ABSTRACT
Background:
Thermogenic activity in the brown adipose tissue (BAT) of obese individuals is reduced, and this condition may be modified by bariatric surgery (BS).
Aim:
To characterize fat deposition in BAT from hypothalamic obese (HyO) rats submitted to duodenal-jejunal-bypass (DJB) surgery.
Methods:
For induction of hypothalamic obesity, newborn male Wistar rats were treated with subcutaneous injections of monosodium glutamate (MSG). The control (CTL) group received saline solution. At 90 days, the HyO rats were submitted to DJB or sham operation, generating the HyO-DJB and HyO-SHAM groups. At 270 days, the rats were euthanized, and the BAT was weighed and submitted to histological analysis.
Results:
Compared to BAT from CTL animals, the BAT from HyO-SHAM rats displayed increased weight, hypertrophy with greater lipid accumulation and a reduction in nucleus number. DJB effectively increased nucleus number and normalized lipid deposition in the BAT of HyO-SHAM rats, similar to that observed in CTL animals.
Conclusion:
DJB surgery avoided excessive lipid deposition in the BAT of hypothalamic obese rats, suggesting that this procedure could reactivate thermogenesis in BAT, and contribute to increase energy expenditure.
HEADINGS:
Bariatric surgery; Thermogenesis; Obesity
RESUMO
Racional:
A atividade termogênica no tecido adiposo marrom (TAM) de indivíduos obesos encontra-se reduzida, condição que pode ser modificada pela cirurgia bariátrica(CB).
Objetivo:
Verificar o efeito da derivação duodeno-jejunal (DDJ) sobre a morfologia do TAM de ratos com obesidade hipotalâmica.
Métodos:
Para indução da obesidade hipotalâmica (OHi), ratos Wistar neonatos receberam injeções subcutâneas de glutamato monossódico (MSG). O grupo controle (CTL) recebeu solução salina. Aos 90 dias, os ratos OHi foram submetidos à DDJ (grupo OHi-DDJ) ou a falsa operação (grupo OHi-FO). Aos 270 dias, eles foram eutanasiados e o TAM foi pesado e submetido à análise histológica.
Resultados:
Em comparação com os animais CTL, o TAM dos ratos OHi-FO apresentou aumento do peso, hipertrofia dos adipócitos com acúmulo de lipídios e redução do número de núcleos. A DDJ reduziu a deposição de gordura e o número de núcleos no TAM de ratos OHi-DDJ em comparação com os OHi-FO, com valores similares aqueles dos animais CTL.
Conclusões:
A DDJ foi capaz de evitar a deposição excessiva de lipídios no TAM de ratos com obesidade hipotalâmica, sugerindo que a cirurgia bariátrica poderia reativar a termogênese neste tecido adiposo, contribuindo para aumentar o gasto energético.
DESCRITORES:
Cirurgia Bariátrica; Termogênese; Obesidade
INTRODUCTION
Obesity results from an imbalance between food intake and energy expenditure is regulated by complex physiological mechanisms which involve the brown adipose tissue (BAT)2828 Vosselman MJ, van Marken Lichtenbelt WD, Schrauwen P. Energy dissipation in brown adipose tissue: From mice to men. Molecular and Cellular Endocrinology 2013;379(1-2), 43-50.. BAT is primarily characterized by its multilocular adipocytes (elevated number of cytoplasmic lipid droplets), with spherical and slightly eccentric nuclei and huge contents of mitochondria, in which are found high levels of mitochondrial uncoupling protein 1 (UCP-1), responsible for the thermogenic capacity of this tissue44 Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiological Reviews 2004;84, 277-359.. Obese rodents have great fat accumulation in the BAT, with expansion of the adipocyte area and a decrease in mitochondria number, as well as in mitochondrial UCP-1 expression77 Commins SP, Watson PM, Padgett MA, Dudley A, Argyropoulos G, Gettys TW. Induction of uncoupling protein expression in brown and white adipose tissue by leptin. Endocrinology 1999; 140, 292-300.. Therefore, BAT activation may have a protective effect against obesity12, 26.
