Abstracts
End-to-end conventional arterial anastomosis is time consuming, requires prolonged clamping times and is associated with focal necrosis, granulocyte infiltration and subsequently, fibrosis and calcification of the arterial wall. Fibrin glue as an alternative for microarterial anastomosis may obviate these lesions, with less adherence to adjacent tissues and better coaptation of the arterial margins. OBJECTIVE: In this study we compared the healing process of conventional to fibrin glue end-to-end anastomosis in larger arteries. METHODS: In 22 rabbits, both carotid arteries were cross sectioned and repaired by end-to-end anastomosis with 4 interrupted sutures and fibrin glue in one side (GI) and with 8 conventional interrupted sutures in the other side (G2). After 3 and 15 days, the animals were randomly allocated for tensile strength, hydroxyproline determination (8 animals), and histologic analysis of the anastomosis (3 animals). Conventional staining procedures (hematoxylin-eosin and Masson methods) and picrosirius red polarization (PSP) technique for collagen type determination were employed. RESULTS: From 3 to 15 days, the tensile strength increased in both groups, from 280.0± 32.6 g to 432.2± 131.2g in Group I and from 221.4± 72.4g to 452.2± 132.0g in Group 2 (p<0.001), with no statistical difference between the groups in each period of the study. The hydroxyproline content, expressed as hydroxyproline /protein ratio, varied from 0.0816 ± 0.0651 to 0.0622 ± 0.0184 in Group l and from 0.0734 ± 0.0577 to 0.0460 ± 0.0271 in Group 2, with no significant difference between periods and groups (p>0.05). Histology showed slight increase of inflammatory and reparative reactions in Group 2. PSP technique demonstrated predominant type I collagen in relation to type III collagen in the anastomosis of both groups, with no significant difference between them. CONCLUSION: Fibrin glue was less harmful to the arterial wall than conventional suture. Even using less sutures in fibrin glue anastomosis, similar tensile strength and healing characteristics were noted in both groups. Completion times for the fibrin glue group was significantly greater than for the conventional anastomosis.
Fibrin glue; Arteries; Wound healing; Sutures; Collagen
A anastomose arterial término-terminal é demorada, requer tempo prolongado de oclusão vascular e esta associada a necrose focal, infiltração leucocitária e, conseqüentemente, à fibrose e calcificação da parede arterial. A cola de fibrina é uma alternativa para a anastomose microvascular e pode evitar estas alterações com menor aderência aos tecidos vizinhos e melhor coaptação das bordas arteriais. OBJETIVO: Comparar o processo cicatricial de anastomoses convencionais com anastomoses feitas com cola de fibrina em artérias maiores. MÉTODOS: Em 22 coelhos, ambas carótidas foram seccionadas transversalmente e reconstruídas por meio de anastomose término-terminal com 4 pontos simples de reparo e cola de fibrina de um lado (G1), e com 8 pontos separados do outro lado (G2). Após 3 e 15 dias, os animais foram destinados aleatoriamente para estudo de força tênsil concentração de hidroxiprolina (8 animais) e avaliação histológica das anastomoses (3 animais). As lâminas histológicas foram coradas pelo HE Masson e Picrossirius polarização (PSP). RESULTADOS: Após 3 e 15 dias a força tênsil aumenta em ambos os grupos, de 280,0± 32,6g para 432,2± 131,2g no Grupo 1 e de 221,4± 72,4g para 452,2± 132,0g no Grupo 2; sem diferença estatística entre os grupos em cada período. A concentração de hidroxiprolina expressa como razão hidroxiprolina/proteína, variou de 0,0816± 0,0651 para 0,0622± 0,0184 no Grupo 1 e de 0,0734± 0,0577 para 0,0460± 0,0271 no Grupo 2; sem diferença estatística entre os períodos e grupos. Os estudos histológicos mostraram discreto aumento das reações de inflamação e reparação no Grupo 2. A técnica PSP mostrou predomínio do colágeno tipo I em relação do colágeno tipo II nas anastomoses de ambos os grupos, sem diferença expressiva entre esses grupos. CONCLUSÃO: A anastomose com a cola de fibrina foi menos lesiva para a parede arterial do que a anastomose convencional. Mesmo usando menos pontos, as características de força tênsil e de cicatrização da anastomose com cola de fibrina foram similares em ambos os grupos. Os tempos de realização das anastomoses foram significativamente maiores do que na anastomose convencional.
