Abstract
The essential oil of Chenopodium ambrosioides L., Amaranthaceae, was obtained by steam distillation in a Clevenger apparatus and characterization was performed using chromatographic and spectroscopic assays (GC-FID, GC/MS, 1H NMR). Two major compounds were identified: p-cymene (42.32%) and ascaridole (49.77%). The ethanolic extract and hydrolate were fractionated by liquid–liquid partitioning and the compounds were characterized by GC/MS. The essential oil, ethanol extract and fractions by partitioning with dicloromethane, ethyl acetate and butanol were tested in tumor cell lines (K562, NALM6, B15, and RAJI). Significant cytotoxic activity was found for essential oil (IC50 = 1.0 µg/ml) for RAJI cells and fraction dicloromethane (IC50 = 34.0 µg/ml) and ethanol extract (IC50 = 47.0 µg/ml) for K562 cells. The activity of the essential oil of C. ambrosioides is probably related to the large amount of ascaridol, since the other major compound, p-cymene, is recognized as a potent anti-inflammatory and has low cytotoxic activity.
Keywords
Chenopodium ambrosioides; Ascaridole; p-Cymene; Essential oil; Cytotoxic; Gas chromatographic
Introduction
Chenopodium ambrosioides L., Amaranthaceae, popularly known as “erva-de-santa-maria” or “mastruço” (Kokanova-Nedialkova et al., 2009Kokanova-Nedialkova, Z., Nedialkov, P.T., Nikolov, S.D., 2009. The genus Chenopodium: phytochemistry, ethnopharmacology and pharmacology. Pharmacogn. Rev. 3, 280-306), has been widely used in folk medicine in the midwest, south and southeast of Brazil and is found mainly in temperate and subtropical countries (Lorenzi and Matos, 2002Lorenzi, H., Matos, F.J.A., 2002. Plantas medicinais no Brasil: nativas e exóticas cultivadas. Instituto Plantarum de Estudos da Flora, Nova Odessa, SP.). The leaves are used as an anthelmintic and vermicide (Alitonou et al., 2012Alitonou, G.A., Sessou, P., Tchobo, F.P., Noudogbessi, J., Avlessi, F., Yehouenou, B., Menut, C., Villeneuve, P., Sohounhloue, D.C.K., 2012. Chemical composition and biological activities of essential oils of Chenopodium ambrosioides L. collected in two areas of Benin. Int. J. Biosci. 2, 58-66) and this species is also used in the treatment of gastrointestinal, respiratory, vascular, and nervous diseases and to combat diabetes and hypercholesterolemia. Furthermore, it presents sedative, antipyretic, and antirheumatic effects (De Feo and Senatore, 1993De Feo, V., Senatore, F., 1993. Medicinal plants and phytotherapy in the Amalfitan Coast, Salerno Province, Campania, southern Italy. J. Ethnopharmacol. 39, 39-51). Due to these properties, in 2009, the Brazil's Health Ministry selected the C. ambrosioides as one of the plants of interest to the Helth Sistem (Renisus) that can be used as an herbal medicine.
The geographical area where C. ambrosioides is obtained, with variable humidity, temperature and general environmental conditions and the degree of evolutionary and genetic variability (the possible existence of chemotypes within the species) are factors that directly influence the chemical composition of the essential oil obtained from this plant (Gobbo-Neto and Lopes, 2007Gobbo-Neto, L., Lopes, N.P., 2007. Plantas Medicinais: Fatores de influência no conteúdo de metabólitos secundários. Quim. Nova. 30, 374-381; Chekem et al., 2010Chekem, M.S.G., Lunga, P.K., Tamokou, J.D.D., Kuiate, R., Tane, P., Vilarem, G., Cerny, M., 2010. Antifungal properties of Chenopodium ambrosioides essential oil against Candida species. Pharmaceuticals. 3, 2900-2909). However, despite this variability, the essential oil consists mainly of mono and sesquiterpenes (Kliks, 1985Kliks, M.M., 1985. Studies on the traditional herbal anthelmintic Chenopodium ambrosioides L.: enthnopharmacological evalution and clinical field strials. Soc. Sci. Med. 21, 879-886; Cruz et al., 2007Cruz, G.V.B., Pereira, P.V.S., Patrício, F.J., Costa, G.C., Sousa, S.M., Frazao, J.B., Aragão-Filho, W.C., Maciel, M.C.G., Silva, L.A., Amaral, F.M.M., Barroqueiro, E.S.B., Guerra, R.N.M., Nascimento, F.R.F., 2007. Increase of cellular recruitment, phagocytosis ability and nitric oxide production induced by hydroalcoholic extract from Chenopodium ambrosioides leaves. J. Ethnopharmacol. 111, 148-154).
