Abstract
Croton echioides Baill., Euphorbiaceae, is a small tree found in Bahia, Northeastern Brazil. Its stem bark has been widely sold as an aphrodisiac and tonic, as a substitute for the roots of Ptychopetalum olacoides Benth. Olacaceae, the Amazon Muira Puama or Marapuama, and C. echioides is characterized as the "Northeastern Marapuama". This contribution describes a morphoanatomical analysis and pharmacognostic study of stem bark of this species. The stem has a thick cortex with compound starch grains and laticifers; a sclerenchymatic sheath which consists of brachysclereids with large crystals externally to the phloem, and abundant fiber in the secondary xylem, as the main features of the species. The data obtained for water content (9.26±0.07%), water-soluble extractives (3.92±0.19%), total ash (1.24±0.06%) and acid-insoluble ash (0.16±0.01%), together with the chromatographic profile obtained by TLC, contribute to the quality control and standardization for the plant drug. The pharmacological screening indicated LD50 values above 500 mg/kg orally and equal to 500 mg/kg by the i.p. route, as well as some stimulant potential, depending on the dose.
Croton echioides; Euphorbiaceae; morphoanatomy; Northeastern Marapuama; pharmacognostic quality control
Morphoanatomy and pharmacognostic study of the wood of Croton echioides, the Northeastern Marapuama
Cláudio R. NovelloI; Luís C. MarquesII; Cristine R. MiyazakiII; Maria A. Milaneze-GutierreIII; Daniela S. Carneiro-TorresIV; Maria H. SarragiottoV; João C. P. de Mello* * Correspondence João Carlos Palazzo de Mello Departamento de Farmácia. Universidade Estadual de Maringá Avenida Colombo, 5790, BR-87020-900, Maringá-PR, Brazil mello@uem.br Tel. + 55 44 3011 4816 Fax: +55 44 3011 5050 ,I
IDepartamento de Farmacia, Universidade Estadual de Maringá, Brazil
IIUniversidade Bandeirante de São Paulo, Brazil
IIIDepartamento de Biologia, Universidade Estadual de Maringá, Brazil
IVUniversidade Estadual de Feira de Santana, Brazil
IVDepartamento de Química, Universidade Estadual de Maringá, Brazil
ABSTRACT
Croton echioides Baill., Euphorbiaceae, is a small tree found in Bahia, Northeastern Brazil. Its stem bark has been widely sold as an aphrodisiac and tonic, as a substitute for the roots of Ptychopetalum olacoides Benth. Olacaceae, the Amazon Muira Puama or Marapuama, and C. echioides is characterized as the "Northeastern Marapuama". This contribution describes a morphoanatomical analysis and pharmacognostic study of stem bark of this species. The stem has a thick cortex with compound starch grains and laticifers; a sclerenchymatic sheath which consists of brachysclereids with large crystals externally to the phloem, and abundant fiber in the secondary xylem, as the main features of the species. The data obtained for water content (9.26±0.07%), water-soluble extractives (3.92±0.19%), total ash (1.24±0.06%) and acid-insoluble ash (0.16±0.01%), together with the chromatographic profile obtained by TLC, contribute to the quality control and standardization for the plant drug. The pharmacological screening indicated LD50 values above 500 mg/kg orally and equal to 500 mg/kg by the i.p. route, as well as some stimulant potential, depending on the dose.
Keywords: Croton echioides; Euphorbiaceae; morphoanatomy; Northeastern Marapuama; pharmacognostic quality control.
Introduction
Products of natural origin with reputed aphrodisiac effects have long been of great interest for therapeutic uses and also to international pharmaceutical companies, and have been sought after and studied for millennia. Some of the sought-after species have proved to be effective, as occurred with yohimbe bark (Corynanthe johimbe K. Schum., Rubiaceae), the roots of Korean ginseng (Panax ginseng C.A. Mey., Araliaceae), and cantharidin obtained from insect cantharides (Lytta vesicatoria L., Meloidae), among others (Riley, 1994; Sandroni, 2001).
A Brazilian plant with popular interest and commercial potential for its aphrodisiac effects is Muira Puama or Marapuama, the root bark of Ptychopetalum olacoides Benth., Olacaceae. This plant drug has been marketed and exported since the beginning of the twentieth century, when it was sometimes counterfeited by substitution of guava roots (Silva, 1925). This was later formalized in the first edition of the Brazilian Pharmacopoeia (Silva, 1926).
