ABSTRACT
This study evaluated the anti-inflammatory and antioxidant activities of Impatiens noli-tangere L., Balsaminaceae, and of Stachys officinalis L., Lamiaceae, polyphenolic-rich extracts obtained by nanofiltration process. Results showed the great potential and efficiency of the nanofiltration process to concentrate the herbal extract's main polyphenolic compounds (over 91% phenolic acids and flavonoids retention). S. officinalis polyphenolic-rich extracts had high antioxidant activities (IC50 2.5 µg/ml) compared to I. noli-tangere polyphenolic-rich extracts (IC50 19.3 µg/ml) and similar with that of ascorbic acid. Polyphenolic-rich extracts were investigated to determine the pro-inflammatory enzymes lipoxygenase, cyclooxygenase-1 and cyclooxygenase-2 and their inhibitory activity. Furthermore, high inhibitory activity of the examined extracts was reported for the first time, for both lipoxygenase (IC50 2.46 and 1.22 µg/ml for I. noli-tangere and S. officinalis polyphenolic-rich extracts, respectively), cyclooxygenase-1 (IC50 18.4 and 10.1 µg/ml for I. noli-tangere and S. officinalis polyphenolic-rich extracts, respectively) and cyclooxygenase-2 (IC50 = 1.9 and 1.2 mg/ml for I. noli-tangere and S. officinalis polyphenolic-rich extracts, respectively). Additionally, the in vivo studies showed that S. officinalis polyphenolic-rich extract has a higher anti-inflammatory effect, the hind-paw volume employed for both models determined that I. noli-tangere polyphenolic-rich extract and is also higher than that of diclofenac. It was noticed that their anti-inflammatory effect persists for more than 24 h. The I. noli-tangere and S. officinalis polyphenolic-rich extracts exert anti-inflammatory and antioxidant activities and these properties can be at least partly assigned to the presence of ursolic acid, caffeic acid, rosmarinic acid, quercetin and also anthocyanidins (genistin). The obtained results indicate the anti-inflammatory potential of the studied herbal extracts.
Keywords:
Polyphenolic-rich extract; Nanofiltration; Antioxidant; Lipoxygenase; Cyclooxygenase; In vivo anti-inflammatory
Introduction
Nowadays, the increasing rate of the inflammatory diseases requires new alternative solutions to the nonsteroidal anti-inflammatory drugs and a wider range of inflammatory products, as well. Inflammation is a normal biological process in response to tissue injury, microbial pathogen infection and chemical irritation. Wound healing is a natural and fundamental histopathological process that restores the function and the integrity of the damaged tissues. This process includes three overlapping phases: hemostasis and inflammation, tissue formation and remodeling (Clark, 1995Clark, R.A.F., 1995. Wound repair: overview and general considerations. In: The Molecular Cellular Biology of Wound Repair, 2nd edition. Plenum Press, NewYork, pp. 513–560.). Therefore, a healing extract should exhibit anti-inflammatory and antioxidant activities in order to have high efficiency healing. Investigations performed over the last years proved that the Lipoxygenase (LOX) and Cyclooxygenase (COX-1 and COX-2) pathways play an important role in the inflammatory disorder's development. In the same way, the drugs that inhibit LOX concurrently with COX-1 and COX-2 may enhance tissue regeneration (Martel-Pelletier et al., 2003Martel-Pelletier, J., Lajeunesse, D., Reboul, P., Pelletier, J., 2003. Therapeutic role of dual inhibitors of 5-LOX and COX, selective and non-selective non-steroidal anti-inflammatory drugs. Ann. Rheum. Dis. 62, 501-509.; Hayashi et al., 2011Hayashi, S., Sumi, Y., Ueno, N., Murase, A., Takada, J., 2011. Discovery of a novel COX-2 inhibitor as an orally potent anti-pyretic and anti-inflammatory drug: design, synthesis, and structure-activity relationship. Biochem. Pharmacol. 82, 755-768.; Steinhilber and Hofmann, 2014Steinhilber, D., Hofmann, B., 2014. Recent advances in the search for novel 5-lipoxygenase inhibitors. Basic Clin. Pharmacol. Toxicol. 114, 70-77.). Leukotriene (LTB4), produced by the oxidation of arachidonic acid by 5-LOX, is known to be an active chemo-attractant for fibroblasts and monocytes and may regulate fibroblast activities in the skin's wound healing (Jovanovic et al., 2001Jovanovic, D., Fernandes, J., Martel-Pelletier, J., Jolicoeur, F., Reboul, P., Laufer, S., Tries, S., Pelletier, J., 2001. In vivo dual inhibition of cyclooxygenase and lipoxygenase by ML-3000 reduces the progression of experimental osteoarthritis: suppression of collagenase 1 and interleukin-1beta synthesis. Arthritis Rheum. 44, 2320-2330.). COX, may also be involved in wound healing because it appears to be associated with directed cell motility and new tissue's growth in airway epithelium, and skin (Savla et al., 2001Savla, U., Appel, H.J., Sporn, P.H., Waters, C.M., 2001. Prostaglandin E2 regulates wound closure in airway epithelium. Am. J. Physiol. Lung Cell. Mol. Physiol. 280, L421-L431.). Antioxidants exert defensive effects against oxidative stress, by scavenging free radicals (Valko et al., 2007Valko, M., Leibfritz, D., Moncol, J., Cronin, M.T.D., Mazur, M., Telser, J., 2007. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 39, 44-84.). Recent researches findings indicated that plant based antioxidants represent great therapeutic agents to combat oxidative stress closely associated with chronic inflammation (Njenga and Viljoen, 2006Njenga, E.W., Viljoen, A.M., 2006. In vitro 5-lipoxygenase inhibition andanti-oxidant activity of Eriocephalus L. (Asteraceae) species. S. Afr. J. Bot. 72, 637-641.). Hence, antioxidants are believed to accelerate wound healing. (Fitzmaurice et al., 2011Fitzmaurice, S.D., Sivamani, R.K., Isseroff, R.R., 2011. Antioxidant therapies for wound healing: a clinical guide to currently commercially available products. Skin Pharmacol. Phys. 24, 113-126.).
Impatiens noli-tangere L., Balsaminaceae, commonly known as touch-me-not balsam is an annual herbaceous plant found in damp places and forests in Europe, Asia and North America. Plants belonging to the genus Impatiens are rich in organic acids, anthraquinones, flavonoids and phenolic acids (Choi and Kim, 2002Choi, B.J., Kim, C.W., 2002. Studies on the constituents of Impatiens noli-tangere L. Korean J. Pharmacogn. 33, 263-266.; Paun et al., 2016Paun, G., Neagu, E., Albu, C., Moroeanu, V., Radu, G.L., 2016. Antioxidant activity and inhibitory effect of polyphenolic-rich extract from Betonica officinalis and Impatiens noli-tangere herbs on key enzyme linked to type 2 diabetes. J. Taiwan Inst. Chem. Eng. 60, 1-7.).