Various studies have been undertaken to characterize the composition of the essential oil of C. ambrosioides by gas chromatography coupled to mass spectrometry (GC/MS). The main compounds found are: (Z)-ascaridole, (E)-ascaridole, carvacrol, p-cymene, α-terpinene and limonene (Cavalli et al., 2004Cavalli, J.F., Tomi, F., Bernardini, A.F., Casanova, J., 2004. Analysis of the essential oil of Chenopodium ambrosioides by GC, GC–MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15, 275-279; Jardim et al., 2008Jardim, C.M., Jham, G.N., Dhingra, O.D., Freire, M.M., 2008. Composition and antifungal activity of the essential oil of the Brazilian Chenopodium ambrosioides L. J. Chem. Ecol. 34, 1213-1218; Chekem et al., 2010Chekem, M.S.G., Lunga, P.K., Tamokou, J.D.D., Kuiate, R., Tane, P., Vilarem, G., Cerny, M., 2010. Antifungal properties of Chenopodium ambrosioides essential oil against Candida species. Pharmaceuticals. 3, 2900-2909; Vieira et al., 2011Vieira, D.F., Azevedo, M.M., Marins, A.K., Pinheiro, P.F., de Queiroz, V.T., Costa, A.V., 2011. Composição química do óleo essencial de Chenopodium ambrosioides L.XI EPG, Anais Encontro latino americano de pós graduação. Alegre – ES, Brasil.). Studies on the essential oil showed antifungal activity against Aspergillus fumigatus, Aspergillus niger, Botryodiplodia theobromae, Fusarium oxysporum, Sclerotium rolfsii, Macrophominapha seolina, Cladosporium cladosporioides, Helminthosporim oryzae, and Pythiumdeba ryanum at a concentration of 100 g/ml (Matos, 2011Matos, J.A.L., (Dissertação de Mestrado) 2011. Potencial Biológico de Chenopodium ambrosioides L. (Erva-de-Santa-Maria). Porto, 51 f. Programa de Pós-graduação em Ciências Farmacêuticas, Universidade Fernando Pessoa.). Monzonte et al. (2007)Monzonte, L., Garcia, M., Montalvo, A., Scull, R., Miranda, M., Abreu, J., 2007. In vitro activity of an essential oil against Leishmania donovani . Phytother. Res. 21, 1055-1058 noted that the essential oil of C. ambrosioides presents in vitro activity against the protozoan Leishmania donovani, causing irreversible inhibition of their growth.
Kinupp (2007)Kinupp, V.F., (Tese de Doutorado) 2007. Plantas alimentícias não-convencionais da região metropolitana de Porto Alegre, RS. 2007. Porto Alegre, 562 f. Faculdade de Agronomia, Universidade Federal do Rio Grande do Sul. describes this species as also being rich in flavonoids and terpenoids and having very diverse pharmacological activities, including antioxidant and chemopreventive effects against cancer, as well as antimicrobial, anti-inflammatory, and analgesic properties (Cruz et al., 2007Cruz, G.V.B., Pereira, P.V.S., Patrício, F.J., Costa, G.C., Sousa, S.M., Frazao, J.B., Aragão-Filho, W.C., Maciel, M.C.G., Silva, L.A., Amaral, F.M.M., Barroqueiro, E.S.B., Guerra, R.N.M., Nascimento, F.R.F., 2007. Increase of cellular recruitment, phagocytosis ability and nitric oxide production induced by hydroalcoholic extract from Chenopodium ambrosioides leaves. J. Ethnopharmacol. 111, 148-154; Dembitsky et al., 2008Dembitsky, V., Shkrob, I., Hanus, L.O., 2008. Ascaridole and related peroxides from the genus Chenopodium . Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2, 209-215; Grassi, 2011Grassi, L.T., (Dissertação de Mestrado) 2011. Chenopodium ambrosioides L. – Erva deSanta Maria (Amaranthaceae): estudo do potencial anti-inflamatório, antinoci-ceptivo e cicatrizante. 2011. Itajaí, 147f. Programa de Pós-graduac¸ ão em CiênciasFarmacêuticas – Universidade do Vale do Itajaí.). According to Hmamouchi et al. (2000)Hmamouchi, M., Lahlou, M., Agoumi, A., 2000. Molluscicidal activity of some Moroccan medicinal plants. Fitoterapia. 71, 308-314, the extract obtained from this species presents potential molluscicidal activity against snails transmitting schistosomiasis, Bulinus truncatus (LC90 = 2.23 mg/l). Patrício et al. (2008)Patrício, F.J., Costa, G.C., Pereira, P.V.S., Aragão-Filho, W.C., Sousa, S.M., Frazão, J.B., Pereira, W.S., Maciel, M.C.G., Silva, L.A., Amaral, F.M.M., Rebêlo, J.M.M., Guerra, R.N.M., Ribeiro, M.N.S., Nascimento, F.R.F., 2008. Efficacy of the intralesional treatment with Chenopodium ambrosioides in the murine infection by Leishmania amazonensis . J. Ethnopharmacol. 115, 313-319, on the other hand, studied the effect of the aqueous crude extract of the leaves of C. ambrosioides on skin ulcers induced by Leishmania amazoniensis in mice. Intralesional treatment was able to inhibit the progression of the ulcer.
Some authors, cited by Matos (2011)Matos, J.A.L., (Dissertação de Mestrado) 2011. Potencial Biológico de Chenopodium ambrosioides L. (Erva-de-Santa-Maria). Porto, 51 f. Programa de Pós-graduação em Ciências Farmacêuticas, Universidade Fernando Pessoa., reported that the anti-inflammatory activity of Chenopodium is due mainly to ascaridole, which is one of the major components of the essential oil of this plant. This same compound can also exhibit antipyretic effects and has been indicated as being responsible for growth inhibition in different tumor cell lines. Its action is so significant in vitro that is a strong candidate for the treatment of cancer (Efferth et al., 2002Efferth, T., Olbrich, A., Sauerbrey, A., Ross, D.D., Gebhart, E., Neugebauer, M., 2002. Activity of ascaridol from the anthelmintic herb Chenopodium anthelminticum L. against sensitive and multidrug-resistant tumor cells. Anticancer Res. 22, 4221-4224).
Given the above, the objective of this study was to obtain and characterize the essential oil and fractions from the hydrolate and ethanol extract of leaves of C. ambrosioides through chromatographic and spectrometric assays (GC-FID, GC/MS, 1H NMR) and evaluate its cytotoxicity in vitro by the MTT method (3(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) in different tumor cell lines: myeloid leukemia (K562), acute B lymphoblastic leukemia (NALM6 and B15), and Burkitt's lymphoma (RAJI), since cancer is a major public health problem worldwide (INCA, 2011INCA, 2011. Estimativa 2012: Incidência de Câncer no Brasil. Instituto Nacional do Câncer http://www.inca.gov.br/estimativa (accessed February 2014).
http://www.inca.gov.br/estimativa...