Several studies have provided evidence of the effects of this plant drug on the central nervous system. Initially it was found to show effects as an anxiogenic (Silva et al., 2002), anticholinesterase (Siqueira et al., 2003), neuroprotective (Siqueira et al., 2004), facilitation of memory recovery in young and old mice (Silva et al., 2004), antioxidant (Siqueira et al., 2007), antidepressant (Paiva et al., 1998; Piato et al., 2009), and anti-stress (Piato et al., 2010). Its aphrodisiac potential has not been confirmed experimentally or clinically, although some of its effects, such as antidepressant and antioxidant activity, may improve sexual performance (Feldman et al., 1994).
Interest in these effects and the folk wisdom of the Brazilian Amazon have kept this species in evidence, together with catuaba and other plants (Charam, 1987; Pozzoli, 1999). Consequently, it has continued to be collected in the wild, marketed, and exported for decades. The continual exploitation of this Amazon endemic species, the use of roots, and the lack of incentives to cultivate it to supply this demand must have led to significant reduction of its natural populations.
When such a situation arises, usually the market seeks alternative sources of supply, eventually triggering the replacement of the true species by another with the same indications, although it may not always be chemically similar, as occurred with the catuaba (Marques, 1998a). In the case of P. olacoides, the commercial substitution led to exploitation of stems, which were initially offered as being from the species itself, as a strategy to prevent the destruction of the specimens by removal of the roots, as reported by plant drug distribution companies. However, it was gradually found that these stems came from the interior of the state of Bahia, probably originating from some species of Croton, Euphorbiaceae, and were completely different from the Amazon Marapuama (Tobias et al., 2007).
The large and diverse genus Croton L. occurs in tropical and subtropical regions worldwide (Webster, 1993), and according to Govaerts et al. (2000) contains about 1200 species. The main compounds found in the genus Croton are clerodane-type diterpenes, flavonoids, and indole alkaloids (Matos, 2011). Many clerodane diterpenes show biological activity, including antibiotic, antitumor, insecticidal, anti-inflammatory, antinociceptive, hypoglycemic, antiulcer (Maciel et al., 1998), and cardiovascular (Silva et al., 2005).
P. olacoides was reported initially to contain behenic acid ester and β-sitosterol (Auterhoff & Pankow, 1968), free fatty acids, and no alkaloids (Toyota et al., 1979), as well as several steroids, the triterpene lupeol, flavonoids, and saponins (Maul et al., 1998). More recently, Colombo et al. (2010) found vanillic acid, protocatechuic acid, and theobromine in the stems of P. olacoides. However, the presence of essential oils, tannins and alkaloids was reported by Bucek et al. (1987), as well as several clerodane diterpenes by Tang et al. (2009, 2011).
By linking these contradictory phytochemical observations with the reported problem of adulteration of commercial species in the Brazilian market, which also extrapolates to the international market, even as raw material for scientific research, it is apparent that some of these studies in fact assessed the stems of the "Northeastern Marapuama".
Taking into account the lack of strict control of the harvesting, identification, and botanical characterization, for chemical or pharmacological quality control for this species of Croton, this study aimed to collect and identify unambiguously the species used as the "Northeastern Marapuama", C. echioides Baill. In addition, we sought to describe the morphoanatomy of the plant drug (stems) and conduct a pharmacognostic analysis, establishing the first scientific specifications for this raw material.
Materials and Methods
Plant material
Through contacts with suppliers of plant drugs for the Brazilian market, we tracked the source location of stems offered as P. olacoides in commercial batches of raw materials. This effort led to José Maria Fernandes Monteiro from the municipality of Lagoa Real, state of Bahia, and we visited him personally to collect, plant material, in December 2008. Mr. Monteiro is a leading supplier of plant drugs for the Brazilian pharmaceutical market.
The flowering aerial parts of Croton echioides Baill., Euphorbiaceae, were collected at Lagoa Real under collection authorization No. 11995-3 of 02.11.2010, registered as IBAMA No. 1844493, under the responsibility of João Carlos Palazzo de Mello. We also collected samples of stems (plant drugs) for use in further studies.