Stachys officinalis L. (syn. Betonica officinalis L.), commonly known as wood betony or Bishop's wort is a perennial herb and is one of the largest genus of the Lamiaceae family. S. officinalis contains various alkaloids, saponins, flavonoids and phenolic acids and has been used in traditional medicine to treat various disorders such as: respiratory tract, gastrointestinal tract, nervous system, inflammatory disease and liver disorders (Háznagy-Radnai et al., 2012Háznagy-Radnai, E., Balogh, Ú., Czigle, S., Máthé, I., Hohmann, J., Blazsõ, G., 2012. Antiinflammatory activities of hungarian stachys species and their iridoids. Phytother. Res. 26, 505-509.; Vogl et al., 2013Vogl, S., Picker, P., Mihaly-Bison, J., Fakhrudin, N., Atanasov, A.G., Heiss, E.H., Wawrosch, C., Reznicek, G., Dirsch, V.M., Saukel, J., Kopp, B., 2013. Ethnopharmacological in vitro studies on Austria's folk medicine – an unexplored lore in vitro anti-inflammatory activities of 71 Austrian traditional herbal drugs. J. Ethnopharmacol. 149, 750-771.; Šliumpaite et al., 2013Šliumpaite, I., Venskutonis, P.R., Murkovic, M., Ragažinskiene, O., 2013. Antioxidant properties and phenolic composition of wood betony (Betonica officinalis L., syn. Stachys officinalis L.). Ind. Crops Prod. 50, 715-722.; Rigat et al., 2015Rigat, M., Vallès, J., Dambrosio, U., Gras, A., Iglésias, J., Garnatje, T., 2015. Plants with topical uses in the ripollès district (pyrenees, catalonia, iberian peninsula): ethnobotanical survey and pharmacological validation in the literature. J. Ethnopharmacol. 164, 162-179..
In Romanian traditional medicine I. noli-tangere and S. officinalis are known for their antioxidative, anti-inflammatory, astringent, hemostatic and wound healing properties (Ardelean and Mohan, 2008Ardelean, A., Mohan, Gh., 2008. Flora medicinala a Romaniei. Ed.ALL, Bucharest.; Vogl et al., 2013Vogl, S., Picker, P., Mihaly-Bison, J., Fakhrudin, N., Atanasov, A.G., Heiss, E.H., Wawrosch, C., Reznicek, G., Dirsch, V.M., Saukel, J., Kopp, B., 2013. Ethnopharmacological in vitro studies on Austria's folk medicine – an unexplored lore in vitro anti-inflammatory activities of 71 Austrian traditional herbal drugs. J. Ethnopharmacol. 149, 750-771.; Jarić et al., 2017Jarić, S., Kostić, O., Mataruga, Z., Pavlović, D., Pavlović, M., Mitrović, M., Pavlović, P., 2017. Traditional wound-healing plants used in the Balkan region (Southeast Europe). J. Ethnopharmacol., http://dx.doi.org/10.1016/j.jep.2017.09.018.
http://dx.doi.org/10.1016/j.jep.2017.09....
). The literature provides limited information about the chemical composition and biological activity of I. noli-tangere. However, the anti-inflammatory effects of I. noli-tangere and S. officinalis polyphenolic-rich extracts have not been studied.
Material and methods
Samples and reagents
Phenolic compound standards were purchased from Sigma Chemical Company (Sigma Aldrich, Germany) and Roth (Carl Roth GmbH, Germany), solvents were purchased from Sigma Chemical Company (Sigma Aldrich, Germany) and kaolin was obtained from Health Chemicals Co. Ltd. (China). Polymeric membranes were purchased from Millipore and Sterlitech. Impatiens noli-tangere L., Balsamiaceae, and Stachys officinalis L., Lamiaceae, herbs were collected from Cluj region, Romania. The voucher specimen no. 640632/1985 (det. Gh. Groza) for I. noli-tangere and voucher specimen no. 658238/2005 (det. L. Filipas) for S. officinalis were deposited in the Botanic Garden of Cluj-Napoca.
Animals
All experiments were performed on adult male rats (n = 32; 197 ± 48 g), purchased from UMF Biobase, Bucharest. They were housed eight per cage in a ventilated cage fitted with wood sawdust bedding, with free access to water and food pellets. The experiments were performed under controlled light/dark cycle conditions (12 h light/12 h dark; lights on at 6 a.m.) in the temperature ranged between 20 and 22 °C and the RH was maintained at 35–45%. All procedures were carried out according to EU Directive 2010/63/UE and with the approval of the Institutional Animal Care and Use Committee (Approval for pre-clinical experimental research based on the protocol no. HERB07/21.01.2016).
Preparation of polyphenolic-rich fractions from plant sources by nanofiltration
Air-dried leaves and stems of herbs were grounded, extracted with 50% EtOH then introduced in a sonication bath (Elma Transsonic T 460, Germany), at a frequency of 35 kHz for 90 min. The herbal's mass concentration in the solvent was 100 g/l. After filtration through Whatman filter paper, the extracts were microfiltred through 0.45 µm pore size microfiltration membrane (MF) (Millipore) as to remove any fine solid particles. The concentration experiments were further carried out by nanofiltration (NF) membrane (Sterlitech membrane, NF90 with cut-off 200–400 Da).
Membrane filtration experiments were performed using a lab cross-flow membrane filtration unit (KMS Laboratory Cell CF-1, Koch Membrane, Germany). NF experiments were performed according to the batch concentration mode, at a 9 bar trans-membrane pressure and at 23 ± 2 °C temperature. The performance of the nanofiltration process was evaluated through the rejection (R, %) of NF membrane toward specific compounds (total phenolic content), calculated as follows:
where cp and cf are the permeate and the feed concentrations of the total phenolics.
Extracts analysis
Determination of the total phenolic content and of the flavonoids
The phenolic total content was determined by the Folin-Ciocalteu method (Singleton et al., 1999Singleton, V.L., Orthofer, R., Lamuela-Raventos, R.M., 1999. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 299, 152-178.), measuring the absorbance at 760 nm, using gallic acid (GAE) as a standard. Total polyphenols contents were obtained from the regression equation of the gallic acid's calibration curve (y = 0.0032x + 0.0073, R2 = 0.9963) and expressed as GAE equivalents.
The total flavonoids content was assessed by the aluminum chloride colorimetric assay with the absorbance measured at 430 nm and quercetin (QE) used as standard (Lin and Tang, 2007Lin, J.-Y., Tang, C.-Y., 2007. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem. 101, 140-147.). Results were expressed as mg QE equivalents/l of extract. Total flavonoid contents were obtained from the regression equation of the quercetin's calibration curve (y = 0.0051x + 0.0286, R2 = 0.9966).
