ABSTRACT
The perception, knowledge and use of non-native and invasive flora by social actors associated with protected areas in central Argentina were analyzed. Contemporary ethnoecology methods were followed. A total of 108 non-native species of botanical interest was documented. An organization of species is proposed according to their Relative Importance, and on an attitude scale in relation to conservation/elimination interest. In the case of invasive species, perceptions on ecology, importance and/or control are discussed based on the profiles of the social actors. The results shed light upon local points of view and nuances of what “non-native” and “invasive” means to the studied rural population. The use spectrum for some of the non-native species has shed light on adaptation to the cultural use of species based on resource availability, not necessarily associated with preference. Additionally, it should be noted that local actors perceive non-native and invasive species differently, which should be considered in the development of management and conservation guidelines.
Keywords:
invasive flora; non-native; perception; relative importance; social actors
Introduction
Invasive plants are naturalized plants that produce reproductive offspring, often in very large numbers, at considerable distances from parent plants, thus having the potential to spread over a considerable area (Richardson et al. 2000Richardson DM, Pysek P, Rejmánek M, Barbour MG, Panetta FD, West CJ. 2000. Naturalization and invasion of alien plants: concepts and definitions. Diversity and Distributions 6: 93-107.). The naturalization of non-native species introduced is a continuous process, which includes as results by means of different terms used in the literature, such as cultivated, naturalized adventitious and invasive species (Hurrell & Deluchi 2013Hurrell J, Deluchi G. 2013. Aportes de la etnobotánica al estudio de las invasiones biológicas. Casos en la Región Rioplatense (Argentina). Historia Natural 3: 61-76.). In particular, non-native species are plants introduced by humans from one geographic area to another non-native one, either intentionally or unintentionally, and which have become established, proliferated and spread out (Das & Duarah 2013Das K, Duarah P. 2013. Invasive alien plant species in the roadside areas of Jorhat, Assam: Their harmful effects and beneficial uses. International Journal of Engineering Research and Applications 3: 353-358.). Such species have also been given greater and greater emphasis due to their negative impact on landscapes, ecosystems, biodiversity and the subsistence of local populations (Bardsley & Edward-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.; Mack et al. 2000Mack RN, Simberloff D, Lonsdale WM, Evans H, Clout M, Bazzaz FA. 2000. Biotic invasions causes, epidemiology, global consequences, and control. Ecological Applications 3: 689-710. ; Busso et al. 2013Busso CA, Bentivegna DJ, Fernández OA. 2013. A review on invasive plants in rangelands of Argentina. Interciência 38: 95-103.; Rapoport 2000Rapoport EH. 2000. Remarks on the biogeography of land invasions. Revista Chilena de Historia Natural 73: 367-380.). The numerous environmental impacts include loss of native species, changes in the structure and the composition of invaded habitats, in the nutrient and water cycle, in the fire regiment and in the ability to produce goods and ecosystemic services (Busso et al. 2013Busso CA, Bentivegna DJ, Fernández OA. 2013. A review on invasive plants in rangelands of Argentina. Interciência 38: 95-103.; Giorgis & Tecco 2014Giorgis MA, Tecco PA. 2014. Árboles y arbustos invasores de la provincia de Córdoba (Argentina): una contribución a la sistematización de bases de datos globales. Boletín de la Sociedad Argentina de Botánica 49: 581-603.; Souza et al. 2018Souza OA, Rodríguez Chaves MPS, Barbosa RI, Clement CR. 2018. Local ecological knowledge concerning the invasion of Amerindian lands in the northern Brazilian Amazon by Acacia mangium (Willd.). Journal of Ethnobiology and Ethnomedicine 14:33. doi: 10.1186/s13002-018-0231-x
https://doi.org/10.1186/s13002-018-0231-...
). In this sense, it is often believed that invasive plants have a destructive impact on their ecosystems, posing threats on native vegetation and species.
However, some of these invasive plants not only generate no costs, they also offer benefits when used by local inhabitants for daily life (for example, firewood, food, medicine or forage), making them holders of ethnobotanical use-value (Bardsley & Edwards-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.; Sundaram et al. 2012Sundaram B, Krishnan S, Hiremath AJ, Joseph G. 2012. Ecology and Impacts of the Invasive Species, Lantana camara, in a Social-Ecological System in South India: Perspectives from Local Knowledge. Human Ecology 40: 931-942. ; Das & Duarah 2013Das K, Duarah P. 2013. Invasive alien plant species in the roadside areas of Jorhat, Assam: Their harmful effects and beneficial uses. International Journal of Engineering Research and Applications 3: 353-358.; Hurrell & Deluchi 2013Hurrell J, Deluchi G. 2013. Aportes de la etnobotánica al estudio de las invasiones biológicas. Casos en la Región Rioplatense (Argentina). Historia Natural 3: 61-76.; Santos et al. 2014Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.; Souza et al. 2018Souza OA, Rodríguez Chaves MPS, Barbosa RI, Clement CR. 2018. Local ecological knowledge concerning the invasion of Amerindian lands in the northern Brazilian Amazon by Acacia mangium (Willd.). Journal of Ethnobiology and Ethnomedicine 14:33. doi: 10.1186/s13002-018-0231-x
https://doi.org/10.1186/s13002-018-0231-...
). For instance, great emphasis has been placed on: a vast number of invasive species due to their medical benefits in India (Wagh & Jain 2018Wagh VV, Jain AK. 2018. Status of ethnobotanical invasive plants in western Madhya Pradesh, India. South African. Journal of Botany 114: 171-180.) and South Africa (Maema et al. 2016Maema LP, Potgieter M, Mahlo SM. 2016. Invasive alien plant species used for the treatment of various diseases in Limpopo province, South Africa. African Journal of Traditional, Complementary and Alternative Medicine 13: 223-231. ), the food use of a wide range of weeds (Díaz-Betancourt et al. 1999Díaz-Betancourt M, Ghermandi L, Ladio A, López-Moreno IR, Raffaele E, Rapoport EH. 1999. Weeds as a source for human consumption. A comparison between tropical and temperate Latin America. Revista de Biología Tropical 47: 329-338.; Rapoport et al. 2009Rapoport EH, Marzocca A, Drausal BS. 2009. Malezas comestibles del Cono Sur y otras partes del planeta. Buenos Aires, Ediciones INTA. ; Santos et al. 2014Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.), the use of fuel species (Martínez 2015Martínez GJ. 2015. Cultural patterns of firewood use as a tool for conservation: A study of multiple perceptions in a semiarid region of Cordoba, Central Argentina. Journal of Arid Environments 121: 84: 99.) or the use of species as a source of forage (Mekoya et al. 2008Mekoya A, Oosting SJ, Fernandez-Rivera S, Zijpp AJ. 2008. Farmers’ perceptions about exotic multipurpose fodder tres and constraints to their adoption. Agroforestry Systems 73: 141-153.). Even though there are some examples, more detailed information is needed about the traditional and ethnobotanical knowledge of non-native plants, especially with regard to the impact of invasive plants on the well-being of traditional and/or indigenous populations. This is so if we consider the need of having control programs based on cost-benefit analysis of non-native species management (Bardsley & Edwards-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.). Or rather, the negative impact exerted on indigenous and/or traditional communities must be considered when setting priorities in control programs.