Botanical identification and herborization
The botanical material was mounted on exsiccates, which were deposited in the herbarium of the Universidade Estadual de Feira de Santana, under HUEFS number 139049, and in the herbarium of the Universidade Estadual de Maringá, under HUEM number 19433. The species was identified by Prof. Dr. Daniela Santos Carneiro Torres, with the aid of the key by Silva et al. (2009).
Pharmacobotany characterization
The organoleptic characterizations, and macroscopic and microscopic examinations of stem segments were performed according to standard methodology (Oliveira et al., 2003; Farmacopeia Brasileira, 2010). Pieces of commercial stems were evaluated, described, and photographed. Young (up to 1 cm diameter) and mature (over 1 cm in diameter) stem segments were dried and evaluated with the naked eye, with regard to the macroscopic structure, rhytidome color, and internal tissues. For the anatomical analysis, fresh stems were cut into smaller portions, fixed in FAA 50, and conserved in 70% ethanol (Johansen, 1940). The histological sections were made on several planes, freehand with the aid of stainless-steel blades, and then bleached with sodium hypochlorite (30% commercial) for approximately 5 min, stained with safranin and Astra blue (1% aqueous solutions in the ratio 9:1 v/v) (Kraus & Arduin, 1997), and mounted as semi-permanent slides with glycerin jelly. For the preparation of the illustrations, the tissues were photographed with a digital camera attached to an Olympus microscope, using the software Image-Pro Plus 4.5.
Pharmacognostic quality control assays
Samples of stems of C. echioides (about 10 kg) were cut into small pieces, dried at room temperature in the PALAFITO-UEM Laboratory, ground in a hammer mill (Tigre ASN5), and stored hermetically sealed and protected from heat and light. The granulometry of the powder was measured by sieves of mesh sizes 150, 180, 212, 300, 600, and 850 μm (Bertel AGT-01). The levels of total ash, acid-insoluble ash, and loss on drying were determined according to methods described by Brazilian Pharmacopeia (Farmacopeia Brasileira, 2010) as were the water-soluble extractives (WHO, 1998), all from the samples of powdered stems.
For thin layer chromatography (TLC), ground or powdered plant drug (1 g) was subjected to reflux with 100 mL of methanol, filtered and concentrated to 10 mL. This solution (20 μL) was applied to silica gel F254 plates (Merck), eluted with n-hexane: dichloromethane: methanol (6:3:1, v/v), and revealed with sulfuric anisaldehyde upon heating to 110 ºC, and the retention factors (Rf) were determined.
Pharmacological screening of the crude extract
We used male Swiss mice aged 2-3 months and weighing from 30-40 g, from the Center for Development of Experimental Models of the Universidade Federal de São Paulo. The animals were kept for a week before the experimental procedures at the Laboratory of Experimental Pharmacology of Uniban, in groups of ten animals per box (24±3 ºC) with a controlled light/dark cycle (12 h) and fresh water and food ad libitum, up to 120 min before the start of the experiments. All experimental procedures were performed according to the ethical principles in animal experimentation adopted by Uniban protocol (No. 20090161). The plant extract was dissolved in water with drops of Tween 80 at the time of testing.
The extracts were prepared from powdered stems of the plant (7.6 kg) by Ultra-Turrax (Ika Works, UTC115KT) using 70% (v/v) hydroalcoholic solvent in the proportion of 10% (w/v). The extraction was performed for 5 min of stirring followed by 2 h of rest, successively, within a period of 48 h. The solvent was removed in a rotavapor under reduced pressure, frozen, and lyophilized to provide the crude extract (CE).
Groups of five young male mice received various doses of CE (1, 10, 100, and 500 mg/kg) by i.p., and 500 mg/kg of CE orally (gavage), together with a control group. Immediately thereafter the mice were placed in wire cages (five per cage), and were observed at the times of 5, 15, and 30 min, 1, 2, 4, and 24 h, noting all behaviors pertinent to the expression of some pharmacological and/or toxic effect. The animals were weighed before the experiment and again after fourteen days of observation.
The animals were evaluated according to the protocol established by Carlini (1972): urination, defecation, abdominal constrictions, rough hair coat, motor activity, tremors, convulsions, ataxia, loss of posture reflex, pain sensitivity, ptosis, Straub tail, abduction of the hind legs, signs of stereotypy, yawning, sleep, excessive grooming, muscle tone, lacrimation, exophthalmia, salivation, and death.
