Abstract
The monoterpenes are secondary metabolites of plants. They have various pharmacological properties including antifungal, antibacterial, antioxidant, anticancer, anti-spasmodic, hypotensive, and vasorelaxant. The purpose of this research was to review the cardiovascular effects of monoterpenes. The data in this resarch were collected using the Internet portals Pubmed, Scopus, and ISI Web of Knowledge between the years 1987 and 2010. In the study 33 monoterpenes were included, which were related to each of the thirteen individual words: artery, cardiovascular, heart, myocyte, vasorelaxant, vessel, hypotension, hypotensive, cardiomyocyte, ventricular, vasodilatory, aorta, and aortic. The research utilized 22 articles published mainly in the journals Phytomedicine, Fundamental Clinical Pharmacology, Planta Medica, Life Science, European Journal of Pharmacology, and Brazilian Journal of Medical and Biological Research. Of the 33 monoterpenes studied surveyed, sixteen of them had already been studied for their effects on the cardiovascular system: carvacrol, citronellol, p-cymene, eucalyptol (1,8-cineole), linalool, menthol, myrtenal, myrtenol, α-pinene, rotundifolone (piperitenone oxide), sobrerol, thymol, α-limonene, α-terpinen-4-ol, α-terpineol, and perillyl alcohol. The main effects observed were vasorelaxation, decreased heart rate and blood pressure. This review showed that the monoterpenes may be considered promising agents for prevention or treatment of diseases of the cardiovascular system.
cardiovascular system; essential oils; monoterpenes; review
Cardiovascular effects of monoterpenes: a review
Márcio R. V. Santos; Flávia V. Moreira; Byanka P. Fraga; Damião P. de Souza; Leonardo R. Bonjardim; Lucindo J. Quintans-Junior
Departamento de Fisiologia, Universidade Federal de Sergipe, Brazil
Correspondence Correspondence Márcio R. V. Santos Departamento de Fisiologia, Universidade Federal de Sergipe 49.100-000 São Cristóvão - SE, Brazil marciorvsantos@pq.cnpq.br Tel.: +55 79 2105 6842 Fax: +55 79 2105 6474
ABSTRACT
The monoterpenes are secondary metabolites of plants. They have various pharmacological properties including antifungal, antibacterial, antioxidant, anticancer, anti-spasmodic, hypotensive, and vasorelaxant. The purpose of this research was to review the cardiovascular effects of monoterpenes. The data in this resarch were collected using the Internet portals Pubmed, Scopus, and ISI Web of Knowledge between the years 1987 and 2010. In the study 33 monoterpenes were included, which were related to each of the thirteen individual words: artery, cardiovascular, heart, myocyte, vasorelaxant, vessel, hypotension, hypotensive, cardiomyocyte, ventricular, vasodilatory, aorta, and aortic. The research utilized 22 articles published mainly in the journals Phytomedicine, Fundamental Clinical Pharmacology, Planta Medica, Life Science, European Journal of Pharmacology, and Brazilian Journal of Medical and Biological Research. Of the 33 monoterpenes studied surveyed, sixteen of them had already been studied for their effects on the cardiovascular system: carvacrol, citronellol, p-cymene, eucalyptol (1,8-cineole), linalool, menthol, myrtenal, myrtenol, α-pinene, rotundifolone (piperitenone oxide), sobrerol, thymol, α-limonene, α-terpinen-4-ol, α-terpineol, and perillyl alcohol. The main effects observed were vasorelaxation, decreased heart rate and blood pressure. This review showed that the monoterpenes may be considered promising agents for prevention or treatment of diseases of the cardiovascular system.
Keywords: cardiovascular system, essential oils, monoterpenes, review
Introduction
It is estimated that about 80% of the world's population uses traditional medicine for its primary health care (Balick et al., 1994). Most of these therapies involve the use of plant extracts or their active compounds, such as terpenoids (Wagner & Elmadfa, 2003).
The terpenoids, also known as isoprenoids, are formed by repetition branched units of five carbons, similar to units of isoprene (Sharkey & Yeh, 2001). They are derived from mevalonic acid and sometimes called active isoprenes (Simões et al., 2004). The generalization that terpenes could be seen as composed of isoprene units became known as the isoprene rule, which investigators used to work out structures of isoprenoids (Sharkey & Yeh, 2001). However, isoprenoids are not made from isoprene. The biological precursors to the isoprenoids are isopentenyl pyrophosphate (IPP) and its isomer dimethylallyl pyrophosphate (DMAPP), sometimes called active isoprenes (Sharkey & Yeh, 2001).