In Argentina, over the last few years, greater interest has been placed on non-native plants found away from their area of natural distribution due to anthropic reasons, either because they are potential invasive species or because of their potential naturalization in the flora of the country, especially, the ornamental species escaped from cultivation (see Cantero et al. 2016Cantero JJ, Barboza GE, Chiarini FE, et al. 2016. Novedades para la flora de la Argentina. Boletín de la Sociedad Argentina de Botánica 51: 183-207. and references therein). Thus, these studies on invasive plants have only focused on ecological aspects and, except for the work reported by Hurrell & Deluchi (2013Hurrell J, Deluchi G. 2013. Aportes de la etnobotánica al estudio de las invasiones biológicas. Casos en la Región Rioplatense (Argentina). Historia Natural 3: 61-76.), neither the ethnobotanical perspectives nor the approaches based on environmental perceptions have been considered.
Concerning perception studies on environmental changes, these have also been researched in some indigenous and peasant populations both in Latin America and worldwide. This has contributed to a variety of environmental views related to different variables, including personal (such as the personal and historic career in relation to the milieu), sociocultural (gender-based work), economic and even political ones (Barri 2012Barri F. 2012. Territorio, percepción ambiental e intereses en conflicto: El caso de la Reserva Vaquerías. PhD Thesis, Universidad Nacional de Córdoba, Córdoba. ). In particular, studies on environmental perception in various local actors offer a diverging approach to ecological studies, showing the view of local people at stake in relation to biodiversity (Mekoya et al. 2008Mekoya A, Oosting SJ, Fernandez-Rivera S, Zijpp AJ. 2008. Farmers’ perceptions about exotic multipurpose fodder tres and constraints to their adoption. Agroforestry Systems 73: 141-153.; Cáceres et al. 2015Cáceres DM, Tapella E, Quétier F, Díaz S. 2015. The social value of biodiversity and ecosystem services from the perspectives of different social actors. Ecology and Society 20(1): 62 doi: 10.5751/ES-07297-200162
https://doi.org/10.5751/ES-07297-200162...
; Martínez & Manzano-Garcia 2016Martínez GJ, Manzano-García J. 2016. Estilos de percepción de la biodiversidad y su conservación en actores sociales de áreas protegidas de córdoba. Revista del Museo de Antropología 9: 135-152.). These studies analyze different topics, including variations in biodiversity and the provision of ecosystemic services in transformed environments; local meaning of living creatures in the environment; local priorities of conservation; transformation in daily life due to changes in resource availability; displacement of local knowledge by scientific-technical knowledge; and transformation from a model based on use of resources to patrimonialization process of nature for conservation practices and policies in protected areas (Santamarina-Campos & Bodí 2012Santamarina-Campos B, Bodí J. 2012. Lugares rurales versus espacios naturalizados. Conocimientos y reconocimientos en las lógicas patrimoniales de las áreas protegidas. Revista de Antropología Iberoamericana 8: 111-138.). Finally, there are few references to studies on environmental perception of invasive species in protected areas. Some examples/cases include the works of Wilgen (2012Wilgen B. 2012. Evidence, perceptions, and trade-offs associated with invasive alien plant control in the Table Mountain National Park, South Africa. Ecology and Society 17: 23. doi: 10.5751/ES-04590-170223
https://doi.org/10.5751/ES-04590-170223...
), and Richardson & Wilgen (2004Richardson DM, Wilgen BW. 2004. Invasive alien plants in South Africa: How well do we understand the ecological impacts? South African Journal of Science 100: 45-52.).
The aim of this work is to analyze the perception, knowledge and use of non-native and invasive non-native flora in social actors associated with two protected areas in Sierras de Córdoba, interpreting the local notion of biological invasion phenomena.
Specific objectives include: Identifying the non-native species used in the regions under study and describe their main applications following categories of ethnobotanical use; characterizing local perceptions on the importance, ecology and/or control of flora that has been “introduced” and is “non-native and invasive” in different social actors associated with protected areas based on qualitative and quantitative indicators; interpreting the ethnobotanical and perceptual information related to practices of conservation, management and/or control of these species in protected areas.
Materials and methods
Study site and cultural background
According to Cabrera (1994Cabrera AL. 1994. Regiones fitogeográficas argentinas. Vol. II Enciclopedia Argentina de Agricultura y Jardinería. Fascicle 1. Buenos Aires, Acme S.A.C.I. ), most of the region of Sierras de Córdoba belongs to the Chaco Serrano, a phytogeographical district in the province of Chaco. According to Torrella & Adámoli (2005Torrella S, Adámoli J. 2005. Situación ambiental en el Chaco Árido y Serrano. In: Brown A, Martínez Ortiz U, Acerbi M, Corchera J. (eds.) La Situación Ambiental Argentina. Buenos Aires, Fundación Vida Silvestre Argentina. p. 75-83.), it is part of the ecoregion of Chaco Seco. The floristic composition of these forests changes with latitude and altitude at a regional scale as well as with edaphic characteristics and the disturbance background at a local scale, turning it into a heterogeneous system with significant variations in the composition of species at short distances (Giorgis et al. 2017Giorgis MA, Cingolani AM, Gurvich DE, et al. 2017. Changes in floristic composition and physiognomy are decoupled along elevation gradients in central Argentina. Applied Vegetation Science 20: 558-571.).
This research was conducted in two protected areas in the mountainous range of Córdoba with different characteristics in relation to practices of conservation, management, and use of natural resources as well as population associated with such areas and their environmental background. One area has been declared the Bamba Natural Water Reserve (Reserva Hídrica Natural Bamba); yet, no plan or strategy for environmental management has been developed accordingly. The other area, called La Rancherita Natural Reserve (Reserva Natural La Rancherita), has been recently created, and its regulations and management are underway.
The Bamba Natural Water and Recreation Reserve, in Colón district, covers around 20,000 hectares (around 49,500 acres) located West of the city of Córdoba, between San Roque Lake and East of the Punilla valley. Its territories as well as La Calera Natural Military Reserve, which is contiguous to the latter, are being considered as potential areas for the creation of a National Park and a Memory Park (see additional information at www.escalera.org.ar). It has been recently declared a National Area of Interest for Conservation (Espacio Nacional de Interés para la Conservación, ENIC). In particular, the Bamba reserve is a very important reservoir that supplies water to the city of Córdoba. Despite this, its background of environmental use is not linked to conservation; so much so that there are different major socio-environmental and territorial challenges in the area, which have been posed by mining, land expropriation for urban development, water pollution, among others. Some independent organizations have recently proposed to issue regulations and develop management plans for the reserve (Palacios et al. 2010Palacios AA, Spicogna JM, Bernasconi J, et al. 2010. Decidirnos a decidir. Experiencias sobre la gestación de la Unidad Reserva Natural Militar la Estancia La Calera y Reserva Hídrica y Recreativa Natural Bamba. Córdoba, Aula de Montaña de la Universidad Nacional de Córdoba.). However, in practice, such measures have not been implemented/applied given the resistance and lack of consent ofby political leaders. This territory features very valuable natural qualities and historical-cultural characteristics, which at times are unknown or not recognized by its residents. Those involved in the composition of this territory are small-scale livestock producers, depeasantized natives and urban population members of different backgrounds that have recently settled down. The total population of Bamba Reserve is estimated to reach 93 inhabitants in the center of the reserve and 850 in the buffer zone.
La Rancherita Multi-Purpose Reserve is located in the Paravachasca region, Santa María district, southwest of Córdoba. It is a protected area covering 45 hectares (112 acres) of mountain forests that are very well preserved. Its community has suggested to start a participation process for issuing regulations as well as creating a land management and organization plan through which a Provincial Multi-Purpose Reserve can be built and the common land altogether, included-this would reach 300 hectares (742 acres). At present, the population living there is made up of about 200 inhabitants with permanent and temporal residence, the peasant population being minority and the remaining population a community of neorural foreign inhabitants especially interested in the practice of conversation and the use for recreation and tourism (Martínez et al. 2016Martínez GJ, Fernández M, Flores C, et al. 2016. Diagnóstico participativo, orientaciones y aportes para la conservación y el manejo de la reserva de usos múltiples de La Rancherita y Las Cascadas. Córdoba, IDACOR-CONICET.).