Results and discussion
Botanical identification
Review of the literature showed that, until the present, C. echioides had not been properly identified and related to the commercial material available throughout the country, and also as raw material for export, since invariably it was labeled and offered as Ptychopetalum olacoides. Thus, the systematic examination proved crucial to unequivocally identify the plant material offered in Brazil as 'Marapuama'.
These observations have clarified the identifications of the main species known as Brazilian aphrodisiacs, including Catuaba stem bark of Trichilia catigua A. Juss. (Marques, 1998a); Brazilian Ginseng roots Pfaffia glomerata (Spreng.) Pedersen (Marques, 1998b); and now Croton echioides as the "Northeastern Marapuama", available commercially as a substitute for the true "Marapuama", P. olacoides.
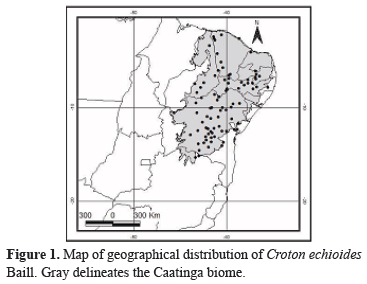
Croton echioides is popularly known in the northeast region of Brazil as "quebra-faca", "caatinga-branca" and "canela-de-velho". It is restricted to the Caatinga biome, occurring in the states of Piauí, Ceará, Rio Grande do Norte, Pernambuco, Paraíba, Alagoas, Bahia, and Minas Gerais in arboreal to shrubby caatinga vegetation, in sandy and clay soils, with rocky outcrops (Figure 1). As do all species of Euphorbiaceae, C. echioides has unisexual flowers and schizocarp fruit, tricarpellar with only one ovule per locule. It is differentiated from other species of Croton of the Caatinga biome in having leaves with the margin entire, the presence of two short-stipitate glands between the petiole and leaf blade, staminate flowers with spatulate petals, and pistillate flowers that are sessile and sparsely arranged along the inflorescence.
Organoleptic, macroscopic and microscopic characterization
The samples are marketed in stem fragments of various sizes, 10-15 cm long and about 1-4 cm wide. The rhytidome is mixed in color, ranging from light gray to light brown with dark-brown spots; there are a few thick cracks, with no regular pattern, but generally longitudinal. In median longitudinal section, the cork appears thin, strongly adhered to other tissues. The wood is light brown, with the central, medullar, dark brown along its entire length; in transverse view, the medullary tissue shows as a central dark spot (Figure 2 and 3). These characteristics are consistent with the strains of C. echioides collected in the municipality of Lagoa Real, as are the organoleptic characteristics, in which the bark has its own aromatic odor and slightly bitter taste; the wood is odorless and tasteless.
The optical-microscopy analysis revealed that the stem of C. echioides shows a periderm composed of 6-7 strata of quadrangular cells in cross section (Figures 4A and 5A), somewhat elongated longitudinally (Figures 4B and 5B) and 10 to 12 strata can occur at sites of defoliation. The cells of the periderm are thicker in the periclinal walls (Figure 5C) than in the opposite walls, and have different dimensions when viewed in longitudinal tangential section (Figure 5D).
The stem cortex of C. echioides is thick, with approximately twenty strata whose cells vary in diameter (Figures 4A and 5A). A longitudinal section revealed somewhat longitudinally elongated cortical cells, sometimes isodiametric, but also with no uniformity in their dimensions (Figures 4B, 5B, and 6B ). This tissue contains compound starch grain (Figure 6C ) and abundant laticifers (Figures 4A, 4B, 5A, 5B, and 6A , 6C , and 6D ). According to Farias et al. (2009), the presence of latex in the cortical portion of the stem is common in many species of the genus Croton. Although few studies describe the anatomical tissues of the stem of species of Brazilian Croton, Silva (2006) reported the occurrence of laticifers located near the region of the stem phloem of C. floribundus. When comparing the Marapuama samples available on the market, the presence of laticifers, which is common in Euphorbiaceae, in contrast to Olacaceae species, serves as a distinctive character between P. olacoides and C. echioides, although it does not distinguish the latter from other members of the genus Croton.
