Abstracts
PURPOSE: The effect of a highly selective 6-hydroxydopamine (6-OHDA)-induced lesion of the nigrostriatal system on the astroglial and microglial activation was analysed in adult Wistar rats after an unilateral striatal injection of the neurotoxin. METHODS: Male rats received an unilateral stereotaxical injection of the 6-OHDA in the left side of the neostriatum and were sacrificed 22 days later. Control animals received the injection of the solvent. The rotational behaviour was registered by a rotometer just before the sacrifice. Immunohistochemistry was employed for visualization of the tyrosine hydroxylase (TH) positive dopamine cells, glial fibrillary acidic protein (GFAP) immunolabeled astrocytes and OX42 immunoreactive microglia. Stereological method employing the optical disector was used to estimate the degree of the changes. RESULTS: The striatal injection of the 6-OHDA induced a massive disappearance (32% of control) of the TH immunoreactive terminals in a defined area within the striatum surrounding the injection site. A disappearance (54% of control) of dopamine cell bodies was observed in a small region of the ipsilateral pars compacta of the substantia nigra (SNc). The GFAP and OX42immunohistochemistry revealed astroglial and microglial reactions (increases in the number and size of the cells) in the ipsilateral neostriatum and SNc of the 6-OHDA injected rats. CONCLUSIONS: The striatal injection of 6-OHDA leads to retrograde degeneration as well as astroglial and microglial activation in the nigrostriatal dopamine pathway. Modulation of activated glial cells may be related to wound repair and to the trophic paracrine response in the lesioned nigrostriatal dopamine system.
Astrocyte; Microglia; Immunohistochemistry; Dopamine lesion; Glial activation
OBJETIVO: O efeito de uma lesão altamente seletiva induzida pela 6-hidroxidopamina (6-OHDA) no sistema nigroestriatal dopaminérgico sobre a ativação astroglial e microglial foi analisado em ratos Wistar adultos após uma injeção unilateral da neurotoxina no estriado. MÉTODOS: Ratos receberam injeção estereotáxica unilateral de 6-OHDA no lado esquerdo do neoestriado e foram sacrificados 22 dias depois. Os animais controles receberam injeção de solvente. O comportamento rotacional foi registrado antes do sacrifício. A técnica da imunohistoquímica foi utilizada para a visualização das células dopaminérgicas positivas à tirosina hidroxilase (TH), dos astrócitos marcados pela proteína glial fibrilar ácida (GFAP) e da microglia imunorreativa ao OX42. O método estereológico empregando o dissector óptico foi usado para estimar o grau das mudanças no sistema nigroestriatal. RESULTADOS: A injeção estriatal de 6-OHDA induziu o desaparecimento massivo (32% do controle) de terminais imunorreativos à TH numa área definida dentro do estriado, ao redor do local da injeção. Desaparecimento de corpos celulares dopaminérgicos (54% do controle) foi observado em pequena região da porção compacta da substância negra (SNc) ipsilateral. A imunohistoquímica da GFAP e da OX42 revelou reação astroglial e microglial (aumentos no número e tamanho das células) no estriado ipsilateral e na SNc ipsilateral de ratos injetados com a 6-OHDA. CONCLUSÕES: A injeção estriatal de 6-OHDA leva à degeneração retrógrada, bem como à ativação astroglial e microglial na via nigroestriatal dopaminérgica. A modulação das células gliais ativadas pode estar relacionada à cicatrização e à resposta trófica parácrina no sistema nigroestriatal lesado.
Astrócito; Microglia; Imunohistoquímica; Lesão dopaminérgica; Ativação glial
3 ORIGINAL ARTICLE
Striatal injection of 6-hydroxydopamine induces retrograde degeneration and glial activation in the nigrostriatal pathway1 1 Article from the Laboratory of Neuroregeneration, Department of Anatomy, University of São Paulo (USP), São Paulo, Brazil.
Striatal Injeção estriatal de 6-hidroxidopamina induz degeneração retrógrada e ativação glial na via nigro-estriatal
Ricardo Wilson Pinho RodriguesI; Vânia Canterucci GomideII; Gerson ChadiIII
IMaster, Department of Anatomy, University of São Paulo (USP), São Paulo, Brazil
IIPhD, Department of Anatomy, University of São Paulo (USP), São Paulo, Brazil
IIIFull Professor, Department of Anatomy, University of São Paulo (USP), São Paulo, Brazil
Correspondence Correspondence to Gerson Chadi Department of Anatomy. ICB. USP Av Prof Lineu Prestes, 2415 CEP: 05508-900 São Paulo SP Brazil Phone: (11)3091-7384 / Fax: (11)3091-7366 E-mail: gerchadi@usp.br
ABSTRACT
PURPOSE: The effect of a highly selective 6-hydroxydopamine (6-OHDA)-induced lesion of the nigrostriatal system on the astroglial and microglial activation was analysed in adult Wistar rats after an unilateral striatal injection of the neurotoxin.
