Abstracts
Low cytoreductive regimen of irradiation associated to unmodified bone marrow infusion (UBM) does not prevent the occurrence of graft versus host disease (GVHD) after transplant. PURPOSE: In this study we evaluated the potential advantages of a long-term immunossupression and T-cell depleted bone marrow infusion (TCDBMI) in preventing the occurrence of GVHD after small bowel transplantation (SBTx). METHODS: Heterotopic SBTX was performed with Lewis rats as recipients and DA as donors and distributed into 5 groups according to the irradiation, duration of immunossupression and the use of UBM or TCDBMI: G1 (n=6), without irradiation and G2 (n=9), G3 (n=4), G4 (n=5) and G5 (n=6) was given 250 rd of irradiation. Groups 1,2,4 and G3 and 5 were infused with 100 x 10(6) UBM and TCDBM respectively. Animals in G1, 2, 3 were immunossupressed with 1mg/ FK506/Kg/IM for 5 days and G4 and G5 for 15 days. Anti CD3 monoclonal antibodies and immunomagnetic beads were used for T-cell depletion.Animals were examined for rejection, GVHD, chimerism characterization and ileal and skin biopsies. RESULTS: Minimal to mild rejection was observed in all groups; however, GVHD were present only in irradiated groups. Long-term immunossupression changed the severity of GVHD in G4 and G5. Rejection was the cause of death in G1 while GVHD in G2, 3, 4 and 5, not avoided by the use of TCDBMI. Total chimerism and T-cell chimerism was statistically higher in irradiated groups when compared to G1. CONCLUSION: Extended immunossupression associated to low dose of irradiation decrease the severity of GVHD, not avoided by the use of TCDBMI.
Intestinal transplant; T-cell depletion; Bone marrow
Baixas doses de irradiação associadas à infusão de células da medula óssea não previnem a ocorrência da reação do enxerto versus hospedeiro após o transplante intestinal. OBJETIVO: Neste estudo foi avaliado a potencial vantagem em estender o regime imunossupressor associado a infusão de células de medula óssea do doador depletadas de células T na prevenção da reação do enxerto versus hospedeiro após o transplante intestinal. MÉTODOS: Transplante heterotópico de intestino delgado foi realizado em ratos Lewis como receptores e DA como doadores, distribuídos em cinco grupos de acordo com a duração da imunossupressão, irradiação e do uso de medula óssea normal ou depletada: G1 (n=6), sem irradiação e G2 (n=9), G3 (n=4), G4 (n=5) e G5 (n=6) foram irradiados com 250 rd. Grupos1, 2, 4 e G3 e 5 foram infundidos com 100 x 10(6) células da medula normal e depletada respectivamente. Animais no G1,2,3 foram imunossuprimidos com 1mg/kg/FK506/ IM por cinco dias e G4 e cinco por 15 dias. Anticorpos monoclonais contra células CD3 e colunas magnéticas foram utilizadas para a depleção da medula óssea. Os animais foram examinados para a presença de rejeição, reação do enxerto versus hospedeiro, chimerismo e biópsias intestinais e da pele. RESULTADOS: Rejeição mínima foi observada em todos os grupos; entretanto, a reação do enxerto versus hospedeiro somente nos animais irradiados. Extensão da imunossupressão alterou a gravidade da reação nos animais dos G4 e 5. Rejeição foi a causa mortis no G1 e a reação do enxerto versus hospedeiro nos Grupos 2,3,4 e 5, não controlada com a infusão de medula óssea depletada. O chimerismo total e de células T do doador foi estatisticamente maior nos grupos irradiados em comparação ao G1. CONCLUSÃO: A extensão do regime de imunossupressão associado a baixas doses de irradiação diminui a gravidade da reação do enxerto versus hospedeiro, não abolida pelo uso de medula óssea depletada.