).
Materials and methods
Plant material
Chenopodium ambrosioides L., Amaranthaceae, was obtained in March 2010 in Cáceres, a city in the state of Mato Grosso, Brazil. Taxonomic identification of the collected material was done by botanist M.Sc. Oscar Benigno Iza by comparison with authentic samples. The exsiccate was deposited in the Herbarium Barbosa Rodrigues (HBR), Itajaí, SC, with the registry number 52802.
Extraction of the essential oil
The essential oil of the leaves of C. ambrosioides, which had been dried and weighed (148 g), was obtained by steam distillation in a Clevenger apparatus. The leaves were extracted with 3 l of distilled water in the flask, and the extraction was performed over a period of 4 h. The oil was removed with a micropipette and, to prevent oxidation reactions due to the remaining water, anhydrous sodium sulfate was added. The oil was stored frozen, protected from light.
Hydrolate and ethanolic extract
After the extraction of the essential oil, the water was filtered to yield about 2.75 l of hydrolate. The leaves used for oil extraction (approximately 185 g) were subjected to maceration in ethanol for 7 days. Approximately 1 l each of the hydrolate and ethanolic extract were subjected to liquid–liquid partitioning with immiscible solvents in increasing order of polarity (dichloromethane, ethyl acetate and butanol). For each solvent, two extractions were performed using first 400 ml and then 200 ml of the solvent. After the separation of the phases, anhydrous sodium sulfate was added to the organic phase to remove the remaining water. The obtained fractions were filtered and concentrated in a rotary evaporator at a maximum temperature of 50 °C to obtain a dry residue. Aliquots of these fractions were sent for analysis by GC/MS.
Analysis by chromatographic and spectroscopic assays
The essential oil obtained from the dried leaves of C. ambrosioides was analyzed by gas chromatography with a flame ionization detector (GC-FID) and by gas chromatography coupled to mass spectrometry (GC/MS Shimadzu QP2010 S). The analysis by GC-FID, with an Rtx-1 capillary column (30 m × 0.25 mm × 0.10 µm), used helium as the carrier gas (0.8 ml/min); the injector temperature was 180 °C and the detector temperature was 250 °C, 1:50 split, using the following temperature program: 80–200 °C at 20 °C/min, 200–300 °C at 15 °C/min; 300–310 °C at 12 °C/min; FID (310 °C); H2: 40 ml/min.
The fractions obtained from the hydrolate and ethanol extract and the essential oil were analyzed by GC/MS with an injector temperature of 250 °C; the temperature program was 80–200 °C at 20 °C/min; 200–300 °C at 15 °C/min, with other conditions equal to those of GC-FID. The identification of the chemical composition of the essential oil and fractions was performed by comparing the mass spectra obtained with the data available in the library (NIST version 8.0).
The 1H NMR spectra were obtained on a Bruker AC-300 MHz 300F. Spectra were obtained in deuterated chloroform (purity 99.8% + 0.05% TMS) obtained from Cambridge Isotope Laboratories Inc., with tetramethylsilane as the internal reference (TMS). Chemical shifts were recorded in dimensionless values δ (ppm) indicating the sign as singlet (s), doublet (d), triplet (t), etc.
1H NMR (CDCl3, 300 MHz) essential oil consisting mainly of ascaridole and p-cymene. Ascaridole δ: 1.03 (d, J = 6.9, H9, H10), 1.39 (s, H7), 1.52 (d, J = 9.0, H5), 1.91 (sept, J = 6.9, H8), 2.07 (d, J = 9.0, H6), 6.43 (d, J = 8.7, H3), 6.51 (d, J = 8.7, H2). p-Cymene δ: 1.26 (d, J = 6.9, H9, H10), 2.34 (s, H7), 2.90 (n, J = 6.9, H8), 7.16 (s, H3, H4, H5, H6).
The essential oil, ethanol extract and dichloromethane, ethyl acetate and butanol fractions from the extract were evaluated for cytotoxicity using K562 (myeloid leukemia), Nalm6 and B15 (acute B lymphoblastic leukemia), and RAJI (Burkitt's lymphoma) cells. Cell viability was assessed by the MTT (3 (4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay, which is based on a method using a dye, i.e. a tetrazolium salt soluble that is in water which is converted to purple formazan after reduction by mitochondrial dehydrogenases in viable cells (Mosmann, 1983Mosmann, T., 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65, 55-63).
Cells were grown in appropriate plastic dishes with DMEM (Dulbecco's Modified Eagle's Minimum Essential Medium), supplemented with fetal calf serum (FBS) 10% inactivated for 1 h at 56 °C, 10 mM of ethanesulfonic acid hydroxyethyl piperazine (HEPES), 1.5 g/l of sodium bicarbonate, 1% of penicillin G (100 U/ml), 100 mg/ml of streptomycin, 50 µg/ml of amphotericin B in an incubator humidified at 37 °C with 5% CO2 emissions. Before the experiments, the number of viable cells was determined by the trypan blue exclusion method, with counts performed in a Neubauer chamber.
Cells were plated using epMotion® 5070 equipment (Eppendorf, Vaudaux, Schonenbuch, Switzerland) which distributed 2 × 104 cells per well in 96-well plates, which were incubated with the test substances at different concentrations (0.01, 0.1, 1, 10, 100 and 1000 mg/ml) for 48 h at 37 °C in 5% CO2. DMSO was used for solubilization of the test substances in a maximum concentration of 0.1% well/treatment; this concentration is not cytotoxic to the cells. Doxorubicin at the same concentrations tested was used as a positive standard.
