Abstracts
OBJECTIVE: A simple method to reduce the ischemia/reperfusion injury that can accompany cardiac surgery would have great clinical value. This study was to investigate the effect of hyperosmotic perfusion on ischemia/reperfusion injury in isolated perfused rat hearts. METHOD: Forty male Sprague-Dawley rats were randomly divided either to have their isolated hearts perfused with normal osmotic buffer or buffer made hyperosmotic by addition of glucose. Hearts were then subjected to 30 min ischemia followed by 30 min reperfusion. Coronary flow, time to ischemic arrest, reperfusion arrhythmia, and ventricular function were recorded. Creatine phosphokinase leakage into the coronary artery, and myocardial content and activity of superoxide dismutase and catalase were also examined. RESULTS: Rat hearts with hyperosmotic perfusion showed higher coronary flow, a prolonged time to ischemic arrest (10.60 vs. 5.63 min, P<0.005), a lower reperfusion arrthythmia score (3.2 vs. 5.3, P<0.001), better ventricular function, and less creatine phosphokinase leakage (340.1 vs. 861.9, P<0.001) than normal osmotic controls. Myocardial catalase content and activity were increased significantly (1435 vs. 917 U/g wet weight, P<0.001) in hearts perfused with hyperosmotic solution in comparison to the normal osmotic controls. CONCLUSION: Pretreatment with hyperosmotic perfusion in normal rat hearts, which is attributed partly to the increased antioxidative activity, could provide beneficial effects from ischemia and reperfusion-induced injury by increasing coronary flow, and decreasing reperfusion arrhythmia.
Ischemia; Reperfusion injury; Myocardial infarction
OBJETIVO: Um método simples para reduzir a lesão de isquemia/reperfusão que pode acompanhar a cirurgia cardíaca teria grande valor clínico. O objetivo deste estudo foi investigar o efeito da perfusão hiperosmótica na isquemia/reperfusão em corações isolados de ratos perfundidos. MÉTODOS: Quarenta ratos machos Sprague-Dawley foram divididos aleatoriamente e tiveram os seus corações isolados perfundidos com tampão osmótico normal ou tampão hiperosmótico com a adição de glucose. Os corações foram então submetidos a 30 minutos de isquemia, seguida de 30 min de reperfusão. O fluxo coronariano, tempo de parada isquêmica, arritmia de reperfusão e da função ventricular foram registrados. Vazamento creatinofosfoquinase na artéria coronária, o miocárdio e atividade de superóxido dismutase e catalase foram também examinados. RESULTADOS: Crações de ratos com perfusão hiperosmótica apresentaram maior fluxo coronariano, tempo prolongado de parada isquêmica (10,60 vs. 5,63 min, P<0,005), menor pontuação de reperfusão arritmica (3,2 vs. 5,3, P<0,001), melhor função ventricular e menos vazamento de creatina fosfoquinase (340,1 vs. 861,9, P<0,001) do que controles normais osmóticos. Teor de catalase e atividade do miocárdio também tiveram aumento significativo (1435 vs. 917 peso U/g de peso fresco, P<0,001) em corações perfundidos com solução hiperosmótica em comparação com os controles normais osmóticos. CONCLUSÃO: O pré-tratamento com perfusão hiperosmótica em corações de ratos normais, o que é atribuído, em parte, ao aumento da atividade antioxidante, pode oferecer efeitos benéficos de isquemia e reperfusão induzida por lesão, aumentando o fluxo coronário e diminuindo a arritmia de reperfusão.