METHODS: Male rats received an unilateral stereotaxical injection of the 6-OHDA in the left side of the neostriatum and were sacrificed 22 days later. Control animals received the injection of the solvent. The rotational behaviour was registered by a rotometer just before the sacrifice. Immunohistochemistry was employed for visualization of the tyrosine hydroxylase (TH) positive dopamine cells, glial fibrillary acidic protein (GFAP) immunolabeled astrocytes and OX42 immunoreactive microglia. Stereological method employing the optical disector was used to estimate the degree of the changes.
RESULTS: The striatal injection of the 6-OHDA induced a massive disappearance (32% of control) of the TH immunoreactive terminals in a defined area within the striatum surrounding the injection site. A disappearance (54% of control) of dopamine cell bodies was observed in a small region of the ipsilateral pars compacta of the substantia nigra (SNc). The GFAP and OX42immunohistochemistry revealed astroglial and microglial reactions (increases in the number and size of the cells) in the ipsilateral neostriatum and SNc of the 6-OHDA injected rats.
CONCLUSIONS: The striatal injection of 6-OHDA leads to retrograde degeneration as well as astroglial and microglial activation in the nigrostriatal dopamine pathway. Modulation of activated glial cells may be related to wound repair and to the trophic paracrine response in the lesioned nigrostriatal dopamine system.
Key words: Astrocyte. Microglia. Immunohistochemistry. Dopamine lesion. Glial activation.
RESUMO
OBJETIVO: O efeito de uma lesão altamente seletiva induzida pela 6-hidroxidopamina (6-OHDA) no sistema nigroestriatal dopaminérgico sobre a ativação astroglial e microglial foi analisado em ratos Wistar adultos após uma injeção unilateral da neurotoxina no estriado.
MÉTODOS: Ratos receberam injeção estereotáxica unilateral de 6-OHDA no lado esquerdo do neoestriado e foram sacrificados 22 dias depois. Os animais controles receberam injeção de solvente. O comportamento rotacional foi registrado antes do sacrifício. A técnica da imunohistoquímica foi utilizada para a visualização das células dopaminérgicas positivas à tirosina hidroxilase (TH), dos astrócitos marcados pela proteína glial fibrilar ácida (GFAP) e da microglia imunorreativa ao OX42. O método estereológico empregando o dissector óptico foi usado para estimar o grau das mudanças no sistema nigroestriatal.
RESULTADOS: A injeção estriatal de 6-OHDA induziu o desaparecimento massivo (32% do controle) de terminais imunorreativos à TH numa área definida dentro do estriado, ao redor do local da injeção. Desaparecimento de corpos celulares dopaminérgicos (54% do controle) foi observado em pequena região da porção compacta da substância negra (SNc) ipsilateral. A imunohistoquímica da GFAP e da OX42 revelou reação astroglial e microglial (aumentos no número e tamanho das células) no estriado ipsilateral e na SNc ipsilateral de ratos injetados com a 6-OHDA.
CONCLUSÕES: A injeção estriatal de 6-OHDA leva à degeneração retrógrada, bem como à ativação astroglial e microglial na via nigroestriatal dopaminérgica. A modulação das células gliais ativadas pode estar relacionada à cicatrização e à resposta trófica parácrina no sistema nigroestriatal lesado.
Descritores: Astrócito. Microglia. Imunohistoquímica. Lesão dopaminérgica. Ativação glial.
Introduction
Reactive microglia and astrocytes undergo hiperplasy and hipertrophy following a brain injury1,2,3,4. Enlargement of the cell body, increased number and size of astroglial processes and the increased synthesis of glial fibrillary acidic protein (GFAP), an intermediate filament protein of the astroglial cytoskeletum, are indications of the state of astroglial activation following brain damage5. Furthermore, microglia also become reactive after central nervous system (CNS) lesion2. Furthermore, number and the size of microglial cytoplasm and processes increase few minutes after brain injury6. When microglia start to undergo phagocytosis, the cells show a round shape and loose their processes. In that state the microglial cells are called ameboid microglia6.