Transplante intestinal; Depleção de células T; Medula óssea
3 - ORIGINAL ARTICLE
Graft versus host disease in a rat small bowel transplant model after T-cell depleted donor specific bone marrow infusion1 1 University of Miami School of Medicine, Division of Liver and GI Transplantation
Reação do enxerto versus hospedeiro apos infusão de celulas T depletadas da medula ossea no transplante de intestino delgado em ratos
Alexandre Bakonyi NetoI, 2 2 Department of Surgery/ Division of Liver and GI Transplantation. Assistant Professor, PhD, Department of Surgery from Botucatu Faculty of Medicine (UNESP); Ex-fellow of Transplant Division at the University of Miami supported by CNPq ; Evangelos P. MisiakosI, 2 2 Department of Surgery/ Division of Liver and GI Transplantation. Assistant Professor, PhD, Department of Surgery from Botucatu Faculty of Medicine (UNESP); Ex-fellow of Transplant Division at the University of Miami supported by CNPq ; Andreas G. TzakisI, 2 2 Department of Surgery/ Division of Liver and GI Transplantation. Assistant Professor, PhD, Department of Surgery from Botucatu Faculty of Medicine (UNESP); Ex-fellow of Transplant Division at the University of Miami supported by CNPq ; Mariana BehroII, 3 3 Department of Pathology/ Division of Immunopathology ; Phillip RuizII, 3 3 Department of Pathology/ Division of Immunopathology ; Joshua MillerIII, 4 4 Department of Surgery/ Division of Kidney and Pancreas Transplantation ; Camilo RicordiIV, 5 5 Diabetes Research Institute and Cell Transplantation Center ; Bonifacio K. TakegawaV, 6 6 Assistant Professor, Department of Surgery from Botucatu Faculty of Medicine
IDepartment of Surgery/ Division of Liver and GI Transplantation. Assistant Professor, PhD, Department of Surgery from Botucatu Faculty of Medicine (UNESP); Ex-fellow of Transplant Division at the University of Miami supported by CNPq
IIDepartment of Pathology/ Division of Immunopathology
IIIDepartment of Surgery/ Division of Kidney and Pancreas Transplantation
IVDiabetes Research Institute and Cell Transplantation Center
VAssistant Professor, Department of Surgery from Botucatu Faculty of Medicine
Address to correspondence Address to correspondence Alexandre Bakonyi Neto Rua Hortência, 66 18603-660 Botucatu, SP E-mail: fefofofo@laser.com.br
ABSTRACT
Low cytoreductive regimen of irradiation associated to unmodified bone marrow infusion (UBM) does not prevent the occurrence of graft versus host disease (GVHD) after transplant.
PURPOSE: In this study we evaluated the potential advantages of a long-term immunossupression and T-cell depleted bone marrow infusion (TCDBMI) in preventing the occurrence of GVHD after small bowel transplantation (SBTx).
METHODS: Heterotopic SBTX was performed with Lewis rats as recipients and DA as donors and distributed into 5 groups according to the irradiation, duration of immunossupression and the use of UBM or TCDBMI: G1 (n=6), without irradiation and G2 (n=9), G3 (n=4), G4 (n=5) and G5 (n=6) was given 250 rd of irradiation. Groups 1,2,4 and G3 and 5 were infused with 100 x 106 UBM and TCDBM respectively. Animals in G1, 2, 3 were immunossupressed with 1mg/ FK506/Kg/IM for 5 days and G4 and G5 for 15 days. Anti CD3 monoclonal antibodies and immunomagnetic beads were used for T-cell depletion.Animals were examined for rejection, GVHD, chimerism characterization and ileal and skin biopsies.
RESULTS: Minimal to mild rejection was observed in all groups; however, GVHD were present only in irradiated groups. Long-term immunossupression changed the severity of GVHD in G4 and G5. Rejection was the cause of death in G1 while GVHD in G2, 3, 4 and 5, not avoided by the use of TCDBMI. Total chimerism and T-cell chimerism was statistically higher in irradiated groups when compared to G1.
CONCLUSION: Extended immunossupression associated to low dose of irradiation decrease the severity of GVHD, not avoided by the use of TCDBMI.