Cola de fibrina; Artérias; Cicatrização; Colágeno; Anastomoses
2 ORIGINAL ARTICLE
END-TO-END ARTERIAL ANASTOMOSIS WITH FIBRIN GLUE IN LARGER ARTERIES: HISTOLOGY, HYDROXYPROLINE CONCENTRATION AND TENSILE STRENGTH STUDY IN CAROTIDS OF RABBITS1 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Winston B. Yoshida2 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Luiz E. Naresse3 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Antonio C. Rodrigues4 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Viciany E. Fabris5 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Aparecida Y. Angeleli6 1 . Trabalho realizado na Faculdade de Medicina de Botucatu - UNESP. 2 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP 3 . Professor Adjunto Livre Docente do Depto de Cirurgia e Ortopedia - Faculdade de Medicina de Botucatu - UNESP. 4 . Professor Adjunto Livre Docente do Depto de Morfologia - Instituto de Biociências de Botucatu - UNESP. 5 . Professor Adjunto Livre Docente do Depto de Patologia - Faculdade de Medicina de Botucatu - UNESP. 6 . Bióloga, Doutora do Depto de Clínica Médica - Faculdade de Medicina de Botucatu - UNESP.
Yoshida WB, Naresse LE, Rodrigues AC, Fabris VE, Angeleli AY. End-to-end arterial anastomosis with fibrin glue in larger arteries: histology, hydroxyproline concentration and tensile strength study in carotids of rabbits. Acta Cir Bras [serial online] 2002 Jan-Feb;17(1). Available from: URL: http://www.scielo.br/acb.
ABSTRACT: End-to-end conventional arterial anastomosis is time consuming, requires prolonged clamping times and is associated with focal necrosis, granulocyte infiltration and subsequently, fibrosis and calcification of the arterial wall. Fibrin glue as an alternative for microarterial anastomosis may obviate these lesions, with less adherence to adjacent tissues and better coaptation of the arterial margins. OBJECTIVE: In this study we compared the healing process of conventional to fibrin glue end-to-end anastomosis in larger arteries. METHODS: In 22 rabbits, both carotid arteries were cross sectioned and repaired by end-to-end anastomosis with 4 interrupted sutures and fibrin glue in one side (GI) and with 8 conventional interrupted sutures in the other side (G2). After 3 and 15 days, the animals were randomly allocated for tensile strength, hydroxyproline determination (8 animals), and histologic analysis of the anastomosis (3 animals). Conventional staining procedures (hematoxylin-eosin and Masson methods) and picrosirius red polarization (PSP) technique for collagen type determination were employed. RESULTS: From 3 to 15 days, the tensile strength increased in both groups, from 280.0± 32.6 g to 432.2± 131.2g in Group I and from 221.4± 72.4g to 452.2± 132.0g in Group 2 (p<0.001), with no statistical difference between the groups in each period of the study. The hydroxyproline content, expressed as hydroxyproline /protein ratio, varied from 0.0816 ± 0.0651 to 0.0622 ± 0.0184 in Group l and from 0.0734 ± 0.0577 to 0.0460 ± 0.0271 in Group 2, with no significant difference between periods and groups (p>0.05). Histology showed slight increase of inflammatory and reparative reactions in Group 2. PSP technique demonstrated predominant type I collagen in relation to type III collagen in the anastomosis of both groups, with no significant difference between them. CONCLUSION: Fibrin glue was less harmful to the arterial wall than conventional suture. Even using less sutures in fibrin glue anastomosis, similar tensile strength and healing characteristics were noted in both groups. Completion times for the fibrin glue group was significantly greater than for the conventional anastomosis.
KEY WORDS: Fibrin glue. Arteries. Wound healing. Sutures. Collagen.
INTRODUCTION
Since conventional vascular anastomosis techniques, whether continuous or interrupted, may be time consuming, require prolonged clamping time and may be responsible for severe focal necrotic lesions of the arterial wall, leading to granulomas, calcification, fibrosis, intimal hyperplasia and other thickenings of the arterial wall1, it has been proposed the use of fibrin glue (FG) for microarterial anastomosis2,3,4,5,6,7. The advantages by using fibrin glue would be good biocompatibility, shortening of operating time, fewer sutures required for the anastomosis, and consequently, less arterial wall damage3-8. Fibrin glue is basically a human plasma fibrinogen activated by thrombin9. Besides being an excellent hemostatic agent10, fibrin glue has been employed successfully to suture or reconstruct a variety of hollow structures11. However, only one investigator used this method on the reconstruction of larger arteries (> 2mm)12. Therefore, the purpose of this study was to compare the healing process of fibrin glue end-to-end anastomosis in arteries larger than 2 mm in diameter to conventional anastomosis by using standard suture techniques.