The nomenclature of terpenoids depends on the number of isoprene structures and can be classified as monoterpene, sesquiterpene, diterpene, triterpene, tetraterpene, and polyterpene (Table 1). The terpenoids present cyclic or acyclic structures, which result from changes of isoprenoid chain reactions as reductions, oxidations, cyclizations (involving the formation of carbocations), ring breaks, or rearrangements (Chappell, 1995). Because of this, they are the most numerous and structurally diverse natural plant products (Zwenger & Basu, 2008).
Monoterpenes can be divided into three subgroups: acyclic (myrcene, linalool, geraniol), monocyclic (α-terpineol and terpinolene), and bicyclic (α-pinene, thujone, camphor, fenchone). In each of these subgroups, there are other classifications: unsaturated hydrocarbons (limonene), alcohols (menthol), aldehydes and ketones (myrtenal, carvone), lactones (monoterpene lactones are called iridoids, ex. nepetalactone), and tropolonas (γ-thujaplicin) (Simões et al., 2004).
Monoterpenes, as well as the sesquiterpenes and diterpenes, are secondary metabolites because they are classified as nonessential for viability; however, they mediate important interactions between plants and their environment (Chappell, 1995). Several monoterpenes are widely used in the agriculture, cosmetic, and food industries, and as a general antiseptic in medical practice (Aeschbach et al., 1994; Lee et al., 1997; Manou et al., 1998). Most of the aroma from citrus fruit oil, cherries, and mint is due to the high content of monoterpenes, but they have no nutritional value because they are exclusively vegetable (Serrano et al.,2006).
Studies have shown that monoterpenes have various pharmacological properties including antifungal, antibacterial, antioxidant, anticancer, and anti-spasmodic (Garcia et al., 2008; Kato et al., 1990; Singh et al., 2010; Karkabounas et al., 2006; Magalhães et al., 1998).
Besides the activities described above, monoterpenes also produce significant effects on the cardiovascular system, promoting, among other actions, vasorelaxation, decreased heart rate, and hypotension (Peixoto-Neves et al., 2010; Bastos et al., 2010; Aydin et al., 2007; Magalhães et al., 2008). Thus, these monoterpenes can be useful as agents for prevention and/or treatment of cardiovascular diseases (CVD).
The CVD are the lead death cause in developed and developing countries (American Heart Association, 2008; Brazilian Society of Cardiology, 2007), causing great impact not only on human health, but also in social and economic areas (Lefkowits & Willerson, 2001). In an attempt to reduce this impact, several research groups in recent decades have worked extensively to seek advances in the treatment of CVD, among them the discovery of new therapies (Lefkowits & Willerson, 2001).
Based on this and knowing that the monoterpenes are the major constituents of various essential oils of medicinal aromatic plants, most recently, pharmacological studies have focused on efforts to investigate the effects of this group of substances on the cardiovascular system. Thus, the aim of this work was to review the effects of the monoterpenes on this system.
Methods
The data in this research were collected using Pubmed, Scopus, ISI Web of Knowledge, and Scifinder portal databases. The search included articles published between 1987 and 2010 in refereed journals, which are written in English and internationally recognized. The search included 33 monoterpenes which were related to each of thirteen individual words: artery, cardiovascular, heart, myocyte, vasorelaxant, vessel, hypotension, hypotensive, cardiomyocyte, ventricular, vasodilatory, aorta, and aortic. The articles that presented results of monoterpenes added to mixtures as well as those that appeared in congress abstracts, monographs, theses, or dissertations were not considered in this review.
Results and Discussion
This study utilized seventeen articles published mainly in the journals Phytomedicine, Fundamental Clinical Pharmacology, Planta Medica, Life Science, European Journal of Pharmacology, and Brazilian Journal of Medical and Biological Research.