Keywords: Intestinal transplant. T-cell depletion. Bone marrow.
RESUMO
Baixas doses de irradiação associadas à infusão de células da medula óssea não previnem a ocorrência da reação do enxerto versus hospedeiro após o transplante intestinal.
OBJETIVO: Neste estudo foi avaliado a potencial vantagem em estender o regime imunossupressor associado a infusão de células de medula óssea do doador depletadas de células T na prevenção da reação do enxerto versus hospedeiro após o transplante intestinal.
MÉTODOS: Transplante heterotópico de intestino delgado foi realizado em ratos Lewis como receptores e DA como doadores, distribuídos em cinco grupos de acordo com a duração da imunossupressão, irradiação e do uso de medula óssea normal ou depletada: G1 (n=6), sem irradiação e G2 (n=9), G3 (n=4), G4 (n=5) e G5 (n=6) foram irradiados com 250 rd. Grupos1, 2, 4 e G3 e 5 foram infundidos com 100 x 106 células da medula normal e depletada respectivamente. Animais no G1,2,3 foram imunossuprimidos com 1mg/kg/FK506/ IM por cinco dias e G4 e cinco por 15 dias. Anticorpos monoclonais contra células CD3 e colunas magnéticas foram utilizadas para a depleção da medula óssea. Os animais foram examinados para a presença de rejeição, reação do enxerto versus hospedeiro, chimerismo e biópsias intestinais e da pele.
RESULTADOS: Rejeição mínima foi observada em todos os grupos; entretanto, a reação do enxerto versus hospedeiro somente nos animais irradiados. Extensão da imunossupressão alterou a gravidade da reação nos animais dos G4 e 5. Rejeição foi a causa mortis no G1 e a reação do enxerto versus hospedeiro nos Grupos 2,3,4 e 5, não controlada com a infusão de medula óssea depletada. O chimerismo total e de células T do doador foi estatisticamente maior nos grupos irradiados em comparação ao G1.
CONCLUSÃO: A extensão do regime de imunossupressão associado a baixas doses de irradiação diminui a gravidade da reação do enxerto versus hospedeiro, não abolida pelo uso de medula óssea depletada.
Descritores: Transplante intestinal. Depleção de células T. Medula óssea.
Introduction
The use of allogeneic bone marrow infusion as a mean of inducing donor-specific tolerance could provide an immunological specific conditioning regimen for organ transplantation. The major limitation to this approach is the toxicity of whole body irradiation necessary to inactivate the host resistance and permit the marrow engraftment1. In data to be publish we demonstrate that even with low doses of irradiation an increased morbidity and mortality due to graft-versus-host disease (GVHD) was observed in recipients after small bowel transplantation. Low cytoreductive regimens of irradiation can successfully decrease the severity of graft rejection but enhance the occurrence of GVHD.
In this study we evaluate whether nonmyeloablactive preconditioning given by low doses of irradiation and associated with unmodified or T-cell depleted (TCD) donor bone marrow (BM) infusions and extended immunosuppression can enhance small bowel allograft and recipient survival.
Methods
Small bowel transplantation was performed using a fully histoincompatible rat strain combination. Male DA (Rt1a) weighing 80 to 100 g were used as donors and female Lewis (Rt11) weighing 180 g as recipients. The animals were obtained from Harlan Sprague Dawley (Indianapolis, IN) and housed at the Division of Veterinarian Resources of the University of Miami.
Small bowel transplantation- Heterotopic small bowel transplant was performed using standard microvascular techniques2. Under metofane anesthesia, the abdomen was opened by a midline incision. The donor small bowel (jejunum-ileum) was removed after intraluminal and intravascular irrigation with cold heparinized saline solution (40C). The bowel was excised with its intact vascular pedicle consisting of the superior mesenteric artery with an aortic cuff and the portal vein. The graft was kept at 40C until transplantation.
The recipient abdomen was opened and the graft was reperfused by anastomosing the aortic cuff to the recipient infrarenal aorta, while the portal vein was anastomosed to the recipient inferior vena cava. The proximal and distal ends of the graft were exteriorized as stomas (Thiry-Vella Loop).