In spite of the fact that gliotic scar may represent a physical barrier to axonal regeneration within the CNS7, reactive astrocytes synthetize increased amount of neurotrophic substances8,3,9 which are related to the paracrine trophic actions to lesioned neurons10.
Many authors have described the mechanisms involved in the cellular paracrine trophic regulation following neuronal lesion11,8.
Chemical interaction between activated microglia and astroglia might influence the survival of damaged neurons12,13,7. It is known that activated microglia synthetize interleukins that stimulate the activation of astrocytes which in turn produce increased amount of substances with neurotrophic properties7.
The degeneration of the nigrostriatal system following a nigral stereotaxical injection of 6-hydroxydopamine (6-OHDA) is a well described method to develop parkinsonism in rats14,15. However, the analysis of the retrograde degeneration of the nigrostriatal pathway following a striatal unilateral injection of 6-OHDA and the related glial activation are remaining to be detailed investigated.
By means of GFAP and OX42 immunohistochemistry which label astrocyte and microglia, respectively, within the CNS, combined to a computer assisted stereological method, this report has quantified the degree of astroglial and microglial activation following a selective injury of the rat nigrostriatal system promoted by a striatal injection of 6-OHDA. The dopaminergic neurons in the pars compacta of the substantia nigra (SNc) and the terminals in the neostriatum were examined by tyrosine hydroxylase (TH) immunohistochemistry. The apomorphine induced stereotyped rotational behaviour was also employed to evaluate the degree of the nigrostriatal dopamine lesion.
Methods
Animal: Adult male Wistar rats [body weight (b.w.) 220-250g] from the Institute of Biomedical Sciences (São Paulo, Brazil) were used in the present study. Rats were kept under controlled temperature and humidity conditions with a standardized light and dark cycle (lights on at 7:00 a.m. and off at 7:00 p.m.) with free access to food pellets and tap water.
Neurosurgery and behaviour: The animals received a chloral hydrate anaesthesia (Merck, 0.6mg/100mg b.w.) and were placed in a stereotaxical apparatus (Kopf). The skull was opened with a drill and 6-hydroxydopamine hydrochloride (8g/4l, Sigma, U.S.A.), dissolved in 0.9% saline containing ascorbic acid (0.2mg/ml), was unilaterally injected during 15 minutes into the left neostriatum (n=4). The coordinates were 1.2mm, lateral 2.5mm, and 4.7 caudal (Figure 1A) according to the atlas of Paxinos and Watson16.
The skin was sutured. Control animals received the solvent in the same fashion of the experimental animals. Twenty two days after surgery the rats were placed in the rotometer to evaluate the degree of stereotyped rotation induced by apomorphine following dopamine lesion. 6-OHDA and solvent rats received the nonselective dopamine agonist apomorphine hydrochloride (0.5mg/kg-1) subcutaneously (s.c.) in the neck and their behaviour was recorded in the rotometer15 that allows the continuous recording of turns to the left or the rigth (Figure 2). The rotational counts (360o turns) were recorded at 5 minutes intervals for a total period of 75 minutes. The results were then trasfered to a host computer for analysis. The rotational behaviour of each animal was plotted as a number of 360o turns 5 min-1 during the entire duration of the recording. The data were plotted and the means ± s.e.m. were presented. After the recording, the animals were deeply anaesthetized and sacrificed by a transcardiac perfusion with 70 ml isotonic saline at room temperature followed by 350 ml of fixation fluid (4oC) during 6 minutes. The fixative17 consisted of 4% paraformaldehyde (w/v) and 0.2% picric acid (v/v) in 0.1M phosphate buffer (pH 6.9). The brains were removed and kept in the fixative solution at 4oC for 90 minutes. Following, the brains were rinsed in 20% sucrose (Merck, Germany) dissolved in 0.1M phosphate buffered saline, pH 7.4 (PBS) during 48 hours, frozen in dry ice-cooled (-40oC) isopentane (Sigma) and stored at -70oC until use.