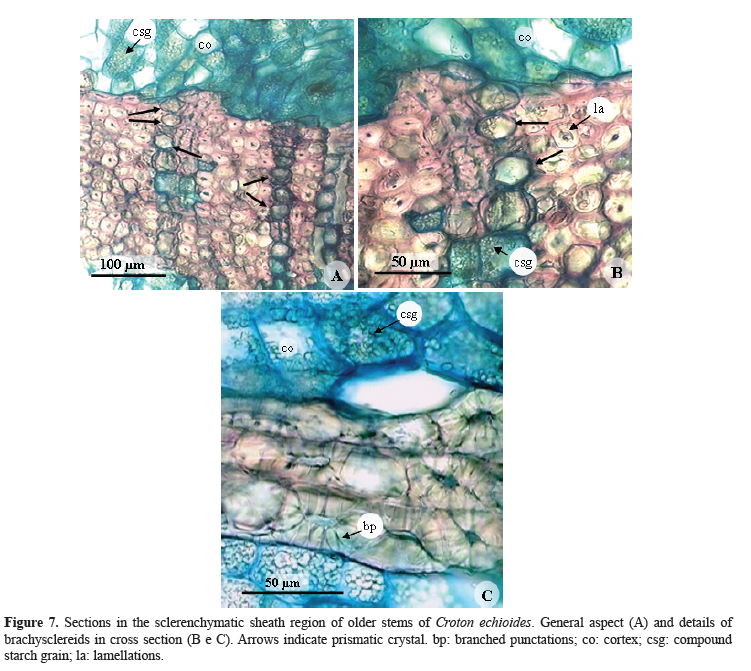
More internally, and involving the vascular system, there is a thick sclerenchymatous sheath, which consists of brachysclereids. In young stem, has 5-6 layers of this cells (Figure 6A ) and 10-17 layers in older stem (Figure 7A). The sclerenchymatic cells are more homogeneous in shape when viewed in cross section (Figures 7A, 7B, and 7C), but show different shapes, tending to be elongated in longitudinal section. In its thick walls, lamellations (Figure 7B) and branched punctations are evident (Figure 7C), similar to those observed by Silva (2006) in the secondary stem of C. floribundus and C. macrobothrys, although in these species they are arranged in a discontinuous ring, interspersed with parenchyma cells. In addition, according to Thomas et al. (1995) in Hevea brasiliensis these sclereids are distributed diffusely or in bundles, from near the periderm to the phloem region. Both the brachysclereids and the cells that compose the parenchyma rays that permeate the sclerenchymatous sheath contain large crystals, which are polygonal to typical prismatic in shape (Figures 6A , 7A, and 7B). The presence of sclerenchyma with large crystals in the secondary phloem was also reported by Farias et al. (2009) in C. draco var. draco, a Mexican species. The druses observed by Silva (2006) in the cortical parenchyma of C. floribundus were not found in C. echioides.
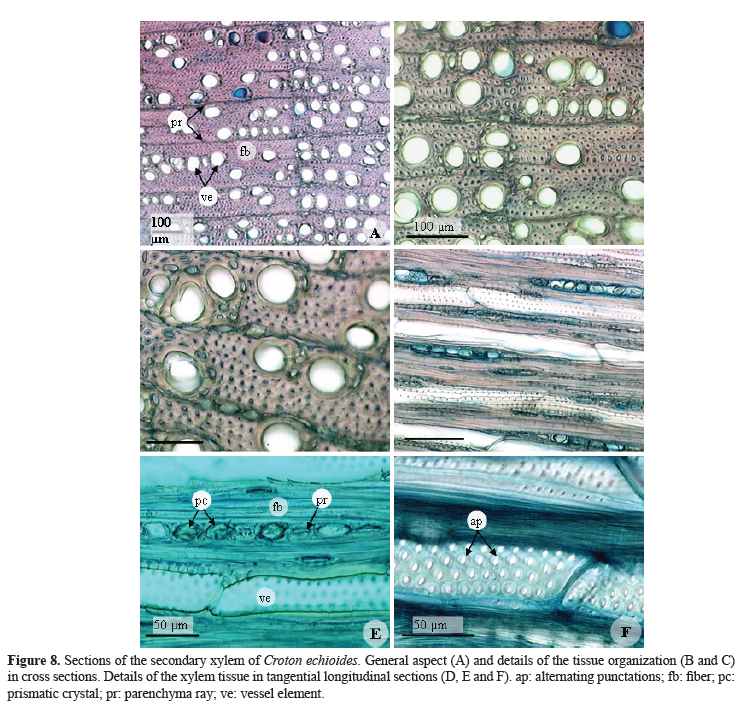
The secondary xylem of C. echioides rarely contains parenchyma cells (Figures 8A, 8B and 8C), which is not common for other members of this taxonomic group. In the secondary xylem, growth rings were evident (Figure 8A) as were vessel elements, most often solitary, although multiple pairs and clusters may also occur (Figures 8A, 8B, and 8C). In these vessel elements the punctuations are areolate and the alternating type (Figure 8F). The fibers that compose the secondary xylem of C. echioides are abundant, with very thick walls, giving them a small lumen (Figures 8B and 8C), and a high degree of rigidity to the mature stems of this species. The gelatinous fibers, common in species of Croton, also appear in abundance in the secondary xylem of youngers stems.