After the treatment period, the medium was removed and 100 µl of the MTT solution was added at 0.5 mg/ml in the culture medium and incubated for 4 h. After that time, the medium was removed and the formazan precipitate was dissolved in 100 µl DMSO/well and reading was performed on a microplate reader at 540 nm (Bio-Tek Power Wave XS). The optical density obtained in the control group, i.e. untreated cells (incubated with growth medium only), was regarded as 100% viable cells in order to establish concentration vs. response curves, and therefore the IC50 (concentration inhibiting 50% growth of cell), according to the equation below. Each experiment was performed in triplicate and repeated at least three times. Results are expressed as mean ± standard deviation values. For IC50 calculation was used nonlinear regression Prism 5.0 software for windows
Results and discussion
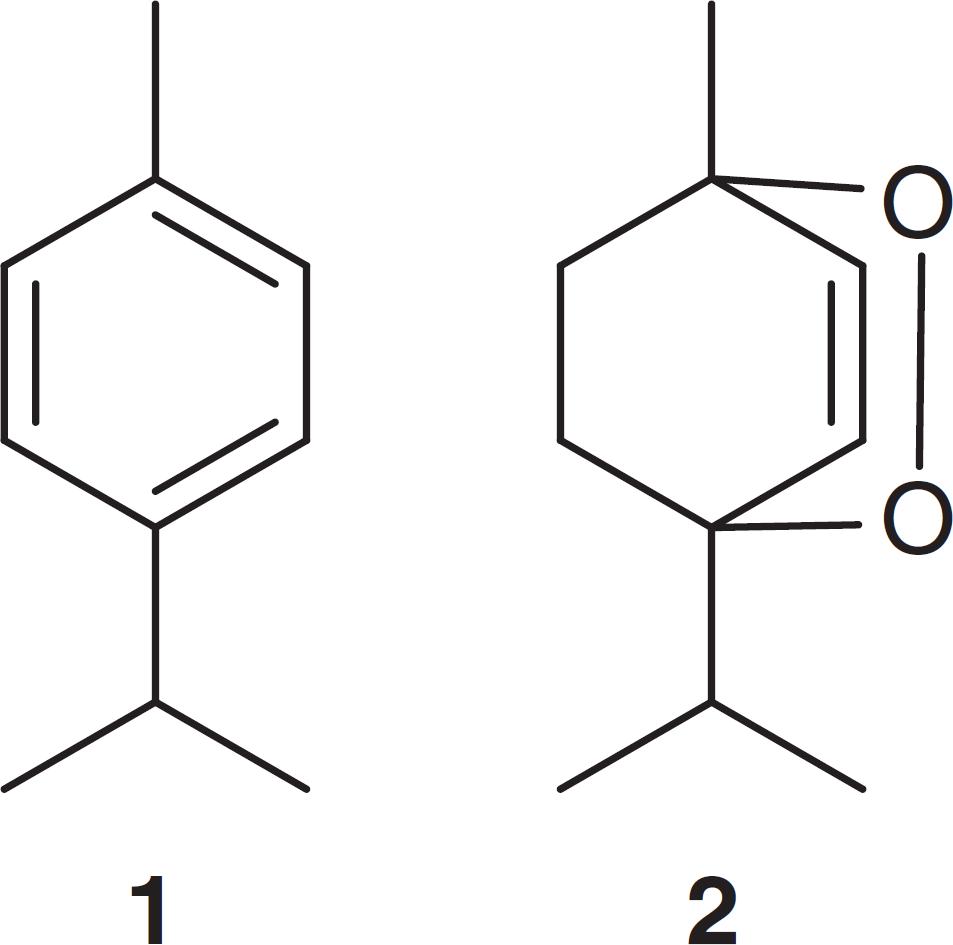
The process of extracting the essential oil from C. ambrosioides was conducted by steam distillation with a Clevenger apparatus. The essential oil obtained was called CAEO, with a yield of 1.15%. After extraction, an aliquot of the oil was analyzed by GC-FID (Fig. 1), displaying two major peaks with retention times of 3.614 and 5.064 min, which were identified by comparison of the mass spectra in the standards of the NIST 8.0 library as 1-isopropyl-4-methyl-benzene (p-cymene or p-cymol 1 ) (42.32%) and 1-isopropyl-4-methyl-2,3 dioxabicyclo [2.2.2] oct-5-ene (ascaridole 2 ) (49.77%), respectively. These two monoterpenes amounted to 92.09% of the essential oil. Of the remaining 7.91% of the essential oil, only two compounds showed percentages above 1%, but these were not identified in NIST library version 8.0.
Chromatogram obtained by GC-FID (180 °C) of the essential oil of Chenopodium ambrosioides (CAEO).
According to the literature, it is known that the chemical composition of the oil varies greatly according to its region of origin and its method of extraction. As a result, variations in the percentage of analyzed compounds can be observed. p-Cymene, shown to make up 42.32% of the essential oil, differed little from a study by Tapondjou et al. (2002)Tapondjou, L.A., Adler, C., Bouda, H., Fontem, D.A., 2002. Efficacy of powder and essential oil from Chenopodium ambrosioides leaves as postharvest grain protectants against six-stored product beetles. J. Stored Prod. Res. 38, 395-402, who found that the oil contained 50% p-cymene. Research by Jardim et al. (2008)Jardim, C.M., Jham, G.N., Dhingra, O.D., Freire, M.M., 2008. Composition and antifungal activity of the essential oil of the Brazilian Chenopodium ambrosioides L. J. Chem. Ecol. 34, 1213-1218 on the essential oil of C. ambrosioides obtained in Brazil, Cavalli et al. (2004)Cavalli, J.F., Tomi, F., Bernardini, A.F., Casanova, J., 2004. Analysis of the essential oil of Chenopodium ambrosioides by GC, GC–MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15, 275-279 in Madagascar, Chekem et al. (2010)Chekem, M.S.G., Lunga, P.K., Tamokou, J.D.D., Kuiate, R., Tane, P., Vilarem, G., Cerny, M., 2010. Antifungal properties of Chenopodium ambrosioides essential oil against Candida species. Pharmaceuticals. 3, 2900-2909 in Cameroon and Singh et al. (2008)Singh, H.P., Batish, D.R., Kohli, R.K., Mittal, S., Yadav, S., 2008. Chemical composition of essential oil from leaves of Chenopodium ambrosioides from Chandigarh, India. Chem. Nat. Compd. 44, 378-379 in India, showed lower concentrations of p-cymene, with percentages of 2.0, 16.2, 23.4 and 25.77%, respectively.