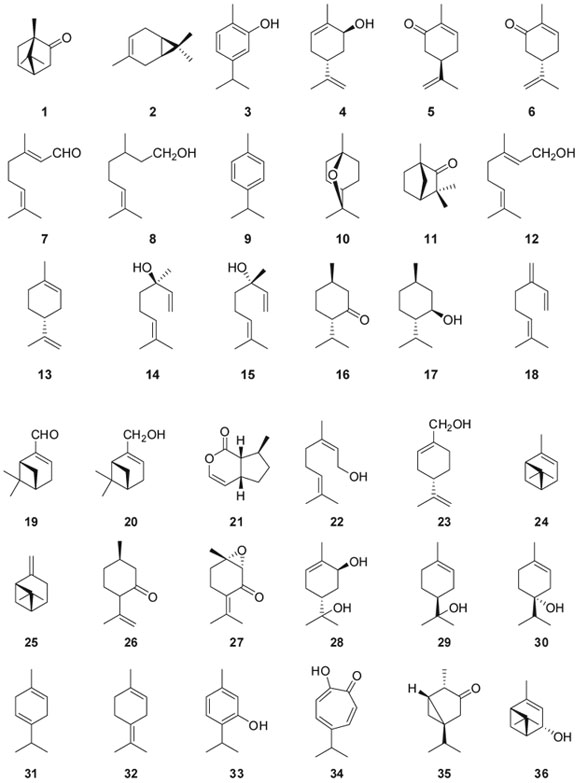
Among the 33 monoterpenes researched because of their cardiovascular activity, sixteen of them were carvacrol (3), citronellol (8), eucalyptol (1,8-cineole) (10), (-)-linalool (14), (+)-linalool (15), menthol (17), myrtenal (19), myrtenol (20), rotundifolone (piperitenone oxide) (27), sobrerol (28), thymol (33), α-limonene (13), α-terpinen-4-ol (30), α-terpineol (29), p-cymene (9), perillyl alcohol (23), α-pinene (24) and β-pinene (25). The seventeen remaining, which have not been found in any publications, were camphor (1), carene (2), carveol (4), (S)-carvone (5), (4R)-carvone (6), citral (7), fenchone (11), γ-thujaplicin (34), geraniol (12), menthone (16), myrcene (18), thujone (35), nerol (22), pulegone (26), γ-terpinene (31), terpinolene (32), nepetalactone (21), and verbenol (36). The sixteen monoterpenes and their cardiovascular effects are summarized in Table 1.
Thymol
Thymol (33) is a phenolic monoterpene carvacrol (3) isomer (Peixoto-Neves et al., 2010). Because it is often used as a general antiseptic and in medical practice, the cardiovascular effects of thymol have been extensively studied (Periago & Moezelaar, 2001). Magyar et al. (2002) showed that thymol induced cardiac arrhythmias in venticular myocytes isolated from dogs. These effects were mediated by inhibition of K+ and Ca2+ currents. Recently, Magyar et al. (2004), studying the effects of thymol in ventricular cardiomyocytes isolated from humans and canines, and using the technique of "patch clamp" in the "whole-cell" configuration, showed that thymol was able to inhibit the currents for L-type Ca2+. Moreover, the effects of thymol were also investigated in perfused guinea pig heart using the Langendorff technique, and in canine ventricular trabecula (Szentandrássy et al., 2004). These studies showed that thymol induced a cardiopressant effect caused by a reduction in the calcium content of sarcoplasmic reticulum, due mainly to the inhibition of the calcium pump (Szentandrássy et al., 2004).
Vasorelaxant effects of thymol (33) were also observed. In isolated rat aorta, Peixoto-Neves et al. (2010) demonstrated that thymol induced an endothelium-independent relaxation, possibly involving inhibition of Ca2+ release from the sarcoplasmic reticulum, reducing the sensitivity of contractile elements to Ca2+ and blocking the influx of Ca2+ across the membrane.
Carvacrol
Carvacrol (3) is a phenolic monoterpene cyclic isomer of the monoterpene 33 (Periago & Moezelaar, 2001). It is commonly used by the food and cosmetic industries as a preservative and antioxidant (Manou et al., 1998). It is present in the essential oil of oregano, which contributes approximately 65% of its composition (Earley et al., 2010). Cardiovascular effects of carvacrol were studied both in vivo and in vitro. In normotensive rats, carvacrol at a dose of 100 mg/kg (i.p.) reduced blood pressure and heart rate, and inhibited the hypertension induced by L-NAME (Aydin et al., 2007). However, in isolated rat aorta, although Aydin et al. (2007) did not observe any significant effect of carvacrol, Peixoto-Neves et al. (2010) demonstrated that this monoterpene induced an endothelium-independent relaxation, possibly involving inhibition of Ca2+ influx through the membrane. In the cerebral artery of rats, carvacrol also caused potent vasodilation, but this effect was endothelium-dependent (Earley et al., 2010). According to Earley et al. (2010), this effect was attributed to the action of carvacrol on the TRPV3 channel. This monoterpene caused an influx of Ca2+ in endothelial cells by increasing intracellular Ca2+ and leading to activation of K+ channels sensitive to Ca2+ medium (IKCa) and low (SKCa) conductance. This activation produces hyperpolarization of the plasma membrane of endothelial cells and vascular smooth muscle, thereby resulting in vasodilatation.