Isquemia; Traumatismo por reperfusão; Infarto do miocárdio
ORIGINAL ARTICLE
Beneficial effects of hyperosmotic perfusion in the myocardium after ischemia/reperfusion injury in isolated rat hearts
Efeitos benéficos da perfusão hiperosmótica no miocárdio após lesão isquemia/reperfusão em corações isolados de ratos
Yong CaoI; Lie WangII; Hong ChenIII; Zhiqian LvIV
IDoctor - No 6th People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China. Participated the experiment and drafted the manuscript
IIDoctor -Research Institute of General Surgery, General Hospital, Fuzhou Military Area Command of PLA, Shanghai, China. Participated the experiment and drafted the manuscript
IIIDoctor - Department of Pharmacology, Shanghai Jiaotong University School of Medicine, Shanghai, China. Designed the experiment and participated in the experiment
IVDoctor -No 6th People's Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China. Provided the research grant and research plan
Correspondence address Correspondence address: Zhiqian Lv and Hong Chen Department of Cardiosurgery, No 6 th People's Hospital Shanghai Jiaotong University School of Medicine, 200233, Shanghai, P R. China E-mail: luzhiqian@gmail.com and hchen100@hotmail.com
ABSTRACT
OBJECTIVE: A simple method to reduce the ischemia/reperfusion injury that can accompany cardiac surgery would have great clinical value. This study was to investigate the effect of hyperosmotic perfusion on ischemia/reperfusion injury in isolated perfused rat hearts.
METHOD: Forty male Sprague-Dawley rats were randomly divided either to have their isolated hearts perfused with normal osmotic buffer or buffer made hyperosmotic by addition of glucose. Hearts were then subjected to 30 min ischemia followed by 30 min reperfusion. Coronary flow, time to ischemic arrest, reperfusion arrhythmia, and ventricular function were recorded. Creatine phosphokinase leakage into the coronary artery, and myocardial content and activity of superoxide dismutase and catalase were also examined.
RESULTS: Rat hearts with hyperosmotic perfusion showed higher coronary flow, a prolonged time to ischemic arrest (10.60 vs. 5.63 min, P<0.005), a lower reperfusion arrthythmia score (3.2 vs. 5.3, P<0.001), better ventricular function, and less creatine phosphokinase leakage (340.1 vs. 861.9, P<0.001) than normal osmotic controls. Myocardial catalase content and activity were increased significantly (1435 vs. 917 U/g wet weight, P<0.001) in hearts perfused with hyperosmotic solution in comparison to the normal osmotic controls.
CONCLUSION: Pretreatment with hyperosmotic perfusion in normal rat hearts, which is attributed partly to the increased antioxidative activity, could provide beneficial effects from ischemia and reperfusion-induced injury by increasing coronary flow, and decreasing reperfusion arrhythmia.
Descriptors: Ischemia. Reperfusion injury. Myocardial infarction.
RESUMO
OBJETIVO: Um método simples para reduzir a lesão de isquemia/reperfusão que pode acompanhar a cirurgia cardíaca teria grande valor clínico. O objetivo deste estudo foi investigar o efeito da perfusão hiperosmótica na isquemia/reperfusão em corações isolados de ratos perfundidos.
MÉTODOS: Quarenta ratos machos Sprague-Dawley foram divididos aleatoriamente e tiveram os seus corações isolados perfundidos com tampão osmótico normal ou tampão hiperosmótico com a adição de glucose. Os corações foram então submetidos a 30 minutos de isquemia, seguida de 30 min de reperfusão. O fluxo coronariano, tempo de parada isquêmica, arritmia de reperfusão e da função ventricular foram registrados. Vazamento creatinofosfoquinase na artéria coronária, o miocárdio e atividade de superóxido dismutase e catalase foram também examinados.
RESULTADOS: Crações de ratos com perfusão hiperosmótica apresentaram maior fluxo coronariano, tempo prolongado de parada isquêmica (10,60 vs. 5,63 min, P<0,005), menor pontuação de reperfusão arritmica (3,2 vs. 5,3, P<0,001), melhor função ventricular e menos vazamento de creatina fosfoquinase (340,1 vs. 861,9, P<0,001) do que controles normais osmóticos. Teor de catalase e atividade do miocárdio também tiveram aumento significativo (1435 vs. 917 peso U/g de peso fresco, P<0,001) em corações perfundidos com solução hiperosmótica em comparação com os controles normais osmóticos.
CONCLUSÃO: O pré-tratamento com perfusão hiperosmótica em corações de ratos normais, o que é atribuído, em parte, ao aumento da atividade antioxidante, pode oferecer efeitos benéficos de isquemia e reperfusão induzida por lesão, aumentando o fluxo coronário e diminuindo a arritmia de reperfusão.
Descritores: Isquemia. Traumatismo por reperfusão. Infarto do miocárdio.