Ascaridole appeared at a concentration of 49.77% in the oil. This percentage was close to that found in a study by Cavalli et al. (2004)Cavalli, J.F., Tomi, F., Bernardini, A.F., Casanova, J., 2004. Analysis of the essential oil of Chenopodium ambrosioides by GC, GC–MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15, 275-279, who found that the oil contained 55.3% ascaridole. This percentage differed greatly compared to that found in a study by Vieira et al. (2011)Vieira, D.F., Azevedo, M.M., Marins, A.K., Pinheiro, P.F., de Queiroz, V.T., Costa, A.V., 2011. Composição química do óleo essencial de Chenopodium ambrosioides L.XI EPG, Anais Encontro latino americano de pós graduação. Alegre – ES, Brasil., which found ascaridole as the major compound (87%). On the other hand, Borges et al. (2012)Borges, R.A., Aires, J.R., Higino, T.M., de Medeiros, M.D., Citó, A.M., Lopes, J.A., de Figueiredo, R.C., 2012. Trypanocidal and cytotoxic activities of essential oils from medicinal plants of Northeast of Brazil. Exp. Parasitol. 132, 123-128 found less ascaridole in the oil obtained from the plant collected in northeastern Brazil (17.1%). In a study on the essential oil from India, ascaridole was present as only 7% of the oil composition (Gupta et al., 2002Gupta, D., Charles, R., Mehta, V.K., Garg, S.N., Kumar, S., 2002. Chemical examination of the essential oil of Chenopodium ambrosioides L. from the southern hills of India. J. Essent. Oil Res. 14, 93-94).
According to Johnson and Croteau (1984)Johnson, M.A., Croteau, R., 1984. Biosynthesis of ascaridole: iodide peroxidase-catalyzed synthesis of monoterpene endoperoxide in soluble extracts of Chenopodium ambrosioides fruit. Arch. Biochem. Biophys. 235, 254-266, ascaridole is a sensitive heat component, which has a biosynthetic pathway related to the formation of α-terpinene. Ascaridole may undergo changes in its conformation and become isoascaridole. Johnson and Croteau (1984)Johnson, M.A., Croteau, R., 1984. Biosynthesis of ascaridole: iodide peroxidase-catalyzed synthesis of monoterpene endoperoxide in soluble extracts of Chenopodium ambrosioides fruit. Arch. Biochem. Biophys. 235, 254-266 assessed the thermal isomerization in ascaridole to isoascaridole by GC analysis of leaves and fruits of C. ambrosioides when the injector temperature was above 200 °C. The thermal structural rearrangement of ascaridole is known, but there have been few studies on this reaction (Boche and Runquist, 1968Boche, J., Runquist, O., 1968. Kinetics of the thermal rearrangement of ascaridole. J. Org. Chem. 33, 4285-4286). Cavalli et al. (2004)Cavalli, J.F., Tomi, F., Bernardini, A.F., Casanova, J., 2004. Analysis of the essential oil of Chenopodium ambrosioides by GC, GC–MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15, 275-279 reported that during the characterization of the essential oil of C. ambrosioides from Madagascar, using GC-FID and GC/MS assays, the partial isomerization of ascaridole into isoascaridole occurred.
In the present study, when the oil was analyzed by GC/MS with an injection temperature of 250 °C it was observed that ascaridole (retention time of 5.550 min) was converted into its isomer, isoascaridole (retention time of 6.001 min). So, the oil was also evaluated by nuclear magnetic resonance of hydrogen (1H NMR) (Fig. 2). The spectrum was obtained at room temperature and no sign was detected that could be attributed to isoascaridole. This technique has become an excellent tool for the analysis of essential oils because it allows for evaluating all hydrogens present in a matrix, which can be complex. All signals observed in the 1H NMR spectrum were assigned to the hydrogens of the two major compounds. The signals relative to the other hydrogens of the compounds were observed as noise in the baseline. From the analysis of the integral of the signals, the ratio between p-cymene and ascaridole could be established, which was 1:1.2. These data are consistent with the information obtained by GC-FID.
After extraction of the essential oil, the hydrolate was submitted to partitioning with solvents of increasing polarity. These fractions were called: partition hydrolate dichloromethane (PHDCM), partition hydrolate ethyl acetate (PHEA), and partition hydrolate butanol (PHB). The first two were subjected to analysis by GC/MS for characterization, while the latter, due to its high polarity, was not analyzed by this technique.