Immunohistochemistry: Coronal brain adjacent serial 60m thick sections were obtained in a cryostat (Leica, CM3000, Germany) from rostro-caudal levels of the midbrain (-4.30mm to -6.30mm) and forebrain (+1.60mm to 0.80mm), respectively, according to the atlas of Paxinos and Watson16. The sections were sampled systematically during sectioning. Five or eigth series in a rostro-caudal order including every fifth or eighth section were taken to immunohistochemistry from midbrain or forebrain, respectively. The immunoreactivity was detected using avidin-biotin peroxidase technique18. Free-floating sections were washed 2x10 minutes in PBS (0.1M, pH 7.4) at room temperature and incubated with normal goat serum (NGS) 5% for 30 minutes at room temperature. Following, series of sections were incubated for 48 hours at 4oC under shaking with a rabbit polyclonal antiserum to GFAP (Dakopatts, Denmark) diluted 1:1500, with a mouse monoclonal antiserum to TH (Incstar, U.S.A., diluted 1:1000) or with a mouse monoclonal antiserum to OX42 (Harlon, U.K., diluted 1:1000), respectively. The antibodies were diluted in PBS containing 0.5% Triton X-100 (Sigma) and 1% bovine serum albumin (Sigma). The sections were washed again in PBS (2x10 minutes) and incubated with biotinylated goat anti-rabbit or horse anti-mouse immunoglobulins (both diluted 1:200, Vector, U.S.A.) for 2 hours. The sections were washed again in PBS and incubated in an avidin-biotin peroxidase complex solution (both diluted 1:100, Vectastain, Vector) for 90 minutes. Visualization of the immunoreactivities was carried out in 3-3-diaminobenzidine tetrahydrocloride (DAB, Sigma) as a chromogen and H2O2 (0.05%, v/v, Sigma) for 8-10 minutes.
For standardization of the immunohistochemical procedure we have used a dilution of the primary antibody and a concentration of the DAB far from saturation and an incubation time adjusted so that, the darkest elements in the brain sections were bellow saturation19.
The sections were analysed in a AX70 Olympus photomicroscope (U.S.A.). The image were digitalized in a Zeiss microscope (Axioskop 2, Germany).
Stereology. analysis of the estimated number: The optical fractionator20,21 was used to sample TH, GFAP and OX42 immunoreactive profiles in sampled fields placed in one section of the SNc and neostriatum (Figures 1A e B ). The striatal section showed the epicenter of injection. The GFAP and OX42 as well as the TH immunoreactive nigral sections were counterstained with cresyl violet to allowed interallia visualization of the nucleus which makes the countings more accurate. The TH immunoreactive striatal sections were not counterstained with cresyl violet. TH immunoreactive varicosities, defined as a dilatation of the TH positive fibers, were counted in the sampled region of the neostriatum. TH dopamine cell bodies counterstained with cresyl violet were counted in the sampled SNc. Astroglial and microglial immunoreactive cells counterstained with cresyl violet were counted in the sampled regions of the neostriatum and SNc.
The stereological analysis was developed using a CAST-system (Computer Assisted Stereological Toolbox). Briefly: an Olympus BX50 microscope (Olympus, Denmark) was interfaced with a computer (IBM 330-P75, U.S.A.) and a colour video camera (JAI 2040, Protec, Japan), both linked to a colour video monitor (G70, IBM). The GRID software package (Interactvision, Silkeborg, Denmark) was used to generate sampling (counting) frames as an overlay image to the microscopic image on the monitor as well as to control the motorized X-Y stage (Lang, Huttenberg, FRG). A microcator (MT12, Heidenhain, FRG) was linked to the microscope to monitor movements in a vertical (Z) direction. The border of the region to be quantified was outlined using a 4x objective (Figure 1A,B ). The step rates were entered (70-150µm depending on the immunoreactivity analysed), after which the program created series of uniformly sampled fields of vision throughout the entire delimitated regions. For counting the profiles, a 100x oil-immersion objective with the numerical aperture 1.4 was used. The counting frames (with a known area, aframe) were created by the GRID software. The sampling volume (disector) in the Z-axis extended 3-20µm deep (height of the disector) after excluding the parts of the section close to the slide and coverslip (3-5µm). Also the total thickness of the section was measured in each sampled field. The estimated number of immunoreactive profiles inside the sampled field was calculated according to the formula: N = Q- . ƒ1 . ƒ2 . The numbers of the ipsilateral and contralateral sides of the 6-OHDA striatal injection were presented. The numbers of ipsilateral and contralateral striatum represent the mean of the estimated number in two sampled fields of each striatal side (Figure 1A).
Statistical analysis: Statistical analysis was performed according to the non-parametric two-tailed Mann-Whitney U-test22. The sham operated and 6-OHDA treated groups were compared.