INTRODUCTION
Each year in the United States, approximately 1 million myocardial infarctions occur, and 700,000 patients undergo cardioplegic arrest for various cardiac surgeries. Minimizing ischemic time in these clinical scenarios has appropriately received a great deal of attention because of the long-established relationship between duration of ischemia and extent of myocardial injury. However after coronary flow is restored, the myocardium is susceptible to another form of insult stemming from reperfusion of the previously ischemic tissue. Given that cardiac ischemia is either unpredictable (myocardial infarction) or inevitable (in the operating room), there is great interest in developing strategies to minimize reperfusion-mediated injury.
Hyperosmotic saline has been used successfully in haemorrhagic and other types of shock to protect different tissues and organs [1,2]. Hyperosmotic saline perfusion has also been shown to decrease ischemia/reperfusion injury in rat hearts [3]. Elevated blood glucose also increases plasma osmolarity, and resistance to ischemia/reperfusion injury is increased in hearts from type 1 diabetic rats that have severe hyperglycaemia [4]. Because both hypertonic saline and, in diabetic rats, hyperglycemia are cardioprotective, we sought to determine whether addition of glucose instead of sodium chloride (NaCl) to produce hyperosmolarity would, in hearts from normal rats, provide protection from ischemia/reperfusion injury. If hyperosmotic glucose perfusion were found to be protective, it might, in the future, be able to be used clinically to attenuate ischemia/reperfusion injury during cardiac bypass surgery.
The present study investigates the effect of hyperosmotic perfusion pretreatment with glucose on ischemia/reperfusion arrhythmia and ventricular function damage in isolated rat hearts in vitro. Coronary creatine phosphokinase leakage (that is, leakage of this cardiac enzyme from damaged myocardial cells into the perfusate from the coronary artery), and myocardial content and activity of two antioxidant enzymes, superoxide dismutase and catalase, were also measured in order to assess the effect of hyperosmotic perfusion with glucose on myocardial cell damage and anti-oxidant activity.
METHODS
Animals
This study was approved by the animal care and use committee of our institution (2011(LW)-13). Forty male Sprague-Dawley rats (originally weighing 220-280 g) were housed under standard conditions with free access to tap water and chow. Rats were randomly divided either to have their isolated hearts perfused with normal Krebs-Henseleit buffer solution (300 mOsm/L) or with Krebs-Henseleit buffer solution made hyperosmotic (360 mOsm/L) by addition of glucose.
Isolated heart preparation and ischemia/reperfusion with normal buffer solution
Rats were anaesthetized with sodium pentobarbital (60 mg/kg, i.p.), injected with 25 mg/kg heparin, and their hearts excised a few minutes later. The hearts were then placed in a Langendorff apparatus [5] and rapidly connected to the aortic cannula of this apparatus for retrograde perfusion with Krebs-Henseleit buffer containing (in mmol/L): NaCl 118.0; KCl 4.7; CaCl2 2.5; MgSO41.2; KH2P4O1.2; NaHCO325.0; glucose 11.0 mM; Na2 -EDTA 0.5. The perfusate was maintained at 37˚C and aerated continuously with 95% O2 and 5% CO2. The left atrium was connected to a cannula and perfused in order to fill the left ventricle, and a catheter was inserted into the left ventricle to record left ventricular pressure (LVP), left ventricular end diastolic pressure (LVEDP), and maximal rate of LVP rise (dp/dtmax) and fall (dp/dtmin), using a data acquisition system (MPA 2000; Alcott Biotech, Shanghai, China). Aortic perfusion pressure was set at 70 mmHg and left atrial pressure at 15 mmHg throughout the experiment. However, cardiac output as it would occur in the living animal cannot be measured with this method. A catheter inserted into a small artery originating in the marginal or oblique branch of the left circumflex coronary artery was used for continuous measurement of coronary flow (calculated from the fluid fractions collected as volume/time) in pre-ischemia perfusion, ischemia, and reperfusion periods. We used this location for catheter insertion because it enables more precise and clean collection of coronary effluent than effluent collected from other locations. The perfusate collected from the coronary artery was used to assay for creatine phosphokinase.