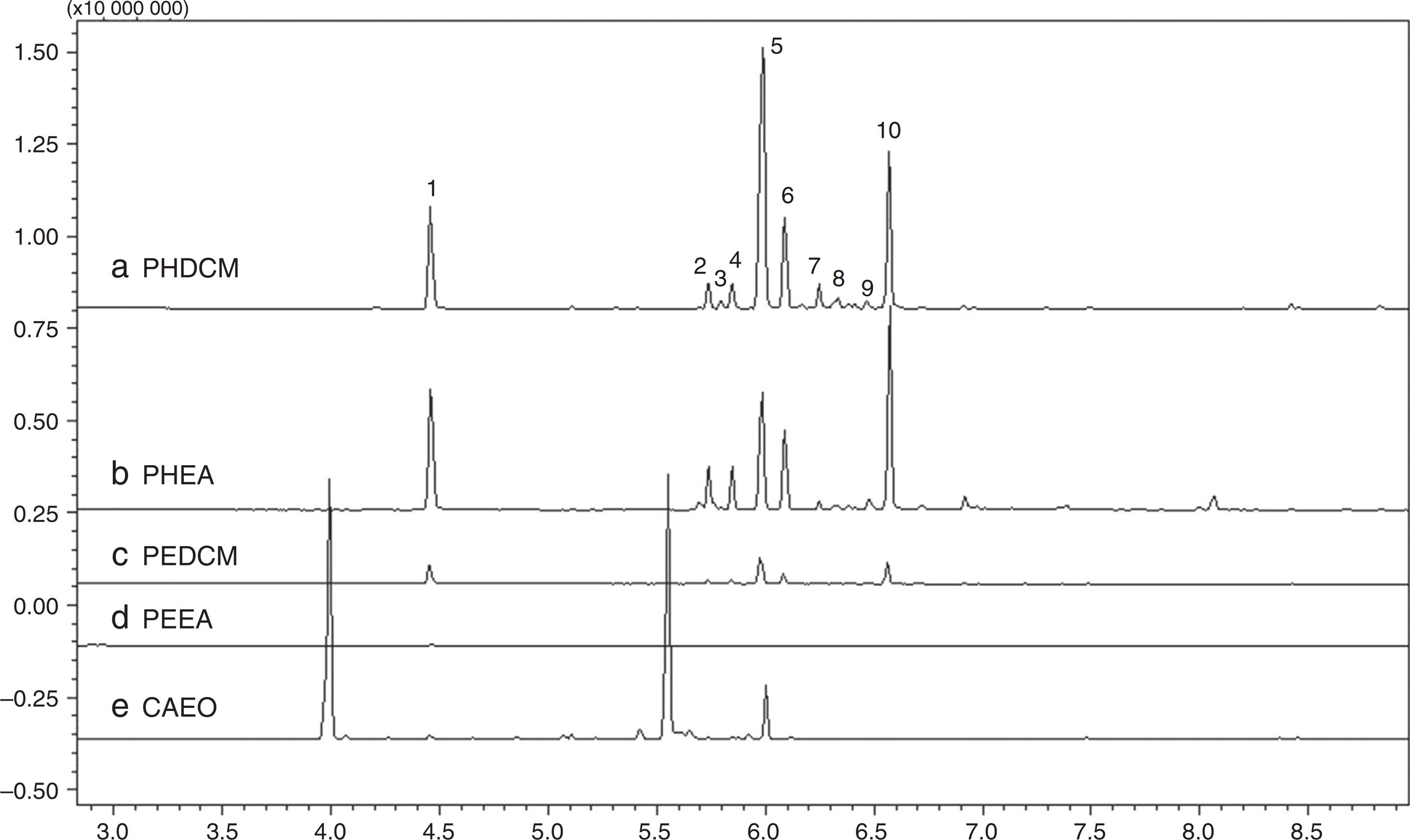
The leaves used for extraction of the oil, in turn, were further subjected to extraction by maceration with ethanol. The extract was also subjected to the liquid–liquid partition process. The fractions were called: partition extract dichloromethane (PEDCM); partition extract ethyl acetate (PEEA); partition extract butanol (PEB). These fractions, except for PEB, were subsequently analyzed by GC/MS. The chromatograms obtained from fractions of the DCM and AE hydrolates and the ethanol extract of C. ambrosioides were compared and are shown in Fig. 3.
Gas chromatogram obtained by GC/MS (250 °C) of samples (a) PHDCM, (b) PHEA, (c) PEDCM, (d) PEEA, (e) CAEO.
The chromatograms of the fractions PHDCM, PHEA, and PEDCM had peaks with similar retention times, indicating a similar chemical composition, differing only in their concentrations (Table 1). Hydrolate partitioning was not efficient because the same compounds were detected in different fractions.
Comparison of percentage of the compounds analyzed by GC/MS in the dichloromethane (PHDCM) and ethyl acetate (PHEA) hydrolate fractions and in the fraction of dichloromethane of ethanol extract (PEDCM).
The mass spectra obtained from the hydrolate fraction DCM was compared with the mass spectra in NIST library version 8.0, but no compound was identified. It is suggested that many of these compounds are derived from the degradation of the main compounds in the essential oil due to heating during extraction. Peaks 5, 6, and 7, with retention times of 5.99, 6.09, and 6.25 min, respectively, correspond to isomers of each other, since they have the same molecular weight (170), one of which is possibly dihydroascaridol, and others its isomers. Compound 10, corresponding to a retention time of 6.57 min, is an isomer of ascaridol, with an identical molecular mass (168).
After the characterization of essential oil and fractions obtained from the ethanol extract and hydrolate, samples CAEO, CAEE, PEDCM, PEEA and PEB were tested on different tumor cell lines: myeloid leukemia (K562), acute B lymphocytic leukemia (NALM6 and B15), and Burkitt's lymphoma (RAJI). The results (Table 2) show the potential cytotoxicity of the tested samples, especially CAEO, which showed an extremely low IC50 of 1 mg/ml on RAJI cells, compared to the IC50 of 13.2 mg/ml of doxorubicin (positive control). The fractions PEDCM and CAEE also showed lower IC50 values in K562 cells compared to doxorubicin (62.5 mg/ml), with values of 34 and 47 mg/ml, respectively.
Cytotoxic effect (IC50 µg/ml) of essential oil and fractions of the extract of Chenopodium ambrosioides and positive control (doxorubicin) against K562, NALM6, B15, and RAJI cells.