Plant collection
The collection of local species of ethnobotanical interest that serve as a reference for both protected areas was conducted in different periods and throughout several years: La Rancherita reserve (2001-2013) and Bamba Natural Water and Recreation Reserve (2009-2013). Furthermore, a database of collections and ethnobotanical uses in Sierras de Córdoba was used by the team as a reference. The material collected was conditioned and deposited in the Herbariums of the School of Agricultural Sciences (ACOR) and the Multidisciplinary Institute of Plant Biology (CORD).
Data collection
Some methodologies used in social and natural sciences were applied following the interdisciplinary approach of ethnoecology (Alexiades 1996Alexiades MN. 1996. Standard techniques for collecting and preparing herbarium species. In: Alexiades MN. (ed.) Selected guidelines for ethnobotanical research: A field manual. New York, Botanical Garden Bronx. p. 99-126.; Berkes 2011Berkes F. 2011. Sacred ecology: traditional ecological knowledge and resource management. New York, Taylor y Francis. ) and by conducting mixed quantitative and qualitative analyses (Albuquerque et al. 2010Albuquerque UP, Lucena RFP, Alencar NL. 2010. Métodos e técnicas para a coleta de dados etnobiologicos. In: Albuquerque UP, Lucena RFP, Cunha LVF. (eds.) Métodos e técnicas na pesquisa etnobiologica e etnoecologica. Recife, NUPEEA. p. 39-63.; Svanverg et al. 2011Svanverg I, Luczaj L, Santayana MP, Pieroni A. 2011. History and current trends of ethnobiological research in Europe. In: Anderson EN, Pearsall DM, Hunn ES, Turner NJ. (eds.) Ethnobiology. New Jersey, Wiley-Blackwell, Inc. Hoboken. p. 189-212.; Arenas & Martínez 2012Arenas P, Martínez GJ. 2012. Estudio etnobotánico en zonas áridas y semiáridas de Argentina y zonas limítrofes. Experiencias y reflexiones metodológicas de un grupo de investigación. In: Arenas P. (ed.) Etnobotánica en zonas áridas y semiáridas del Cono Sur de Sudamérica. Buenos Aires, CEFIBO-CONICET. p. 11-43. ). To this end, the following plan was carried out:
Open and in-depth interviews in 12 households. The content of the narratives obtained were interpreted by means of qualitative analysis (Valles 1999Valles M. 1999. Técnicas cualitativas de investigación social. Reflexión metodológica y práctica profesional. Madrid, Síntesis S.A. ; Guber 2005Guber R. 2005. El salvaje metropolitano, Reconstrucción del conocimiento social en el trabajo de campo. Buenos Aires, Paidos. )
Semi-structured and structured interviews (with precategorized data) to native, resident and foreign populations, among others, with a total of 44 interviewees chosen intentionally by the snowball method. For semi-structured interviews, in order to establish the sample size, we adopted the data saturation criterion. Additionally, it was intended to avoid redundancy and represent the wide range of actors as well as their views on conservation and its association with biodiversity. To make a more detailed analysis, based on a previous demographic and cultural characterization of the interviewees, three types of social actors were determined, including: a) native population (PN)- members of the population born there or having been lived for long there, peasant population currently associated with subsistence activities (peasants) or associated with these in the recent past (the depeasantized); b) foreign or neorural population (PF)- non-native population coming from urban areas, newly arrived, temporal or sporadic residents, non-peasant population; c) other actors (OA)- external actors settled in the area and associated with conservation organisms and local environmental groups, or real estate and tour businesses and state/governmental entities. In addition, a socio-economic characterization was taken into account for the analysis, for which, from the continuum of economic capitalization, the following polar categories were defined: more capitalization (C), and less capitalization with features of a subsistence economy (S). Table 1 shows the number of inhabitants by area and the type of social actors.
The semi-structured interviews helped carry out different quantitative studies, such as: amount of species and ethnobotanical uses of non-native flora; distribution of the most common uses; Relative Importance: the Relative Importance of Species (RI) adapted from Bennett & Prance (2000)Bennett BC, Prance GT. 2000. Introduced plants in the indigenous Pharmacopoeia of Northern South America. Economic Botany 54: 90-102. was calculated. This values species according to the number of ethnobotanical areas (EA) attributed to the taxon (e.g., 2: medicinal and edible), and the number of uses (NU) to which it is applied (e.g., 3: 2 different medicinal uses and 1 as a firewood species): RI = ((Rel EA + Rel NU) % 2) x 100; =0.25, where: Rel EA: relative number of ethnobotanical areas of uses attributed to a species (normalized to a maximum value of 1); Rel NU: relative number of ethnobotanical uses of a given species (normalized to a maximum value of 1).
An organization of non-native species in a positive and negative ranking for non-native species based on the mention of species based on perception of: importance due to use (+); relevance for conservation (+); retraction or environmental pressure (+); need for control/elimination (−)
Analysis of the perception from visual stimuli: Based on a task consisting of identifying a landscape that, due to its characteristics, the subjects would eliminate or change, interpretations of the perception on landscapes from visual stimuli were made (choosing pictures of different environmental areas). A Multiple Correspondence Analysis was also applied, as described below. To this end, a matrix of qualitative data was built, consisting of three variables or categories with their respective features, namely: a) type of actor (native, foreign and others); b) type of landscape to be eliminated or controlled; and c) socio-economic profile (capitalized and subsistence one). The analysis was made in the statistical program Infostat 2017 (Di Rienzo et al. 2017Di Rienzo JA, Balzarini M, Gonzalez L, et al. 2017. InfoStat software estadístico. Versión 2017. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar. 10 Jul. 2018.
http://www.infostat.com.ar...
).
Participation workshops on diagnosis and environmental planning (held at La Rancherita reserve): complementary information on environmental perceptions was gathered in seven workshops. Here, excerpts from narratives and/or accounts referring to the issue of invasive plants as well as the role of emblematic non-native plants were transcribed.
Participant observation (Guber 2005Guber R. 2005. El salvaje metropolitano, Reconstrucción del conocimiento social en el trabajo de campo. Buenos Aires, Paidos. ).
Terminology
To define the status of plants, we adopted the terminology proposed by Richardson et al. (2000Richardson DM, Pysek P, Rejmánek M, Barbour MG, Panetta FD, West CJ. 2000. Naturalization and invasion of alien plants: concepts and definitions. Diversity and Distributions 6: 93-107.). In that study they consider exotic plants, non-native plants, introduced plants and non-indigenous plants, the plant taxa in a given area whose presence there is attributed to intentional or accidental introduction from human activity. Naturalized plants are also alien plants that reproduce consistently and sustain population over many life cycles without direct intervention by humans; they often recruit offspring freely, usually close to adult plants, and do not necessarily invade natural, seminatural or human-made ecosystems. Finally, invasive plants are naturalized plants that produce reproductive offspring, often in very large numbers, at considerable distances from parent plants, thus having the potential of spreading over a considerable area.
