Abstracts
Purpose: To show that a non-laser light source can be constructed, using a 500 W Tungsten lamp and optical filters, and that this light source produces photodynamic effect via protoporphyrin IX (PpIX) similar to the effect produced by HeNe laser light. Methods: The broad band spectrum from a Tungsten lamp was filtered. Infrared and blue part of the spectrum was discarded by absorption process and the fraction of the spectrum, centered at the red portion, was filtered by an interference filter. Photodynamic effect was studied by the activity on endogenous PpIX of Harderian glands of Wistar rats. Twenty rats were used for the experiment. Each animal had its two Harderian gland surgically exposed, so one of them was treated with the system while the other was kept as control. After a 30 minutes period of treatment, the animals were sacrificed and their glands were removed for histological analysis. This analysis was compared to earlier published results obtained with HeNe laser light. Results: The resultant light source emission was centered around (636 ± 6.5) nm and gives up to 11.3 mW/cm² power density. It produces photodynamic effect in Harderian gland, observed either by fluorescence spectroscopy or by histological microscopy. Conclusion: There is no noticeable difference in Photodynamic effect results if activated by HeNe laser or by the proposed non-laser light source emitting at the red portion of the spectrum.
Photodynamic Therapy; Lasers; Harderian gland
Objetivo: Descrever a construção de uma fonte de luz não-laser, a partir de uma lâmpada de Tungstênio e filtros óticos adequados e demonstrar que sua eficiência em estudos fotodinâmicos, mediados por protoporfirina IX, é semelhante a do laser de Hélio Neônio. Métodos: As regiões Infravermelha e Azul do espectro de emissão óptica de uma lâmpada de Tungstênio foram convenientemente descartadas por processos de absorção, enquanto que a fração centrada na região do vermelho foi removida com o uso de um filtro de interferência. O efeito fotodinâmico foi estudado, em glândula Harderiana de ratos Wistar em razão da produção endógena de protoporfirina IX (PpIX) por estas glândulas. Foram utilizados 20 ratos. Cada animal teve as duas glândulas expostas cirurgicamente, sendo uma delas tratada com a fonte não-laser e a outra mantida como controle. Após tratamento por 30 minutos os animais foram sacrificados e suas glândulas removidas para estudo histológico. Os resultados foram comparados a estudos realizados com laser de Hélio Neônio, já publicados. Resultados: A luz produzida pelo equipamento está centrada em torno de (636 ± 6,5) nm, fornecendo uma densidade de potência de 11,3 mW/cm². Os efeitos fotodinâmicos produzidos na glândula Harderiana, podem ser observados tanto por espectroscopia de fluorescência como por microscopia ótica. Conclusão: Não foram observadas diferenças significativas nos resultados do efeito fotodinâmico obtidos com a fonte de luz não-laser proposta, em comparação aos resultados conhecidos com o uso do laser de Hélio Neônio.
Fotoquimioterapia; Lasers; Glândula de Harder
ORIGINAL ARTICLE
Construction and evaluation of a non-laser optical system for photodynamic process excitation1 1 Work developed at the Laser Laboratory of the Center for Medicine and Experimental Surgery (NMCE) School of Medical Science (FCM), State University of Campinas - UNICAMP, Campinas, SP, Brazil.
Construção e avaliação de um sistema óptico não-laser para excitação de processos fotodinâmicos
Rickson Coelho MesquitaI; Edmyr Rosa dos ReisII; Jorge Humberto NicolaIII; Ester Maria Danielli NicolaIV
IFormer fellowship of FAPESP at Laser Laboratory of NMCE. Actually MSc student at the Institute of Physics "Gleb Wataghin" - UNICAMP
IIDoctor in Biomedical Science, Biologist of the Laser Laboratory of NMCE
IIIProfessor Doctor of the Physical Institute Gleb Wataghin (retired) and Professor of the Postgraduate program in Biomedical Science FCM UNICAMP
IVProfessor Doctor of the OFT / ENT Department, FCM-UNICAMP. Head of the Laser Laboratory NMCE, FCM-UNICAMP
Correspondence Correspondence to Prof. Dr. Ester Maria Danielli Nicola Dep. of Otolaryngology, Head and Neck Surgery School of Medical Sciences State University of Campinas - UNICAMP Campinas - SP Brazil Phone: (55-19)3788-7903 enicola@fcm.unicamp.br
ABSTRACT
Purpose: To show that a non-laser light source can be constructed, using a 500 W Tungsten lamp and optical filters, and that this light source produces photodynamic effect via protoporphyrin IX (PpIX) similar to the effect produced by HeNe laser light.