The parenchyma rays of secondary xylem are uniseriate and are almost obliterated in mature stem segments (Figures 8C, 8D, and 8E). In this figures can also be seen crystals, as in the phloem, present in nearly all the cells of the ray parenchyma.
The pith, even in young stem, is compose by sclerenchymatic sheath which consists of brachysclereids, with a square to polygonal shape in cross section (Figures 9A and 9B), most of them slightly elongated compared to the longer axis of the organ (Figures 9C and 9D). In this cell type with very thick walls, the punctuations are, in most cases, branched (Figure 9D).
Pharmacognostic quality-control data
The results for the analysis of the physicochemical quality control of the stems of C. echioides are seen in Table 1. The levels are within the parameters found in most plant drugs in the pharmacopeia, with the exception of the particle-size test, which is specific for this plant.
The thin-layer chromatographic analysis showed a different chromatographic profile, as revealed with sulfuric anisaldehyde (Figure 10). In addition, there were light-brown and purple spots with the same Rf values 0.24 and 0.52, compared to the reference compounds diterpene [15,16-epoxy-3,13(16),14-neo-clerodatrien-17-carboxy-18-methyl carboxylate] and lupeol, respectively, as shown in Figure 9. Notably, lupeol is also found in P. olacoides (Auterhoff & Momberger, 1971; Ito et al., 1995), and therefore is not a useful standard for the differentiation of the species P. olacoides and C. echioides.
Pharmacological screening results
The CE showed a preliminary stimulatory effect at low doses, by oral and i.p.; toxicity was evident for the larger i.p. doses, with 100% mortality after administration of 500 mg/kg (Table 2). Given this profile, it is suggested that more specific pharmacological tests be used with doses ranging from 1 to 100 mg/kg, in order to determine more precisely the levels of toxicity. One case of Straub tail was observed, which indicated stimulation of opioid receptors; it would be interesting to apply tests for analgesia or others aimed at evaluating this potential effect. Most of the groups lost weight, although not in statistically significant amounts.
Conclusion
The data presented here confirmed that the stems of Croton echioides are the "Northeastern Marapuama" that is widely marketed within and outside Brazil. The pharmacognostic data obtained in this study resulted in an effective basic characterization and standardization of this plant material, contributing to the differentiation from the plant drugs obtained from roots or stems of Ptychopetalum olacoides.
The LD50 values above 500 mg/kg orally and equal to 500 mg/kg by the i.p. route indicated that the species has low acute toxicity (Zucker, 1985), although new pharmacological and toxicological studies would be appropriate to better understand these aspects. Therefore, further investigations are needed to confirm whether there are effective therapeutic and pharmacological similarities between the two Brazilian species popularly named "Marapuama".
Acknowledgments
This work was supported by the Brazilian granting agencies CNPq, CAPES/Proap, INCT_if, FINEP and Fundação Araucária. The authors are grateful to Dr. Janet W. Reid, JWR Associates, Trumansburg, New York, for the English language revision. In memorian Prof. Dr. Gentil José Vidotti.
Received 23 Jan 2012
Accepted 11 Apr 2012
- Auterhoff H, Pankow E 1968. Inhaltsstoffe von muira puama. Archiv der Pharmazie 7: 481-489.
