Abstracts
In the skin, the concept of reperfusion injury is well established. The application of this knowledge to deal with skin flap surgery problems, has a great prophylactic potential. This experimental study was performed to evaluate the action of mannitol as a scavenger of oxygen-free radicals, after an ischemia-reperfusion injury on skin island flaps. Thirty six male Wistar rats were divided into three test groups (n = 12): a non-ischemic group (group I), and two others (groups II and III) which were subjected to nine hours of ischemia following by 30 minutes of reperfusion. After seven days, all animals of group II, treated with saline, showed full skin flap necrosis. The assessment of group III, that received a 20% solution of mannitol prior to the onset of reperfusion, revealed 75% (9/12) of flap viability. These results suggest that pre-treatment with mannitol is able to enhance flaps survival with significantly less tissue necrosis (p <0.02).
Reperfusion; Free radicals; Surgical flaps; Mannitol
O conhecimento acerca da lesão decorrente da reperfusão na pele, já encontra-se consolidado. A aplicação destes conceitos revela uma perspectiva muito promissora na profilaxia de problemas cirúrgicos resultantes do manuseio de retalhos cutâneos em ilha. Este estudo experimental foi realizado com o objetivo de avaliar a ação do manitol, na qualidade de inativador dos radicais oxigênio livres, após isquemia e reperfusão sobre retalhos cutâneos em ilha. Trinta e seis ratos machos, do tipo Wistar, foram divididos em três grupos (n =12, cada) com a seguinte distribuição: Grupo I - sem isquemia, grupos II e III - submetidos durante nove horas a isquemia seguida por 30 minutos de reperfusão. Após sete dias, todos os animais do grupo II, tratados com solução salina, apresentaram necrose e em toda extensão dos retalhos. Na análise do grupo III, que recebeu solução de manitol a 20% previamente ao inicio da reperfusão, verificou-se viabilidade de 75% (9/12) dos retalhos. Estes resultados sugerem que o pré tratamento com manitol é capaz de melhorar a sobrevida dos retalhos, com uma expressiva redução da necrose tecidual (p <0.02).
Reperfusão; Radicais livres; Retalhos cirúrgicos; Manitol
Mannitol in reperfusion skin island flaps injury1 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Alberto Schanaider2 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Antônio Ambrósio de Oliveira Neto3 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Ilson Rosique da Costa4 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Gustavo Luiz Simões Leite5 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Daniela Peixoto Considera6 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Anna Karyna Pereira Lopes6 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Alessandra Rodrigues Silva6 1 Work performed at the Department of Surgery and Department of Pathology, and University Hospital Clementino Fraga Filho of the Federal University of Rio de Janeiro. 2 Associate Professor of Surgery, PhD, Researcher I of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). 3 Assistant Professor of Department of Pathology. 4 Master Degree in Medicine. 5 Students at the Faculty of Medicine (FM) of Federal University of Rio de Janeiro (UFRJ). CNPq scientific initiation scholarship. 6 Students at the Faculty of Medicine of Federal University of Rio de Janeiro (FM-UFRJ).
Schanaider A, Oliveira Neto AA, Costa IR, Leite GLS, Considera DP, Lopes AKP, Silva AR. Mannitol in reperfusion skin flaps injury. Acta Cir Bras [serial online] 1999 Jul-Sept;14(3). Available from: URL: http://www.scielo.br/acb .
SUMMARY: In the skin, the concept of reperfusion injury is well established. The application of this knowledge to deal with skin flap surgery problems, has a great prophylactic potential. This experimental study was performed to evaluate the action of mannitol as a scavenger of oxygen-free radicals, after an ischemia-reperfusion injury on skin island flaps. Thirty six male Wistar rats were divided into three test groups (n = 12): a non-ischemic group (group I), and two others (groups II and III) which were subjected to nine hours of ischemia following by 30 minutes of reperfusion. After seven days, all animals of group II, treated with saline, showed full skin flap necrosis. The assessment of group III, that received a 20% solution of mannitol prior to the onset of reperfusion, revealed 75% (9/12) of flap viability. These results suggest that pre-treatment with mannitol is able to enhance flaps survival with significantly less tissue necrosis (p <0.02).
SUBJECT HEADINGS: Reperfusion. Free radicals. Surgical flaps. Mannitol.
