanas, larger ground birds, etc. The numerous potential fruit- and seed-eaters available to fruits disposed at moderate heights may explain the high number of endozoochorous species found in the scrub layer. Synzoochory is strongest represented in the ground layer. Ten of the eleven synzoochorous species are dispersed by ants (myrmecochory); Anacardium humile is dispersed by birds and mammals. Epizoochorous species are the best represented among the ground layer. Ground animals move predominantly horizontally, reinforcing the effectiveness of this dispersal mode. In the tree layer, where animals move both horizontally and vertically, epizoochory as a passive event appears to function less efficiently. Our study also indicated that polychory, dispersal by several means or, in our case, by more than one animal group, predominated when we consider all species together (Gottsberger & Silberbauer-Gottsberger 2006bGottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.). As described for the temperate zones (see, e.g., Snow & Snow 1988Snow B, Snow D. 1988. Birds and berries. Calton., T & AD Poyser.; Pirl 2001Pirl M. 2001. Reproduktionsökologische Untersuchungen an Sträuchern und Bäumen zentraleuropäischer Gehölzfluren. Ertragskundliche, phänologische, nahrungs- und verhaltensökologische Studien zu Bestäubung und Ausbreitung als tiervermittelter Schlüsselprozesse im Lebenszyklus von Blütenpflanzen. Frankfurt am Main, Verlag Neue Wissenschaft. ), in the Cerrado, a large number of animals appear adapted for feeding on fleshy diaspores. The animals are of adequate size and physically able to reach and swallow these diaspores. Most animals appear to be generalists and the fruits constitute only part of their diet, but these fruits may be essential for survival when other sources are scarce.
The majority of pollinating or seed dispersing animals in the Cerrado is closely associated with mesic gallery forests. Gallery forests are the primary living habitat for most of them, also serving as corridors for their migration. Many animals enter the Cerrado mostly for feeding. For their reproduction and the major part of their food supply, they depend on the gallery forests. In a sense, Cerrado is often a secondary habitat for many flower-visiting and seed-dispersing forest animals. In terms of pollinators and seed dispersers, Cerrado is often “nourished” by the surrounding forests. Without this surrounding forests, animal and plant life in Cerrado would be much poorer. Quite certainly there is a continual migration not only of birds and mammals, but probably also of many insects (especially strong flyers) from the forest to the Cerrado and back again to the gallery forests on a daily basis (Gottsberger & Silberbauer-Gottsberger 1983Gottsberger G, Silberbauer-Gottsberger I. 1983. Dispersal and distribution in the cerrado vegetation of Brazil. Sonderband naturwissenschaftlicher Verein für Hamburg 7: 315-352. ). Such movements and flight patterns are important to the understanding of the role “surrounding” vegetation plays in the dynamics and maintenance of Cerrado.
Acknowledgements
The authors thank the authorities of their former university in Botucatu for support during the years of their employment (1968 to 1981), their field assistants Clemente José Campos, Valdir Factori, Antonio Suman, Benedito Gonçalo Alves, Antonio Chiquinato, Oswaldo Rodrigues and Jorge de Almeida, as well as the specialists who identified the plant and animal material. Graciela Hintze kindly prepared the figures and Felipe Amorim and Paulo Eugenio Oliveira were extremely helpful in improving the manuscript.
References
- Batalha MA, Mantovani W. 2000. Reproductive phenological patterns of cerrado plant species at the Pe-de-Gigante reserve (Santa Rita do Passa Quatro, SP, Brazil): a comparison between the herbaceous and woody floras. Revista Brasileira de Biologia 60: 129-145.
- Batalha MA, Martins FR. 2004. Reproductive phenology of the cerrado plant community in Emas National Park (Central Brazil). Australian Journal of Botany 52: 149-161.
- Bawa KS, Bullock SH, Perry DR, Coville RE, Grayum MH. 1985. Reproductive biology of tropical lowland rain forest trees. II. Pollination systems. American Journal of Botany 72: 346-356.
- Chazdon RL, Careaga S, Webb C, Vargas O. 2003. Community and phylogenetic structure of reproductive traits of woody species in wet tropical forests. Ecological Monographs 73: 331-348.
- Coutinho LM. 1978. O conceito de cerrado. Revista Brasileira de Botânica 1: 17-23.
- Dulmen A. 2001. Pollination and phenology of flowers in the canopy of two contrasting rain forest types in Amazonia, Colombia. Plant Ecology 153: 73-85.
- Eiten G. 1972. The cerrado vegetation of Brazil. Botanical Review: 201-341.
- Eiten G. 1978. Delimitation of the cerrado concept. Vegetatio 36: 169-178.
- Eiten G. 2001. Vegetação natural do Distrito Federal. Brasília, Editora da Universidade de Brasília.
- Frankie GW. 1975. Tropical forest phenology and pollinator plant coevolution. In: Gilbert LE, Raven PH. (eds.) Coevolution of animals and plants. Austin/ London, University of Texas Press. p. 192-209.