Some studies have shown that the thermogenic activity in BAT can be modulated by bariatric surgery (BS)22 Bucerius J, Vijgen GHEJ, Brans B, Bouvy ND, Bauwens M, Rudd JHF, Havekes B, Fayad ZA, van Marken Lichtenbelt WD, Mottaghy FM. Impact of Bariatric Surgery on Carotid Artery Inflammation and the Metabolic Activity in Different Adipose Tissues. Medicine 2015;94(20): e725.,1010 Hui SCN, Wong SKH, Ai Q, Yeung DKW, Ng EKW, Chu WCW. Observed changes in brown, white, hepatic and pancreatic fat after bariatric surgery: Evaluation with MRI. European Radiology 2019;29: 849.,2020 Rachid B, Van De Sande-Lee S, Rodovalho S, Folli F, Beltramini GC, Morari J, Amorim BJ, Pedro T, Ramalho AF, Bombassaro B, Tincani AJ, Chaim E, Pareja JC, Geloneze B, Ramos CD, Cendes F, Saad MJ, Velloso LA. Distinct regulation of hypothalamic and brown/beige adipose tissue activities in human obesity. International Journal of Obesity(2015).39(10), 1515-1522.,2121 Rodovalho S, Rachid B, De-Lima-Junior JC, Van De Sande-Lee S, Morari J, Carvalho HM, Amorim BJ, Tincani AJ, Chaim E, Pareja JC, Saad MJ, Folli F, Ramos CD, Geloneze B, Velloso LA. Impairment of body mass re, duction-associated activation of brown/beige adipose tissue in patients with type 2 diabetes mellitus. International Journal of Obesity2017;41(11), 1662-1668.,2727 Vijgen GHEJ, Bouvy ND, Teule GJJ, Brans B, Hoeks J, Schrauwen P, van MarkenLichtenbelt WD. Increase in Brown Adipose Tissue Activity after Weight Loss in Morbidly Obese Subjects. The Journal of Clinical Endocrinology & Metabolism 2012; 97(7):1229-1233.. The BS is usually effective for achieving weight loss and energy homeostasis reestablishment in morbidly obese patients88 Dadson P, Hannukainen JC, Din MU, Lahesmaa M, Kalliokoski KK, Iozzo P, Pihlajamäki J, Karlsson HK, Parkkola R, Salminen P, Virtanen KA, Nuutila P. Brown adipose tissue lipid metabolism in morbid obesity: Effect of bariatric surgery-induced weight loss. Diabetes, Obesity and Metabolism 2018;20(5): 1280-1288.,1313 Mango VL, Frishman WH. Physiologic, psychologic, and metabolic consequences of bariatric surgery. Cardiology Review 2006;14(5): 232-237.,1818 Nonino CB, Oliveira BAP, Chaves RCP, Silva LTP, Pinhel MAS, Ferreira FC, Rocha GC, Donadelli SP, Marchini JS, Salgado-Junior W, Nicoletti CF. Is there any change in phenotypic characteristics comparing 5 to 10 years of follow-up in obese patients undergoing Roux-en-Y gastric bypass? ABCD Arq Bras Cir Dig.; 2019;32(3):e1453,1919 Porcelli Ilma CDS, Corsi NM, Fracasso MDLC, Pascotto RC, Cardelli AAM, Poli-Frederico RC et al. Oral Health Promotion In Patients With Morbid Obesity After Gastroplasty: A Randomized Clinical Trial. ABCD, arq. bras. cir. dig. 2019;32( 2 ): e1437.,2929 Zilberstein B, Santo MA,Carvalho MH. Critical analysis of surgical treatment techniques of morbid obesity. ABCD, arq. bras. cir. dig. 2019;32,3.. The duodenal-jejunal bypass (DJB), a procedure that maintains the volume of the stomach, but avoids the passage of food through the duodenum and part of the jejunum, improves glucose and lipids homeostasis11 Bonfleur ML, Ribeiro RA, Pavanello A, Soster R, Lubaczeuski C, Cezar Faria Araujo A, Boschero A C, Balbo SL. Duodenal-Jejunal Bypass Restores Insulin Action and beta-Cell Function in Hypothalamic-Obese Rats. Obesity Surgery2014;25(4): 656-665.,55 Cantelli KR, Soares GM, Ribeiro RA, Balbo SL, Lubaczeuski C, Boschero AC, Araújo ACF, Bonfleur ML. Duodenal-jejunal bypass normalizes pancreatic islet proliferation rate and function but not hepatic steatosis in hypothalamic obese rats. Brazilian Journal of Medical and Biological Research2017;50(5).,1111 Jurowich CF, Rikkala PR, Thalheimer A, Wichelmann C, Seyfried F, Sander V, Kreissl M, Germer C-T, Koepsell H, Otto C. Duodenal-Jejunal Bypass Improves Glycemia and Decreases SGLT1-Mediated Glucose Absorption in Rats With Streptozotocin-Induced Type 2 Diabetes. Annals of Surgery 2013;258(1):89-97.,2424 Soares GM, Cantelli KR, Balbo SL, Ribeiro RA, Alegre-Maller ACP, Barbosa-Sampaio HC, Boschero AC, Araújo ACF, Bonfleur ML. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sciences2017;188, 68-75., however, the effects of DJB on BAT have never been studied yet.