METHODS
Twenty-two NORFOLK rabbits, weighing from 2000 to 2500 g were anesthetized with intravenous pentobarbital (30 mg per kg), with an additional 5mg per kg pentobarbital given as needed to maintain anesthesia. All procedures with the animals were performed in strict adherence to the guidelines of the U.S. National Research Council's "Guide for the Care and Use of Laboratory Animals" (NRC publication - 1996) and were approved by the local "Ethical Committee for Animal Research". The animals were operated on in a supine position, attached on a Claude Bernard board. Proper oxygenation was insured by placing a small tubing in the oral cavity, carrying oxygen at 1 liter per minute.
The carotid arteries of both sides were approached by a midline cervical incision, and were encircled with vessel loops. The diameter of carotid arteries varied from 2.0 to 2.5 mm after lidocaine application. Although not strictly necessary, loupe magnification of 2.5 times was used for a better technical result. Each artery was clamped and divided, the arterial stumps irrigated with heparinized saline (100 USP units/ml) and the adventitious tissue excised.
Using standard vascular technique, each carotid artery was reconstructed by end-to-end anastomosis with 8 interrupted sutures (Group 2) using 7-0 Prolene on a 10 mm needle or 4 interrupted stay sutures with fibrin glue application between each set of sutures (Group I). The fibrin glue used in the present experiment was prepared as by Dresdale et al13, without aprotinin inclusion14. Right or left, the artery which would receive fibrin glue or conventional sutures, was randomly chosen. After the completion of the anastomosis, gentle local digital pressure was applied for hemostasis. The time necessary for the completion of the anastomosis and hemostasis was recorded. All the anastomosis were performed by the same surgeon (WBY). After wound closure, the animals received 600 units /IM of benzatin penicillin and no anticoagulant, antiplatelet or vasodilator agents were used.
The animals were allowed to recover from anesthesia and then returned to their individual cages, with standard rabbit chow and water provided ad libitum. Dipirone was diluted in the water of the animals during the first postoperative day for analgesia.
At 3 and 15 days, 8 animals of each period were chosen at random, and studied by using angiography to determine the anastomotic patency. Five milliliters of ditriazoate-meglumine contrast were injected through a 23-scalp needle in each proximal carotid artery, followed by serial pictures. After angiographic evaluation, both carotid arteries were ligated proximally and distally and excised en bloc, including the anastomotic site. The animals were then sacrificed with pentobarbital.
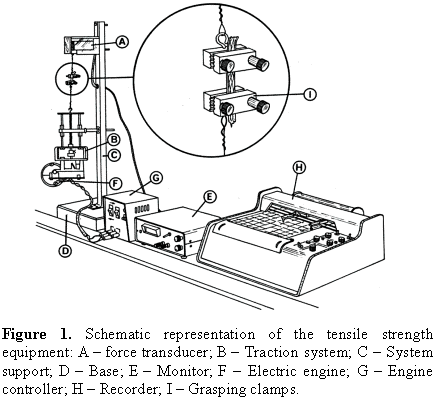
The arteries were immersed in a saline solution of papaverine (0.25 mg/ml) at 37°C to standardize the level of muscle relaxation/contraction. Tensile strength was measured in a tensiometer developed in the author's laboratory15. Briefly, the equipment was fitted with a transducer to which one of the operated extremities was attached. The other extremity was connected to a vertical moving platform, driven by a vertical rotating screw, driven in turn by a synchronous electrical motor at a controlled velocity (Figure 1). The tensile strength was displayed digitally on the monitor and recorded on a strip chart recorder. The extremities of the carotid arteries were stretched longitudinally to the point of vessel rupture (tensile strength values were expressed in gram).
Following tensile strength measurement, the ruptured arterial segments were weighted and hydroxyproline content determined by acid hydrolysis with HC1 6N for 24 hours, neutralization with saturated LiOH (pH=7.0), oxidation with chloramine T (7% w/v in H2O), and analysed by spectrophotometry with Ehrlich reagent16. Total protein was also determined by treating the acid hydrolysate with alkaline cupper solution (l % w/v in H2O) and then allowed to react with Folin phenol reagent17. The results were expressed as hydroxyproline/protein ratio.