HPLC–MS analyses of phenolic, anthocyanidins and anthocyanins compounds
The polyphenol's measurement method was based on the previous HPLC analysis to evaluate the compounds in the extracts (Cristea et al., 2009Cristea, V., Deliu, C., Oltean, B., Brummer, A., Albu, C., Radu, G.L., 2009. Soilless cultures for pharmaceutical use and biodiversity conservation. Acta Hort. 843, 157-164.). The chromatographic measurements were performed using a complete HPLC Shimadzu system, through a C18 Nucleosil 3.5, 4.6 × 50 mm, Zorbax column. The system was coupled to a MS detector, LCMS-2010 detector (liquid chromatography mass spectrometer), equipped with an ESI interface. The mobile phase consists of formic acid in water (pH 3.0) as solvent A and formic acid in acetonitrile (pH 3.0) as solvent B. The polyphenolic compound's separation was performed using binary gradient elution: 0 min 5% solvent B; 0.01–20 min 5–30% solvent B; 20–40 min 30% solvent B; 40.01–50 min 30–50% solvent B; 50.01–52 min 50–5% solvent B. The flow rate was: 0–5 min 0.1 ml/min; 5.01–15 min 0.2 ml/min; 15.01–35 min 0.1 ml/min; 35.01–50 min 0.2 ml/min; 50–52 min 0.1 ml/min. ESI source and negative ionization mode have been used. The gallic acid, chlorogenic acid, ellagic acid, caffeic acid, rutin, rosmarinic acid, luteolin, quercetin, quercetin 3-β-d-glucoside, apigenin, umbelliferone and kaempferol were used as reference standard. Full scan acquisition mode was initially used in m/z range, between 50 and 1000. Further, selected ion monitoring (SIM) mode was used to search for some particular ions.
The identification and quantification of anthocyanidins and peonidin 3-O-glucoside have been conducted according to the previously reported HPLC–MS method (Albu Birsan et al., 2017Albu Birsan, C., Eremia, S.A.V., Penu, R., Vasilescu, I.M.I., Litescu, S.C., Radu, G.L., 2017. Characterization of the phenolics and free-radical scavenging of Romanian red wine. Anal. Lett., http://dx.doi.org/10.1080/00032719.2016.1192641.
http://dx.doi.org/10.1080/00032719.2016....
), using a mobile phase water-formic acid (95:5, v/v) (solvent A) and methanol-formic acid (95:5, v/v) (solvent B). The compounds were separated on a Nucleosil 100-3.5C18, Kromasil, 100 × 2.1 mm column after 47 min. The analyses were performed at 40 °C, at the 0.15 ml/min flow rate, using binary gradient elution: 0 min 6% solvent B; 0.01–10 min 6–30% solvent B; 10.01–25 min 30–50% solvent B; 25.01–30 min 50–60% solvent B; 30.01–36 min 60–6% solvent B; 36.01–47 min 6% solvent B. ESI source and positive ionization mode were used for anthocyanidins and anthocyanin compound's assessment. The MS data acquisition with the ESI interface was performed under the following condition: nebulizing gas (N2) flow rate, 1.5 l/min; curved desolvation line (CDL) voltage, 40 V; CDL temperature, 150 °C; heat block, 120 °C; detector voltage, 1.5 kV, interface voltage 4.5 kV.
Antioxidant activity-scavenging effect
The antioxidant activity was measured with the UV-Vis spectrophotometer (Jasco V-630), using the DPPH radical scavenging activity (Bondet et al., 1997Bondet, V., Brand-Williams, W., Berset, C., 1997. Kinetics and mechanisms of antioxidant activity using the DPPH• free radical method. LWT - Food Sci. Technol. 30, 609-615.). Briefly, 100 µl samples of different concentrations of extract were mixed with 1000 µl of the freshly prepared 0.25 mM DPPH in methanol and 1900 µl methanol. Absorbance at 516 nm was determined after 10 min. The decreasing of the DPPH radical absorption by the antioxidants action is used to evaluate the antioxidative activity. The antiradical activity was calculated using the equation:
where: A0 = blank absorbance; As = sample absorbance. Ascorbic acid (vitamin C) was used as standard antioxidant. The calculated IC50 values denote the concentration of the sample required to decrease by 50% the absorbance at 516 nm.
Anti-inflammatory activity
In vitro cyclooxygenase (COX) and lipoxygenase (LOX) inhibition assays
The I. noli-tangere and S. officinalis polyphenolic-rich extracts were first tested in order to investigate the anti-inflammatory response by inhibiting the COX-1, COX-2 and LOX enzymes. The COX (EC 1.14.99.1) activity was monitored on a spectrophotometer at 611 nm, as the result of the N,N,N′,N′-tetramethyl-p-phenylenediamine (TMPD) oxidation reaction with arachidonic acid. This assays were performed according to Amessis-Ouchemoukh et al. (2014)Amessis-Ouchemoukh, N., Madani, K., Falé, P.L.V., Serralheiro, M.L., Araújo, M.E.M., 2014. Antioxidant capacity and phenolic contents of some mediterranean medicinal plants and their potential role in the inhibition of cyclooxygenase-1 and acetylcholinesterase activities. Ind. Crops Prod. 53, 6-15. and Petrovic and Murray (2010)Petrovic, N., Murray, M., 2010. Using N,N,N′,N′-tetramethyl-p-phenylenediamine (TMPD) to assay cyclooxygenase activity in vitro. Methods Mol. Biol. 594, 129-140. with slight modifications. The efficacy of extracts and reference compound (ibuprofen) to inhibit both COX-1 and COX-2 isoenzymes was determined as the concentration causing 50% enzyme inhibition (IC50). COX-2 selectivity indices (SI) were also calculated as COX-1 (IC50)/COX-2 (IC50).
Inhibition of LOX from Glycine max (type I-B; EC 1.13.11.12) was determined by monitoring the absorbance's increase at 234 nm (Granica et al., 2013Granica, S., Czerwińska, M.E., Piwowarski, J.P., Ziaja, M., Kiss, A.K., 2013. Chemical composition, antioxidative and anti-inflammatory activity of extracts prepared from aerial parts of Oenothera biennis L. and Oenothera paradoxa Hudziok obtained after seeds cultivation. J. Agric. Food Chem. 61, 801-810.). Briefly, 50 µl of the different extract's concentrations was pre-incubated with 100 µl of soybean LOX solution (1000 U/ml in borate buffer solution, pH 9) and 3000 µl of borate buffer at 25 °C for 15 min. After pre-incubation 100 µl of linoleic acid (substrate) was added to each tube to start the reaction. Inhibition percentage of LOX was determined using the formula (E − S)/E × 100, where E is the activity of enzyme without test sample and S is the activity of enzyme with test sample. Ibuprofen and quercetin were used as reference compounds. IC50 values – the inhibitory concentration of the extracts required to decrease by 50% the enzyme's activity – were determined from the plotted graphs of enzyme inhibition (%) against the concentrations of the extracts.