Results and discussion
The amount of species and ethnobotanical uses of non-native flora
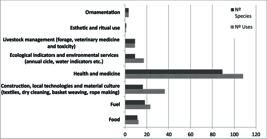
A total of 209 applications corresponding to 108 non-native species of ethnobotanical interest belonging to 41 botanical families were recorded (Fig. 1). Among these, 16.11 % (29 species) correspond to non-native invasive species according to a database for the province of Cordoba, Argentina (Giorgis & Tecco 2014Giorgis MA, Tecco PA. 2014. Árboles y arbustos invasores de la provincia de Córdoba (Argentina): una contribución a la sistematización de bases de datos globales. Boletín de la Sociedad Argentina de Botánica 49: 581-603.). It was found that the most frequent applications are those associated with health and medicine fields (108 uses/88 species). These are important medical resources since they are often used in home orchards and gardens, which gives the peasant population access to them as well as the easy availability of adventitious species of wide dissemination. Following the medicinal plants we can find those used for construction, technologies and material culture (36 uses/15 species), fuel species (23 uses/17 species), ecological indicators and environmental services (such as shade and water indicator) (17 uses/9 species), followed by other uses.
Absolute frequency of the amount of species and ethnobotanical uses of exotic species (both adventitious and cultivated).
As stated by Santos et al. (2014Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.), herbaceous species can complement the stock of local wild vegetable species, which often represent critical elements in local nutritional profiles, and even fulfill the therapeutic needs that native medical plants cannot meet. In this sense, Santos et al. (2014)Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300. argue that it is wrong to assume that invasive species only have a completely negative biocultural influence because, in some cases, these species are more useful than non-invasive species. They have many uses, but the main focus is often on the category of forage for animals and medications (Stepp & Moerman 2001Stepp JR, Moerman DE. 2001. The importance of weeds in ethnopharmacology. Journal of Ethnopharmacology 75: 25-31.; Vieyra-Odilon & Vibrans 2001Vieyra-Odilon L, Vibrans H. 2001. Weeds as crops. The value of maize weeds in the valley of Toluca, Mexico. Economic Botany 52: 426-443.; Stepp 2004Stepp JR. 2004. The role of weeds as sources of pharmaceuticals. Journal of Ethnopharmacology 92: 163-166; Blanckaert et al. 2007Blanckaert I, Vancraeynest K, Swennen RL, Espinosa-García FJ, Piñero D, Lira-Saade R. 2007. Non-crop resources and the role of indigenous knowledge in semi-arid production of Mexico. Agriculture. Ecosystems and Environment 119: 39-48.). By comparing our records with others, in a study on non-native and invasive plants in India, Wagh & Jain (2018Wagh VV, Jain AK. 2018. Status of ethnobotanical invasive plants in western Madhya Pradesh, India. South African. Journal of Botany 114: 171-180.) documented 102 medicinal plants belonging to 38 botanical families, with assessment values similar to ours. Furthermore, Das & Duarah (2013Das K, Duarah P. 2013. Invasive alien plant species in the roadside areas of Jorhat, Assam: Their harmful effects and beneficial uses. International Journal of Engineering Research and Applications 3: 353-358.) kept a record of 18 medical and invasive species for another region in this country. Blanckaert et al. (2007)Blanckaert I, Vancraeynest K, Swennen RL, Espinosa-García FJ, Piñero D, Lira-Saade R. 2007. Non-crop resources and the role of indigenous knowledge in semi-arid production of Mexico. Agriculture. Ecosystems and Environment 119: 39-48. reported that, in Mexico, 91.9 % of invasive plants in a region keep one or more uses, forage being the most relevant one, followed by medicinal, food and ornamental uses.
As shown in the preceding examples, these species, in addition to bringing benefits to the inhabitants, can affect environmental systems on a temporal and spatial scale. However, the assessment made by villagers can be found on a scale of individuals, that is, they value species that grow in their peridomestic zones, which they make use of given that such species are not perceived as troublesome. In this sense, we consider that the control of invasive and non-native species must weigh both time and landscape scales. This implies establishing a reference line for management purposes, which will depend on the capacity of multiplication and the invasiveness of each species through a detailed study of all species.
Most common uses distribution
Figure 2 shows that in the ranking of non-native species with the most applications, there are some that include various medical uses, such as Ruta chalepensis (ruda), Malva sylvestris/M: parviflora (malva) and Plantago major (llantén), along with other species from the Old World with wide domestic medicine dissemination, such as Marrubium vulgare (marrubio), Rosmarinus officinalis (romero), Aloe spp. (aloe) and Artemisia absinthium (ajenjo). All of them, except for Aloe spp. and Rosmarinus officinalis, are also listed as invasive species.
It has been noted that species considered as “plagues” or “invasive” from an ecological viewpoint are important for a number of non-medical uses, such as Melia azedarach (paraíso) as a natural insecticide, Ligustrum lucidum (siempreverde) for shade, Gleditsia triacanthos (acacia negra) as fodder and for shade, Robinia pseudoacacia (acacia, acacia blanca) for poles and for its edible flowers, and Salix fragilis (mimbre) for shade. Among these species, Melia azederach, Gleditsia triacanthos, Robinia pseudoacacia and Ligustrum lucidum have been cited as invasive and problematic flora in Córdoba by Furey et al. (2013Furey C, Tecco PA, Perez-Harguindeguy N, Giorgis MA, Grossi M. 2013. The importance of native and exotic plant identity and dominance on decomposition patterns in mountain woodlands of central Argentina. Acta Oecologica 54: 13-20.), Ferreras et al. (2014Ferreras AE, Funes G, Galetto L. 2014. Evaluación interanual de las estrategias regenerativas de la especie exótica invasora Gleditsia triacanthos en relación a la nativa Acacia aroma en el bosque chaqueño serrano de Córdoba (Argentina). Bosque 35: 155-162.), Giorgis & Tecco (2014Giorgis MA, Tecco PA. 2014. Árboles y arbustos invasores de la provincia de Córdoba (Argentina): una contribución a la sistematización de bases de datos globales. Boletín de la Sociedad Argentina de Botánica 49: 581-603.) and Ferreras et al. (2015)Ferreras AE, Giorgis MA, Tecco PA, Cabido M, Funes G. 2015. Impact of Ligustrum lucidum on the soil seed bank in invaded subtropical seasonally dry woodlands (Córdoba, Argentina). Biological Invasions 17: 3547 - 3561.. Moreover, Plantago major (llantén) has been classified as invasive for meadows in Argentina by Busso et al. (2013Busso CA, Bentivegna DJ, Fernández OA. 2013. A review on invasive plants in rangelands of Argentina. Interciência 38: 95-103.).
In particular, the tree called Gleditsia triacanthos (Fabaceae) is the most frequent invader of invaded environments in Chaco Serrano of Córdoba (Giorgis et al. 2011bGiorgis MA, Tecco PA, Cingolani AM, Renison D, Marcora P, Paiaro V. 2011b. Factors associated with woody alien species distribution in a newly invaded mountain system of central Argentina. Biological Invasions 13: 1423-1434.), where it tampers with the floristic composition and processes associated with the nutrient cycle. The non-native firewood species that spread out and create monospecific vegetation patches are Ligustrum lucidum, Gleditsia triacanthos and Pinus spp. (Gavier & Bucher 2004Gavier G, Bucher EH. 2004. Deforestación de las Sierras Chicas de Córdoba (Argentina) en el periodo 1970-1997. Academia Nacional de Ciencias Miscelánea 101: 1-27.; Hoyos et al. 2010Hoyos LE, Gavier-Pizarro GI, Kuemmerle T, et al. 2010. Invasion of glossy privet (Ligustrum lucidum) and native forest loss in the Sierras Chicas of Córdoba, Argentina. Biological Invasions 12: 3261-3275; Giorgis et al. 2011aGiorgis MA, Cingolani F, Chiarini J, et al. 2011a. Composición florística del Bosque Chaqueño Serrano de la provincia de Córdoba, Argentina. Kurtziana 36: 9-43.; bGiorgis MA, Tecco PA, Cingolani AM, Renison D, Marcora P, Paiaro V. 2011b. Factors associated with woody alien species distribution in a newly invaded mountain system of central Argentina. Biological Invasions 13: 1423-1434.; Gavier-Pizarro et al. 2012Gavier-Pizarro GI, Kuemmerle T, Hoyos LE, et al. 2012. Monitoring the invasion of an exotic tree (Ligustrum lucidum) from 1983 to 2006 with Landsat TM/ETM + satellite data and Support Vector Machines in Córdoba, Argentina. Remote Sensing of Environment 122: 134-145.).