Electrodes were inserted into the myocardium at the base and apex of the heart, and heart rate (HR) and electrocardiogram were recorded continuously with a data-acquisition system (MPA 2000; Alcott Biotech, Shanghai, China). Premature ventricular beats, ventricular tachycardia and ventricular fibrillation during reperfusion were recorded. The severity of arrhythmias was scored using the Curtis arrhythmia scoring system [6]. This system uses a scale of 0-9, where 0 indicates occasional ectopic beats and 9 indicates ventricular fibrillation occurring during the first 60s of reperfusion. The time from the clamping of the atrial inflow and aortic outflow to the cessation of ventricular contractions was recorded as the time to arrest, and the time from the beginning of reperfusion to the start of ventricular contractions was recorded as the time to recovery.
All hearts were allowed to beat spontaneously. After the 10 min normal buffer equilibration perfusion and 20 min pre-treatment normal buffer or hyperosmotic glucose perfusion (the total pre-ischemia period was 30 min), the hearts were subjected to 30 min global ischemia by clamping both atrial inflow and aortic outflow. The heart was thus in contact with hyperosmotic glucose buffer for a total of 50 min. The heart was kept warm during the ischemic period. At the end of this period, the above flow catheters were opened for a 30 min reperfusion period.
Hyperosmotic perfusion
The procedure for the rat hearts in the hyperosmotic perfusion group was the same as that described above for normally perfused hearts except that the osmolarity of the Krebs-Henseleit buffer was increased to 360 mOsmol/L during the 20 min pre-treatment period by adding 60 mOsmol/L of glucose and thus increasing the glucose concentration to 71 mM.
Biochemical determinations and Western blot analysis
At the end of the 30 min reperfusion period, the front portions of the left ventricles were freeze-clamped for use in biochemical assays and Western blotting. Creatine phosphokinase (CPK) from the coronary artery, and superoxide dismutase (SOD) and catalase from myocardial tissue homogenate, were determined with assay kits (Jian-Cheng Biomedical Engineering, Nanjing, China) according to the instructions of the manufacturer.
For Western blot analysis, the freeze-clamped sections of the left ventricle were homogenized in lysis buffer (pH 7.4) containing (in mmol/L): Tris-HCl 50; sucrose 150; EDTA-Na25; EGTA2 ; Na3VO41; NaF 50; phenylmethanesulphonyl fluoride 0.1; leupeptin 1 mg/L. Actin was used as the protein standard. Protein samples were loaded onto 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDSPAGE) acrylamide gels and transferred to polyvinylidene difluoride membranes. Blots were incubated with antibodies to either catalase (Calbiochem, San Jose, CA, USA) or SOD (Kagaard & Perry Laboratories). The membranes were then incubated with corresponding horseradish peroxidaseconjugated secondary antibody, and immunoreactive bands were visualized using electrochemoluminescence (Pierce; Thermo Fisher Scientific Inc., Rockford, IL, USA). Relative levels of proteins were semi-quantified with densitometry (Leica).
Statistics
Normally distributed continuous variables between control and hyperosmotic glucose groups were compared using an independent two sample t test. Differences between ischemia and reperfusion were determined using a paired t test. Repeated measurement of ANOVA was used for coronary flow. Data are presented as means ± SE. All statistical assessments were two-sided and evaluated at the 0.05 level of significant difference. Statistical analyses were performed using SPSS 15.0 statistics software (SPSS Inc, Chicago, IL).
RESULTS
Coronary flow and correlation with hyperosmotic perfusion
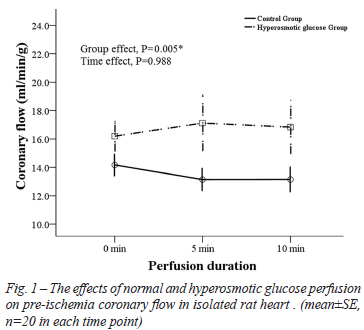
Compared to normal osmotic perfusion, hyperosmotic glucose perfusion significantly increased pre-ischemia basal coronary flow in isolated rat heart after 5 and 10 minutes of perfusion (Figure 1). After ischemia and reperfusion treatment, coronary flow was also significantly higher with hyperosmotic glucose than normal osmotic perfusion over the 30 minutes of perfusion (Figure 2).