- Frankie GW, Haber WA. 1983. Why bees move among mass-flowering neotropical trees. In: Jones CE, Little RJ. (eds.) Handbook of experimental pollination biology. New York, Van Nostrand Reinhold. p. 361-372.
- Frankie GW, Haber WA, Opler PA, Bawa KS. 1983. Characteristics and organization of the large bee pollination system in the Costa Rican dry forest. In: Jones CE, Little RJ. (eds.) Handbook of experimental pollination biology . New York, Van Nostrand Reinhold . p. 411-447.
- Frisch RO. 1988. Fruchtbiologische Untersuchungen an einem amazonischen Regenwald der Terra Firme. Doctoral Thesis, University of Vienna, Vienna.
- Gentry AH. 1995. Diversity and floristic composition of neotropical dry forests. In: Bullock SH, Mooney HA, Medina E. (eds.) Seasonally dry tropical forests. Cambridge/ New York/ Melbourne, Cambridge University Press. p. 146-194.
- Girão LC, Lopes AV, Tabarelli M, Bruna EM. 2007. Changes in tree reproductive traits reduce functional diversity in a fragmented Atlantic forest landscape. PLOS ONE 9(8): e908. doi: 10.1371/journal.pone.0000908
» https://doi.org/10.1371/journal.pone.0000908 - Goodland R. 1971. A physiognomic analysis of the “cerrado” vegetation of Central Brazil. Journal of Ecology 59: 411-419.
- Goodland R, Ferri G. 1979. Ecologia do Cerrado. Belo Horizonte/ São Paulo, Livraria Itatiaia Editora, Editora Universidade São Paulo.
- Goodland R, Pollard R. 1973. The Brazilian cerrado vegetation: a fertility gradient. Journal of Ecology 61: 219-224.
- Gottsberger G, Camargo JMF, Silberbauer-Gottsberger I. 1988. A bee-pollinated tropical community: the beach dune vegetation of Ilha de São Luís, Maranhão, Brazil. Botanische Jahrbücher für Systematik 109: 469-500.
- Gottsberger G, Silberbauer-Gottsberger I. 1983. Dispersal and distribution in the cerrado vegetation of Brazil. Sonderband naturwissenschaftlicher Verein für Hamburg 7: 315-352.
- Gottsberger G, Silberbauer-Gottsberger I. 2006a. Life in the cerrado: a South American tropical seasonal ecosystem. Vol. I. Origin, structure, dynamics and plant use. Ulm, Reta.
- Gottsberger G, Silberbauer-Gottsberger I. 2006b. Life in the cerrado: a South American tropical seasonal ecosystem . Vol. II. Pollination and seed dispersal. Ulm, Reta.
- Griz LMS, Machado ICS. 2001. Fruiting phenology and seed dispersal syndromes in caatinga, a tropical dry forest in the Northeast of Brazil. Journal of Tropical Ecology 17: 303-321.
- Guariguata MR, Chazdon RL, Denslow JS, Dupuy JM, Anderson L. 1997. Structure and floristics of secondary and old-growth forest stands in lowland Costa Rica. Plant Ecology 132: 107-120.
- Gumbert A, Kunze J. 1999. Inflorescence height affects visitation behavior of bees - a case study of an aquatic plant community in Bolivia. Biotropica 31: 466-477.
- Haber WA, Frankie GW. 1989. A tropical hawkmoth community: Costa Rican dry forest Sphingidae. Biotropica 21: 155-172.
- Hallé F, Oldeman RAA, Tomlinson PB. 1978. Tropical trees and forests. Berlin/ Heidelberg/ New York, Springer.
- Kanstrup J, Olesen JM. 2000. Plant-flower visitor interactions in a Neotropical rain forest canopy: community structure and generalisation level. Det Norske Videnskaps-Akademi. I. Mat.-Naturv. Klasse Skrifter, Ny Serie 39: 33-41.
- Kress WJ, Beach JH. 1994. Flowering plant reproductive systems. In: McDade LA, Bawa KS, Hespenheide HA, Hartshorn GS. (eds.). La Selva - Ecology and natural history of a Neotropical rain forest. Chicago/ London, University of Chicago Press. p. 161-182.
- Levey DJ, Moermond TC, Denslow JS. 1984. Fruit choice in Neotropical birds: the effect of distance between fruits on preference patterns. Ecology 65: 844-850.
- Lowmann MD, Rinker HB. 2004. Forest canopies. 2nd. edn. Burlington, Elsevier Academic Press.
- Mantovani W. 1983. Composição e similaridade florística, fenologia e espectro biológico do cerrado da Reserva Biológica de Mogi Guaçu, Estado São Paulo. MSc thesis, Universidade de Campinas, Campinas.
- Mantovani W, Martins FR. 1988. Variações fenológicas das espécies do cerrado da Reserva Biológica de Moji Guaçu, Estado de São Paulo. Revista Brasileira de Botânica 11: 101-112.