In contrast, from an ethnobotanical view, and as an example, peasants use the fruit of Melia azederach as a pediculicide, and as a forage for herds. It is valued as it provides environmental services, such as shade; it is used as wood for constructions; and it makes good firewood when no other choices are available. Likewise, Ligustrum lucidum and Gleditsia triacanthos are used as forage for livestock, in constructions and as firewood.
Relative importance
Table 2 shows, in descending order, the non-native species in the areas under study based on their Relative Importance. Three species considered as invasive are first on the list: Melia azederach, Salix fragilis and Ligustrum lucidum. These species feature multiple uses in different ethnobotanical areas. Other invasive species that also have considerable RI values for peasants include Morus alba var. alba, Ruta chalepensis, Malva parviflora / M. sylvestirs, Gleditsia triacanthos, Robinia pseuo-acacia, Salix babylonica and Pyracantha angustifolia, which is indicative of variety of uses. Other species with high RI values include plants with several medicinal uses, such as Ruta chalepensis and Malva spp., already mentioned within the species most frequently used.
Organization of exotic and invasive species based on their Relative Importance (RI) in descending order. [(*) Invasive species according to the National System of Information on Exotic and Invasive Species (Sistema Nacional de Información de Especies Exóticas Invasoras 2018Sistema Nacional de Información sobre Especies Exóticas Invasoras. 2018. http://www.inbiar.uns.edu.ar/ . 14 Aug. 2018.
http://www.inbiar.uns.edu.ar/... )].
A thorough floristic survey of the vegetable communities of Chaco Serrano of Córdoba, which included forests in different stages of succession, meadows and damaged shrublands, found Gleditsia triacanthos and Ligustrum lucidum as the major invasive species (Giorgis et al. 2011aGiorgis MA, Cingolani F, Chiarini J, et al. 2011a. Composición florística del Bosque Chaqueño Serrano de la provincia de Córdoba, Argentina. Kurtziana 36: 9-43.; bGiorgis MA, Tecco PA, Cingolani AM, Renison D, Marcora P, Paiaro V. 2011b. Factors associated with woody alien species distribution in a newly invaded mountain system of central Argentina. Biological Invasions 13: 1423-1434.). Moreover, the studies on Pyracantha angustifolia indicate that the plant coverage of this species includes even richer native and non-native species in comparison with other treatments, such as that of native Condalia montana or other shrubs, with Ligustrum lucidum as the one that was recruited with the most abundance and density in its area of coverage (Tecco et al. 2006Tecco PA, Gurvich DE, Díaz S, Pérez Harguindeguy N, Cabido M. 2006. Positive interactions between invasive plants: The influence of Pyracantha angustifolia on the recruitment of native and exotic woody species. Austral Ecology 31: 293-300. ). This species gains ethnobotanical importance because of its ornamental use and the ecosystemic services that it provides, such as shade and food for birds.
Perception on species control
After conducting an in-depth analysis of the species that have been mentioned the most as for their control/elimination (Fig. 3), three groups of species can be found: Species such as Pyracantha and Crataegus spp. (Crataegus) valued negatively by foreign population and other actors; Species such as Melia azedarach (paraíso) and Ligustrum lucidum (siempreverde) for which the need for control/elimination has been more emphasized by foreign population and other actors than by the native population, who shows more acceptance towards these species. Melia azedarach was the first species on the list, based on Relative Importance, given the wide range of applications by the population; Species for which control is suggested in the same manner or exclusively by small-scale native producers, such as Eucalyptus spp. (eucaliptus), Gleditsia triacanthos (acacia negra), and native species, Acacia aroma (tusca), considered as disturbing for livestock activities due to its thorns.
Species that have been mentioned the most as for their “control/elimination” according to the perception of various local actors.
These differences perceived by the population was pointed out by Bardsley & Edwards-Jones (2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.), who compared perception of the benefits of invasive species among ecologists and non-ecologists.
Perception and overall assessment of species mentioned as for control/elimination, considering assessment of their importance and conservation.
Figure 4 shows, on the left, the negative characteristics related to the need for control/elimination species; positive assessment related to species the importance and conservation of species is shown on the right. The lighter shades refer to non-native species; the darker shades denote native species with negative perceptions and for which control is deemed necessary.
Assessment of exotic and native species based on interviews of environmental perception for importance/conservation/retraction (positive assessment) and control/elimination (negative assessment). (n=44 only spp. was presented with two mentions; native species with darker tones).
Based on the perception interview related to the species that should be controlled or eliminated in the area, we can find: Some non-native species for which assessment was only negative (Ulmus sp., Pyracantha spp. /Crataegus and Gleditsia triacanthos); Non-native species that, according to interviewees, had a negative assessment. Yet, in other cases, they were considered important/relevant for conservation (Melia azederach, Ligustrum lucidum). In some other cases, they were assessed mainly positively (Pinus spp., Morus spp., Eucalyptus spp.); Although native species were assessed mostly positively, they were considered as being potentially “invasive” as they hinder (in general, due to its thorns) the development of subsistence activities (livestock, access to the field), as in the case of Acacia aroma, Celtis ehrenbergiana and Condalia buxifolia.
From these results, the notion that peasants have of “invasive”, not always in agreement with the definition provided by the Convention of Biological Biodiversity, also includes native species that hinder daily household activities, such as those mentioned above. In this sense, the biological invasion by non-native species must be evaluated not only from the perspective of ecological sustainability, but also from a socio-environmental viewpoint/angle, for which a cost-benefit relation must be valued (Bardsley & Edwards-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.). Finally, we agree with Santos et al. (2014Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.) who argue that it is wrong to assume that invasive species have a completely negative biocultural influence, since in some cases these species are more useful than non-invasive ones.
Perception of landscapes that various social actors would eliminate or change.
The preference criteria of various actors in relation to the landscapes that they would eliminate were identified by means of correspondence analysis (Fig. 5). Such analysis was conducted by asking about the immediate environment that these actors would change or eliminate, showing it as a tool for ecological control. To this end, photographic material was used as visual stimulus, including 8 pictures of environments that differ in their floristic, physiognomic-visual composition, involving: native vegetation (Nt), such as Chaco Serrano (Nt-ChS1, Nt-ChS2) Espinal (Nt-Espn) and environments with silvopastoral systems (Nt-Mnj); exotic vegetation (Ex), such as gardens cultivated (Ex-Clt); and mixed vegetation (Mx), such as Chaco Serrano with pine trees (Mx-ChSpin) and grasslands with pine trees (Mx-Ptzpin1, Mx-Ptzpin2). The idea of adding this was to obtain not only the interviewees’ opinion, but also their criteria and perceptual styles, including interpretative aspects (such as sensitivity, visual impact, taste and preference). The BiPlot shows the difference in preference for the elimination of certain landscapes among the three types of actors, since each is in a different quadrant. Therefore, regarding axis 1, a contrast can be found between the profile of the native population (PN) and the profile of subsistence (S), on the one hand, and between the profile of the foreign population (PF) and the profile of the capitalized one (C), on the other. Whereas the former group shows preference for not eliminating any landscape or for eliminating native landscapes with Espinal (Nt.Espn), the latter group prefers eliminating the exotic cultivated landscapes (ExClt). Furthermore, the other actors prefer/favor eliminating mixed landscapes, such as meadows with pine trees (Mx. Ptzpin2). These differences are probably attributed to the different perception styles, according to the socioeconomic and cultural situation and the role played by the different social actors. The native population is more dependent on resources for subsistence, while the other actors perceive nature from a conservationist perspective. This indicates that there is not a unique and clear-cut way of thinking about what is non-native, invasive or what needs to be eliminated or controlled. This goes in line with research reported by Cáceres et al. (2015Cáceres DM, Tapella E, Quétier F, Díaz S. 2015. The social value of biodiversity and ecosystem services from the perspectives of different social actors. Ecology and Society 20(1): 62 doi: 10.5751/ES-07297-200162
https://doi.org/10.5751/ES-07297-200162...