Ventricular function during ischemia and reperfusion
Rat hearts perfused with hyperosmotic solution took a longer time (10.6±1.5 vs. 5.6±0.7 min) to stop beating during ischemia and a shorter time (13.0±2.5 vs. 19.2±1.5 sec) to resume beating during reperfusion than control hearts. In other words, hyperosmotic hearts were asystolic about 32 minutes as opposed to 43 minutes for control hearts. The reperfusion arrhythmia score (3.3±1.7) and ventricular tachycardia-fibrillation period (15.0±1.2 min) were significantly reduced in hyperosmotic hearts compared to control hearts (5.3±1.2, 24.3±2 min respectively) (Table 1). There were no significant differences in pre-ischemia HR, LVP, LVEDP, dP/dtmax and dP/dtminbetween the 2 groups. However, HR, LVP, dP/dtmax and dP/dtmin in both groups significantly decreased during reperfusion (P<0.05, Table 2).
Creatine phosphokinase (CPK) leakage from myocardial tissue and myocardial anti-oxidant enzymes
CPK leakage into the coronary artery, an indication of myocardial cell damage, was significantly reduced after reperfusion in hyperosmotic compared to control hearts, as shown by biochemical assays (360±115 vs. 860±124 U/g). Of the two anti-oxidant enzymes measured, hyperosmotic hearts showed a significant increase in myocardial catalase in both biochemical assay and Western blotting (Table 1 and Figure 3A, C). However, myocardial contents of SOD in hyperosmotic hearts were similar to those of normal control hearts in both biochemical assay and Western blotting (Table 1 and Figure 3B, D).
DISCUSSION
In the current study, hyperosmotic perfusion pretreatment with glucose of rat hearts subjected to global ischemia resulted in better ventricular contractile and electrophysiological function, increased coronary flow, and less myocardial damage than was seen in normal rat hearts. Hyperosmotic perfusion pretreatment also increased levels of catalase, although levels of a second anti-oxidant enzyme, SOD, were similar to those seen in control hearts.
Endothelial nitric oxide (NO) production has previously been shown to be responsible for the increase in coronary blood flow seen after bolus injection of hypertonic saline in anaesthetized pigs [7]. This is a possible mechanism for the increased coronary flow seen with hypertonic perfusion pretreatment of glucose in our study, but we did not measure NO production.
Previous research by others has shown that an important change responsible for ischemia/reperfusion damage is the increase in intracellular calcium that occurs during ischemia [8]. Anoxia decreases pH, and the increased Na/H exchange used by the cell to bring the pH back to normal affects Na/Ca exchange in a way that increases intracellular calcium [8]. Myocardial perfusion with fluid made hypertonic with either NaCl or sucrose has been shown to decrease intracellular calcium by decreasing the anoxia-induced increase in Na/H exchange [9].
Reactive oxygen species (ROS) have also been implicated in the pathogenesis of ischemia/reperfusion injury [8]. In our study, hyperosmotic hearts showed an increase in the activity of the anti-oxidant enzyme, catalase, but no increase in SOD, the second enzyme studied. A study of diabetic hearts from rats with severe hyperglycaemia also showed an increase in catalase, but not SOD, compared to normal hearts [4]. And a study of hyperosmotic sodium chloride perfusion of normal and stroke-prone spontaneously hypertensive rat hearts also showed increased catalase but not increased SOD, although in this study raising the osmolarity to 400 mOsm/L was able to increase SOD slightly [10]. In our study, levels of catalase were increased 20%, but levels of the marker of myocardial damage, CPK, however, were decreased 60%. It seems doubtful, therefore, that this modest increase in catalase activity could, by itself, cause such a profound lessening of myocardial damage.
An unanswered question is whether beneficial effects for heart by solution made hypertonic by the addition of glucose differs in any way from that provided by solution made hypertonic by the addition of NaCl. Hyperosmotic glucose differs from hypertonic saline in that glucose is a substrate for energy metabolism in the myocardium. In normal conditions, myocardium utilizes fatty acids for aerobic metabolism but in anoxic conditions, it uses glucose for anaerobic glycolysis. Anaerobic metabolism plays a crucial role in protecting myocardium from death [11]. Hyperosmotic glucose perfusate provides more glucose for the myocardium to produce ATP through glycolysis, an action beneficial for maintaining the activity of Na+-K+ATPase and the ATP-dependent K channel. This action may attenuate myocardium injury. However, because the manner in which the myocardium utilizes metabolic substrates is complex, the possibility that increases in extracellular glucose availability can be beneficial to heart needs further investigation.