- Auterhoff H, Momberger B 1971. Lipophilic constituent of Muira puama 3. Archiv der Pharmazie 3: 223-228.
- Bucek EU, Fournier G, Dadoun H 1987. Volatile constituent of Ptychopetalum olacoides root oil. Planta Med 53: 231-231.
- Carlini EA 1972. Screening farmacológico de plantas brasileiras. Rev Bras Biol 32: 265-274.
- Charam I 1987. Há ações afrodisíacas nas plantas medicinais do Brasil? A Folha Médica 94: 303-309.
- Colombo R, Batista ANL, Bomfim GCC, Burgos RCR, Cavalheiro AJ, Bolzani VS, Silva DHS, Reimberg MCH 2010. Validated high-performance liquid chromatographic method for the standardization of Ptychopetalum olacoides Benth., Olacaceae, commercial extracts. Rev Bras Farmacogn 20: 781-788.
- Farias FR, Williamson JS, Rodrígues SV, Angeles G, Portugal VO 2009. Bark anatomy in Croton draco var. draco (Euphorbiaceae). Am J Bot 96: 2155-2167.
- Farmacopeia Brasileira 2010. Ministério da Saúde, 5. ed. Brasília: Agência Nacional de Vigilância Sanitária
- Feldman HA, Goldstein I, Hatzichristou DG, Krane RJ, McKinlay JB 1994. Impotence and its medical and psychosocial correlates: results of the Massachusetts male aging study. J Urology 151: 54-61.
- Govaerts R, Frodin DG, Radcliffe-Smith A 2000. World Checklist and Bibliography of Euphorbiaceae (with Pandaceae) 1-4. Great Britain: The Royal Botanic Gardens, Kew.
- Ito Y, Hirayama F, Aikawa Y, Kondo H, Sagara K, Shoji J 1995. Constituents from Muira-puama (the roots of Ptychopetalum olacoides). Nat Med 49: 487-495.
- Johansen DA 1940. Plant microtechnique New York: McGraw-Hill.
- Kraus JE, Arduin M 1997. Manual básico de métodos em morfologia vegetal. Rio de Janeiro: Editora Universidade Rural.
- Maciel MAM, Pinto AC, Brabo SN, Da Silva MN 1998. Terpenoids from Croton cajucara. Phytochemistry 49: 823-828.
- Marques LC 1998a. Contribuição ao esclarecimento da identidade botânica da droga vegetal catuaba. Rev Racine 8: 8-11.
- Marques LC 1998b. Avaliação da ação adaptógena das raízes de Pfaffia glomerata (Sprengel) Pedersen - Amaranthaceae. São Paulo, 145 p. PhD Thesis. Escola Paulista de Medicina.
- Matos LMM 2011. Química de espécies nativas de Croton L. (Euphorbiaceae). São Paulo, 123 p. Dissertation. Instituto de Biociências, Universidade de São Paulo.
- Maul AA, Pena RS, Polakiewicz B, Kachan GC 1998. Extração de matérias primas das raízes da Muirapuama (Ptychopetalum olacoides) por fluido supercrítico. Rev Farm Quim 31: 41-44.
- Oliveira F, Akisue G 2003. Farmacognosia São Paulo: Atheneu.
- Paiva LAF, Rao VSN, Silveira ER 1998. Effects of Ptychopetalum olacoides extract on mouse behaviour in forced swimming and open field tests. Phytother Res 12: 294-296.
- Piato AL, Rizon LP, Martins BS, Nunes DS, Elisabetsky E 2009. Antidepressant profile of Ptychopetalum olacoides Bentham (Marapuama) in mice. Phytother Res 23: 519-524.
- Piato AL, Detanico BC, Linck VM, Herrmann AP, Nunes DS, Elisabetsky E 2010. Anti-stress effects of the "tonic" Ptychopetalum olacoides (Marapuama) in mice. Phytomedicine 17: 248-253.
- Pozzoli C 1999. Sù con le piante! Quattro antidote tradizionali contro le disfunzioni erettili. Erboristeria Domani 10: 52-58.
- Riley AJ 1994. Yohimbine in the treatment of erectile disorder. Br J Clin Pract 48: 1363-1366.