Results
Rotational behaviour: Apomorphine induced an increased number of contralateral rotation in the unilateral 6-OHDA striatal injected rats 22 days after the lesion (Figure 3). Contralateral turning was not seen in the solvent striatal injected animals following apomorphine stimulation (Figure 3). Furthermore, ipsilateral rotation was not found either in the 6-OHDA or solvent striatal injected rats after apomorphine injection by 22 days after surgery (Figure 3).
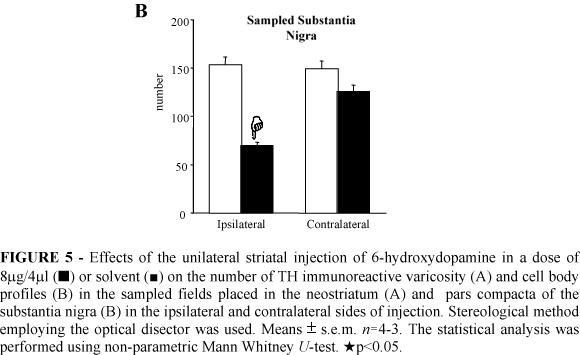
TH immunoreactivity: TH immunoreactive terminals were found to be disappeared in a defined area within the ipsilateral neostriatum surrounding the site of the 6-OHDA injection 22 days after the surgery (Figures 4A e B). A slight disappearance of the TH immunoreactive fibers was seen in the ipsilateral neostriatum of the solvent injected rats only close to the needle track (not shown). Furthermore, a disappearance of dopamine cell bodies was observed in a defined region of the ipsilateral SNc of the 6-OHDA lesioned rats (Figure 4D). No changes in the TH immunoreactivity were seen within the contralateral neostriatum and SNc of the 6-OHDA injected rats. Also, no disappearance of dopamine cells was found in the SNc of the solvent treated rats (Figure 4C).
The stereological analysis demonstrated a 32% reduction in the number of TH immunoreactive varicosities in the ipsilateral neostriatum of the 6-OHDA injected rats (Figure 5A). There was a decrease of 54% in the number of TH immunoreactive neurons in the ipsilateral SNc of the 6-OHDA lesioned rats (Figure 5B). No changes in the number of TH immunoreactive varicosity and of the TH immunoreactive neurons were found in the sampled contralateral neostriatum and SNc, respectively, of the 6-OHDA striatal injected rats 22 days after surgery (Figure 5).
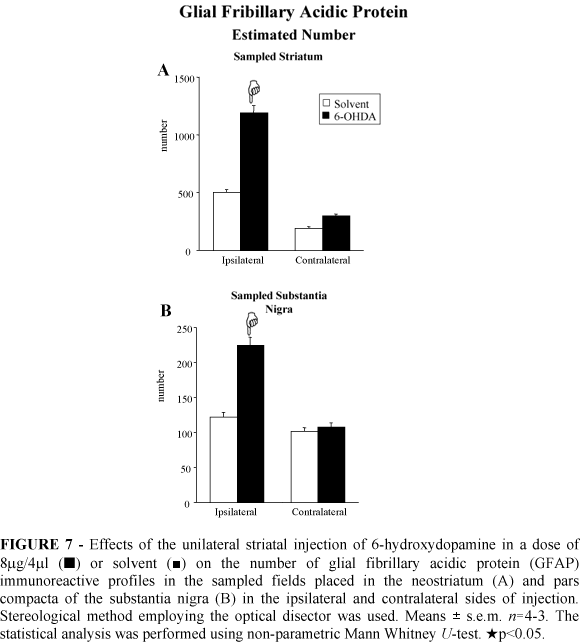
GFAP immunoreactivity: The GFAP immunohistochemistry revealed an increased density of the astroglial GFAP immunoreactive profiles in the ipsilateral neostriatum and SNc of the 6-OHDA lesioned rats (illustrated in the figure 6B). The reactive astrocytes possessed increased size of the cytoplasm and number of processes (Figure 6B). An astrocytic reaction was found in the neostriatum of the solvent injected rats only bordering the needle track (not shown). The GFAP immunoreactivity was not changed within the SNc of the solvent treated rats.
The stereological analysis demonstrated a significant increased number of GFAP immunoreactive astroglial cells in the ipsilateral neostriatum (137%) and ipsilateral SNc (83%) of the 6-OHDA injected rats (Figure 7). The number of astroglial immunoreactive profiles did not change in the contralateral neostriatum and SNc after striatal 6-OHDA injection (Figure 7).