In vivo anti-inflammatory activity
In vivo studies were performed on an experimental model of paw inflammation in male rats. Two models of inflammation were employed in order to assess the anti-inflammatory effect of S. officinalis and I. noli-tangere extracts: kaolin and dextran induced hind-paw edema. Four groups of eight rats, each received intraplantar in the left hind-paw 0.2 ml dextran solution 0.6% and in the right hind-paw 0.2 ml kaolin suspension 10%. Group 1 (control) received no treatment, while groups 2–4 received topical treatment: diclofenac gel 1% (group 2-reference group), S. officinalis extract (group 3-E1), I. noli-tangere extract (group 4-E2). Each extract (5 ml) and of diclofenac gel were applied immediately after the inflammation induction (extracts with a pre-set standard composition: polyphenolic compounds 14 mg/5 ml) and maintained in contact with the inflammatory region for 24 h using sterile bandage. The initial paw volume (baseline – V 0) was assessed. The volume of the induced edema was determined after 1, 2, 3, 4, 5, 24 h from the inflammatory agent's intraplantar administration. The volumes were determined using an Ugo Basile Plethysmometer. The growth percentage (Gper) of the edema was calculated and compared with control group as well as with reference group, according to Eqs.:
Ps represent the paw size of the sample and Psc represents the paw size of the control.
Statistical analysis
All measurements were performed in triplicate and Excel 2007 was used for statistical processing. Standard deviation (STDV) was <10%. Correlation analyses were performed using a two-tailed Pearson's correlation test and Student's t-test.
Statistical analysis for in vivo anti-inflammatory activity was performed using GraphPad Prism version 5.00 for Windows (GraphPad Software, San Diego California, USA). The type of distribution of the animal response was established with D’Agostino & Pearson test. Data are reported as means ± standard error of the mean (SEM) and were statistically analyzed using parametric Student's t-test. A confidence interval (CI) of 90% and p values of 0.05 or less were considered to be significant.
Results and discussion
Phytochemical characterization and antioxidant activity of polyphenolic-rich extract
The performance of the nanofiltration process used to obtain herbal extracts enriched in polyphenolic compounds was evaluated by the total polyphenol and flavonoids content's determination and also by the DPPH scavenging activity.
Table 1 shows that after NF step, the retention of the total phenolic compounds and also of the total flavonoids was higher than 91%, indicating also a good concentration performance of the membrane. This result is in accordance with the previously made studies to evaluate the potential of the nanofiltration process for the concentration of flavonoids. Conidi et al. (2012)Conidi, C., Cassano, A., Drioli, E., 2012. Recovery of phenolic compounds from orange press liquor by nanofiltration. Food Bioprod. Process. 90, 867-874. reported 0.89 retention value (R) in the concentration of flavonoids in orange press liquor and Arriola et al. (2014)Arriola, N.A., dos Santos, G.D., Prudêncio, E.S., Vitali, L., Petrus, J.C.C., Castanho Amboni, R.D.M., 2014. Potential of nanofiltration for the concentration of bioactive compounds from watermelon juice. Int. J. Food Sci. Technol. 49, 2052-2060. achieved R value of about 0.96 in the concentration of flavonoids in the watermelon juice.
Phenolic compounds and flavonoids content in the extract fractions of the Impatiens noli-tangere and Stachys officinalis.
In this study, the presence and content of eleven polyphenols and four anthocyanidin and anthocyanin compounds were studied by HPLC–MS analysis in the MF and NF retentate fractions (Table 2).
Presented data from Table 2 show that the fractions from I. noli-tangere contain mainly flavonoids, especially isoquercitrin and rutin, while in S. officinalis fractions, the phenolic acids are the dominant components, especially the chlorogenic, caffeic and rosmarinic acids, in agreement with the ones reported in literature (Choi and Kim, 2002Choi, B.J., Kim, C.W., 2002. Studies on the constituents of Impatiens noli-tangere L. Korean J. Pharmacogn. 33, 263-266.; Šerá et al., 2005Šerá, B., Vrchotová, N., Tříska, J., 2005. Phenolic compounds in the leaves of alien and native Impatiens plants. In: Alford, D.V., Backhaus, G.F. (Eds.), Plant Protection and Plant Health in Europe: Introduction and Spread of Invasive Species. British Crop Production Council, Alton, pp. 281–282.; Šliumpaite et al., 2013Šliumpaite, I., Venskutonis, P.R., Murkovic, M., Ragažinskiene, O., 2013. Antioxidant properties and phenolic composition of wood betony (Betonica officinalis L., syn. Stachys officinalis L.). Ind. Crops Prod. 50, 715-722.; Paun et al., 2016Paun, G., Neagu, E., Albu, C., Moroeanu, V., Radu, G.L., 2016. Antioxidant activity and inhibitory effect of polyphenolic-rich extract from Betonica officinalis and Impatiens noli-tangere herbs on key enzyme linked to type 2 diabetes. J. Taiwan Inst. Chem. Eng. 60, 1-7.).
The total concentrations of anthocyanin and of anthocyanidin compounds in NF retentate fractions were higher than those found in the microfiltrate fractions. Peonidin 3-O-glucoside was the major anthocyanin found in I. noli-tangere extracts, while the S. officinalis extracts contain genistin and cyanidin as main anthocyanidins. This is the first report on the presence of the anthocyanin and anthocyanidin compounds in I. noli-tangere and S. officinalis herbs extract.
Our study's significant contribution stays in that of new polyphenol's identification and quantification for the studied herbal extracts. In this regard, for both I. noli-tangere and S. officinalis the ursolic acid is reported for the first time in the literature, while umbelliferone is also first time reported in S. officinalis.
Since inflammatory diseases are considered to be caused by lipid peroxidation process where free radicals are involved (Bouarroudja et al., 2016Bouarroudja, K., Tamendjaria, A., Larbatb, R., 2016. Quality, composition and antioxidant activity of Algerian wild olive (Olea europaea L. subsp. oleaster) oil. Ind. Crops Prod. 69, 21-28.), antioxidant properties of the extract fractions were also studied. The nanofiltration retentate were found to be more effective, as both of the plant's extract showed the DPPH free radical scavenging activity higher than those of the microfiltrate and of the standard reference, ascorbic acid (Table 3). This is also in accordance to the phenolic and flavonoid contents of the nanofiltrate retentate.
DPPH scavenging activity and anti-inflammatory activity represented by IC50 (µg/ml) of Impatiens noli-tangere and Stachys officinalis extract fractions and its representative compounds.
Anti-inflammatory activity
In the first experimental part, the extract fractions from both medicinal plants were evaluated for their in vitro anti-inflammatory activity using LOX, COX-1 and COX-2, three enzymes involved in inflammatory process, an important stage of the wound repair in response to the tissue damage. COX is one of the most significant enzyme in pharmacology, since COX inhibition is the mechanism of action for most of the nonsteroidal anti-inflammatory drugs.