To study the pathophysiological mechanisms involved in obesity, the neonatal administration of monosodium glutamate (MSG) in rodents is frequently used to induce hypothalamic lesions in these animals, resulting in obesity33 Bunyan J, Murrell EA, Shah PP. The induction of obesity in rodents by means of monosodium glutamate. British Journal of Nutrition 1976;35(01):25.,1515 Morris M, Tortelli C, Filippis A, Proietto J. Reduced BAT function as a mechanism for obesity in the hypophagic, neuropeptide Y deficient monosodium glutamate-treated rat. Regulatory Peptides 1998.75-76, 441-447.. In addition to the excessive fat accumulation and similar to the observed in obese patients, hypothalamic obesity (HyO) rodents99 HochbergI, Hochberg Z. Expanding the definition of hypothalamic obesity. Obesity Reviews 2010;11(10):709-721. display hyperinsulinemia, insulin resistance and dyslipidemia55 Cantelli KR, Soares GM, Ribeiro RA, Balbo SL, Lubaczeuski C, Boschero AC, Araújo ACF, Bonfleur ML. Duodenal-jejunal bypass normalizes pancreatic islet proliferation rate and function but not hepatic steatosis in hypothalamic obese rats. Brazilian Journal of Medical and Biological Research2017;50(5).,2424 Soares GM, Cantelli KR, Balbo SL, Ribeiro RA, Alegre-Maller ACP, Barbosa-Sampaio HC, Boschero AC, Araújo ACF, Bonfleur ML. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sciences2017;188, 68-75..Moreover, these animals present an increase in BAT mass1515 Morris M, Tortelli C, Filippis A, Proietto J. Reduced BAT function as a mechanism for obesity in the hypophagic, neuropeptide Y deficient monosodium glutamate-treated rat. Regulatory Peptides 1998.75-76, 441-447.,1717 Moss D, Ma A, Cameron DP. Defective thermoregulatory thermogenesis in monosodium glutamate-induced obesity in mice. Metabolism1985;34(7): 626-630. and lipid content, and further a reduction in thermogenesis induced by cold1717 Moss D, Ma A, Cameron DP. Defective thermoregulatory thermogenesis in monosodium glutamate-induced obesity in mice. Metabolism1985;34(7): 626-630.. As such, we sought to characterize the effects of DJB on BAT morphology using HyO rats.
METHOD
Animals
All experimental procedures were previously approved by the Unioeste’s Animal Ethics Committee (CEUA/November 15/2015). All rats were maintained under controlled luminosity (light 8:00-20:00h) and temperature (22±1° C) and had free access to rodent standard chow (BioBase, SC, Brazil) and water.
Induction of hypothalamic obesity
Male newborn Wistar rats received one subcutaneous injection per day of monosodium glutamate (4 mg/g body weight) during the first five days of life, (MSG; n=34). During the same period, another group of newborns received an equimolar solution of saline (1.25 mg/g body weight) forming rats control group (CTL, n=17)
DJB surgery
At 90 days of life, HyO rats were randomly submitted to DJB (HyO-DJB group, n=17) or sham operations (HyO-SHAM, n=17). Preoperative procedures were performed as reported by Meguid et al. (2004)1414 Meguid M. A surgical rat model of human roux-en- gastric bypass. Journal of Gastrointestinal Surgery 2004;8(5), 621-630., and the DJB surgery was executed as described by Rubino and Marescaux (2004)2222 Rubino F, Marescaux J. Effect of Duodenal-Jejunal Exclusion in a Non-obese Animal Model of Type 2 Diabetes. Annals of Surgery 2004; 239(1): 1-11.. Sham operated rats were submitted to laparotomy and had their intestines massaged without section2424 Soares GM, Cantelli KR, Balbo SL, Ribeiro RA, Alegre-Maller ACP, Barbosa-Sampaio HC, Boschero AC, Araújo ACF, Bonfleur ML. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sciences2017;188, 68-75..
Histological analysis
At the sixth month after the bariatric procedure, animals’ body weight was registered, rats were euthanized by decapitation. After laparotomy, the BAT was excised and weighed. Subsequently, BAT samples were fixed in 10% formalin for 24 h, dehydrated in alcohol, permeabilized with xylene and then embedded in Paraplast® (Sigma-Aldrich, MO, USA). Sections of 5 µm in thickness were stained with H&E. For the assay, three sections from each BAT were analyzed using a light microscope (Olympus DP71; Tokyo, Japan) with a 40X magnification lens. The Image J software (Bethesda, MD, USA) was used for image analyses. The nuclei proliferation in BAT was verified by counting the number of these ones. For this, a quadrant (501 µm) was selected and the total nuclei in each field was registered. The hypertrophy of adipocytes was evaluated by measuring the adipocytes size (µm). In addition, using the Image J software’s tool “count and measure objects”, the percentage of area occupied by nuclei and fat was evaluated. Additionally, the percentage occupied by the remaining area, which probably represented vascularization, cytosol and extracellular tissue, was calculated and denominated VCE.
Statistical analysis
Data were analyzed by one-way analysis of variance (ANOVA) followed by the Tukey post-test (p<0.05), using GraphPad Prism software (GraphPad Inc., CA, USA).
RESULTS
The body weight of the HyO-SHAM and HyO-DJB rats were approximately 25% lower than that of the CTL group (p<0.0001, Table 1). In addition, no difference in body weight was observed between the HyO-DJB and HyO-SHAM rats. Figure 1 shows the effects of DJB on BAT in HyO rats and its histological aspects. The BAT weight was 135.1% greater in HyO-SHAM animals, compared to CTL rats (p<0.0084, Figure 1d). DJB surgery did not affect the weight of this fat depot (Figure 1d) in relation to both other groups, CTL and HyO-SHAM.
Representative photomicrography of BAT (H&E 40x): a) CTL; b) HyO-SHAM; c) HyO-DJB; d) BAT weight; e) adipocyte nucleus proliferation; f) adipocyte size.