In studies using the technique of patch clamp in the whole-cell configuration, it was shown that, like thymol (33), carvacrol (3) was also able to inhibit the currents for L-type Ca2+ in cardiomyocytes isolated from canine and human ventricles (Magyar et al., 2004).
Eucalyptol (1,8-cineole)
Due to its pleasant aroma and spicy taste, eucalyptol (10) has been extensively used in the food industry as a flavoring and flavor enhancer (Santos & Rao, 2001). Its cardiovascular effects were studied by Lahlou et al. (2002), using a combined approach in vivo and in vitro. In this research, the authors demonstrated that intravenous administration of eucalyptol significantly reduced the blood pressure of both conscious and anesthetized rats. In the same paper, an assay with isolated rat aorta showed that eucalyptol has vasorelaxant activity which led the authors to suggest that the hypotensive effect was probably due to a reduction in peripheral vascular resistance caused by direct relaxation of vascular smooth muscle. Recently, Pinto et al. (2009) demonstrated that this vasorelaxation seems to be dependent on the integrity of the vascular endothelium and nitric oxide releasing.
Furthermore, Soares et al. (2005) investigated eucalyptol effects on the papillary muscle preparations from rat ventricle. In these preparations, the eucalyptol produced a relaxation, possibly caused by inhibition of Ca2+ influx through the membrane.
Menthol
Menthol (17), which can be extracted from plants of the Mentha genus, is widely used in food, beverages and toiletries for its fragrance and its well-known effect of eliciting a refreshing and cooling sensation (Baylie et al., 2010). In study performed by Johnson et al. (2009), was demonstrated that menthol is able to cause a profound dilatation in human forearm cutaneous vessels. This effect appears to involve activation of muscarinic receptors and/or production of nitric oxide. However, in contrast to the human studies, the major part of vasodilatation induced by menthol, in rat arteries appears to be independent of cholinergic/nitric oxide pathways (Johnson et al., 2009) and mediated by activation of transient receptor potential melastatin 8 (TRPM8) (Venkatachalam & Montell, 2007; Johnson et al., 2009).
In studies using the technique of patch clamp in the whole-cell configuration, it was shown that, menthol was also able to inhibit the currents for L-type Ca2+ in right ventricular myocytes isolated from rabbits (Baylie et al., 2010).
Rotundifolone (piperitenone oxide)
Rotundifolone (27) is the main constituent of the essential oil of Mentha x villosa, an aromatic herb known as "the mint-leaf-girl" and extensively used for the treatment of worms (Guedes et al., 2002). In this oil, rotundifolone contributes approximately 63% of its content (Guedes et al., 2002). According to Guedes et al. (2002), intravenous administration of rotundifolone in conscious rats significantly reduced blood pressure and heart rate. To investigate the mechanisms involved in these responses, Guedes et al. (2002) performed experiments in vitro using isolated preparations of atrial and aortic rings, both from rats. These experiments have demonstrated that rotundifolone was able to induce negative inotropic and chronotropic effects in atrium (Guedes et al., 2002) and in aorta vasorelaxation (Guedes et al., 2004). The vasorelaxation was due to inhibition of Ca2+ influx through the membrane and the release of Ca2+ from intracellular stores (Guedes et al. 2004). From these results, the authors concluded that the hypotensive effect was possibly due to a reduction in heart rate associated to a reduction of peripheral vascular resistance, both due to muscarinic activation.
α-Terpineol
The cardiovascular effects of α-terpineol (29) were first reported by Saito et al. (1996). The authors demonstrated that α-terpineol had a hypotensive effect in rats at a dose of 5 mg/kg administered intravenously. Magalhães et al. (2008), using perfused rat mesenteric vascular bed, showed that α-terpineol also induces vasorelaxation which was abolished in the presence of L-NAME, suggesting the involvement of NO in this vasorelaxation. Possibly the hypotensive effects reported by Saito et al. (1996) may have been caused by a reduction in peripheral vascular resistance as a result of vasorelaxation (Magalhães et al., 2008).