Animals surviving less than 48 hours were considered technical failures and excluded. All animals were fed with a regular laboratory diet and water beginning immediately after transplant.
Experimental design- Thirty male DA rats and thirty female Lewis rats were used as donors and recipients respectively, for a heterotopic small bowel transplant. Animals were distributed into 5 groups according to the use of radiation, duration of immunosuppression and the use of unmodified (UBM) or TCD bone marrow: group 1 (n=6), without irradiation and groups 2 (n=9), 3 (n=4) , 4 (n=5) and group 5 (n=6) was given 250 rd of total body irradiation (TBI) from a cesium-137 source 1 to 2 hours prior the small bowel transplant. Groups 1, 2, 4 and groups 3 and 5 were infused with 100 x 106 UBM cells and TCD BM cells respectively. Animals in groups 1,2,3 were immunosupressed with 1 mg/kg/FK 506/ IM/ for 5 days and in groups 4 and 5 for 15 days, started on the day of the transplant.
Bone marrow preparation- Fresh donor bone marrow was obtained from femurs and tibias of exsanguinated DA rats. Bone marrow was harvested from the long bones by flushing with RPMI 1640 medium (Life Technologies, Grand Island, NY), using a 23-gauge needle. The bone marrow cells were gently ressuspended using an 18-gauge needle and filtered through a sterile nylon mesh. The viability of the cells was checked by trypan blue exclusion and counted before antibody treatment for T cell depletion. Trypan blue exclusion test always showed > 95% viability before infusion. The bone marrow cells were then pelleted by centrifugation at 1500 rpm for 10 min.
For T-cell depletion (TCD), mouse anti-rat CD3 monoclonal antibodies was added to the cell suspension and allowed to incubate at 4oC for 30 minutes. Unbound antibody was removed by washing twice in RPMI medium containing 2% fetal calf serum (Gibco, Grand Island, NY). Cells were then combined with magnetic beads coated with sheep anti-mouse IgG (Dynabeads M-450, Dynal, Lake Success, NY) and gently agitated at 4oC for 1 hour. The final bead:cell ratio was 1:4.
Negative selection of a CD3 positive T-cells was accomplished by exposing the flask containing the labeled marrow to a hand-held magnet for 3 minutes. Elution of the supernatant yielded the final product. Marrow was then ressuspended and cell concentration was adjusted so that the total number of cells (80-100 x 106) was injected in a 1 ml volume in the femoral vein after dissection immediately after transplant.
Adequacy of T cell depletion was confirmed using flow cytometry for detection of cells expressing DA rats class I antigen (haplotype RT1a), with a two color staining cocktail (FITC-conjugated Mab to RT1a and PE-conjugated anti-rat CD3, both from PharMingen, San Diego, CA) before and after depletion.
Immunomagnetic bead-based T-cell depletion were considered efficient when post-depletion of donor CD3+ cells content was greater than 99% of the lymphoid gate.
Postoperative follow-up- Rats were observed daily for clinical signs of graft-versus-host disease (GVHD), such as diffuse erythema (particularly of the ears and extremities), hyperkeratosis of the footpads, skin rash, anorexia, diarrhea and unkempt appearance. The abdomen was palpated daily to detect development of a palpable abdominal mass, a characteristic signal of graft rejection.
Postoperative assessment of chimerism by flow cytometric analysis- Blood samples were collected weekly by tail puncture into anti-coagulated (EDTA) plastic serum vials after light anesthesia. Flow cytometry detection of cells expressing DA rats class I antigen (haplotype RT1a), were done with a two color staining protocol for whole blood. Briefly, 100 ml of a two color cocktail (FITC-conjugated Mab to RT1a and PE-conjugated anti-rat CD3, both from PharMingen, San Diego, CA) were incubated with 100 ml of whole blood. After an incubation of 30 minutes, the red blood cells (RBC) were lysed (with lysing reagent for whole blood Becton/Dickinson San Jose, CA). The presence of donor cells in the recipient Lewis rat (DA cells circulating in the Lewis blood stream) were classified as T cells and non-T cells.