Histological analyses showed that in the BAT of the CTL group presented characteristics of multilocular adipose tissue, since adipocyte cells contained small lipid droplets of different sizes. In contrast, in HyO-SHAM animals, the cells in the BAT were expanded and displayed a higher fat content, almost ceasing to be multilocular and becoming unilocular Differently, in BAT of HyO-DJB group contained some fat droplets, but the cells were more similar to those of the CTL group. The spherical nucleus of cells is located centrally or eccentrically in all groups, despite being reduced in the HyO-SHAM BAT. In addition, the cytoplasm of the BAT cells of the CTL group appeared to contain numerous mitochondria and a rich supply of capillaries between the cells, since these regions were stained with hematoxylin (in purple). However, in the BAT of HyO-SHAM animals these regions have been reduced, while in HyO-DJB BAT it was similar to that of CTL rats.
HyO-SHAM BAT presented a reduction of 48.74% in nucleus number (Figure 1e; p<0.0001) and a larger (71.11%) adipocyte size, in relation to the BAT of CTL rats (Figure 1f; p<0.0001). Interestingly, in BAT from HyO-DJB animals, an increase of 62.16% in nucleus number was observed, when compared to BAT from the HyO-SHAM group (Figure 1e; p<0.0001), no significant differences from that observed in the CTL group. Adipocyte size in the BAT of HyO-DJB rats was similar to that observed in the BAT of CTL rats (Figure 1f).
Figure 2a demonstrates the effects of DJB surgery on the percentages of nuclei, WAT and VCE occupation per field in the BAT of CTL and HyO rats. The percentage of area occupied by nuclei in HyO-SHAM BAT was approximately 76% lower in relation to the same parameter in BAT from CTL rats (Figure 2b; p<0.0012). The percentage area of nuclei in the HyO-DJB BAT was nearly 197% higher than in the BAT of the HyO-SHAM group (p<0.0012, Figure 2b), statistically resembling the CTL group. The percentage of fat content per field in the BAT from the HyO-SHAM group was 109% and 32% higher than the fat percentage content found in the BAT from CTL and HyO-DJB groups, respectively (Figure 2c, p<0.0001). However, the area occupied by lipids in HyO-DJB BAT remained 57% greater, when compared to the fat percentage area in BAT from CTL animals (Figure 2c; p<0.0001). Consequently, the percentage of VCE area in the BAT from HyO-SHAM rats was 46.49% and 28.73% lower, respectively, in relation to the same area in BAT from CTL and HyO-DJB rats (Figure 2d; p<0.0001). HyO-DJB BAT also presented reduced percentage (25%) in the VCE area, compared to the BAT from CTL rats (p<0.0001, Figure 2d).
Representative photomicrography of BAT (H&E 40x 50.0µm scale): the columns represent % of nucleus occupation; % of WAT occupation; % of VCE, respectively. Black arrows in the first column indicate adipocyte nuclei; in the second indicate adipocyte in WAT; in the third indicate BAT.
DISCUSSION
Obesity due to lesions in the hypothalamus has negative impacts on survival and quality of life of patients and BS can represent a therapeutic alternative for this syndrome99 HochbergI, Hochberg Z. Expanding the definition of hypothalamic obesity. Obesity Reviews 2010;11(10):709-721.. Herein, using MSG obese rats to mimic hypothalamic obesity syndrome, we demonstrated that adipocytes of BAT from HyO rats lost the multilocular droplets lipids profile and presented a reduction in nucleus number and augment in fat content. For the first time, we observed that at six months after DJB operation, the BAT morphology in HyO-DJB rats returned to a similar morphology to that of BAT from CTL animals.
BAT is an important site of cold-induced non-shivering thermogenesis2828 Vosselman MJ, van Marken Lichtenbelt WD, Schrauwen P. Energy dissipation in brown adipose tissue: From mice to men. Molecular and Cellular Endocrinology 2013;379(1-2), 43-50.. The sympathetic nervous system (SNS) is responsible for activating lipolysis and fatty acid ß-oxidation in BAT. Therefore, the proton gradient generated by this process is diverted to ATP-synthase through UCP-1, and the energy generated is dissipated as heat44 Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiological Reviews 2004;84, 277-359.. Reductions in SNS activity and UCP1 expression contribute to lower energy expenditure and higher adiposity in BAT77 Commins SP, Watson PM, Padgett MA, Dudley A, Argyropoulos G, Gettys TW. Induction of uncoupling protein expression in brown and white adipose tissue by leptin. Endocrinology 1999; 140, 292-300.. HyO mice exhibited hypertrophy of BAT with an 85% increase in wet weight and lipid content and did not mobilize BAT lipids after cold exposure to 4º C for 6 h1717 Moss D, Ma A, Cameron DP. Defective thermoregulatory thermogenesis in monosodium glutamate-induced obesity in mice. Metabolism1985;34(7): 626-630.. Another study showed a reduction in GLUT44 Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiological Reviews 2004;84, 277-359. transporter levels in BAT from HyO rats1515 Morris M, Tortelli C, Filippis A, Proietto J. Reduced BAT function as a mechanism for obesity in the hypophagic, neuropeptide Y deficient monosodium glutamate-treated rat. Regulatory Peptides 1998.75-76, 441-447.. Additionally, the type II thyroxine 5-deiodinase (T2) activity in BAT from HyO mice was reduced after cold and norepinephrine stimulation2525 Tsukahara F, Uchida Y, Ohba K, Nomoto T, Muraki T. Defective Stimulation of Thyroxine 5´-Deiodinase Activity by Cold Exposure and Norepinephrine in Brown Adipose Tissue of Monosodium Glutamate-Obese Mice. Hormone and Metabolic Research 1997;29(10), 496-500.. A decrease in retroperitoneal sympathetic nerve activity and lower adrenal catecholamine stores have also been reported in HyO mice2323 Scomparin DX, Gomes RM, Grassiolli S, Rinaldi W, Martins AG, de Oliveira JC, Gravena C, de Freitas Mathias PC. Autonomic activity and glycemic homeostasis are maintained by precocious and low intensity training exercises in MSG-programmed obese mice. Endocrine2009;36(3): 510-517.. As such, modifications in BAT morphology in the obese rats, observed in the present study, may be due to the low SNS activity associated with norepinephrine stimulation reduction, which could alter the function of BAT in HyO animals.