The results of the LOX and COX inhibitory activity (IC50) are presented in Table 3. Results showed for both of the studied herbs that the NF retentate fractions possess the highest inhibitory activity comparable to the standard ibuprofen, versus the MF fractions. Amongst the fractions, the S. officinalis retentate NF fraction exhibited high inhibitory activity with IC50 value of 1.22 ± 0.1 µg/ml for LOX and 10.1 ± 1.0 µg/ml for COX-1. The both fraction of I. noli-tangere extract and S. officinalis retentate NF fraction showed better COX-2 isozyme inhibition (IC50 6.2 ± 0.5, 1.9 ± 0.3 and 1.2 ± 0.2 µg/ml, in sequent) than ibuprofen. Moreover, the I. noli-tangere retentate NF fraction (SI = 9.12) and S. officinalis retentate NF fraction (SI = 8.42) exhibited the highest COX-2 selectivity index. These results support the traditional use of I. noli-tangere and S. officinalis in remedies for inflammatory diseases.
Previous papers suggested that the anti-inflammatory activity of the plants is greatly correlated to their content of the phenolic acids, flavonoids, anthocyanidins and anthocyanins. The anti-inflammatory effects are attributed to their inhibition capacity of the inflammatory reaction in its different stages, including COX and LOX activity (Cai et al., 2004Cai, Y., Luo, Q., Sun, M., Corke, H., 2004. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 74, 2157-2184.; Nagasaka et al., 2007Nagasaka, R., Chotimarkorn, C., Shafiqul, I.M., Hori, M., Ozaki, H., Ushio, H., 2007. Anti-inflammatory effects of hydroxycinnamic acid derivatives. Biochem. Biophys. Res. Commun. 358, 615-619.; Patel et al., 2013Patel, K., Jain, A., Patel, K.D., 2013. Medicinal significance, pharmacological activities, and analytical aspects of anthocyanidins ‘delphinidin’: a concise report. J. Acute Dis. 2, 169-178.). Some studies suggest a relationship between LOX inhibition and the ability of the inhibitors to reduce Fe3+ at the active site to the catalytically inactive Fe2+. This inhibition is related to their ability to reduce the iron species in the active site to the catalytically inactive ferrous form (Muller, 1994Muller, K., 1994. 5-Lipoxygenase and 12-lipoxygenase: attractive targets for the development of novel antipsoriatic drugs. Arch Pharm. 327, 3-19.). The anti-inflammatory properties of S. officinalis and I. noli-tangere may be attributed to the presence of ursolic acid, caffeic acid, rosmarinic acid, quercetin, rutin, cyanidin and genistin previously studied for their anti-inflammatory properties (Liu, 1995Liu, J., 1995. Pharmacology of oleanolic acid and ursolic acid. J. Ethnopharmacol 49, 57-68.; Ikeda et al., 2008Ikeda, Y., Murakami, A., Ohigashi, H., 2008. Ursolic acid: an anti- and pro-inflammatory triterpenoid. Mol. Nutr. Food Res. 52, 26-42.; Mulabagal et al., 2009Mulabagal, V., Lang, G.A., DeWitt, D.L., Dalavoy, S.S., Nair, M.G., 2009. Anthocyanin content, lipid peroxidation and cyclooxygenase enzyme inhibitory activities of sweet and sour cherries. J. Agric. Food Chem. 57, 123-1246.; Kashyap et al., 2016Kashyap, D., Tuli, H.S., Sharma, A.K., 2016. Ursolic acid (UA): a metabolite with promising therapeutic potential. Life Sci. 146, 201-213.; Kim et al., 2016Kim, S., Jeong, K., Cho, S.K., Park, J., Park, W., 2016. Caffeic acid, morin hydrate and quercetin partially attenuate sulfur mustard-induced cell death by inhibiting the lipoxygenase pathway. Mol. Med. Rep. 14, 4454-4460.; Ganeshpurkar et al., 2017Ganeshpurkar, A., Ajay, K., Saluja, A.K., 2017. The pharmacological potential of rutin. Saudi Pharm. J. 25, 149-164.).
Furthermore, there was a correlation between the relatively high amount of total phenol and LOX inhibition as well as DPPH free radical scavenging activity, because lipid hydroperoxide formations are usually inhibited as a result of the scavenging of lipid-oxy- or lipid-peroxy radicals formed in the course of enzymatic peroxidation.
Increasing evidence showed that oxidative stress is generated via free radicals that play an important role in inflammatory processes whereas antioxidants and free radical scavengers played a vital role in attenuation of inflammation. Reactive oxygen species (ROS) are implicated in cell signaling, playing an essential role in signal transduction. In inflammatory diseases high levels of ROS are produced and accumulated. Previous studies have shown an important role of antioxidant and/or radical-scavenging mechanism in various anti-inflammatory, anti-arthritic, neuroprotective, and wound healing potential of drugs (Mahajan and Tandon, 2004Mahajan, A., Tandon, V.R., 2004. Antioxidants and rheumatoid arthritis. J. Indian Rheumatol. Assoc. 12, 139-142.; Arulselvan et al., 2016Arulselvan, P., Fard, M.T., Tan, W.S., Gothai, S., Fakurazi, S., Norhaizan, M.E., Kumar, S.S., 2016. Role of antioxidants and natural products in inflammation. Oxid. Med. Cell. Longev., http://dx.doi.org/10.1155/2016/5276130.
http://dx.doi.org/10.1155/2016/5276130...
; Tambewagh et al., 2017Tambewagh, U.U., Kandhare, A.D., Honmore, V.S., Kadam, P.P., Khedkar, V.M., Bodhankar, S.L., Rojatkar, S.R., 2017. Anti-inflammatory and antioxidant potential of guaianolide isolated from cyathocline purpurea: role of COX-2 inhibition. Int. Immunopharmacol. 52, 110-118.).