INTRODUCTION
Active species of oxygen have been increasingly implicated as mediators of acute island skin flaps injury1,8, specially in the reconstructive surgery, the major source of microvascular flap morbidity. These are generated either during ischemia/reperfusion syndromes or under inflammatory conditions13, ultimately promoting the sequence of events which results in flap necrosis, if the defence system is overwhelmed by an exceeding rate of oxygen radicals production. Mannitol is an exogenous antioxidant that acts as a hydroxyl radical (OH) scavenger10. Treatment with this agent has the potential to increase the survival of rat skin flaps21. The objective of this study is to assess the real effectiveness of mannitol and to achieve a better understanding of flap pathophysiology.
METHOD
Thirty six male Wistar rats weighing 370 to 400 g, were divided into three groups (n=12 each): G I -control, GII - ischemia and reperfusion and GIII - ischemia and reperfusion plus mannitol. All animals were anaesthetised with ether and after abdominal skin preparation, an intraperitoneal infusion of sodium pentobarbital (35 mg/kg) was performed. An island skin ventral flap, measuring, 3X6 cm2, was created in the right abdominal side with the animal's midline as the medial border12. The distal part of deep femoral vessels, the muscular branches together with the nerves were ligated and cut. The nerves were transected in order to exclude adrenergic action. Thus the skin flap supply was provided only by the right inferior superficial epigastric pedicle. After elevating the flap completely on its pedicle, it was sutured back at its original place by an interrupted suture. The rats were randomly divided into three groups. In group I, as showed above, flap was created without vascular occlusion. The other groups, had the same surgical procedure, but after the flap dissection, microvascular clamps (ABB-2V and 2A) were placed proximal to the epigastric pedicle. They were removed after nine hours of ischemia and the blood flow through the flap was restored during 30 minutes. Fifteen minutes before reperfusion, either isotonic saline solution or mannitol 20% (3 ml/kg) were injected into the left femoral vein, distal to the femoral ligament, respectively to the groups II and III. After seven days the flap was detached and histologic evaluation was performed.
The flaps survival were assessed by the Fisher Test. It was established 0,05 or 5% (p< 0,05) as the level to reject the hypotheses of nullity, pointing out the significant values.
RESULTS
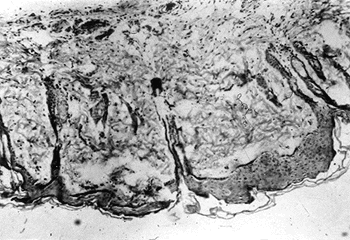
At the end of day 7, all flaps in group II (saline infusion) were black and hardened, showing a considerable loss of tissue due to complete necrosis. Seventy five per cent (9/12) of the island skin flaps receiving mannitol before reperfusion (group III) exhibited macroscopic integrity (p < 0.02), similar of those observed in the group I, characterised by pink and soft skin seeming entirely normal (Fig. 1). The remaining rats of the group III showed full flap necrosis. The histologic evaluation in group II revealed advanced tissue necrosis with degeneration of skeletal muscular cells and also vascular thrombosis (Fig. 2). In 9 flaps of group III, only slight deem and dermal fibroplasia were observed (Fig. 3).
Final results after seven days. In saline treated group there is a complete skin flap necrosis with partial loss of tissue area. In mannitol treated group the skin is viable.
DISCUSSION
Recent advances in plastic surgery such as microsurgery, have been improving the free skin flaps viability, although no/slow-reflow phenomenon continues to be a significant cause of morbidity. A variety of ethiological factors, including clinical causes (hypoxia, hypotension, hypovolemia, vasculitis, arteriosclerosis, and infection), and surgical complications (stricture, compression, torsion, and traumatic intimal dissection) could be responsible for tissue injury. However, most of them are adequately managed if a suitable therapy, with hemodynamic stabilization, or a good operative technique, are performed9. Attempts with extrinsic modulators of vascular tone such as dibutryl cyclic AMP, prostacycline, pentoxyifylline and others have been experimented, but results are inconsistent and unreliable10. Nevertheless also the presence of humoral mediators, specially the oxygen-derived free radicals, have been implicated to explain this complex pathogenic mechanism2,6,13. Abundant evidence indicates a major role for intravascular neutrophil sequestration early in reperfusion with free oxygen species overproduction6,13. Free radicals are chemical species that have one or more unpaired electron in its outer orbital. These molecules are extremely unstable and reactive and tends to initiate a chain of reactions that plays a central role in tissue injury. Hydroxyl radical when produced in the intracellular milieu, starts to attack polyunsaturated fatty acid side chains, located in phospholipid membranes, resulting in lipid radicals synthesis. Thus lipid peroxidation occurs and results in an increase in membrane permeability, and loss of its integrity. In addition, lipid hydroperoxides play a role in the endothelial injury triggering an inflammatory response. This process not only arrests activated leukocytes that adhere to microvascular endothelium, but also produces neutrophil-derived free radicals (hypochlorous acid and monochloramine) accompanied by proteases and elastases. This intricated reaction mediated by cytokines and platelet activating factor, results in vascular dysfunction (increase resistance, hyperpermeability), aggregation and sludging of red cells and platelets with the formation of microthrombi, and also extravasation of blood2,13,15. It is not surprising that the rat's endothelium is a prominent target in reperfusion injury considering that it has a high level of xanthine oxidase activity and it is the site of early neutrophil adherence4. Furthermore, free iron released from extravasated hemolysed red cells and injured parenchyma, catalyses a generation of hydroxyl radicals by HABER-WEISS and FENTON reactions7.