Recently, studies performed by Ribeiro et al. (2010), by using combined functional and biochemical approaches, demonstrated that hypotension and vasorelaxation induced by α-terpineol (29) were mediated, at least in part, by the endothelium, most likely via NO release and activation of the NO-cGMP pathway.
α-Terpinen-4-ol
The monoterpene α-terpinen-4-ol (30) is the major constituent of the essential oil of the species Alpinia zerumbet or Alpinia speciosa (Blume) D. Dietr., Zingiberaceae. This medicinal plant is popularly known as Colony and is widely used by the population as a tea in the treatment of arterial hypertension. Studies have shown that intravenous administration of α-terpinen-4-ol caused immediate blood pressure reduction in a dose-dependent manner in both normotensive (Lahlou et al., 2002) and hypertensive rats (Lahlou et al., 2003). In preparations of isolated rat aorta pre-contracted with depolarizing solution of K+, α-terpinen-4-ol was able to induce a concentration-dependent vasorelaxation (Lahlou et al., 2003).
Linalool
Linalool (14 and 15) is usually found in nature in the form of a racemic blend of various herbs. Its use is common in the cosmetic and food industries as perfumes and food flavorings (Peana et al, 2006). Studies by Höferl et al. (2006), which sought to investigate the effects of linalool on stress, showed that this monoterpene has significant effects on the cardiovascular system of humans. In this study, the effects of the optical isomers (+) and (-)-linalool (14) and 15) on blood pressure and heart rate, administered by inhalation, were independently evaluated in 24 subjects. Interestingly, the results showed that the optical isomers had opposite effects. While (+)-linalool (14) showed stimulating effect on the cardiovascular system, (-)-linalool (15) had a depressing effect.
Menezes et al. (2010) evaluated the hypotensive activity of (±)linalool in non-anaesthetized normotensive rats and showed that this monoterpene was able to induce hypotension associated with tachycardia, which could be suggestive of an effect on the peripheral vascular resistance with consequent baroreflex response.
Citronellol
Citronellol (8) is a monoterpene found in some plants used in popular medicine with antihypertensive agents, including Cymbopogon citratus (Abegaz et al., 1983), Cymbopogon winterianus (Quintans-Júnior et al., 2008), and Lippia alba (Tavares et al., 2005). Bastos et al. (2010) and Menezes, et al. (2010) showed that citronellol, administered intravenously, produced hypotension and tachycardia in conscious rats. Seeking to investigate the mechanisms involved in these responses, Bastos et al. (2010) performed experiments in vitro using preparations of isolated rings of superior mesenteric artery of rats, and showed that citronellol was able to induce vasorelaxation. The vessel relaxation was due to inhibition of Ca2+ influx through the membrane and the release of Ca2+ from intracellular stores (Bastos et al., 2010). From these results, the authors concluded that the hypotensive effect is probably because of a reduction of peripheral vascular resistance due to a direct effect on vascular smooth muscle.
Limonene and sobrerol
D-limonene (13) is one of the most common terpenes in nature. It is the major constituent in several citrus oils (orange, lemon, mandarin, lime, and grapefruit) (Sun, 2007).
Touvay et al. (1995) studied the effects of limonene (13) and sobrerol (28) on pulmonary hypertension and right ventricular hypertrophy induced by monocrotaline (MCT) in rats. After daily oral administrations at a dose of 400 mg/rat, both limonene and sobrerol were able to significantly decrease the changes induced by MCT. Moreover, both monoterpenes also reduced the increase in medial thickness of the pulmonary artery (Touvay et al., 1995).
Pinene and cymene
α-Pinene (23) and p-cymene (9) monoterpenes are present in the oil essential of Nigella sativa. Studies about the pharmacological actions of the volatile oil of the Black seed Nigella sativa in both, rats and guinea-pigs revealed its ability in doses of (4-32 µL/kg) (i.v.) to decrease the arterial blood pressure and induce bradycardia in a dose-dependent manner (El Tahir et al., 1993; El Tahir & Ageel, 1994).