Completion time, tensile strength values and hydroxyproline/protein ratios were statistically analyzed by two-way analysis of variance and the Tukey test was used for differences between means.
Three animals representing different time periods of the experiment were randomly chosen for histologic study. After angiography, their carotid arteries were excised and fixed in 10% formalin solution. Hematoxilin-eosin and thrichromic Masson staining methods were used for conventional histology. Determination of collagen in the anastomosis was performed by the picrosirius polarization (PSP) method18. Briefly, 5 µm sections of isotonic 10% formalin material were deparaffinized, hydrated with water, and stained during one hour in a 0,1% solution of Sirius Red , dissolved in aqueous saturated picric acid. The sections were rapidly washed in running tap water and counterstained with fresh Harris hematoxylin for 6 hours. A conventional optical microscope with a strong light source and two polaroid filters were used to study the birefringence of the stained collagen.
RESULTS
The angiographic appearance of the two anastomosis was similar (Figure 2) and no difference was demonstrated between the two groups related to stenosis. There were no thrombosis in this experiment. Completion time was significantly greater (p<0.001) in the fibrin glue group (Figure 3).
Tensile strength increased significantly in both groups from the 3rd to the l5th postoperative day (p<0.01). However, there was no statistical difference between the groups in each period of the study (Figure 4).
Hydroxyproline/protein ratios did not change during the study and no significant difference was demonstrated between the groups in each period (Figure 5).
On the third postoperative day, conventional histology showed no differences between the groups regarding inflammatory reaction and focal necrosis (Figure 6 A and 6 B). Subjectively there was the impression that the fibrin glue anastomosis group showed less inflammatory reaction than the conventional anastomoses. However, this finding was not consistent, for all animals of the fibrin glue group. There was evidence of mural platelet thrombus with fibrin near the anastomosis in both study groups. Edema and fibrin-leukocyte exsudate was also seen in this area. Both types of anastomosis showed similar reparative processes with granulation tissue, giant cells surrounding sutures and intimal hyperplasia near anastomotic sites on the fifteenth day (Figures 6C and D). Interruption of the elastic laminae was noted uniformly and calcification was not found in any of the specimens. Adventitial fibrosis occurred in both groups, perhaps less pronounced in the fibrin glue group. In both groups and periods, the PSP technique demonstrated predominantly type I collagen and a lesser amount of type III collagen in the anastomosis (Figure 7).
DISCUSSION
The search for a quick and efficient tissue glue has been sought for the past three decades9. Desirable characteristics include a hemostatic effect, strong adherence to tissue, no interference with the normal healing process, without carcinogenic potential. A fibrin glue composed of fibrinogen, factor XIII, aprotinin, fibronectin, calcium chloride and thrombin is commercially available in Europe under a variety of trade names, but has not been approved in the United States because of the concern for viral transmission, based upon its origin from pooled human plasma.
In order to overcome this problem, some authors developed other types of fibrin glue processing, utilizing blood from a single donor12,13 or from the patient himself (autologous)19,20.
Though the risk of viral transmission was decreased or eliminated, processing could take from 90 minutes to 2 days.
Ikossi-OConnor et al12, studied the use of fibrin glue for end-to-end anastomosis in canine femoral arteries. They observed a significant decrease in the number of sutures required to achieve hemostasis and also observed a decreased in foreign body reaction. Similar findings were also noted in our study. Good patency have been associated with fibrin glue anastomosis2,4-8 and this was as well observed in our study in both experimental groups.
The healing process of the arterial anastomosis performed either by FG or conventional suture is usually similar. After an initial period of inflammatory reaction, including focal necrosis and endothelial loose, a granulomatous foreign body type reaction and fibrosis appear along with intimal hyperplasia in these arteries1-3. This sequence was noted in the study by Ikossi-OConnor et al12, and was reproduced in the present study for both types of anastomosis. However, there was tendency to less inflammatory reaction and less adventitial fibrosis in the fibrin glue anastomoses . Other authors have also observed that fibrin glue anastomosis was associated with less injury to the arterial wall2,11.
Another point on the vascular healing process is related to the mechanical properties of the anastomotic tissue. Our tensile strength measurement showed comparable findings for both the fibrin glue anastomosis and the conventional suture group.
Collagen is an important component of the healing process, the most important being types I, III and IV18,21. Previous studies with the PSP method in normal arteries showed that collagen type I appears as thick, bright, strongly birefringent yellow or red fibers in the tunica adventitia; in contrast, type III collagen appears as thin, pale, weakly birefringent, greenish fibers in the media arterial layer18,21. In the present study, those characteristics were also observed in normal areas of the arteries, far from the anastomotic site. However, type I collagen was predominant in the entire width of the arterial wall of both groups and study periods at the healing area.