The morphologic changes found in this study, such as cellular oedema, intravascular obstruction and necrosis, became apparent only after biochemical system derangements. Progressive damage to plasma membrane with massive influx of calcium, cytoskeletal abnormalities, inactivation of mitochondria, depletion of ATP synthesis and denaturation of proteins, and lysosomal disruption, cause irreversible cell injury and necrosis15. Autocannibalisation of the abdominal rat skin flap model was not observed.
Mannitol (C6H14O6), was chosen because of its action against hydroxyl radicals resulting in a more stable and non-reactive dimmer, breaking up the free radical chain reaction11. It seems that this hyperosmolar agent does not produce haemodynamic changes interfering with its local action. The experimental results showed a beneficial effect when mannitol was administered in a single dose, 15 minutes before the reperfusion. Although it provides endothelial integrity, it can not protect against oxidants effects inside the tissue. It could explain the lack of a complete improvement in skin flap survival found in this study. Moreover it is possible that the radicals production during the inflammatory response, may have contributed to the flap injury. Direct intra-arterial flap injection was not considered because of the risk of vasospasm, embolisation, or blood clots. Furthermore, it would demand heparin administration, superimposing an additional protection, since this drug could regulate the release of inflammatory mediators3.
Many studies indicate that antioxidants effectively attenuate flap reperfusion induced injury5,8,12,14, but factors such as drugs half-lives and blood levels, interfere with the choice of an ideal timing of their administration. It is widely accepted that the critical period of oxygen free radical generation is about five minutes after reperfusion and the scavenger administration must begin just before it. Despite of these arguments it is not yet clear if the treatment should begin at the onset of the ischaemic period. The study developed by FU and JIAO5 showed that mannitol and anisodamin increased the survival rate of flaps after eight hours ischemia, when each one were administered five minutes before the release of vascular occlusion. It was also demonstrated that the combination of both drugs was not more effective than either agent alone. MANSON and co-workers12 reported that treatment with superoxide dismutase, a superoxide radical scavenging enzyme, prior to (15 minutes), or almost immediately (one to three minutes) following the onset of reperfusion, significantly enhanced island flap survival. Growing evidence suggests that better results could be achieved if multiple scavenger therapy, capable of acting in different steps of oxygen-derived free radicals synthesis pathway, was established. Although neither a single scavenger prophylactic therapy nor a rat model provide definitive data for clinical approaches, these findings present mannitol as a useful scavenger which plays a relevant role in preventing reperfusion injury of island skin flaps.
CONCLUSIONS
Mannitol acting as an oxygen free radical scavenger was able to protect 75% of the flaps from lesions that occurs after reperfusion. Its effectiveness was not complete considering that ischemia-reperfusion injury damaged the other 25% island flaps of this group.
Schanaider A, Oliveira Neto AA, Costa IR, Leite GLS, Considera DP, Lopes AKP, Silva AR. Manitol na reperfusão de retalhos cutâneos em ilha. Acta Cir Bras [serial online] 1999 Jul-Sept;14(3) . Available from: URL: http://www.scielo.br/acb.