El Tahir et al. (2003) observed that after intravenous administration of α-pinene and p-cymene in urethane anaesthetized rats, both showed hypotension and bradycardia. This effect was probably due to inhibition of vasomotor centre with the consequent decrease in the sympathetic outflow reaching the peripheral blood vessels and the heart resulting in decreases in both the arterial blood pressure and the heart rate (El Tahir et al., 1993). On the other hand, Menezes et al. (2010) demonstrated that the (+)-α-pinene (23) and (-)-β-pinene (24) showed a hypotensive effect associated with tachycardia in non-anaesthetized normotensive rats (i.v.).
Myrtenal, myrtenol, and perillyl alcohol
It was observed by Saito et al. (1996) that myrtenal (19), myrtenol (20), and perillyl alcohol (25) possess hypotensive effects at doses of 1 and 5 mg/kg, when administered intravenously in rats.
Conclusion
This review showed the therapeutic potential of monoterpenes in the prevention or treatment of cardiovascular diseases. The main objective of this review was to make researchers aware of the importance of monoterpenes and to stimulate the search for new drugs, as well as to provide a scientific basis for their use. Thus, in the future monoterpenes may be considered as the object of clinical studies, pharmaceutical applications, and as adjuvants in medicine.
Received 8 Oct 2010
Accepted 26 Jan 2011
- Abegaz B, Yohannes PG, Dieter RK 1983. Constituents of the essential oil of Ethiopian Cymbopogon citratus Stapf. J Nat Prod 46: 424-426.
- Aeschbach R, Loliger J, Scott BC, Murcia A, Butler J, Halliwell B, Aruoma OI 1994. Antioxidant actions of thymol, carvacrol, 6-gingerol, zingerone and hydroxytyrosol. Food Chem Toxicol 32: 31-36.
- American Heart Association 2008. Heart Disease and Stroke Statistics - 2008 Update. Circulation 117: e25-e146.
- Aydin Y, Kutlay O, Ari S, Duman S, Uzuner K, Aydin S 2007. Hypotensive effects of carvacrol on the blood pressure of normotensive rats. Planta Med 73: 1365-1371.
- Balick M, Duke J, Kaptchuk T, Mc Coleb R, Pavek R, Pellin C, Reed J, Swyers 1994. Herbal Medicine Washington, DC: National Institutes of Health.
- Baylie RL, Cheng H, Langton PD, James AF 2010. Inhibition of the cardiac L-type calcium channel current by the TRPM8 agonist, (-)-menthol. J Physiol Pharmacol 61: 543-550.
- Bastos JF, Moreira IJ, Ribeiro TP, Medeiros IA, Antoniolli AR, De Sousa DP, Santos MR 2010. Hypotensive and vasorelaxant effects of citronellol, a monoterpene alcohol, in rats. Basic Clin Pharmacol Toxicol 106: 331-337.
- Chappell J 1995. Biochemistry and molecular biology of the isoprenoid biosynthetic pathway in plants. Annu Rev Plant Physiol Plant Mol Biol 46: 521-547.
- Earley S, Gonzales AL, Garcia ZI 2010. A dietary agonist of transient receptor potential cation channel V3 elicits endothelium-dependent vasodilation. Mol Pharmacol 77: 612-620.
- El Tahir KEH, Ashour MM, Al-Harbi MM 1993. The cardiovascular actions of the volatile oil of the black seed (Nigella sayiva) in rats: Elucidation of the mechanism of action. Gen Pharmacol 24: 1123-1131.
- El Tahir KEH, Ageel AM 1994. Effect of the volatile oil of Nigella sativa on the arterial blood pressure and heart rate of the guinea-pig. Saudi Pharm J 2: 163-168.
- El Tahir KEH, Al-Ajmi MF, Al-Bekairi AM 2003. Some cardiovascular effects of the dethymoquinonated Nigella sativa volatile oil and its major components α-pinene and p-cymene in rats. Saudi Pharm J 113: 104-110.
- Garcia R, Alves ESS, Santos MP, Aquije GMFV, Fernandes AAR, Santos RB, Ventura JA, Fernandes PMB 2008. Antimicrobial activity and potential use of monoterpenes as tropical fruits preservatives. Braz J Microbio 39: 163-168.
- Guedes DN, Silva DF, Barbosa-Filho JM, Medeiros IA 2004. Calcium antagonism and the vasorelaxation of the rat aorta induced by rotundifolone. Braz J Med Biol Res 37: 1881-1887.
- Guedes DN, Silva DF, Barbosa-Filho JM, Medeiros IA 2002. Muscarinic agonist properties involved in the hypotensive and vasorelaxant responses of rotundifolone in rats. Planta Med 68: 700-704.