) who, by means of an iterative method, showed how various social actors perceived the provision of several ecosystemic services in different natural or changed environments.
BiPlot of the Multiple Correspondence Analysis between images (visual stimulus) of environmental change or elimination (blue shapes), type of social actors (red shapes), and socioeconomic profile (yellow shape). (Inertia or Chi-square value divided; grand total of contingency table: 52%; : Native vegetation (Nt), such as Chaco Serrano (Nt-ChS1, Nt-ChS2) Espinal (Nt-Espn), and environments with silvopastoral systems (Nt-Mnj); exotic vegetation (Ex), such as gardens that are cultivated (Ex-Clt); and Chaco Serrano with pine trees (Mx-ChSpin) and grasslands with pine trees (Mx-Ptzpin1, Mx-Ptzpin2).
Analysis of local accounts and narratives
The perception of non-native and “invasive” plants-a category often used in the field of ecology-is not consistent among actors, which is also the case when dealing with other topics, divergences and tensions as regards academic discourse.
Among the native population of the Bamba reserve, for instance, it is not unusual to refer to or assess some non-native or “invasive” plants in term of the ecosystemic services that they provide.
The species that are highly valued include those that give shade-Melia azedarach (paraíso), Morus alba var. alba (mora), Broussonetia papyrifera (mora turca)-, resources for material culture (rods), forage-Ligustrum lucidum (siempreverde) and Melia azedarach (paraíso), and even ornaments-Pyracantha angustifolia (crataegus), whose segments are multiplied in gardens with fences or “decorations”. Moreover, after having delved into this topic, some members of the peasant population stated that some autochthonous species are invasive or need to be controlled, including Acacia aroma (tusca) or Acacia caven (espinillo), tampering with livestock activities. We agree with Santos et al. (2014Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.) in their categories for native plants, including both non-native and native species.
“I think that the reason why so many native plants are dying out, such as “peperina” or even “tomillo”, is because they are being attacked by several types of little ants. It doesn’t seem to be real, but the truth is that these plants are telling us that something is wrong and that, quite on the contrary, I believe that plants, such as” paraíso”, invasive plants as they are called, “mora”, “siempreverde”; look around, G., look around! Imagine if this “paraíso” or these “mora” plants were not here, check out those invasive plants, what would there be? This would be a wasteland. So, for me this whole thing about the invasive plants, and the stupid fights that come up are not important, because there are some nature protectors that say that the plants this and that... when it comes to afforestation and what not. I think that they should start by finding out why these native plants are dying out! If you analyze this situation thoroughly, even the “mora” plant has its attacker, did you know that? (L., other actors Pje. Boca Dos, Bamba Natural Water Reserve [RHNB])
“Paraíso is extremely strong to be used as a post.” (P., member of the native population in Villa El Diquecito, RHNB)
"Can you guess which plants have spread out? Espinillo and tusca, because, in the past, there were chivas, the chivas themselves have sowed them, they eat algarrobo tree, broad chaucha (the fruit). Walking in the field is nearly impossible." (R., member of the native population, El Diquecito, RHNB)
“A plant that really thrived was the “siempreverde”. It’s useful for the birds, they have leaves all year round, the little gray dove uses it as shelter, the song thrush takes the “grateus”, it can become a nice ornament; it is useful to build fences.” (H., member of the foreign population, El Diquecito, RHNB)
The previous statements can be compared with those uttered by the population in La Rancherita, mostly members of the foreign population, whose socioeconomic profile includes more schooling years. With a discourse closer to that of academic ecology, the interviewees highlight the difference between what is native and what is non-native, and they show their differential impact in relation to water consumption as well as how important it is to control the latter to avoid biological invasions. This is indicated through the results from the participation workshops held in the community. Such results reveal that the spreading of Iris pseudoacorus (lirio amarillo) on the stream bed, or the propagation the species Pyracantha spp., Crataegus sp. and Gleditsia triacanthos has been cumbersome. During these workshops, control and elimination measures to deal with some of them were put forth.
“To get firewood, I make use of plants that are not from here-paraíso, olmos, acacia negra, which are plagues...” (H., La Rancherita)
“There are rushes and weeds that retain a lot of water, mud is formed.... that place was beautiful. It should be restored for touristic purposes. When you leave the reserve and you get to the bed stream, you can head for the water fall and walk past all native plants first, and then you can walk past all the non-native ones. It’s a wholly different landscape! It’s full of water flags, honey locusts and rushes. And it’s full of this one with small red balls (crataegus). You cannot move forward, there are times when you cannot see the river. There are some animals in that area, but the number is decreasing more and more, since there is no water for them. In Villa Giardino, the yellow flag (lirio amarillo) is used for water improvement, but here it’s out of control. The non-native plants should be controlled, and changed little by little." (Participants of the 3rd. “Rooting Paravachasca” Workshop, La Rancherita)
Based on the narratives, “invasive” species are those that “are not from here”, “spread out” or “thrive”, showing the predominance of both a phytogeographic and an ecological abundance criteria. By contrast, narratives make no reference to reproductive mechanisms or propagation form of species, key to understand invasions (Giorgis et al. 2016Giorgis MA, Cingolani AM, Tecco PA, Cabido M, Poca M, Wehrden H. 2016. Testing alien plant distribution and habitat invasibility in mountain ecosystems: growth form matters biological invasions. Biological Invasions 18: 2017-2028.). This indicates that local perceptions on an ecological phenomenon, such as invasions, do not go in line with the academic ecology criterion. In order to gain local support, it is vital that environmental scientists do not discredit all non-native species that could potentially be invasive, but rather, the focus should be on programs and policies on the study of individual species that cause the most negative social and ecological impact (Bardsley & Edwards-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.). Many studies (including DiTomaso 2000DiTomaso J. 2000. Invasive weeds in rangelands: Species, impacts, and management. Weed Science 48: 255-265. and Bardsley & Edwards-Jones 2006Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.) suggest that the strategic holistic plans should be developed carefully with key species and ecosystems, applying techniques of mechanical, cultural, biological, and chemical management. Moreover, the estimates used to evaluate underlying factors in attitudes, such as perceptions or individual assessment, may help understand public viewpoints. They may also help facilitate the development of environmental management policies and the application of communication strategies mo4re widely accepted by the public sphere in conservation biology (Fischer & Wal 2007Fischer A, Wal R. 2007. Invasive plant suppresses charismatic seabird- the construction of attitudes towards biodiversity management options. Biological Conservation 135: 256-267.).
Conclusions
Despite the negative features that have been widely advertised (for example, rivalry with native species, which damages either plants for cultivation or productivity), “invasive weeds” help enrich the local species reserve that are useful in rural areas and some urban communities. In this study, it has been shown that local populations value both non-native and invasive species due to their relevance. The use spectrum of some non-native species contributes to their adaptation to the cultural use of species based on resource availability, not necessarily associated with preference (such as firewood obtained from “paraíso” or forage obtained from “acacia negra”). It should also be noted that the use of non-native invasive plants may be useful to traditional medicine, a need that autochthonous plants alone would have fulfilled before.