In vivo anti-inflammatory activity
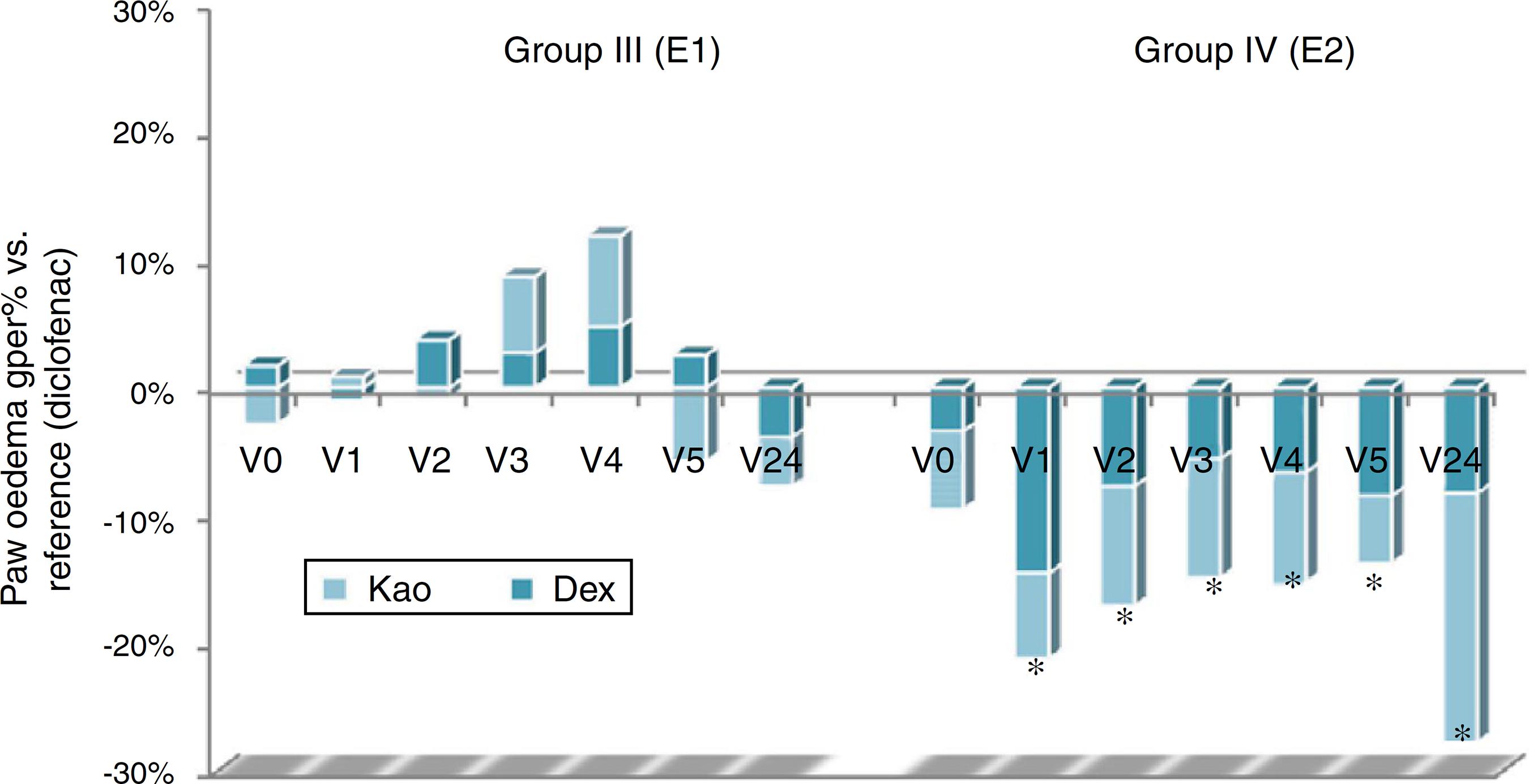
The volume of the paws initially and after inducing inflammation, for both dextran (Dex) and kaolin (Kao) are given in Table 4, while Gper (%) vs. control and Gper (%) vs. reference are illustrated in Figs. 1 and 2, respectively.
Growth percentage of the edema vs. control group, baseline (V0) and after 1 h (V1), 2 h (V2), 3 h (V3), 4 h (V4), 5 h (V5), 24 h (V24) following inflammation induction with dextran (Dex) or kaolin (Kao). *p < 0.05 vs control group for both models of inflammation (t-test Student, CI 90%).
Growth percentage of the edema vs. reference group, baseline (V0) and after 1 h (V1), 2 h (V2), 3 h (V3), 4 h (V4), 5 h (V5), 24 h (V24) following inflammation induction with dextran (Dex) or kaolin (Kao). *p < 0.05 vs control group for both models of inflammation (t-test Student, CI 90%).
Dextran is a polysaccharide with variable molecular weight, which induces inflammation by osmotic mechanism. Our results are similar to those found in literature. An inflammatory response appears shortly after Dextran's administration (about 30 min–1 h). This response is characterized by the accumulation of fluid with few proteins, partial degranulation of mast cells and a small number of neutrophils. Dextran-induced inflammation develops gradually in the early hours of the proinflammatory agent intraplantar administration (p < 0.05, Student's t test). The inflammation is reduced and the edema is much diminished after 24 h from the administration of dextran (Gulati et al., 1983Gulati, O.P., Sharma, S.C., Hammersen, F., 1983. Hemodynamic, biochemical and morphological changes in the dextran and d-galactosamine-induced edemas in rats. Arch. Int. Pharmacodyn. Ther. 263, 272-287.; Motoyama et al., 2016Motoyama, K., Tanida, Y., Hata, K., Hayashi, T., Hashim, I.I.A., Higashi, T., Ishitsuka, Y., Kondo, Y., Irie, T., Kaneko, S., Arima, H., 2016. Anti-inflammatory effects of novel polysaccharide sacran extracted from cyanobacterium Aphanothece sacrum in various inflammatory animal models. Biol. Pharm. Bull. 39, 1172-1178.).
Kaolin is an aluminosilicate with a stronger, longer lasting inflammatory effect than dextran. The most likely mechanism of the kaolin-induced inflammation is mediated by prostaglandins, not by histamine or by serotonin. Kaolin-induced inflammation develops gradually in the first hours after intraplantar administration of the proinflammatory agents (p < 0.05, Student's t-test) and it persists even after 24 h (Motoyama et al., 2016Motoyama, K., Tanida, Y., Hata, K., Hayashi, T., Hashim, I.I.A., Higashi, T., Ishitsuka, Y., Kondo, Y., Irie, T., Kaneko, S., Arima, H., 2016. Anti-inflammatory effects of novel polysaccharide sacran extracted from cyanobacterium Aphanothece sacrum in various inflammatory animal models. Biol. Pharm. Bull. 39, 1172-1178.).
A similar behavior is found for the group treated with diclofenac sodium, used as an anti-inflammatory reference. The initial volume's increase is significantly lower compared to that of the control, due to the diclofenac, a non-steroidal anti-inflammatory, non-selective COX inhibitor. The reduction of edema is significant compared to the control, both in the kaolin- and dextran-induced inflammation for the first five measurements. This is consistent with the literature data, which report 30 min–1 h after administration for the onset of action and the effect time of 6–8 h. 24 h after the edema induction, the measurements are similar to the control group (Rana et al., 2016Rana, M.S., Walia, R., Dixit, A., Raina, K., 2016. To compare and evaluate the anti-inflammatory efficacy of Terminalia arjuna (aqueous extract of bark) with diclofenac sodium on rats. Int. J. Basic Clin. Pharmacol. 5, 692-695.; Mundhava et al., 2016Mundhava, S.G., Mehta, D.S., Thaker, S.J., 2016. A comparative study to evaluate anti-inflammatory and analgesic activity of commonly used proteolytic enzymes and their combination with diclofenac in rats. Int. J. Pharm. Sci. Res. 7, 2615-2619.).
I. noli-tangere polyphenolic-rich extract shows a weak anti-inflammatory effect, the hind-paw volume determined for both of the employed models being lower than the control group. The differences are not statistically significant. Anti-inflammatory effect of this extract is significantly weaker than that of the reference – diclofenac.
S. officinalis polyphenolic-rich extract has a high anti-inflammatory activity, in both models, which suggests a complex mechanism (involving, among others, inhibition of prostaglandin synthesis as supported by our in vitro data). Furthermore, the anti-inflammatory effect is significantly higher than that of diclofenac and persists for more than 24 h, as proved by the obtained results in case of the kaolin-induced inflammation model.