Currently, BS is frequently used as a treatment in morbidly obese patients1313 Mango VL, Frishman WH. Physiologic, psychologic, and metabolic consequences of bariatric surgery. Cardiology Review 2006;14(5): 232-237... However, there are few studies showing the effects of BS on BAT and these reports present contrasting data regarding the surgery’s benefits22 Bucerius J, Vijgen GHEJ, Brans B, Bouvy ND, Bauwens M, Rudd JHF, Havekes B, Fayad ZA, van Marken Lichtenbelt WD, Mottaghy FM. Impact of Bariatric Surgery on Carotid Artery Inflammation and the Metabolic Activity in Different Adipose Tissues. Medicine 2015;94(20): e725.,1010 Hui SCN, Wong SKH, Ai Q, Yeung DKW, Ng EKW, Chu WCW. Observed changes in brown, white, hepatic and pancreatic fat after bariatric surgery: Evaluation with MRI. European Radiology 2019;29: 849.,2020 Rachid B, Van De Sande-Lee S, Rodovalho S, Folli F, Beltramini GC, Morari J, Amorim BJ, Pedro T, Ramalho AF, Bombassaro B, Tincani AJ, Chaim E, Pareja JC, Geloneze B, Ramos CD, Cendes F, Saad MJ, Velloso LA. Distinct regulation of hypothalamic and brown/beige adipose tissue activities in human obesity. International Journal of Obesity(2015).39(10), 1515-1522.,2121 Rodovalho S, Rachid B, De-Lima-Junior JC, Van De Sande-Lee S, Morari J, Carvalho HM, Amorim BJ, Tincani AJ, Chaim E, Pareja JC, Saad MJ, Folli F, Ramos CD, Geloneze B, Velloso LA. Impairment of body mass re, duction-associated activation of brown/beige adipose tissue in patients with type 2 diabetes mellitus. International Journal of Obesity2017;41(11), 1662-1668.,2727 Vijgen GHEJ, Bouvy ND, Teule GJJ, Brans B, Hoeks J, Schrauwen P, van MarkenLichtenbelt WD. Increase in Brown Adipose Tissue Activity after Weight Loss in Morbidly Obese Subjects. The Journal of Clinical Endocrinology & Metabolism 2012; 97(7):1229-1233.. Obese subjects showed increased non-shivering thermogenesis in BAT one year after surgery-induced weight loss, demonstrating that BAT can be recruited after bariatric procedures in humans2727 Vijgen GHEJ, Bouvy ND, Teule GJJ, Brans B, Hoeks J, Schrauwen P, van MarkenLichtenbelt WD. Increase in Brown Adipose Tissue Activity after Weight Loss in Morbidly Obese Subjects. The Journal of Clinical Endocrinology & Metabolism 2012; 97(7):1229-1233.. Additionally, BS displayed a beneficial impact on the metabolic activity of BAT in morbidly obese patients22 Bucerius J, Vijgen GHEJ, Brans B, Bouvy ND, Bauwens M, Rudd JHF, Havekes B, Fayad ZA, van Marken Lichtenbelt WD, Mottaghy FM. Impact of Bariatric Surgery on Carotid Artery Inflammation and the Metabolic Activity in Different Adipose Tissues. Medicine 2015;94(20): e725.. The increase in brown/beige adipose tissue activity related to surgery-induced weight loss occurs independently of changes in hypothalamic activity2020 Rachid B, Van De Sande-Lee S, Rodovalho S, Folli F, Beltramini GC, Morari J, Amorim BJ, Pedro T, Ramalho AF, Bombassaro B, Tincani AJ, Chaim E, Pareja JC, Geloneze B, Ramos CD, Cendes F, Saad MJ, Velloso LA. Distinct regulation of hypothalamic and brown/beige adipose tissue activities in human obesity. International Journal of Obesity(2015).39(10), 1515-1522. and BAT activity was found to be increased in obese non-diabetic and unchanged in obese diabetic subjects submitted to bariatric operation2121 Rodovalho S, Rachid B, De-Lima-Junior JC, Van De Sande-Lee S, Morari J, Carvalho HM, Amorim BJ, Tincani AJ, Chaim E, Pareja JC, Saad MJ, Folli F, Ramos CD, Geloneze B, Velloso LA. Impairment of body mass re, duction-associated activation of brown/beige adipose tissue in patients with type 2 diabetes mellitus. International Journal of Obesity2017;41(11), 1662-1668..