Efferth et al. (2002)Efferth, T., Olbrich, A., Sauerbrey, A., Ross, D.D., Gebhart, E., Neugebauer, M., 2002. Activity of ascaridol from the anthelmintic herb Chenopodium anthelminticum L. against sensitive and multidrug-resistant tumor cells. Anticancer Res. 22, 4221-4224 isolated ascaridole from a commercial preparation of the essential oil of C. ambrosioides and analyzed in it using different strains of tumor cells in vitro (CCRF-CEM–human lymphoblastic leukemia T cells, HL60 – promyelocytic leukemia, and MDA MB-231 – breast cancer). According to their results, it was concluded that ascaridole showed cytotoxicity in the analyzed cells. This study was the first to demonstrate the importance of ascaridole as a possible candidate for the treatment of cancer, and reveals the need for further study of its cytotoxic action. Thus, it can be suggested that the antitumor effect of the essential oil in Burkitt's lymphoma cells (RAJI) in the present study may be related to the high concentration of ascaridole.
Nascimento et al. (2006)Nascimento, F.R.F., Cruz, G.V.B., Pereira, P.V.S., Maciel, M.C.G., Silva, L.A., Azevedo, A.P.S., Barroqueiro, E.S.B., Guerra, R.N.M., 2006. Ascitic and solid Ehrlich tumor inhibition by Chenopodium ambrosioides L. treatment. Life Sci. 78, 2650-2653, on the other hand, investigated the treatment effect of a hydroalcoholic extract of C. ambrosioides in Ehrlich ascites tumor cells and observed that this species was able to inhibit tumor growth when administered prior to tumor implantation, i.e. two days after implantation. The authors also suggested that this effect may be related to the antioxidant properties of C. ambrosioides.
The activity of the essential oil of C. ambrosioides is probably related to the large amount of ascaridol, since the other major compound, p-cymene, is recognized as a potent anti-inflammatory (Chen et al., 2013Chen, L., Zhao, L., Zhang, C., Lan, Z., 2013. Protective effect of p-cymene on lipopolysaccharide-induced acute lung injury in mice. Inflammation. 2, 358-364) and has low cytotoxic activity (Koba et al., 2009Koba, K., Catherine, G., Raynaud, C., Chaumont, J.P., Sanda, K., Laurence, N., 2009. Chemical composition and cytotoxic activity of Chenopodium ambrosioides L. essential oil from Togo. Bangladesh J. Sci. Ind. Res. 44, 435-440).
Through the analysis of the essential oil of C. ambrosioides by GC/MS and 1H NMR, it was possible to identify two major compounds, i.e. ascaridole and p-cymene, which were identified in previous research and have been found in different concentrations. This indicates the dependence of the region of collection and extraction conditions on the isolation of these compounds.
The essential oil of C. ambrosioides and its fractions showed cytotoxic potential. However, there are still few studies investigating antitumor potential of C. ambrosioides. Studies further to the present study toward identifying the mechanisms involved in inhibiting the proliferation of K562 and Raji tumor cells are required.
Acknowledgments
The authors thank CNPq, Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina and the Universidade do Vale de Itajaí for providing financial support (ProBIC ProPPEC/UNIVALI).
Ethical disclosures
-
Confidentiality of data . The authors declare that no patient data appear in this article.
-
Right to privacy and informed consent . The authors declare that no patient data appear in this article.
-
Protection of human and animal subjects . The authors declare that no experiments were performed on humans or animals for this study.
References
- Alitonou, G.A., Sessou, P., Tchobo, F.P., Noudogbessi, J., Avlessi, F., Yehouenou, B., Menut, C., Villeneuve, P., Sohounhloue, D.C.K., 2012. Chemical composition and biological activities of essential oils of Chenopodium ambrosioides L. collected in two areas of Benin. Int. J. Biosci. 2, 58-66
- Boche, J., Runquist, O., 1968. Kinetics of the thermal rearrangement of ascaridole. J. Org. Chem. 33, 4285-4286
- Borges, R.A., Aires, J.R., Higino, T.M., de Medeiros, M.D., Citó, A.M., Lopes, J.A., de Figueiredo, R.C., 2012. Trypanocidal and cytotoxic activities of essential oils from medicinal plants of Northeast of Brazil. Exp. Parasitol. 132, 123-128
- Cavalli, J.F., Tomi, F., Bernardini, A.F., Casanova, J., 2004. Analysis of the essential oil of Chenopodium ambrosioides by GC, GC–MS and 13C-NMR spectroscopy: quantitative determination of ascaridole, a heat-sensitive compound. Phytochem. Anal. 15, 275-279
- Chekem, M.S.G., Lunga, P.K., Tamokou, J.D.D., Kuiate, R., Tane, P., Vilarem, G., Cerny, M., 2010. Antifungal properties of Chenopodium ambrosioides essential oil against Candida species. Pharmaceuticals. 3, 2900-2909
- Chen, L., Zhao, L., Zhang, C., Lan, Z., 2013. Protective effect of p-cymene on lipopolysaccharide-induced acute lung injury in mice. Inflammation. 2, 358-364
- Cruz, G.V.B., Pereira, P.V.S., Patrício, F.J., Costa, G.C., Sousa, S.M., Frazao, J.B., Aragão-Filho, W.C., Maciel, M.C.G., Silva, L.A., Amaral, F.M.M., Barroqueiro, E.S.B., Guerra, R.N.M., Nascimento, F.R.F., 2007. Increase of cellular recruitment, phagocytosis ability and nitric oxide production induced by hydroalcoholic extract from Chenopodium ambrosioides leaves. J. Ethnopharmacol. 111, 148-154
- De Feo, V., Senatore, F., 1993. Medicinal plants and phytotherapy in the Amalfitan Coast, Salerno Province, Campania, southern Italy. J. Ethnopharmacol. 39, 39-51
- Dembitsky, V., Shkrob, I., Hanus, L.O., 2008. Ascaridole and related peroxides from the genus Chenopodium . Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2, 209-215
- Efferth, T., Olbrich, A., Sauerbrey, A., Ross, D.D., Gebhart, E., Neugebauer, M., 2002. Activity of ascaridol from the anthelmintic herb Chenopodium anthelminticum L. against sensitive and multidrug-resistant tumor cells. Anticancer Res. 22, 4221-4224
- Gobbo-Neto, L., Lopes, N.P., 2007. Plantas Medicinais: Fatores de influência no conteúdo de metabólitos secundários. Quim. Nova. 30, 374-381
- Grassi, L.T., (Dissertação de Mestrado) 2011. Chenopodium ambrosioides L. – Erva deSanta Maria (Amaranthaceae): estudo do potencial anti-inflamatório, antinoci-ceptivo e cicatrizante. 2011. Itajaí, 147f. Programa de Pós-graduac¸ ão em CiênciasFarmacêuticas – Universidade do Vale do Itajaí.