As far as our literature survey could ascertain, the pro-inflammatory enzyme's inhibitory potentials of the studied plants have not yet been reported elsewhere. Therefore, data presented in this section could be assumed to be an original contribution to the literature.
In our study, the Pearson correlation test showed that total phenolic content for both species was strong correlated with antioxidant assays (DPPH assays) (r = 0.929), with lipoxygenase inhibition (r = −0.887) and also with cyclooxygenase inhibition (r = −0.899), and significance (p < 0.05). There is also a moderate correlation between total flavonoid content, antioxidant assays (r = 0.653) and enzyme inhibitory activity (r = −0.599 for LOX and r = −0.797 for COX, respectively) and significance (p < 0.05) (Table 5).
Conclusion
This study presents for the first time the investigation on the anti-inflammatory activities of I. noli-tangere and S. officinalis extracts, as well as their polyphenolic, anthocyanidins and anthocyanins profile.
Nanofiltration process using the NF90 membrane with cut-off 200–400 Da, was successfully applied for concentration of the ethanolic I. noli-tangere and S. officinalis extracts, achieving a phenolic acids and flavonoids compounds enriched extract. The target of this study consists in obtaining more effective antioxidant and anti-inflammatory products. The anti-inflammatory activity is demonstrated here for the first time. It is assigned to the pro-inflammatory mediator's inhibitory potential developed by the antioxidant and phenolic components of I. noli-tangere and S. officinalis polyphenolic-rich extracts.
The results of this study confirm the folklore use of I. noli-tangere and S. officinalis extracts as a natural anti-inflammatory agent and justify its ethnobotanical use.
Ethical disclosures
-
Protection of human and animal subjects. The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data.Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Acknowledgements
This work was supported by a grant of the Romanian National Authority for Scientific Research, CNDI-UEFISCDI, project number PN-II-PT-PCCA-2013-4-0034.
References
- Amessis-Ouchemoukh, N., Madani, K., Falé, P.L.V., Serralheiro, M.L., Araújo, M.E.M., 2014. Antioxidant capacity and phenolic contents of some mediterranean medicinal plants and their potential role in the inhibition of cyclooxygenase-1 and acetylcholinesterase activities. Ind. Crops Prod. 53, 6-15.
- Albu Birsan, C., Eremia, S.A.V., Penu, R., Vasilescu, I.M.I., Litescu, S.C., Radu, G.L., 2017. Characterization of the phenolics and free-radical scavenging of Romanian red wine. Anal. Lett., http://dx.doi.org/10.1080/00032719.2016.1192641
» http://dx.doi.org/10.1080/00032719.2016.1192641 - Ardelean, A., Mohan, Gh., 2008. Flora medicinala a Romaniei. Ed.ALL, Bucharest.
- Arriola, N.A., dos Santos, G.D., Prudêncio, E.S., Vitali, L., Petrus, J.C.C., Castanho Amboni, R.D.M., 2014. Potential of nanofiltration for the concentration of bioactive compounds from watermelon juice. Int. J. Food Sci. Technol. 49, 2052-2060.
- Arulselvan, P., Fard, M.T., Tan, W.S., Gothai, S., Fakurazi, S., Norhaizan, M.E., Kumar, S.S., 2016. Role of antioxidants and natural products in inflammation. Oxid. Med. Cell. Longev., http://dx.doi.org/10.1155/2016/5276130
» http://dx.doi.org/10.1155/2016/5276130 - Bondet, V., Brand-Williams, W., Berset, C., 1997. Kinetics and mechanisms of antioxidant activity using the DPPH• free radical method. LWT - Food Sci. Technol. 30, 609-615.
- Bouarroudja, K., Tamendjaria, A., Larbatb, R., 2016. Quality, composition and antioxidant activity of Algerian wild olive (Olea europaea L. subsp. oleaster) oil. Ind. Crops Prod. 69, 21-28.
- Cai, Y., Luo, Q., Sun, M., Corke, H., 2004. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 74, 2157-2184.
- Conidi, C., Cassano, A., Drioli, E., 2012. Recovery of phenolic compounds from orange press liquor by nanofiltration. Food Bioprod. Process. 90, 867-874.
- Choi, B.J., Kim, C.W., 2002. Studies on the constituents of Impatiens noli-tangere L. Korean J. Pharmacogn. 33, 263-266.
- Clark, R.A.F., 1995. Wound repair: overview and general considerations. In: The Molecular Cellular Biology of Wound Repair, 2nd edition. Plenum Press, NewYork, pp. 513–560.
- Cristea, V., Deliu, C., Oltean, B., Brummer, A., Albu, C., Radu, G.L., 2009. Soilless cultures for pharmaceutical use and biodiversity conservation. Acta Hort. 843, 157-164.
- Fitzmaurice, S.D., Sivamani, R.K., Isseroff, R.R., 2011. Antioxidant therapies for wound healing: a clinical guide to currently commercially available products. Skin Pharmacol. Phys. 24, 113-126.
- Ganeshpurkar, A., Ajay, K., Saluja, A.K., 2017. The pharmacological potential of rutin. Saudi Pharm. J. 25, 149-164.
- Granica, S., Czerwińska, M.E., Piwowarski, J.P., Ziaja, M., Kiss, A.K., 2013. Chemical composition, antioxidative and anti-inflammatory activity of extracts prepared from aerial parts of Oenothera biennis L. and Oenothera paradoxa Hudziok obtained after seeds cultivation. J. Agric. Food Chem. 61, 801-810.
- Gulati, O.P., Sharma, S.C., Hammersen, F., 1983. Hemodynamic, biochemical and morphological changes in the dextran and d-galactosamine-induced edemas in rats. Arch. Int. Pharmacodyn. Ther. 263, 272-287.
- Hayashi, S., Sumi, Y., Ueno, N., Murase, A., Takada, J., 2011. Discovery of a novel COX-2 inhibitor as an orally potent anti-pyretic and anti-inflammatory drug: design, synthesis, and structure-activity relationship. Biochem. Pharmacol. 82, 755-768.
- Háznagy-Radnai, E., Balogh, Ú., Czigle, S., Máthé, I., Hohmann, J., Blazsõ, G., 2012. Antiinflammatory activities of hungarian stachys species and their iridoids. Phytother. Res. 26, 505-509.
- Ikeda, Y., Murakami, A., Ohigashi, H., 2008. Ursolic acid: an anti- and pro-inflammatory triterpenoid. Mol. Nutr. Food Res. 52, 26-42.