Chu et al. [12] also that ischemic myocardium in alloxaninduced-diabetic (DM) group displayed higher expression of cell survival proteins including phospho eNOS, heat shock protein 27, NFkB, and mTOR as compared with ischemic myocardium in normoglycemic (ND), whereas in non-ischemic tissue, expression of these proteins was similar or lower in the DM group. The expression of total Erk 1/2 was similar between ND and DM groups, whereas the expression of phospho-Erk 1/2(Thr202/Tyr204) was lower in the DM group as compared to the ND group [12]. The expression of SAPK/JNK was lower in the DM group [12]. The current study shows that normal rat hearts prefused with hyperosmotic glucose can provide beneficial effects to heart, but these beneficial effects could very well be due to the hyperosmolarity itself rather than glucose.
In our study, rat hearts prefused with hyperosmotic glucose before ischemia/reperfusion increased coronary artery flow and decreased coronary artery resistance, increased left ventricular function, and increased levels of catalase but not SOD. In Shen's et al. [10] study, prefused with hyperosmolar NaCl gave similar results: increased coronary artery flow and decreased coronary artery resistance, increased left ventricular function, and increased levels of catalase but not SOD. Shen et al. [10] also compared the effects of a 2 hour perfusion of rats hearts with solution made hyperosmolar with 4 different osmolytes (NaCl, glucose, mannitol, and raffinose) and found no significant differences in levels of the cellular constituents measured. They did not, however look at left ventricular function. Our findings and Shen's et al. [10], therefore, may be due entirely to the hyperosmolarity.
However, one must also consider the use for which a solution is intended. Studies comparing the success of solutions used to protect hearts used for transplantation have focused on protecting cellular energy metabolism [13,14]. Despite the crystalloid solutions studied are not fully able to suppress the deleterious effects of ischemia and reperfusion in the rat heart, Lima et al. [14] had also found that a solution may contain the mannitol, instead of glucose, and the concentrations of K+ (15 mmol/L) and Ca2 + (0.25 mmol/L) of solution can contribute to a better performance by promoting the depolarizing arrest without contributing to an overload of intracellular calcium during the ischemic period. We were investigating hyperosmotic solutions might be beneficial to normal rat heart ex vivo to against ischemia/reperfusion injury during cardiac bypass surgery. In this situation, the physiologic effect of the hyperosmotic fluid used on non-cardiac tissue must be considered, and the use of glucose instead of NaCl as the osmolyte might have advantages.
A limitation of this study is that we should compare the use of glucose to mannitol, an osmotically active but metabolically inactive compound, to see whether hyperosmolarity alone was responsible for the beneficial effects of glucose to heart or not. Perfusion with hypertonic mannitol has been shown to increase catalase levels in normal rat hearts [11] but there are no reports to date about its actions on ventricular function and enzymes during ischemia/reperfusion. Other limitations are that NO production, intracellular calcium, and ROS should be measured to elucidate the mechanism behind of these beneficial effects of hyperosmotic perfusion pretreatment. We also like to study the protein profiles of cell survival signaling and mitogen-activated protein kinase signaling to see whether changes in any of these parameters might be involved in beneficial effects of hyperosmotic perfusion pretreatment with glucose to normal rat heart.
In summary, the present study had shown that hyperosmotic perfusion pretreatment may provide beneficial effects to heart from ischemia and reperfusion injury, increase coronary flow, and decrease reperfusion arrhythmia. The beneficial effects induced by hyperosmotic perfusion pretreatment with glucose may be attributed partly to the increased antioxidative activity. It is possible that hyperosmotic perfusion pretreatment may be able in the future to be applied during cardiac bypass surgery for its beneficial effects against ischemia/reperfusion injury.
ACKNOWLEDGEMENTS
This study was supported by grants from the National Natural Science Foundation of China (No. 31171099, 30971154 and 30770848).