RESUMO: O conhecimento acerca da lesão decorrente da reperfusão na pele, já encontra-se consolidado. A aplicação destes conceitos revela uma perspectiva muito promissora na profilaxia de problemas cirúrgicos resultantes do manuseio de retalhos cutâneos em ilha. Este estudo experimental foi realizado com o objetivo de avaliar a ação do manitol, na qualidade de inativador dos radicais oxigênio livres, após isquemia e reperfusão sobre retalhos cutâneos em ilha. Trinta e seis ratos machos, do tipo Wistar, foram divididos em três grupos (n =12, cada) com a seguinte distribuição: Grupo I - sem isquemia, grupos II e III - submetidos durante nove horas a isquemia seguida por 30 minutos de reperfusão. Após sete dias, todos os animais do grupo II, tratados com solução salina, apresentaram necrose e em toda extensão dos retalhos. Na análise do grupo III, que recebeu solução de manitol a 20% previamente ao inicio da reperfusão, verificou-se viabilidade de 75% (9/12) dos retalhos. Estes resultados sugerem que o pré tratamento com manitol é capaz de melhorar a sobrevida dos retalhos, com uma expressiva redução da necrose tecidual (p <0.02).
DESCRITORES: Reperfusão. Radicais livres. Retalhos cirúrgicos. Manitol.
Address for correspondence:
Alberto Schanaider
Rua Eurico Cruz, 33/603
22461-200 Rio de Janeiro-RJ
Data do recebimento: 09/07/99
Data da revisão: 10/08/99
Data da aprovação: 15/09/99
- 1. Angel MF, Narayanan K, Swartz WM, Ramasastry SS, Kuhns DB, Basford, RE, Futrell JW. Deferoxamine increases skin flap survival: additional evidence of free radical involvement in ischaemic flap surgery. Br J Plast Surg 1986;39:469-72.
- 2. Angel MF, Ramasastry SS, Swartz WN, Basford RE, Futrell JW. Free radicals: basic concepts concerning their chemistry. Pathophysiology and relevance to plastic surgery. Plast Reconstr Surg 1987;79:990-7.
- 3. Cooley BC, Tadych KL, Gould JS. Perfusion of free flaps with heparinized whole blood during ischemic storage. J Reconstr Microsurg 1990;6:49-53.
- 4. Engerson TD, Mc Kelvey TG, Rhyne DB, Boggio EB, Snyder SJ, Jones HP. Conversion of xanthine dehydrogenase to oxidase in ischemic rat tissues. J Clin Invest 1987;79:1564-70.
- 5. Fu W, Jiao X. The effect of mannitol and anisodamin on the prevention of free radical injury to post-ischemia flaps: an experimental study. Br J Plast Surg 1995;48:218-21.
- 6. Granger DN, Kvietys PR, Perry MA. Leukocyte-endothelial cell adhesion induced by ischemia and reperfusion. Can J Physiol Pharmacol 1993;71:57-75.
- 7. Halliwell B, Gutteridge JMC. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 1984;219:1-14.
- 8. Im MJ, Manson PN, Bulkley GB, Hoopes JE. Effects of superoxide dismutase and allopurinol on the survival of acute island skin flaps. Ann Surg 1985;201:357-9.
- 9. Kerrigan CL. Skin flap failure: pathophysiology. Plast Reconstr Surg 1983;6:766-77.
- 10. Kusumoto K, Isshiki N, Suzuki S, Ohtsuka M, Nose K. Increase in length of experimental skin flaps that survive with dibutryl cyclic AMP. Scand J Plast Reconstr Surg Hand Surg 1995;29:111-6.
- 11. Magovern GJ, Bolling SS, Casali AS, Bulkley BN, Gardner J. The mechanism of mannitol in reducing ischemia injury, hyperosmolarity or hydroxyl scavenger? Circulation 1984;70:91-5.
- 12. Manson PN, Anthenelli RM, Im MJ, Bulkley GB, Hoopes JE. The role of oxygen-free radicals in ischemic tissue injury in island skin flaps. Ann Surg 1983;198:87-90.
- 13. Schanaider A. Inativadores dos radicais livres derivados do oxigęnio na isquemia intestinal [Dissertaçăo]. Rio de Janeiro: UNI-RIO; 1990.
- 14. Suzuki S, Yoshioka N, Isshiki N, Hamanaka H, Miyachi Y. Involvement of reactive oxygen species in post-ischaemic flap necrosis and its prevention by antioxidants. Br J Plast Surg 1991;44:130-4.
- 15. Willms-Kretschmer K, Majno G. Ischemia of the skin: electron microscopic study of vascular injury. Am J Pathol. 1969;54:327-42.
Publication Dates
-
Publication in this collection
28 Oct 1999 -
Date of issue
Sept 1999
History
-
Accepted
15 Sept 1999 -
Reviewed
10 Aug 1999 -
Received
09 July 1999