Beside from a qualitative analysis, collagen can be quantified by hydroxyproline determinations, which estimate newly synthesized, mature and lysed collagens16. The hydroxyproline/total protein ratio is utilized to eliminate errors due to dilution of collagen by edema. In the present study the hydroxyproline/protein ratio did not change from the 3rd to 15th postoperative day. However, tensile strength increased in both groups. We believe that this difference is explained as tensile strength being a physical measurement that reflects intermolecular collagen cross-linking, rather than biosynthesis. Hydroxyproline determination estimates the total collagen concentration and does not reflect collagen cross-linkage and stability.
Most previous articles showed fibrin glue arterial reconstruction to be faster than conventional anastomosis. That was not our case, because we had to put several layers of the fibrin glue on the anastomosis to stop the blood leak between the stitches. Maybe the method of fibrin glue preparation would explain the difference.
CONCLUSION
Vascular anastomosis employing fibrin glue required less sutures and was comparable in their healing and tensile strength to those anastomosis constructed with conventional suture technique. Fibrin glue anastomosis was significantly more time consuming to perform.
ACKNOWLEDGMENT
We are greatful to Dr. Herbert Dardik, MD and Dr. Alexandre Bakonyi, MD, for the revision of the text and for their suggestions.
REFERENCES
Yoshida WB, Naresse LE, Rodrigues AC, Fabris VE, Angeleli AY. Anastomose arterial término-terminal com cola de fibrina em artérias maiores: estudo histológico, da concentração de hidroxiprolina e força tênsil em carótidas de coelhos. Acta Cir Bras [serial online] 2002 Jan-Fev;17(1). Disponível em: URL: http://www.scielo.br/acb.
RESUMO: A anastomose arterial término-terminal é demorada, requer tempo prolongado de oclusão vascular e esta associada a necrose focal, infiltração leucocitária e, conseqüentemente, à fibrose e calcificação da parede arterial. A cola de fibrina é uma alternativa para a anastomose microvascular e pode evitar estas alterações com menor aderência aos tecidos vizinhos e melhor coaptação das bordas arteriais. OBJETIVO: Comparar o processo cicatricial de anastomoses convencionais com anastomoses feitas com cola de fibrina em artérias maiores. MÉTODOS: Em 22 coelhos, ambas carótidas foram seccionadas transversalmente e reconstruídas por meio de anastomose término-terminal com 4 pontos simples de reparo e cola de fibrina de um lado (G1), e com 8 pontos separados do outro lado (G2). Após 3 e 15 dias, os animais foram destinados aleatoriamente para estudo de força tênsil concentração de hidroxiprolina (8 animais) e avaliação histológica das anastomoses (3 animais). As lâminas histológicas foram coradas pelo HE Masson e Picrossirius polarização (PSP). RESULTADOS: Após 3 e 15 dias a força tênsil aumenta em ambos os grupos, de 280,0± 32,6g para 432,2± 131,2g no Grupo 1 e de 221,4± 72,4g para 452,2± 132,0g no Grupo 2; sem diferença estatística entre os grupos em cada período. A concentração de hidroxiprolina expressa como razão hidroxiprolina/proteína, variou de 0,0816± 0,0651 para 0,0622± 0,0184 no Grupo 1 e de 0,0734± 0,0577 para 0,0460± 0,0271 no Grupo 2; sem diferença estatística entre os períodos e grupos. Os estudos histológicos mostraram discreto aumento das reações de inflamação e reparação no Grupo 2. A técnica PSP mostrou predomínio do colágeno tipo I em relação do colágeno tipo II nas anastomoses de ambos os grupos, sem diferença expressiva entre esses grupos. CONCLUSÃO: A anastomose com a cola de fibrina foi menos lesiva para a parede arterial do que a anastomose convencional. Mesmo usando menos pontos, as características de força tênsil e de cicatrização da anastomose com cola de fibrina foram similares em ambos os grupos. Os tempos de realização das anastomoses foram significativamente maiores do que na anastomose convencional.
Descritores: Cola de fibrina. Artérias. Cicatrização. Colágeno. Anastomoses.