- Sandroni P 2001. Aphrodisiacs past and present: a historical review. Clin Auton Res 11: 303-307.
- Silva RAD 1925. Plantas medicinais brasileiras: estudo botânico e pharmacognóstico Muirapuama. Rev Bras Med Pharm 1: 37-41. Republished in 1989 by Rev Bras Farm 70: 34-35.
- Silva RAD 1926. Pharmacopeia dos Estados Unidos do Brasil. São Paulo: Nacional.
- Silva AL, Bardini S, Nunes DS, Elisabetsky E 2002. Anxiogenic properties of Ptychopetalum olacoides Benth. (Marapuama). Phytother Res 16: 223-226.
- Silva AL, Pinto ALS, Bardini S, Netto CA, Nunes DS, Elizabetsky E 2004. Memory retrieval improvement by Ptychopetalum olacoides in young and aging mice. J Ethnopharmacol 95: 199-203.
- Silva RM, Oliveira FA, Cunha KMA, Maia JL, Maciel MAM, Pinto AC, Nascimento NRF, Santos FA, Rao VSN 2005. Cardiovascular effects of trans-dehydrocrotonin, a diterpene from Croton Cajucara in rats. Vascular Pharmacol 43: 11-18.
- Silva BO 2006. Análise estrutural e Histoquímica de Croton macrobothrys Baillon e Croton floribundus Spreng (Euphorbiaceae) Rio de Janeiro, 150 p. Dissertation. Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro.
- Silva JS, Sales MF, Carneiro-Torres DS 2009. O gênero Croton (Euphorbiaceae) na microrregião do vale do Ipanema, Pernambuco, Brasil. Rodriguésia 60: 879-901.
- Siqueira IR, Fochesatto C, da Silva AL, Nunes DS, Battastini AM, Netto CA, Elisabetsky E 2003. Ptychopetalum olacoides, a traditional Amazonian "nerve tonic", possesses anticholinesterase activity. Pharmacol Biochem Behav 75: 645-650.
- Siqueira IR, Cimarosti H, Fochesatto C, Nunes DS, Salbego C, Elisabetsky E, Netto CA 2004. Neuroprotective effects of Ptychopetalum olacoides Bentham (Olacaceae) on oxygen and glucose deprivation induced damage in rat hippocampal slices. Life Sci 75: 559-568.
- Siqueira IR, Fochesatto C, Torres IL, da Silva AL, Nunes DS, Elisabetsky E, Netto CA 2007. Antioxidant activities of Ptychopetalum olacoides ("Muirapuama") in mice brain. Phytomedicine 14: 763-769.
- Tang W, Kubo M, Harada K, Hioki H, Fukuyama Y 2009. Novel NGF-potentiating diterpenoids from a Brazilian medicinal plant Ptychopetalum olacoides. Bioorg Med Chem Lett 19: 882-886.
- Tang W, Harada K, Kubo M, Hioki H, Fukuyama Y 2011. Eight new clerodane diterpenoids from the bark of Ptychopetalum olacoides. Nat Prod Commun 6: 327-332.
- Thomas V, Premakumari D, Reghu CP, Panikkar AON, Amma S 1995. Anatomical and histochemical aspects of bark regeneration in Hevea brasiliensis. Ann Botany 75: 421-426.
- Tobias ML, Oliveira F, Oliveira KP, Marques LC 2007. Controle de qualidade de drogas vegetais de farmácias de manipulação de Maringá (Paraná - Brasil). Rev Eletr Farm 4: 95-103.
- Toyota A, Ninomiya R, Kobayashi H, Kawanishi K, Uhara Y, Kato A, Hashimoto Y 1979. Studies of Brazilian crude drugs (1): Muira-puama. Shoyakugaku Zasshi 33: 57-64.
- Webster GL 1993. A provisional synopsis of the section of the genus Croton (Euphorbiaceae). Taxon 42: 793-823.
- World Health Organization 1998. Quality control methods for medicinal plant materials. Geneva.
- Zucker E 1985. Hazard evaluation division: standard evaluation procedure acute toxicity test for freshwater fish Washington: USEPA Publication 540/9-85-006.
Publication Dates
-
Publication in this collection
15 May 2012 -
Date of issue
Oct 2012
History
-
Received
23 Jan 2012 -
Accepted
11 Apr 2012