- Martins FQ, Batalha MA. 2005. Vertical and horizontal distribution of pollination systems in ocerrado fragments of Central Brazil. In: The 2005 meeting of the association for tropical biology and conservation, frontiers in tropical biology and conservation. Abstracts.Universidade Federal de Uberlândia. p. 120.
- Maruyama PK, Oliveira GM, Ferreira C, Dalsgaard B, Oliveira PE. 2013. Pollination syndromes ignored: importance of non-ornithophilous flowers to Neotropical savanna hummingbirds. Naturwissenschaften 100:1061-1068.
- Morellato LPC, Leitão-Filho HF. 1992. Padrões de frutificacão e dispersão na Serra do Japi. In: Morellato LPC. (ed.) Histório natural da Serra do Japi: Ecologia e preservação de uma área florestal no sudeste do Brasil. Campinas, Editora UNICAMP/FAPESP. p. 112-140.
- Oliveira PE, Gibbs PE. 2000. Reproductive biology of woody plants in a cerrado community of Central Brazil. Flora 195: 311-329.
- Oliveira PE, Gibbs PE. 2002. Pollination and reproductive biology in cerrado plant communities. In: Oliveira PS, Marquis RJ. (eds.). The Cerrados of Brazil. Ecology and natural history of a Neotropical savanna. New York, Columbia University Press. p. 329-347.
- Pirl M. 2001. Reproduktionsökologische Untersuchungen an Sträuchern und Bäumen zentraleuropäischer Gehölzfluren. Ertragskundliche, phänologische, nahrungs- und verhaltensökologische Studien zu Bestäubung und Ausbreitung als tiervermittelter Schlüsselprozesse im Lebenszyklus von Blütenpflanzen. Frankfurt am Main, Verlag Neue Wissenschaft.
- Ramirez N. 2004a. Pollination specialization and time of pollination on a tropical Venezuelan plain: variations in time and space. Botanical Journal of the Linnean Society 145: 1-16.
- Ramirez N. 2004b. Ecology of pollination in a tropical Venezuelan savanna. Plant Ecology 173: 171-189.
- Ribeiro JF. (ed.) 1998. Cerrado: matas de galeria. Planaltina, Embrapa-CPAC.
- Richards PW. 1996. The tropical rainforest: an ecological study. 2nd. edn. Cambridge, Cambridge University Press.
- Roubik DW. 1993. Tropical pollinators in the canopy and understory : Field data and theory for stratum "preferences". Journal of Insect Behavior 6: 659-673.
- Sarmiento G. 1983. The savannas of tropical America. In: Boulièr F. (ed.) Tropical savannas (Ecosystems of the world 13). Amsterdam, Elsevier.
- Silberbauer-Gottsberger I, Eiten G. 1983. Fitossociologia de um hectare de cerrado. Brasil Florestal 54: 55-70.
- Silberbauer-Gottsberger I, Eiten G. 1987. A hectare of cerrado. I. General aspects of trees and thick-stemmed shrubs. Phyton(Horn, Austria) 27: 55-91.
- Silberbauer-Gottsberger I, Gottsberger G. 1984. Cerrado - Cerradão. A comparison with respect to number of species and growth forms. Phytocoenologia 12: 293-303.
- Silberbauer-Gottsberger I, Gottsberger G. 1988. A polinização de plantas do cerrado. Revista Brasileira de Biologia 48: 651-663.
- Snow B, Snow D. 1988. Birds and berries. Calton., T & AD Poyser.
- Varassin IG, Silva WR. 1999. Padrões estacionais de frutificação e germinação de sementes em cerrado, Minas Gerais. Boletim do Museu de Biologia Mello Leitão 10: 13-28.
- Vicente A, Santos AMM, Tabarelli M. 2003. Variação no modo de dispersão de espécies lenhosas em um gradiente de precipitação entre floresta seca e úmida no nordeste do Brasil. In: Leal IR, Tabarelli M, Silva JMC. (eds.) Ecologia e conservação da caatinga. Recife, Universidade Federal de Pernambuco. p. 565-592.
- Vieira DLM, Aquino FG, Brito MA, Fernandes-Bulhão C, Henriques RPB. 2002. Síndromes de dispersão de espécies arbustivo-arbóreas em cerrado strictu sensu do Brasil Central e savanas amazônicas. Revista Brasileira de Botânica 25: 215-220.
- Warming E. 1892. Lagoa Santa. Kjøbenhavn, Bianco Lunos.
- Yamamoto LF, Kinoshita LS, Martins FR. 2007. Síndromes de polinização e de dispersão em fragmentos da Floresta Estacional Semidecídua Montana, SP, Brasil. Acta Botanica Brasilica 21: 553-573.
-
1
Unfortunately, in 1982, after more than ten years of study, nearly the total area was cut by the owners of the sugarcane company São Manuel, and replanted mainly with sugarcane.
Publication Dates
-
Publication in this collection
30 July 2018 -
Date of issue
Jul-Sep 2018
History
-
Received
23 May 2018 -
Accepted
13 June 2018