- Jarić, S., Kostić, O., Mataruga, Z., Pavlović, D., Pavlović, M., Mitrović, M., Pavlović, P., 2017. Traditional wound-healing plants used in the Balkan region (Southeast Europe). J. Ethnopharmacol., http://dx.doi.org/10.1016/j.jep.2017.09.018
» http://dx.doi.org/10.1016/j.jep.2017.09.018 - Jovanovic, D., Fernandes, J., Martel-Pelletier, J., Jolicoeur, F., Reboul, P., Laufer, S., Tries, S., Pelletier, J., 2001. In vivo dual inhibition of cyclooxygenase and lipoxygenase by ML-3000 reduces the progression of experimental osteoarthritis: suppression of collagenase 1 and interleukin-1beta synthesis. Arthritis Rheum. 44, 2320-2330.
- Kashyap, D., Tuli, H.S., Sharma, A.K., 2016. Ursolic acid (UA): a metabolite with promising therapeutic potential. Life Sci. 146, 201-213.
- Kim, S., Jeong, K., Cho, S.K., Park, J., Park, W., 2016. Caffeic acid, morin hydrate and quercetin partially attenuate sulfur mustard-induced cell death by inhibiting the lipoxygenase pathway. Mol. Med. Rep. 14, 4454-4460.
- Lin, J.-Y., Tang, C.-Y., 2007. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem. 101, 140-147.
- Liu, J., 1995. Pharmacology of oleanolic acid and ursolic acid. J. Ethnopharmacol 49, 57-68.
- Mahajan, A., Tandon, V.R., 2004. Antioxidants and rheumatoid arthritis. J. Indian Rheumatol. Assoc. 12, 139-142.
- Martel-Pelletier, J., Lajeunesse, D., Reboul, P., Pelletier, J., 2003. Therapeutic role of dual inhibitors of 5-LOX and COX, selective and non-selective non-steroidal anti-inflammatory drugs. Ann. Rheum. Dis. 62, 501-509.
- Motoyama, K., Tanida, Y., Hata, K., Hayashi, T., Hashim, I.I.A., Higashi, T., Ishitsuka, Y., Kondo, Y., Irie, T., Kaneko, S., Arima, H., 2016. Anti-inflammatory effects of novel polysaccharide sacran extracted from cyanobacterium Aphanothece sacrum in various inflammatory animal models. Biol. Pharm. Bull. 39, 1172-1178.
- Mulabagal, V., Lang, G.A., DeWitt, D.L., Dalavoy, S.S., Nair, M.G., 2009. Anthocyanin content, lipid peroxidation and cyclooxygenase enzyme inhibitory activities of sweet and sour cherries. J. Agric. Food Chem. 57, 123-1246.
- Muller, K., 1994. 5-Lipoxygenase and 12-lipoxygenase: attractive targets for the development of novel antipsoriatic drugs. Arch Pharm. 327, 3-19.
- Mundhava, S.G., Mehta, D.S., Thaker, S.J., 2016. A comparative study to evaluate anti-inflammatory and analgesic activity of commonly used proteolytic enzymes and their combination with diclofenac in rats. Int. J. Pharm. Sci. Res. 7, 2615-2619.
- Nagasaka, R., Chotimarkorn, C., Shafiqul, I.M., Hori, M., Ozaki, H., Ushio, H., 2007. Anti-inflammatory effects of hydroxycinnamic acid derivatives. Biochem. Biophys. Res. Commun. 358, 615-619.
- Njenga, E.W., Viljoen, A.M., 2006. In vitro 5-lipoxygenase inhibition andanti-oxidant activity of Eriocephalus L. (Asteraceae) species. S. Afr. J. Bot. 72, 637-641.
- Paun, G., Neagu, E., Albu, C., Moroeanu, V., Radu, G.L., 2016. Antioxidant activity and inhibitory effect of polyphenolic-rich extract from Betonica officinalis and Impatiens noli-tangere herbs on key enzyme linked to type 2 diabetes. J. Taiwan Inst. Chem. Eng. 60, 1-7.
- Patel, K., Jain, A., Patel, K.D., 2013. Medicinal significance, pharmacological activities, and analytical aspects of anthocyanidins ‘delphinidin’: a concise report. J. Acute Dis. 2, 169-178.
- Petrovic, N., Murray, M., 2010. Using N,N,N′,N′-tetramethyl-p-phenylenediamine (TMPD) to assay cyclooxygenase activity in vitro Methods Mol. Biol. 594, 129-140.
- Rana, M.S., Walia, R., Dixit, A., Raina, K., 2016. To compare and evaluate the anti-inflammatory efficacy of Terminalia arjuna (aqueous extract of bark) with diclofenac sodium on rats. Int. J. Basic Clin. Pharmacol. 5, 692-695.
- Rigat, M., Vallès, J., Dambrosio, U., Gras, A., Iglésias, J., Garnatje, T., 2015. Plants with topical uses in the ripollès district (pyrenees, catalonia, iberian peninsula): ethnobotanical survey and pharmacological validation in the literature. J. Ethnopharmacol. 164, 162-179.
- Savla, U., Appel, H.J., Sporn, P.H., Waters, C.M., 2001. Prostaglandin E2 regulates wound closure in airway epithelium. Am. J. Physiol. Lung Cell. Mol. Physiol. 280, L421-L431.
- Šerá, B., Vrchotová, N., Tříska, J., 2005. Phenolic compounds in the leaves of alien and native Impatiens plants. In: Alford, D.V., Backhaus, G.F. (Eds.), Plant Protection and Plant Health in Europe: Introduction and Spread of Invasive Species. British Crop Production Council, Alton, pp. 281–282.
- Singleton, V.L., Orthofer, R., Lamuela-Raventos, R.M., 1999. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods Enzymol. 299, 152-178.
- Šliumpaite, I., Venskutonis, P.R., Murkovic, M., Ragažinskiene, O., 2013. Antioxidant properties and phenolic composition of wood betony (Betonica officinalis L., syn. Stachys officinalis L.). Ind. Crops Prod. 50, 715-722.
- Steinhilber, D., Hofmann, B., 2014. Recent advances in the search for novel 5-lipoxygenase inhibitors. Basic Clin. Pharmacol. Toxicol. 114, 70-77.
- Tambewagh, U.U., Kandhare, A.D., Honmore, V.S., Kadam, P.P., Khedkar, V.M., Bodhankar, S.L., Rojatkar, S.R., 2017. Anti-inflammatory and antioxidant potential of guaianolide isolated from cyathocline purpurea: role of COX-2 inhibition. Int. Immunopharmacol. 52, 110-118.
- Valko, M., Leibfritz, D., Moncol, J., Cronin, M.T.D., Mazur, M., Telser, J., 2007. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 39, 44-84.
- Vogl, S., Picker, P., Mihaly-Bison, J., Fakhrudin, N., Atanasov, A.G., Heiss, E.H., Wawrosch, C., Reznicek, G., Dirsch, V.M., Saukel, J., Kopp, B., 2013. Ethnopharmacological in vitro studies on Austria's folk medicine – an unexplored lore in vitro anti-inflammatory activities of 71 Austrian traditional herbal drugs. J. Ethnopharmacol. 149, 750-771.
Publication Dates
-
Publication in this collection
Jan-Feb 2018
History
-
Received
5 July 2017 -
Accepted
18 Oct 2017