Additionally, it should be pointed out that local actors perceived non-native and invasive species differently, with significant divergences regarding their ecological knowledge. This should be taken into account during the development of management and conservation guidelines.
It is also worth mentioning considering species individually for the management and control plans of “invasive species”, and special emphasis should be placed on those of interest and which are ethnobotanically relevant in terms of how useful they are for the peasant population when it comes to material culture, household activities and ecological services.
As regards invasive species, the narratives highlight that, although they have a significant impact on landscapes, ecosystems and biodiversity levels, not only negative impacts are included, since non-native invasive species provide the local population with both costs and benefits. As a result of the growth of these species, they can be the basis for many systems that could help manage economically relevant resources, such as agriculture, horticulture, afforestation and ornamental or garden landscapes. In spite of the economic importance of many invasive species, most research studies published so far have dealt with the biological aspects of invasions. However, this is not enough, seeing that the invasion processes of non-native species are a natural and social process. Indeed, individuals make choices increasing or decreasing the likelihood of a species becoming invasive. Our results show precisely how important it is to consider the social side of these processes. In sum, it is necessary/needed to deconstruct and resignify the use of the terms “non-native” and “invasive” in conservation practices while taking into account the diversity of local actors and the social side of invasiveness.
As for the different actors, the PNs do not perceive issues which come from complex ecological processes or ecosystemic changes, and which do not tamper with production activities conspicuously (for example, non-native and invasive species). Concerning biological invasions, the idea of “invasive/invader” applies both to non-native flora and native species that tamper with household activities (livestock breeding, collection in forests). It is not unusual for some species listed in academic contexts as “invasive” to have a fairly positive assessment due to their ethnobotanical applications (shade, enjoyment, ornamentation). By contrast, PNS and OAs perceive environmental issues associated with ecological processes that are not always conspicuous (as in the relation between water shortage and the spreading of invasive species, such as Melia azedarach, Gleditsia triacanthos and Pyracantha spp.).
Acknowledgements
We would like to thank IDACOR and Museum of Anthropology. We are also grateful to the Herbarium of the Botanical Museum (CORD). We are particularly thankful to the native people of the region who, with extraordinary generosity, have informed about the topic of the research. The authors would like to thank Lic. Carolina Mosconi for helping with the English version and to Dra. Melisa Girogis for the advices. This work was supported through a research grant from CONICET and ANPCyT/FONCyT project, Pict 1001 and 1633.
References
- Alexiades MN. 1996. Standard techniques for collecting and preparing herbarium species. In: Alexiades MN. (ed.) Selected guidelines for ethnobotanical research: A field manual. New York, Botanical Garden Bronx. p. 99-126.
- Albuquerque UP, Lucena RFP, Alencar NL. 2010. Métodos e técnicas para a coleta de dados etnobiologicos. In: Albuquerque UP, Lucena RFP, Cunha LVF. (eds.) Métodos e técnicas na pesquisa etnobiologica e etnoecologica. Recife, NUPEEA. p. 39-63.
- Arenas P, Martínez GJ. 2012. Estudio etnobotánico en zonas áridas y semiáridas de Argentina y zonas limítrofes. Experiencias y reflexiones metodológicas de un grupo de investigación. In: Arenas P. (ed.) Etnobotánica en zonas áridas y semiáridas del Cono Sur de Sudamérica. Buenos Aires, CEFIBO-CONICET. p. 11-43.
- Bardsley D, Edward-Jones G. 2006. Stakeholders’ perceptions of the impacts of invasive exotic plant species in the Mediterranean región. GeoJournal 65: 199-210.
- Barri F. 2012. Territorio, percepción ambiental e intereses en conflicto: El caso de la Reserva Vaquerías. PhD Thesis, Universidad Nacional de Córdoba, Córdoba.
- Bennett BC, Prance GT. 2000. Introduced plants in the indigenous Pharmacopoeia of Northern South America. Economic Botany 54: 90-102.
- Berkes F. 2011. Sacred ecology: traditional ecological knowledge and resource management. New York, Taylor y Francis.
- Blanckaert I, Vancraeynest K, Swennen RL, Espinosa-García FJ, Piñero D, Lira-Saade R. 2007. Non-crop resources and the role of indigenous knowledge in semi-arid production of Mexico. Agriculture. Ecosystems and Environment 119: 39-48.
- Busso CA, Bentivegna DJ, Fernández OA. 2013. A review on invasive plants in rangelands of Argentina. Interciência 38: 95-103.
- Cabrera AL. 1994. Regiones fitogeográficas argentinas. Vol. II Enciclopedia Argentina de Agricultura y Jardinería. Fascicle 1. Buenos Aires, Acme S.A.C.I.
- Cáceres DM, Tapella E, Quétier F, Díaz S. 2015. The social value of biodiversity and ecosystem services from the perspectives of different social actors. Ecology and Society 20(1): 62 doi: 10.5751/ES-07297-200162
» https://doi.org/10.5751/ES-07297-200162 - Cantero JJ, Barboza GE, Chiarini FE, et al 2016. Novedades para la flora de la Argentina. Boletín de la Sociedad Argentina de Botánica 51: 183-207.
- Das K, Duarah P. 2013. Invasive alien plant species in the roadside areas of Jorhat, Assam: Their harmful effects and beneficial uses. International Journal of Engineering Research and Applications 3: 353-358.
- Díaz-Betancourt M, Ghermandi L, Ladio A, López-Moreno IR, Raffaele E, Rapoport EH. 1999. Weeds as a source for human consumption. A comparison between tropical and temperate Latin America. Revista de Biología Tropical 47: 329-338.
- DiTomaso J. 2000. Invasive weeds in rangelands: Species, impacts, and management. Weed Science 48: 255-265.
- Di Rienzo JA, Balzarini M, Gonzalez L, et al 2017. InfoStat software estadístico. Versión 2017. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar 10 Jul. 2018.
» http://www.infostat.com.ar - Ferreras AE, Funes G, Galetto L. 2014. Evaluación interanual de las estrategias regenerativas de la especie exótica invasora Gleditsia triacanthos en relación a la nativa Acacia aroma en el bosque chaqueño serrano de Córdoba (Argentina). Bosque 35: 155-162.
- Ferreras AE, Giorgis MA, Tecco PA, Cabido M, Funes G. 2015. Impact of Ligustrum lucidum on the soil seed bank in invaded subtropical seasonally dry woodlands (Córdoba, Argentina). Biological Invasions 17: 3547 - 3561.
- Fischer A, Wal R. 2007. Invasive plant suppresses charismatic seabird- the construction of attitudes towards biodiversity management options. Biological Conservation 135: 256-267.
- Furey C, Tecco PA, Perez-Harguindeguy N, Giorgis MA, Grossi M. 2013. The importance of native and exotic plant identity and dominance on decomposition patterns in mountain woodlands of central Argentina. Acta Oecologica 54: 13-20.
- Gavier G, Bucher EH. 2004. Deforestación de las Sierras Chicas de Córdoba (Argentina) en el periodo 1970-1997. Academia Nacional de Ciencias Miscelánea 101: 1-27.
- Gavier-Pizarro GI, Kuemmerle T, Hoyos LE, et al 2012. Monitoring the invasion of an exotic tree (Ligustrum lucidum) from 1983 to 2006 with Landsat TM/ETM + satellite data and Support Vector Machines in Córdoba, Argentina. Remote Sensing of Environment 122: 134-145.