- Höferl M, Krist S, Buchbauer G 2006. Chirality influences the effects of linalool on physiological parameters of stress. Planta Med 72: 1188-1192.
- Johnson CD, Melanaphy D, Purse A, Stokesberry SA, Dickson P, Zholos AV 2009. Transient receptor potential melastatin 8 channel involvement in the regulation of vascular tone. Am J Physiol Heart Circ Physiol 296: 1868-1877.
- Karkabounas S, Kostoula OK, Daskalou T, Veltsistas P, Karamouzis M, Zelovitis I, Metsios A, Lekkas P, Evangelou AM, Kotsis N, Skoufos I 2006. Anticarcinogenic and antiplatelet effects of carvacrol. Exp Oncol 28: 121-125.
- Kato T, Iijima H, Ishihara K, Kaneko T, Hirai K, Naito Y, Okuda K 1990. Antibacterial effects of Listerine on oral bacteria. Bull Tokyo Dent Coll 31: 301-307.
- Lahlou S, Figueiredo AF, Magalhães PJ, Leal-Cardoso JH 2002. Cardiovascular effects of 1,8-cineole, a terpenoid oxide present in many plant essential oils, in normotensive rats. Can J Physiol Pharmacol 80: 1125-1131.
- Lahlou S, Interaminense LF, Leal-Cardoso JH, Duarte GP 2003. Antihypertensive effects of the essential oil of Alpinia zerumbet and its main constituent, terpinen-4-ol, in DOCA-salt hypertensive conscious rats. Fundam Clin Pharmacol 17: 323-330.
- Lee S, Tsao R, Peterson C, Coats JR 1997. Insecticidal activity of monoterpenoids to western corn rootworm (Coleoptera: Chrysomelidae) two-spotted spider mite (Acari: Tetranychidae), and house fly (Diptera: Muscidae). J Econ Entomol 90: 883-892.
- Lefkowits RJ, Willerson J 2001. Prospects for cardiovascular research. JAMA 285: 581-587.
- Magalhães PJ, Lahlou S, Jucá DM, Coelho-De-Souza LN, Da Frota PT, Da Costa AM, Leal-Cardoso JH 2008. Vasorelaxation induced by the essential oil of Croton nepetaefolius and its constituents in rat aorta are partially mediated by the endothelium. Fundam Clin Pharmacol 22: 169-177.
- Magalhães PJC, Criddle DN, Tavares RA, Melo EM, Mota TL, Leal-Cardoso JH 1998. Intestinal myorelaxant and antispasmodic effects of the essential oil of Croton nepetaefolius and its constituents cineole, methyl-eugenol and terpineol. Phytother Res 12: 172-177.
- Magyar J, Szentandrássy N, Bányász T, Fülöp L, Varró A, Nánási PP 2004. Effects of terpenoid phenol derivatives on calcium current in canine and human ventricular cardiomyocytes. Eur J Pharmacol 487: 29-36.
- Magyar J, Szentandrássy N, Bányász T, Fülöp L, Varró A, Nánási PP 2002. Effects of thymol on calcium and potassium currents in canine and human ventricular cardiomyocytes. Br J Pharmacol 136: 330-338.
- Manou L, Bouillard L, Devleeschouwer MJ, Barel AO 1998. Evaluation of the preservative properties of Thymus vulgaris essential oil in topically applied formulations under a challenge test. J Appl Microbiol 84: 368-376.
- Menezes IAC, Barreto CMN, Antoniolli AR, Santos, MRV, De Sousa DP 2010. Hypotensive activity of terpenes found in essential oils. J Bioscience 65: 562-566.
- Peana AT, Rubattu P, Piga GG, Fumagalli S, Boatto G, Pippia P, De Montis MG 2006. Involvement of adenosine A1 and A2A receptors in (-)-linalool-induced antinociception. Life Sci 78: 2471-2474.
- Peixoto-Neves D, Silva-Alves KS, Gomes MD, Lima FC, Lahlou S, Magalhães PJ, Ceccatto VM, Coelho-De-Souza AN, Leal-Cardoso JH 2010. Vasorelaxant effects of the monoterpenic phenol isomers, carvacrol and thymol, on rat isolated aorta. Fundam Clin Pharmacol 24: 341-350.
- Periago PM, Moezelaar R 2001. Combined efect of nisin and carvacrol at diferent pH and temperature levels on the viability of diferent strains of Bacillus cereus. Int J Food Microbiol 68: 141-148.