Mice submitted to BS by several techniques presented increased BAT thermogenesis, mediated by higher levels of growth hormone and insulin growth factor 166 Chen Y, Yang J, Nie X, Song Z, Gu Y. Effects of Bariatric Surgery on Change of Brown Adipocyte Tissue and Energy Metabolism in Obese Mice. Obesity Surgery 2018; 28, 820.. On the other hand, no significant difference was observed in BAT volume at 6 and 12 months after bariatric procedures in patients with morbid obesity1010 Hui SCN, Wong SKH, Ai Q, Yeung DKW, Ng EKW, Chu WCW. Observed changes in brown, white, hepatic and pancreatic fat after bariatric surgery: Evaluation with MRI. European Radiology 2019;29: 849..
No study has demonstrated the effects of bariatric procedure in the BAT from HyO model. This obesity disorder is caused by damage to the hypothalamus, leading to metabolic and endocrine disturbances. Traditional treatments of obesity are not effective for patients with this disturbance99 HochbergI, Hochberg Z. Expanding the definition of hypothalamic obesity. Obesity Reviews 2010;11(10):709-721.. Using MSG-treated rats as an experimental model to study HyO, our group has demonstrated that DJB surgery ameliorates glucose homeostasis and insulin sensitivity, normalizes pancreatic islet function and decreases islet-cell proliferation, as well as improving lipid profile and hepatic steatosis11 Bonfleur ML, Ribeiro RA, Pavanello A, Soster R, Lubaczeuski C, Cezar Faria Araujo A, Boschero A C, Balbo SL. Duodenal-Jejunal Bypass Restores Insulin Action and beta-Cell Function in Hypothalamic-Obese Rats. Obesity Surgery2014;25(4): 656-665.,55 Cantelli KR, Soares GM, Ribeiro RA, Balbo SL, Lubaczeuski C, Boschero AC, Araújo ACF, Bonfleur ML. Duodenal-jejunal bypass normalizes pancreatic islet proliferation rate and function but not hepatic steatosis in hypothalamic obese rats. Brazilian Journal of Medical and Biological Research2017;50(5).,2424 Soares GM, Cantelli KR, Balbo SL, Ribeiro RA, Alegre-Maller ACP, Barbosa-Sampaio HC, Boschero AC, Araújo ACF, Bonfleur ML. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sciences2017;188, 68-75.. In the present study, we contribute a little more to the understanding of the effects of DJB surgery on Hy obesity. We, herein, observed lower lipid accumulation in BAT, an increase in nucleus number and the reestablishment of the percentage of area occupied by nuclei in HyO-DJB. Taken together, these results suggest that DJB surgery in HyO animals had a proliferative effect on BAT and could elevate the thermogenic activity in this adipose tissue, probably by normalizing insulin levels and sensitivity, through ameliorating the SNS tonus.
CONCLUSION
DJB procedure in HyO rats reduces lipid accumulation and adipocyte size and increases nucleus number in BAT, suggesting reactivation of BAT thermogenesis. The morphological changes induced by DJB surgery in the BAT of obese rats reflects the enhancement of BAT metabolic capacity.
REFERENCES
-
1Bonfleur ML, Ribeiro RA, Pavanello A, Soster R, Lubaczeuski C, Cezar Faria Araujo A, Boschero A C, Balbo SL. Duodenal-Jejunal Bypass Restores Insulin Action and beta-Cell Function in Hypothalamic-Obese Rats. Obesity Surgery2014;25(4): 656-665.
-
2Bucerius J, Vijgen GHEJ, Brans B, Bouvy ND, Bauwens M, Rudd JHF, Havekes B, Fayad ZA, van Marken Lichtenbelt WD, Mottaghy FM. Impact of Bariatric Surgery on Carotid Artery Inflammation and the Metabolic Activity in Different Adipose Tissues. Medicine 2015;94(20): e725.
-
3Bunyan J, Murrell EA, Shah PP. The induction of obesity in rodents by means of monosodium glutamate. British Journal of Nutrition 1976;35(01):25.
-
4Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiological Reviews 2004;84, 277-359.
-
5Cantelli KR, Soares GM, Ribeiro RA, Balbo SL, Lubaczeuski C, Boschero AC, Araújo ACF, Bonfleur ML. Duodenal-jejunal bypass normalizes pancreatic islet proliferation rate and function but not hepatic steatosis in hypothalamic obese rats. Brazilian Journal of Medical and Biological Research2017;50(5).