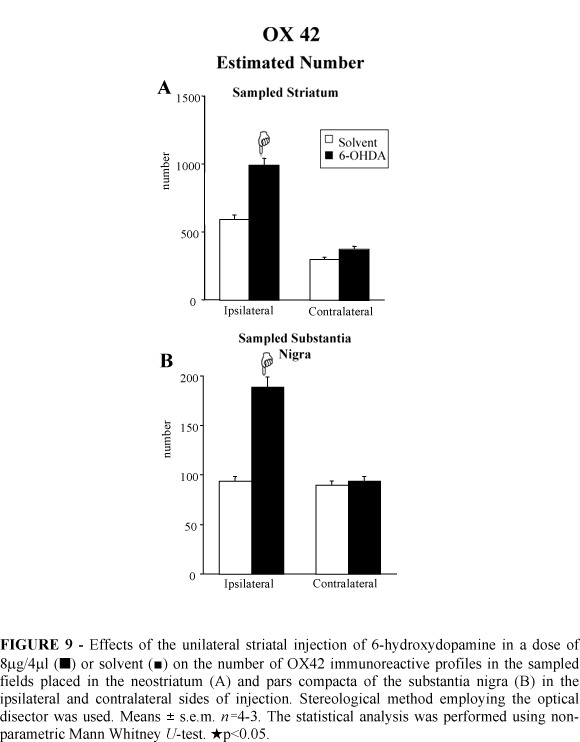
OX42 immunoreactivity: The OX42 immunohistochemistry showed an increased number of microglial OX42 immunoreactive cells in the ipsilateral neostriatum and SNc of 6-OHDA injected rats (illustrated in the Figure 8). In the solvent control group the OX42 immunoreactivity was not changed in the ipsilateral neostriatum of the solvent injected rats as well as in the contralateral neostriatum of the rats of both 6-OHDA and solvent groups (not shown).
The stereological analysis revealed increases of 67% and 100% in the number of microglial OX42 immunoreactive cells in the ipsilateral neostriatum and SNc, respectively, of the 6-OHDA lesioned rats (Figure 9). The counts demonstrated no changes in the number of microglial immunoreactive profiles in the contralateral neostriatum and SNc after 6-OHDA injection (Figure 9).
Discussion
6-OHDA injection in the substantia nigra triggering a massive disappearance of dopamine neurons in the ipsilateral nigrostriatal pathway and apomorphine-induced contralateral rotation23,14 is classical model to promote experimental parkinsonism in rats. Recently, 6-OHDA-induced lesion in the nigrostriatal dopamine pathway has been achieved after striatal injection of 6-OHDA23,24.
The present paper demonstrated the effects of 6-OHDA striatal injection on the degeneration of the nigrostriatal TH immunoreactivity as well as on the degree of apomorphine-induced stereotyped rotational behavioral in rats. The subsequent astroglial and microglial activation following dopamine lesion was also analysed.
Stereotyped rotational behaviour is triggered in 6-OHDA lesioned rats after the apomorphine administration. The circling behaviour is archieved when the striatal post-synaptic dopamine receptors become supersensitive after dopamine lesion25,26. The present paper showed increases in the mean of contralateral turns in lesioned rats 22 days after 6-OHDA striatal injection. Reductions of the number of TH positive varicosities in the neostriatum and dopamine neurons in the SNc in the ipsilateral side of the 6-OHDA injection were also described in this paper. These findings are in agreement with previous reports which demonstrated decreases in the dopamine levels within the neostriatum after a local injection of 6-OHDA27,23 and a disappearance of TH immunoreactive cell bodies within the ipsilateral side of SNc24. Furthermore, Cadet and co-workers28 have shown a diminution of TH immunoreactivity in the neostriatum and in the SNc after a striatal injection of 6-OHDA28.
Taken together, the results of previous papers and the findings of this work indicate the occurence of a retrograde degeneration of the dopamine nigrostriatal system following a striatal injection of 6-OHDA in rats. Furthermore, the degree of the dopamine lesion can be measured behaviorally and using stereological method combined with TH immunoreactivity.
Several papers have demonstrated astroglial reaction after a brain lesion29,30,5. Astroglial activation has been observed close to the lesion site3 and also accompaining the lesioned neuronal pathways2. Once activated, astroglial cells synthesize increased amount of GFAP and undergo morphological changes31.
The present report described intense astroglial reaction in the nigrostriatal dopamine system 22 days after an unilateral 6-OHDA striatal injection. These findings indicate that the astroglial activation occurs not exclusively close the lesion site of the 6-OHDA striatal injection where the astroglial reaction can be related to the scar and wound repair but also in the ipsilateral SNc, accompaining the retrograde degeneration of the nigrostriatal projection.