The complete analysis was obtained by gating on mononuclear cells by size and granularity first, then segregating the RT1a positive cells over a background of a non transplanted Lewis (negative control) and within the spectrum of a normal DA (positive control). An additional segregation was done on this last to determined which where also expressing CD3 epitopes. All the analysis were done in a Facs Calibur flow cytometer, San Jose (CA).
Post-transplant biopsies and pathologic studies- Biopsies from the ileostomy and skin were taken every fifteen and thirty days respectively after transplant to assess the presence of rejection and GVHD. Tissue samples were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 4 mm and stained with hematoxylin and eosin. For each biopsy a combination of histological parameters were applied to assess acute and chronic rejection. Acute rejection was graded as mild, moderate and severe based on a semiquantitative scoring system.
The graded features included surface and crypt epithelial injury (reactive epithelial atypia, cryptitis, apoptotic bodies and presence of crypt abscesses), villous blunting, mucosal ulceration, and vasculitis. A diagnosis of chronic rejection was rendered when prominent obliterative vasculopathy was noted.
Statistics- The significance of differences between means was determined by using the Anova and Duncan' s multiple range test. Values were considered statistically significant for p < 0.05.
Results
Histopathologic studies- Ileal biopsies obtained on day 15 after transplant showed minimal to mild acute rejection in all groups. After 30 days, severe acute and early chronic rejection was observed only in non-irradiated animals (group 1) while was considered as mild to moderate in groups 2 and 3. The long-term immunosuppression groups (4 and 5) showed the same histologic pattern of minimal to mild acute rejection during the experiment. Skin biopsies showed the characteristic pattern of GVHD, and were present only in groups submitted to irradiation. GVHD was histologically characterized as moderate to severe in groups 2 and 3 (short course immunosuppression) and as mild in groups 4 and 5 (long-term immunosuppression).
Survival- Clinical evidence of GVHD after transplant, such as diffuse erythema (particularly on the ears and extremities), hyperkeratosis of the footpads, skin infections, anorexia, diarrhea, unkempt appearance and weight loss were the common pattern in irradiated groups, started 3 weeks after transplant. Median survival time was 54,36, 45, 78 and 70 days in groups 1,2,3, 4 and 5 respectively. With exception of group 1 where rejection was the cause of death, all animals in groups 2, 3, 4 and 5 died of GVHD.
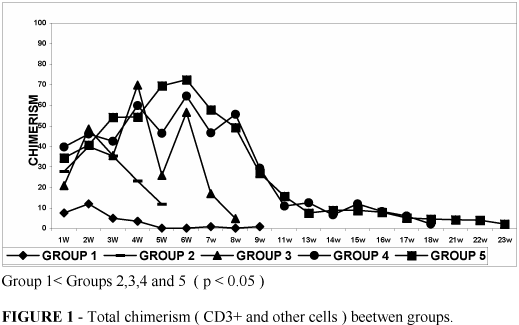
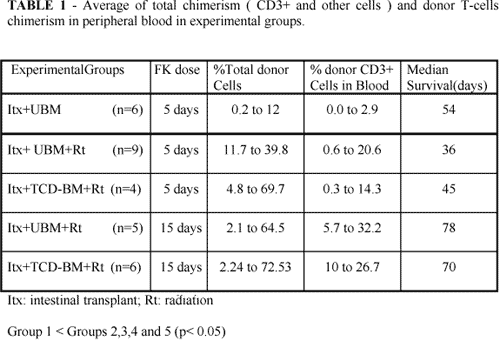
Chimeric studies-Figure 1 and Table 1 shows the total chimerism levels (CD3+ and others cells) in all groups.