-
6Chen Y, Yang J, Nie X, Song Z, Gu Y. Effects of Bariatric Surgery on Change of Brown Adipocyte Tissue and Energy Metabolism in Obese Mice. Obesity Surgery 2018; 28, 820.
-
7Commins SP, Watson PM, Padgett MA, Dudley A, Argyropoulos G, Gettys TW. Induction of uncoupling protein expression in brown and white adipose tissue by leptin. Endocrinology 1999; 140, 292-300.
-
8Dadson P, Hannukainen JC, Din MU, Lahesmaa M, Kalliokoski KK, Iozzo P, Pihlajamäki J, Karlsson HK, Parkkola R, Salminen P, Virtanen KA, Nuutila P. Brown adipose tissue lipid metabolism in morbid obesity: Effect of bariatric surgery-induced weight loss. Diabetes, Obesity and Metabolism 2018;20(5): 1280-1288.
-
9HochbergI, Hochberg Z. Expanding the definition of hypothalamic obesity. Obesity Reviews 2010;11(10):709-721.
-
10Hui SCN, Wong SKH, Ai Q, Yeung DKW, Ng EKW, Chu WCW. Observed changes in brown, white, hepatic and pancreatic fat after bariatric surgery: Evaluation with MRI. European Radiology 2019;29: 849.
-
11Jurowich CF, Rikkala PR, Thalheimer A, Wichelmann C, Seyfried F, Sander V, Kreissl M, Germer C-T, Koepsell H, Otto C. Duodenal-Jejunal Bypass Improves Glycemia and Decreases SGLT1-Mediated Glucose Absorption in Rats With Streptozotocin-Induced Type 2 Diabetes. Annals of Surgery 2013;258(1):89-97.
-
12Kopecky J, Clarke G, Enerbäck S, Spiegelman B, Kozak LP. Expression of the mitochondrial uncoupling protein gene from the aP2 gene promoter prevents genetic obesity. Journal of Clinical Investigation 1995;6, 2914-2923.
-
13Mango VL, Frishman WH. Physiologic, psychologic, and metabolic consequences of bariatric surgery. Cardiology Review 2006;14(5): 232-237.
-
14Meguid M. A surgical rat model of human roux-en- gastric bypass. Journal of Gastrointestinal Surgery 2004;8(5), 621-630.
-
15Morris M, Tortelli C, Filippis A, Proietto J. Reduced BAT function as a mechanism for obesity in the hypophagic, neuropeptide Y deficient monosodium glutamate-treated rat. Regulatory Peptides 1998.75-76, 441-447.
-
16Morrison JFB, Shehab S, Sheen R, Dhanasekaran S, Shaffiullah M, Mensah-Brown E. Sensory and autonomic nerve changes in the monosodium glutamate-treated rat: a model of type II diabetes. Experimental Physiology 2007;93(2):213-222.
-
17Moss D, Ma A, Cameron DP. Defective thermoregulatory thermogenesis in monosodium glutamate-induced obesity in mice. Metabolism1985;34(7): 626-630.
-
18Nonino CB, Oliveira BAP, Chaves RCP, Silva LTP, Pinhel MAS, Ferreira FC, Rocha GC, Donadelli SP, Marchini JS, Salgado-Junior W, Nicoletti CF. Is there any change in phenotypic characteristics comparing 5 to 10 years of follow-up in obese patients undergoing Roux-en-Y gastric bypass? ABCD Arq Bras Cir Dig.; 2019;32(3):e1453
-
19Porcelli Ilma CDS, Corsi NM, Fracasso MDLC, Pascotto RC, Cardelli AAM, Poli-Frederico RC et al. Oral Health Promotion In Patients With Morbid Obesity After Gastroplasty: A Randomized Clinical Trial. ABCD, arq. bras. cir. dig. 2019;32( 2 ): e1437.
-
20Rachid B, Van De Sande-Lee S, Rodovalho S, Folli F, Beltramini GC, Morari J, Amorim BJ, Pedro T, Ramalho AF, Bombassaro B, Tincani AJ, Chaim E, Pareja JC, Geloneze B, Ramos CD, Cendes F, Saad MJ, Velloso LA. Distinct regulation of hypothalamic and brown/beige adipose tissue activities in human obesity. International Journal of Obesity(2015).39(10), 1515-1522.
-
21Rodovalho S, Rachid B, De-Lima-Junior JC, Van De Sande-Lee S, Morari J, Carvalho HM, Amorim BJ, Tincani AJ, Chaim E, Pareja JC, Saad MJ, Folli F, Ramos CD, Geloneze B, Velloso LA. Impairment of body mass re, duction-associated activation of brown/beige adipose tissue in patients with type 2 diabetes mellitus. International Journal of Obesity2017;41(11), 1662-1668.
-
22Rubino F, Marescaux J. Effect of Duodenal-Jejunal Exclusion in a Non-obese Animal Model of Type 2 Diabetes. Annals of Surgery 2004; 239(1): 1-11.