After a CNS lesion microglial cells trigger phagocytosis12 In that state microglia become ameboid, retract their processes and replicate. In the present work we have observed microglial activation close to the striatal site of 6-OHDA injection and also in the ipsilateral SNc. These data are similar to that found by Akiyama and co-workers32 that observed reactive microglia surrounding lesioned dopamine neurons32. Microglial activation in the lesioned neostriatum and in the ipsilateral SNc may be related to the elimination of neuronal debris.
It has to be emphasized that the degree of astroglial and microglial activation was very similar as it was the regions i.e. ipsilateral 6-OHDA lesioned nigrostriatal pathway where both glial cells underwent reaction. These observations already indicate a possible modulation between these two glial cell populations7. The release of microglial interleukins has been mentioned to be involved in the activation of near astrocytes which in turn synthesize increased amounts of substances of neurotrophic properties7,8. Thus, the findings of the present paper are in line with previous reports that have shown that chemical modulation of reactive glial cells may regulate immunological response in the lesioned CNS, triggering neuronal survival or death12,33. The modulation of reactive microglia and astrocytes may have triggered trophic responses in the lesioned nigrostriatal pathway that may have interfered with the degree of dopamine lesion. These phenomena is summarized in the Figure 10.
Conclusion
In conclusion, striatal injection of 6-OHDA leads to the lesion of local dopamine terminals and retrograde degeneration of nigral dopamine neurons. The lesion of the nigrostriatal system is accompained by an astroglial and microglial reaction in the nigrostriatal pathway which may be related to the paracrine trophic actions on remaining dopamine neurons.
Acknowledgements
We wish to thank Ms. Patrícia R. de Campos for excellent technical assistance.
Data do recebimento: 28/03/2003
Data da revisão: 13/04/2003
Data da aprovação: 24/04/2003
Conflict of interest: none
Financial sources: FAPESP and CNPq
- 1. Bignami A, Dahl D. The astroglial response to stabbing. Immunofluorescence studies with antibodies to astrocyte-specific protein (GFA) in mammalian and submammalian vertebrates. Neuropathol Appl Neurobiol 1976;2: 99-111.
- 2. Cerutti S. M., Chadi G. S100 immunoreactivity is increased in reactive astrocytes of the visual pathways following a mechanical lesion of the rat occipital cortex. Cell Biol Int 2000;24: 35-49.
- 3. Gomide V. C., Chadi G. The trophic factors S-100beta and basic fibroblast growth factor are increased in the forebrain reactive astrocytes of adult callosotomized rat. Brain Res 1999;835: 162-74.
- 4. Shao Y, McCarthy KD. Plasticity of astrocytes. Glia 1994;11: 147-55.
- 5. Goss JR, Morgan DG. Enhanced glial fibrillary acidic protein RNA response to fornix transection in aged mice. J Neurochem 1995;64: 1351-60.
- 6. Streit WJ, Graeber MB Kreutzberg GW. Functional plasticity of microglia: a review. Glia 1988;1: 301-7.
- 7. Giulian D, Vaca K ,Corpuz M. Brain glia release factors with opposing actions upon neuronal survival. J Neurosci 1993;13: 29-37.
- 8. Chadi G, Cao Y, Pettersson RF, Fuxe K. Temporal and spatial increase of astroglial basic fibroblast growth factor synthesis after 6-hydroxydopamine-induced degeneration of the nigrostriatal dopamine neurons. Neuroscience 1994;61: 891-910.
- 9. Frautschy SA, Walicke PA, Baird A. Localization of basic fibroblast growth factor and its mRNA after CNS injury. Brain Res 1991;553: 291-9.
- 10. Fawcett J. Astrocytes and axon regeneration in the central nervous system. J Neurol 1994;242: S25-8.
- 11. Chadi G, Rosen L, Cintra A, Tinner B, Zoli M., Pettersson RF, Fuxe K. Corticosterone increases FGF-2 (bFGF) immunoreactivity in the substantia nigra of the rat. Neuroreport 1993a;4: 783-6.
- 12. Giulian D. Ameboid microglia as effectors of inflammation in the central nervous system. J Neurosci Res 1987;18: 155-71, 132-3.
- 13. Hetier E, Ayala J, Denefle P, Bousseau A, Rouget P, Mallat M, Prochiantz A. Brain macrophages synthesize interleukin-1 and interleukin-1 mRNAs in vitro. J Neurosci Res 1988;21: 391-7.