- Giorgis MA, Cingolani F, Chiarini J, et al 2011a. Composición florística del Bosque Chaqueño Serrano de la provincia de Córdoba, Argentina. Kurtziana 36: 9-43.
- Giorgis MA, Tecco PA. 2014. Árboles y arbustos invasores de la provincia de Córdoba (Argentina): una contribución a la sistematización de bases de datos globales. Boletín de la Sociedad Argentina de Botánica 49: 581-603.
- Giorgis MA, Tecco PA, Cingolani AM, Renison D, Marcora P, Paiaro V. 2011b. Factors associated with woody alien species distribution in a newly invaded mountain system of central Argentina. Biological Invasions 13: 1423-1434.
- Giorgis MA, Cingolani AM, Tecco PA, Cabido M, Poca M, Wehrden H. 2016. Testing alien plant distribution and habitat invasibility in mountain ecosystems: growth form matters biological invasions. Biological Invasions 18: 2017-2028.
- Giorgis MA, Cingolani AM, Gurvich DE, et al 2017. Changes in floristic composition and physiognomy are decoupled along elevation gradients in central Argentina. Applied Vegetation Science 20: 558-571.
- Guber R. 2005. El salvaje metropolitano, Reconstrucción del conocimiento social en el trabajo de campo. Buenos Aires, Paidos.
- Hoyos LE, Gavier-Pizarro GI, Kuemmerle T, et al 2010. Invasion of glossy privet (Ligustrum lucidum) and native forest loss in the Sierras Chicas of Córdoba, Argentina. Biological Invasions 12: 3261-3275
- Hurrell J, Deluchi G. 2013. Aportes de la etnobotánica al estudio de las invasiones biológicas. Casos en la Región Rioplatense (Argentina). Historia Natural 3: 61-76.
- Mack RN, Simberloff D, Lonsdale WM, Evans H, Clout M, Bazzaz FA. 2000. Biotic invasions causes, epidemiology, global consequences, and control. Ecological Applications 3: 689-710.
- Maema LP, Potgieter M, Mahlo SM. 2016. Invasive alien plant species used for the treatment of various diseases in Limpopo province, South Africa. African Journal of Traditional, Complementary and Alternative Medicine 13: 223-231.
- Martínez GJ. 2015. Cultural patterns of firewood use as a tool for conservation: A study of multiple perceptions in a semiarid region of Cordoba, Central Argentina. Journal of Arid Environments 121: 84: 99.
- Martínez GJ, Fernández M, Flores C, et al 2016. Diagnóstico participativo, orientaciones y aportes para la conservación y el manejo de la reserva de usos múltiples de La Rancherita y Las Cascadas. Córdoba, IDACOR-CONICET.
- Martínez GJ, Manzano-García J. 2016. Estilos de percepción de la biodiversidad y su conservación en actores sociales de áreas protegidas de córdoba. Revista del Museo de Antropología 9: 135-152.
- Mekoya A, Oosting SJ, Fernandez-Rivera S, Zijpp AJ. 2008. Farmers’ perceptions about exotic multipurpose fodder tres and constraints to their adoption. Agroforestry Systems 73: 141-153.
- Palacios AA, Spicogna JM, Bernasconi J, et al 2010. Decidirnos a decidir. Experiencias sobre la gestación de la Unidad Reserva Natural Militar la Estancia La Calera y Reserva Hídrica y Recreativa Natural Bamba. Córdoba, Aula de Montaña de la Universidad Nacional de Córdoba.
- Rapoport EH. 2000. Remarks on the biogeography of land invasions. Revista Chilena de Historia Natural 73: 367-380.
- Rapoport EH, Marzocca A, Drausal BS. 2009. Malezas comestibles del Cono Sur y otras partes del planeta. Buenos Aires, Ediciones INTA.
- Richardson DM, Pysek P, Rejmánek M, Barbour MG, Panetta FD, West CJ. 2000. Naturalization and invasion of alien plants: concepts and definitions. Diversity and Distributions 6: 93-107.
- Richardson DM, Wilgen BW. 2004. Invasive alien plants in South Africa: How well do we understand the ecological impacts? South African Journal of Science 100: 45-52.
- Santamarina-Campos B, Bodí J. 2012. Lugares rurales versus espacios naturalizados. Conocimientos y reconocimientos en las lógicas patrimoniales de las áreas protegidas. Revista de Antropología Iberoamericana 8: 111-138.
- Santos LL, Nascimento ALB, Vieira FJ, Silva VA, Voeks R, Albuquerque UP. 2014. The cultural value of invasive species: A Case study from Semi-Arid Northeastern Brazil. Economic Botany 68: 283-300.
- Sistema Nacional de Información sobre Especies Exóticas Invasoras. 2018. http://www.inbiar.uns.edu.ar/ . 14 Aug. 2018.
» http://www.inbiar.uns.edu.ar/ - Souza OA, Rodríguez Chaves MPS, Barbosa RI, Clement CR. 2018. Local ecological knowledge concerning the invasion of Amerindian lands in the northern Brazilian Amazon by Acacia mangium (Willd.). Journal of Ethnobiology and Ethnomedicine 14:33. doi: 10.1186/s13002-018-0231-x
» https://doi.org/10.1186/s13002-018-0231-x - Stepp JR. 2004. The role of weeds as sources of pharmaceuticals. Journal of Ethnopharmacology 92: 163-166
- Stepp JR, Moerman DE. 2001. The importance of weeds in ethnopharmacology. Journal of Ethnopharmacology 75: 25-31.
- Sundaram B, Krishnan S, Hiremath AJ, Joseph G. 2012. Ecology and Impacts of the Invasive Species, Lantana camara, in a Social-Ecological System in South India: Perspectives from Local Knowledge. Human Ecology 40: 931-942.
- Svanverg I, Luczaj L, Santayana MP, Pieroni A. 2011. History and current trends of ethnobiological research in Europe. In: Anderson EN, Pearsall DM, Hunn ES, Turner NJ. (eds.) Ethnobiology. New Jersey, Wiley-Blackwell, Inc. Hoboken. p. 189-212.
- Tecco PA, Gurvich DE, Díaz S, Pérez Harguindeguy N, Cabido M. 2006. Positive interactions between invasive plants: The influence of Pyracantha angustifolia on the recruitment of native and exotic woody species. Austral Ecology 31: 293-300.
- Torrella S, Adámoli J. 2005. Situación ambiental en el Chaco Árido y Serrano. In: Brown A, Martínez Ortiz U, Acerbi M, Corchera J. (eds.) La Situación Ambiental Argentina. Buenos Aires, Fundación Vida Silvestre Argentina. p. 75-83.
- Valles M. 1999. Técnicas cualitativas de investigación social. Reflexión metodológica y práctica profesional. Madrid, Síntesis S.A.
- Vieyra-Odilon L, Vibrans H. 2001. Weeds as crops. The value of maize weeds in the valley of Toluca, Mexico. Economic Botany 52: 426-443.
- Wagh VV, Jain AK. 2018. Status of ethnobotanical invasive plants in western Madhya Pradesh, India. South African. Journal of Botany 114: 171-180.
- Wilgen B. 2012. Evidence, perceptions, and trade-offs associated with invasive alien plant control in the Table Mountain National Park, South Africa. Ecology and Society 17: 23. doi: 10.5751/ES-04590-170223
» https://doi.org/10.5751/ES-04590-170223
Publication Dates
-
Publication in this collection
19 June 2019 -
Date of issue
Apr-Jun 2019
History
-
Received
21 Sept 2018 -
Accepted
18 Feb 2019