- Gupta, D., Charles, R., Mehta, V.K., Garg, S.N., Kumar, S., 2002. Chemical examination of the essential oil of Chenopodium ambrosioides L. from the southern hills of India. J. Essent. Oil Res. 14, 93-94
- Hmamouchi, M., Lahlou, M., Agoumi, A., 2000. Molluscicidal activity of some Moroccan medicinal plants. Fitoterapia. 71, 308-314
- INCA, 2011. Estimativa 2012: Incidência de Câncer no Brasil. Instituto Nacional do Câncer http://www.inca.gov.br/estimativa (accessed February 2014).
» http://www.inca.gov.br/estimativa - Jardim, C.M., Jham, G.N., Dhingra, O.D., Freire, M.M., 2008. Composition and antifungal activity of the essential oil of the Brazilian Chenopodium ambrosioides L. J. Chem. Ecol. 34, 1213-1218
- Johnson, M.A., Croteau, R., 1984. Biosynthesis of ascaridole: iodide peroxidase-catalyzed synthesis of monoterpene endoperoxide in soluble extracts of Chenopodium ambrosioides fruit. Arch. Biochem. Biophys. 235, 254-266
- Kinupp, V.F., (Tese de Doutorado) 2007. Plantas alimentícias não-convencionais da região metropolitana de Porto Alegre, RS. 2007. Porto Alegre, 562 f. Faculdade de Agronomia, Universidade Federal do Rio Grande do Sul.
- Kliks, M.M., 1985. Studies on the traditional herbal anthelmintic Chenopodium ambrosioides L.: enthnopharmacological evalution and clinical field strials. Soc. Sci. Med. 21, 879-886
- Koba, K., Catherine, G., Raynaud, C., Chaumont, J.P., Sanda, K., Laurence, N., 2009. Chemical composition and cytotoxic activity of Chenopodium ambrosioides L. essential oil from Togo. Bangladesh J. Sci. Ind. Res. 44, 435-440
- Kokanova-Nedialkova, Z., Nedialkov, P.T., Nikolov, S.D., 2009. The genus Chenopodium: phytochemistry, ethnopharmacology and pharmacology. Pharmacogn. Rev. 3, 280-306
- Lorenzi, H., Matos, F.J.A., 2002. Plantas medicinais no Brasil: nativas e exóticas cultivadas. Instituto Plantarum de Estudos da Flora, Nova Odessa, SP.
- Matos, J.A.L., (Dissertação de Mestrado) 2011. Potencial Biológico de Chenopodium ambrosioides L. (Erva-de-Santa-Maria). Porto, 51 f. Programa de Pós-graduação em Ciências Farmacêuticas, Universidade Fernando Pessoa.
- Monzonte, L., Garcia, M., Montalvo, A., Scull, R., Miranda, M., Abreu, J., 2007. In vitro activity of an essential oil against Leishmania donovani . Phytother. Res. 21, 1055-1058
- Mosmann, T., 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65, 55-63
- Nascimento, F.R.F., Cruz, G.V.B., Pereira, P.V.S., Maciel, M.C.G., Silva, L.A., Azevedo, A.P.S., Barroqueiro, E.S.B., Guerra, R.N.M., 2006. Ascitic and solid Ehrlich tumor inhibition by Chenopodium ambrosioides L. treatment. Life Sci. 78, 2650-2653
- Patrício, F.J., Costa, G.C., Pereira, P.V.S., Aragão-Filho, W.C., Sousa, S.M., Frazão, J.B., Pereira, W.S., Maciel, M.C.G., Silva, L.A., Amaral, F.M.M., Rebêlo, J.M.M., Guerra, R.N.M., Ribeiro, M.N.S., Nascimento, F.R.F., 2008. Efficacy of the intralesional treatment with Chenopodium ambrosioides in the murine infection by Leishmania amazonensis . J. Ethnopharmacol. 115, 313-319
- Singh, H.P., Batish, D.R., Kohli, R.K., Mittal, S., Yadav, S., 2008. Chemical composition of essential oil from leaves of Chenopodium ambrosioides from Chandigarh, India. Chem. Nat. Compd. 44, 378-379
- Tapondjou, L.A., Adler, C., Bouda, H., Fontem, D.A., 2002. Efficacy of powder and essential oil from Chenopodium ambrosioides leaves as postharvest grain protectants against six-stored product beetles. J. Stored Prod. Res. 38, 395-402
- Vieira, D.F., Azevedo, M.M., Marins, A.K., Pinheiro, P.F., de Queiroz, V.T., Costa, A.V., 2011. Composição química do óleo essencial de Chenopodium ambrosioides L.XI EPG, Anais Encontro latino americano de pós graduação. Alegre – ES, Brasil.
Publication Dates
-
Publication in this collection
Jan-Feb 2016
History
-
Received
13 May 2015 -
Accepted
18 Aug 2015