-
23Scomparin DX, Gomes RM, Grassiolli S, Rinaldi W, Martins AG, de Oliveira JC, Gravena C, de Freitas Mathias PC. Autonomic activity and glycemic homeostasis are maintained by precocious and low intensity training exercises in MSG-programmed obese mice. Endocrine2009;36(3): 510-517.
-
24Soares GM, Cantelli KR, Balbo SL, Ribeiro RA, Alegre-Maller ACP, Barbosa-Sampaio HC, Boschero AC, Araújo ACF, Bonfleur ML. Liver steatosis in hypothalamic obese rats improves after duodeno-jejunal bypass by reduction in de novo lipogenesis pathway. Life Sciences2017;188, 68-75.
-
25Tsukahara F, Uchida Y, Ohba K, Nomoto T, Muraki T. Defective Stimulation of Thyroxine 5´-Deiodinase Activity by Cold Exposure and Norepinephrine in Brown Adipose Tissue of Monosodium Glutamate-Obese Mice. Hormone and Metabolic Research 1997;29(10), 496-500.
-
26Tupone D, Madden CJ, Morrison SF. Autonomic regulation of brown adipose tissue thermogenesis in health and disease: potential clinical applications for altering BAT thermogenesis. Frontiers in neuroscience 2014;8(14): 1-14.
-
27Vijgen GHEJ, Bouvy ND, Teule GJJ, Brans B, Hoeks J, Schrauwen P, van MarkenLichtenbelt WD. Increase in Brown Adipose Tissue Activity after Weight Loss in Morbidly Obese Subjects. The Journal of Clinical Endocrinology & Metabolism 2012; 97(7):1229-1233.
-
28Vosselman MJ, van Marken Lichtenbelt WD, Schrauwen P. Energy dissipation in brown adipose tissue: From mice to men. Molecular and Cellular Endocrinology 2013;379(1-2), 43-50.
-
29Zilberstein B, Santo MA,Carvalho MH. Critical analysis of surgical treatment techniques of morbid obesity. ABCD, arq. bras. cir. dig. 2019;32,3.
-
Financial souce:
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001 -
Mensagem central
The thermogenic activity of brown adipose tissue in obese rats may be compromised due to histomorphological changes in the tissue. However, bariatric surgery is able to prevent these changes, suggesting the reactivation of thermogenesis -
Perspectiva
Showing that bariatric surgery in the obese rats is effective to restore the function of brown adipose tissue, reducing the deposition of lipids and favoring energy expenditure, suggests that the search for treatments that promote the activation of brown adipose tissue and, consequently, thermogenesis can be a protective alternative against obesity and its comorbidities.
Publication Dates
-
Publication in this collection
08 July 2020 -
Date of issue
2020
History
-
Received
20 Aug 2019 -
Accepted
12 Nov 2019



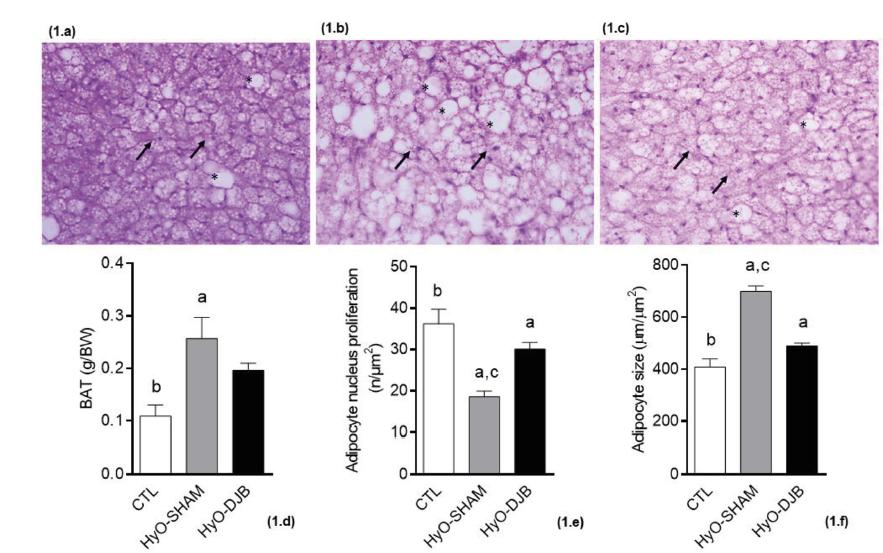 Black arrows indicate adipocyte nuclei; “*” Indicates lipiddroplets; different letters above the bars represent statistical differences between the groups. One-way ANOVA with Tukey post-test (p< 0.05, n=5-6)
Black arrows indicate adipocyte nuclei; “*” Indicates lipiddroplets; different letters above the bars represent statistical differences between the groups. One-way ANOVA with Tukey post-test (p< 0.05, n=5-6)
 Graphs=means±SEM; different letters above the bars represent statistical differences between the groups. One-way ANOVA with Tukey post-test (p< 0.05, n=5-6)
Graphs=means±SEM; different letters above the bars represent statistical differences between the groups. One-way ANOVA with Tukey post-test (p< 0.05, n=5-6)