Conflito de interesses: nenhum
Fontes de financiamento: CNPq
Address for correspondence:
Winston Bonetti Yoshida
Universidade Estadual Paulista
Faculdade de Medicina de Botucatu - Depto. de Cirurgia e Ortopedia
Botucatu - SP
18618-970
Fax: (14) 821-7428
e-mail: winston@fmb.unesp.br
Data do recebimento: 12/09/2001
Data da revisão: 23/10/2001
Data da aprovação: 03/12/2001
- Lidman D, Daniel RK. The normal healing process of microvascular anastomosis. Scand J Plast Reconstr Surg 1981;15:103-10.
- Warter A, Betelli E, Schmitthausler R. Histopathologie de la réparation artérielle microchirurgicale: aport des cryoprécipités fibrineux. Ann Pathol 1987;7:31-40.
- Han SK, Kim SW, Kim WK. Microvascular anastomosis with minimal suture and fibrin glue: experimental and clinical study. Microsurgery 1998;18:306-11.
- Aksik IA, Kikut KP, Apshkalne DL. Extra-intracranial anastomosis performed by means of biological gluing materials: experimental and clinical study. Microsurgery 1986;7:2-8.
- Dowbak GM, Rohrich RJ, Robinson JB. Effectiveness of a new non-thrombogenic bio-adhesive in microvascular anastomosis. J Reconstr Microsurg 1994;10:383-6.
- Isogal N, Cooley BC, Kamiishi H. Clinical outcome of digital replantation using the fibrin glue-assisted microvascular anastomosis technique. J Hand Surg 1996;21b:573-5.
- Saitoh S, Nakatsuchi Y. Long-term results of vein grafts interposed in arterial defects using the telescoping anastomotic technique and fibrin glue. J Hand Surg 1998;21b:47-52.
- Padubri NA, Browne E, Kononov A. Fibrin glue-assisted end-to-side anastomosis of rat femoral vessels: comparison with conventional suture method. Ann Plast Surg 1996;37:41-7.
- Cooper CW, Falb RD. Surgical adhesives. Ann N Y Acad Sci 1968;146:214-24.
- Raccuia JS, Simonian G, Dardik M, Hallac D, Raccuia SV, Stahl R, Dardik H. Comparative study of topical hemostatic agents in a rat kidney model. Am J Surg 1992;63:234-8.
- Detweiller MB, Deweiller JG, Fenton J. Sutareless and reduced suture anastomosis of hollow vessels with fibrin glue: a review. J Invest Surg 1999;12:245-62.
- Ikossi-O'Connor MG, Ambrus JL, Rao U. The role of fibrin adhesive in vascular surgery. J Surg Oncol 1983;23:151-2.
- Dresdale A, Rose EA, Jeevanandam V, Reemtsma K, Bowman FO, Malm JR. Preparation of fibrin glue from single-donor fresh-frozen plasma. Surgery 1985;750-5.
- Durham LM, Willat DJ, Yung MW, Jones I, Stevenson PA, Radaman MF. A method for preparation of fibrin glue. J Laringol Otol 1987;101:1182-6.
- Yoshida WB, Müller SS, Carvalho I, Fabris VE, Naresse LE, Maffei FHA. Tensile strength and histological changes of abdominal aorta of malnourished rats. Cardiovasc Surg 1995;3:437-9.
- Bergman I, Loxley R. The determination of hydroxyproline in urine hidrolysates. Clin Chim Acta 1970;27:347-9.
- Lowry OH, Rosenbrough NJ, Fara AL. Protein measurement with folin phenol reagent. J Biol Chem 1951;193:265-75.
- Montes GS, Junqueira LCU. The use of picrosirius-polarization method for the study of biopathology of collagen. Mem Inst Oswaldo Cruz 1991;86(supll III):1-11.
- Silberstein LE, Williams LJ, Hughlett MA, Magge DA, Weisman RA. An autologous fibrinogen-based adhesive for use in otologic surgery. Transfusion 1988;319-21.
- Dahlstron KK, Wis-Fogh VS, Medgyesi S, Rostgaard J, Sorensen, H. The use of autologous fibrin adhesive in skin transplantation. Plast Reconstr Surg 1992;89:968-76.
- Montes GS, Nicolecis MAL, Brentani S, Furvie SS. Collagen fibril diameters in arteries of mice: a comparison of manual and computed-aided morphometric analysis. Acta Anat 1989;135:57-61.
Publication Dates
-
Publication in this collection
03 Sept 2003 -
Date of issue
Feb 2002
History
-
Accepted
03 Dec 2001 -
Received
12 Sept 2001 -
Reviewed
23 Oct 2001