CONFLICT OF INTEREST
No conflict of interest declared.
Article received on September 18th, 2012
Article accepted on October 1st, 2012
The work was carried out at No 6th People's Hospital and Department of Pharmacology, Shanghai Jiaotong University School of Medicine, Shanghai, China.
- 1. Gurfinkel V, Poggetti RS, Fontes B, Costa Ferreira Novo F, Birolini D. Hypertonic saline improves tissue oxygenation and reduces systemic and pulmonary inflammatory response caused by hemorrhagic shock. J Trauma. 2003;54(6):1137-45.
- 2. Oliveira RP, Velasco I, Soriano FG, Friedman G. Clinical review: hypertonic saline resuscitation in sepsis. Crit Care. 2002;6(5):418-23.
- 3. Chen LB, Liu T, Wu JX, Chen XF, Wang L, Fan CL, et al. Hypertonic perfusion reduced myocardial injury during subsequent ischemia and reperfusion in normal and hypertensive rats. Acta Pharmacol Sin. 2003;24(11):1077-82.
- 4. Chen H, Shen WL, Wang XH, Chen HZ, Gu JZ, Fu J, et al. Paradoxically enhanced heart tolerance to ischaemia in type 1 diabetes and role of increased osmolarity. Clin Exp Pharmacol Physiol. 2006;33(10):910-6.
- 5. Skrzypiec-Spring M, Grotthus B, Szelag A, Schulz R. Isolated heart perfusion according to Langendorff: still viable in the new millennium. J Pharmacol Toxicol Methods. 2007;55(2):113-26.
- 6. Curtis MJ, Walker MJ. Quantification of arrhythmias using scoring systems: an examination of seven scores in an in vivo model of regional myocardial ischaemia. Cardiovasc Res. 1998;22(9):656-65.
- 7. Vacca G, Papillo B, Battaglia A, Grossini E, Mary DA, Pelosi G. The effects of hypertonic saline solution on coronary blood flow in anaesthetized pigs. J Physiol. 1996;491(Pt 3):843-51.
- 8. Murphy E, Steenbergen C. Mechanisms underlying acute protection from cardiac ischemia-reperfusion injury. Physiol Rev. 2008;88(2):581-609.
- 9. Ho HS, Liu H, Cala PM, Anderson SE. Hypertonic perfusion inhibits intracellular Na and Ca accumulation in hypoxic myocardium.Am J Physiol Cell Physiol. 2000;278(5):C953-64.
- 10. Shen WL, Chen LB, Zhao JX, Guo SJ, Chen YC, Wang LP, et al. Effects of hyperosmotic sodium chloride perfusion on ischemia/reperfusion injury in isolated hearts of normal and stroke-prone spontaneously hypertensive rats. J Clin Exper Cardiol. 2011;2(7):146.
- 11. Gong J, Li LH, Pei WD, Wang HY, Zheng YL, Zhou GY, et al. Glycolytic and fatty acid metabolic enzyme changes early after acute myocardial ischemia. Zhonghua Xin Xue Guan Bing Za Zhi. 2006;34(6):546-50.
- 12. Chu LM, Osipov RM, Robich MP, Feng J, Oyamada S, Bianchi C, et al. Is hyperglycemia bad for the heart during acute ischemia? J Thorac Cardiovasc Surg. 2010;140(6):1345-52.
- 13. Silveira Filho LM, Petrucci O Jr, Carmo MR, Oliveira PP, Vilarinho KA, Vieira RW, et al. Trimetazidine as cardioplegia addictive without pre-treatment does not improve myocardial protection: study in a swine working heat model. Rev Bras Cir Cardiovasc. 2008,23(2):224-34.
- 14. Lima ML, Fiorelli AI, Vassallo DV, Pinheiro BB, Stolf NA, Gomes OM. Comparative experimental study of myocardial protection with crystalloid solutions for heart transplantation. Rev Bras Cir Cardiovasc. 2012;27(1):110-6.
Correspondence address:
Publication Dates
-
Publication in this collection
28 May 2013 -
Date of issue
Mar 2013
History
-
Received
18 Sept 2012 -
Accepted
01 Oct 2012