- 14. Ungerstedt U. Postsynaptic supersensitivity after 6-hydroxy-dopamine induced degeneration of the nigro-striatal dopamine system. Acta Physiol Scand Suppl 1971;367: 69-93.
- 15. Ungerstedt U, Arbuthnott GW. Quantitative recording of rotational behavior in rats after 6-hydroxy-dopamine lesions of the nigrostriatal dopamine system. Brain Res 1970;24: 485-93.
- 16. Paxinos G, Watson C. The rat brain: in stereotaxic coordinates. 1986.
- 17. Zamboni I, DeMartino C. Buffered picric acid formaldehyde: a new rapid fixative for electron microscopy. J Cell Biol 1967;35: 148A.
- 18. Hsu SM, Raine L, Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 1981;29: 577-80.
- 19. Zoli M, Zini I, Agnati LF, Guidolin D, Ferraguti F, Fuxe K. Aspects of neuronal plasticity in the central nervous system. I.Computer-assisted image analysis methods. Neurochem Int 1990;16: 383-418.
- 20. Janson AM., Moller A. Chronic nicotine treatment counteracts nigral cell loss induced by a partial mesodiencephalic hemitransection: an analysis of the total number and mean volume of neurons and glia in substantia nigra of the male rat. Neuroscience 1993;57: 931-41.
- 21. Chadi G, Moller A, Rosen L, Janson AM, Agnati LA, Goldstein M, Ogren SO, Pettersson R, Fuxe K. Protective actions of human recombinant basic fibroblast growth factor on MPTP-lesioned nigrostriatal dopamine neurons after intraventricular infusion. Exp Brain Res 1993b;97: 145-58.
- 22. Hollander M, Wolfe DA. Non-parametric statistical methods. 1973.
- 23. Przedborski S, Levivier M, Jiang H, Ferreira M, Jackson-Lewis V, Donaldson D, Togasaki DM. Dose-dependent lesions of the dopaminergic nigrostriatal pathway induced by intrastriatal injection of 6-hydroxydopamine. Neuroscience 1995;67: 631-47.
- 24. Kirik D, Rosenblad C, Bjorklund A. Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat. Exp Neurol 1998;152: 259-77.
- 25. Schwarting RK, Huston JP. The unilateral 6-hydroxydopamine lesion model in behavioral brain research. Analysis of functional deficits, recovery and treatments. Prog Neurobiol 1996;50: 275-331.
- 26. Hefti F, Melamed E, Wurtman RJ. Partial lesions of the dopaminergic nigrostriatal system in rat brain: biochemical characterization. Brain Res 1980;195: 123-37.
- 27. Lee CS, Sauer H, Bjorklund A. Dopaminergic neuronal degeneration and motor impairments following axon terminal lesion by instrastriatal 6-hydroxydopamine in the rat. Neuroscience 1996;72: 641-53.
- 28. Cadet JL, Last R, Kostic V, Przedborski S, Jackson-Lewis V. Long-term behavioral and biochemical effects of 6-hydroxydopamine injections in rat caudate-putamen. Brain Res Bull 1991;26: 707-13.
- 29. Baumann N, Baron-Van Evercooren A, Jacque C, Zalc B. Glial biology and disorders. Curr Opin Neurol Neurosurg 1993;6: 27-33.
- 30. Niquet J, Ben-Ari Y, Represa A. Glial reaction after seizure induced hippocampal lesion: immunohistochemical characterization of proliferating glial cells. J Neurocytol 1994;23: 641-56.
- 31. Stromberg I, Bjorklund H, Dahl D, Jonsson G, Sundstrom E, Olson L. Astrocyte responses to dopaminergic denervations by 6-hydroxydopamine and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine as evidenced by glial fibrillary acidic protein immunohistochemistry. Brain Res Bull 1986;17: 225-36.
- 32. Akiyama H, McGeer PL. Microglial response to 6-hydroxydopamine-induced substantia nigra lesions. Brain Res 1989;489: 247-53.
- 33. Giulian D, Li J, Leara B, Keenen C. Phagocytic microglia release cytokines and cytotoxins that regulate the survival of astrocytes and neurons in culture. Neurochem Int 1994;25: 227-33.
Publication Dates
-
Publication in this collection
21 Aug 2003 -
Date of issue
Aug 2003
History
-
Received
28 Mar 2003 -
Reviewed
13 Apr 2003 -
Accepted
24 Apr 2003