- Pinto NV, Assreuy AM, Coelho-De-Souza AN, Ceccatto VM, Magalhães PJ, Lahlou S, Leal-Cardoso JH 2009. Endothelium-dependent vasorelaxant effects of the essential oil from aerial parts of Alpinia zerumbet and its main constituent 1,8-cineole in rats. Phytomedicine 16: 1151-1155.
- Quintans-Júnior LJ, Souza TT, Leite BS, Lessa NMN, Bonjardim LR, Santos MRV 2008. Phythochemical screening and anticonvulsant activity of Cymbopogon winterianus Jowitt (Poaceae) leaf essential oil in rodents. Phytomedicine 15: 619-624.
- Ribeiro TP, Porto DL, Menezes CP, Antunes AA, Silva DF, De Sousa DP, Nakao LS, Braga VA, Medeiros IA 2010. Unravelling the cardiovascular effects induced by α-terpineol: A role for the nitric oxide-cGMP pathway. Clin Exp Pharmacol P 37: 811-816.
- Saito K, Okabe T, Inamori Y, Tsujibo H, Miyake Y, Hiraoka K, Ishida N 1996. The biological properties of monoterpenes: Hypotensive effects on rats and antifungal activities on plant pathogenic fungi of monoterpenes. Mokuzai Gakkaishi 42: 677-680.
- Santos FA, Rao VS 2001. 1,8-Cineol, a food flavoring agent, prevents ethanol-induced gastric injury in rats. Digest Dis Sci 46: 331-337.
- Serrano MED, López ML, Espuñes T del RS 2006. Componentes bioactivos de alimentos funcionales de origen vegetal. Rev Mex Ciênc Farm 37: 58-68.
- Sharkey TD, Yeh S 2001. Isoprene emission from plants. Annu Rev Plant Physiol Plant Mol Biol 52: 407-436.
- Simões CMO, Spitzer V 2004. Óleos Voláteis. In: Simões CMO, Schenkel EP, Gosmanm G, Mello JCP, Mentz LA, Petrovick PR. Farmacognosia da planta ao medicamento. 5. ed. Porto Alegre/Florianópolis: Editora da UFRGS & UFSC, p. 467-495.
- Singh P, Shukla R, Prakash B, Kumar A, Singh S, Mishra PK, Dubey NK 2010. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, DL-limonene. Food Chem Toxicol 48: 1734-1740.
- Soares MC, Damiani CE, Moreira CM, Stefanon I, Vassallo DV 2005. Eucalyptol, na essential oil, reduces contractile activity in rat cardiac muscle. Braz J Med Biol Res 38: 453-461.
- Sociedade Brasileira de Cardiologia 2007. V Diretrizes Brasileiras de Hipertensão Arterial. Arq Bras Cardio 89: e24-e79.
- Sun J 2007. D-limonene:safety and clinical applications. Altern Med Rev 12: 259-264.
- Szentandrássy N, Szigeti G, Szegedi C, Sárközi S, Magyar J, Bányász T, Csernoch L, Kovács L, Nánási PP, Jóna I 2004. Effect of thymol on calcium handling in mammalian ventricular myocardium. Life Sci 74: 909-921.
- Tavares ES, Julião LS, Lopes D, Bizzo HR, Lage CLS, Leitão SG 2005. Análise do óleo essencial de folhas de três quimiotipos de Lippia alba (Mill.) N. E. Br. (Verbenaceae) cultivados em condições semelhantes. Rev Bras Farmacogn 15: 1-5.
- Touvay C, Vilain B, Carré C, Mencia-Huerta JM, Braquet P 1995. Effect of limonene and sobrerol on monocrotaline-induced lung alterations and pulmonary hypertension. Int Arch Allergy Immunol 107: 272-274.
- Venkatachalam K, Montell C 2007. TRP channels. Annu Rev Biochem 76: 387-417.
- Wagner KH, Elmadfa I 2003. Biological relevance of terpenoids. Ann Nutr Metab 47: 95-106.
- Zwenger S, Basu C 2008. Plant terpenoids: applications and future potentials. Biotechnol Mol Biol Rev 3: 1-7.
Publication Dates
-
Publication in this collection
08 July 2011 -
Date of issue
Aug 2011
History
-
Received
08 Oct 2010 -
Accepted
26 Jan 2011