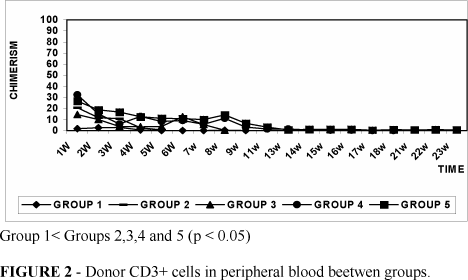
The median chimerism varied from 0.24% to 12% in group 1, 11.78% to 39.84% in group 2, 4.81% to 69.78% in group 3, 2.17% to 64.56% in group 4 and 2.24% to 72.53% in group 5. The total chimerism and the donor T-cell (CD3+ ) chimerism (Figure 2 and Table 1 ) reached the high levels in irradiated groups when compared to non-irradiated group ( p<0.05 ). No differences in total chimerism levels were observed between irradiated groups.
Discussion
These results suggest that a nonmyeloablative regimen based in low dose of irradiation and an extended course of immunosuppression did not enhance the allograft and recipient survival after small bowel transplantation. Extended immunosuppression was the critical factor in reducing the severity of rejection and GVHD after transplant when associated with low dose of irradiation. The occurrence of GVHD was the common pattern in irradiated groups, not avoided by the strategy used in this study to deplete de donor bone marrow from mature T-cells when compared to UBM. Rat bone marrow contains sufficient number of mature T-cells, the most important effector cell subsets in initiating a rejection response against the recipient3. Therefore, removal of these cells effectively prevent the occurrence of GVHD. Previous reports4 showed that permanent donor-specific tolerance can be achieved for vascularized allografts through mixed chimerism without evidence of GVHD.
However, when BM are used after small bowel transplantation, the characteristics and dose of transferred hematopoietic and lymphoid tissue within the graft requires associated strategies in order to immunomodulate the response after transplant. The passenger leukocytes in the intestine have been shown to have inferior tolerogenic qualities compared with BM cells; in addition, have a lineage profile that predisposed to GVHD5.
The high levels of donor CD3+ cells in peripheral blood in the first two weeks after transplant observed in this study were probably the critical factor in the occurrence of GVHD after transplant. Despite the use of TCDBM in G3 and G5, no further decrease in the levels of peripheral donor T cells was observed, suggesting that T cells from the intestinal graft (rather than from the bone marrow), are the cells responsible for the GVHD development. Therefore, immunomodulatory preconditioning regimens addressing the intestinal graft before the transplant seems to be a logical approach in order to enhance the allograft and recipient survival.
We are now evaluating if the combination of donor preconditioning with antilymphocyte serum with the current model can further improve the results after small bowel transplantation.
Conclusion
Extended immunossupression associated to low doses of irratiation decrease the severity of GVHD, not avoided by the use of TCDBMI.
Data do recebimento: 10/11/2002
Data da revisão: 02/12/2002
Data da aprovação: 18/12/2002
Conflict of interest: none
Financial source: none
- 1. Sharabi Y, Sachs DH. Mixed chimerism and permanent specific transplantation tolerance induced by a nonlethal preparative regimen. J Exp Med 1989;169:493-9.
- 2. Monchik GJ, Russel A. Transplantation of small bowel in the rat: technical and immunological considerations. Surgery 1971; 70:693-7.
- 3. Exner BG, Acholonu I, Ildstad ST. Hematopoiet chimerism, tolerance induction and graft-versus-host disease: considerations for composite tissue transfer. Transplant Proc 1998; 30: 2718-22.
- 4. Colson Yl, Zadach K, Nalesnik M, Ildstad ST. Mixed allogeneic chimerism in the rat. Transplantation 1995; 60 (9): 971-8.
- 5. Murase N, Ye Q, Lee RG, Demetris AJ, Abu-Elmagd K, Reyes J, Starzl TE. Immunomodulation of intestinal transplant with allograft irradiation and simultaneous donor bone marrow infusion. Transplant Proc 1999; 40: 565-70.
Publication Dates
-
Publication in this collection
03 Sept 2003 -
Date of issue
Mar 2003
History
-
Accepted
18 Dec 2002 -
Reviewed
02 Dec 2002 -
Received
10 Nov 2002