Methods: The broad band spectrum from a Tungsten lamp was filtered. Infrared and blue part of the spectrum was discarded by absorption process and the fraction of the spectrum, centered at the red portion, was filtered by an interference filter. Photodynamic effect was studied by the activity on endogenous PpIX of Harderian glands of Wistar rats. Twenty rats were used for the experiment. Each animal had its two Harderian gland surgically exposed, so one of them was treated with the system while the other was kept as control. After a 30 minutes period of treatment, the animals were sacrificed and their glands were removed for histological analysis. This analysis was compared to earlier published results obtained with HeNe laser light.
Results: The resultant light source emission was centered around (636 ± 6.5) nm and gives up to 11.3 mW/cm2 power density. It produces photodynamic effect in Harderian gland, observed either by fluorescence spectroscopy or by histological microscopy.
Conclusion: There is no noticeable difference in Photodynamic effect results if activated by HeNe laser or by the proposed non-laser light source emitting at the red portion of the spectrum.
Key words: Photodynamic Therapy. Lasers. Harderian gland.
RESUMO
Objetivo: Descrever a construção de uma fonte de luz não-laser, a partir de uma lâmpada de Tungstênio e filtros óticos adequados e demonstrar que sua eficiência em estudos fotodinâmicos, mediados por protoporfirina IX, é semelhante a do laser de Hélio Neônio.
Métodos: As regiões Infravermelha e Azul do espectro de emissão óptica de uma lâmpada de Tungstênio foram convenientemente descartadas por processos de absorção, enquanto que a fração centrada na região do vermelho foi removida com o uso de um filtro de interferência. O efeito fotodinâmico foi estudado, em glândula Harderiana de ratos Wistar em razão da produção endógena de protoporfirina IX (PpIX) por estas glândulas. Foram utilizados 20 ratos. Cada animal teve as duas glândulas expostas cirurgicamente, sendo uma delas tratada com a fonte não-laser e a outra mantida como controle. Após tratamento por 30 minutos os animais foram sacrificados e suas glândulas removidas para estudo histológico. Os resultados foram comparados a estudos realizados com laser de Hélio Neônio, já publicados.
Resultados: A luz produzida pelo equipamento está centrada em torno de (636 ± 6,5) nm, fornecendo uma densidade de potência de 11,3 mW/cm2. Os efeitos fotodinâmicos produzidos na glândula Harderiana, podem ser observados tanto por espectroscopia de fluorescência como por microscopia ótica.
Conclusão: Não foram observadas diferenças significativas nos resultados do efeito fotodinâmico obtidos com a fonte de luz não-laser proposta, em comparação aos resultados conhecidos com o uso do laser de Hélio Neônio.
Descritores: Fotoquimioterapia. Lasers. Glândula de Harder.
Introduction
First information about photodynamic applied to cancer therapy dates from the seventies. Since then, many patients with certain malignant tumors have been treated with Photodynamic Therapy (PDT), presenting results that justify its efficiency1,2,3.
Photodynamic process is obtained by the light action on a photoactive substance, which in the presence of oxygen can causes the destruction of the hostess cell. This technique, characterized by a sequence of physical, chemical and biological effects, has increased its use in cancer treatment.
Molecular oxygen can exist in two possible states: singlet (1O2) and triplet (3O2). The latter is the fundamental and stable state of the molecule, and is mostly found in any cell. Chemically, singlet oxygen is more reactive than triplet oxygen, forming new compounds and releasing energy. Transition from the fundamental state of the oxygen (triplet) to singlet state, results in a destruction of the hostess cell by oxidation of its constituents. This transition (TripletSinglet) corresponds to the absorption of an energy equivalent to 1.27mm, and is not allowed to occur by direct absorption, according to conservation laws of physics (spin conservation). This transition is only possible by non-radiative energy absorption (Fermi resonance). Some substances, known as photosensitizer, like Hematoporphyrin derivative (HpD) and Protoporphyrin IX (PpIX), can absorb photons (around 632 nm) directly and, after a sequence of intersystem energy conversion can promote neighbors triplet oxygen to the excited singlet state3,4,5.
In the specific case of PDT, the photosensitizer is injected in the patient and is kept inside cancer cells for a time longer than in normal cells. So, the photon absorption and the consequent energy transfer to oxygen will occur only inside cancer cells, as explained above.
In the Harderian gland6,7,8 model, for photodynamic studies, the presence of endogenous protoporphyrin IX eliminates the need of photosensitizer injection9.
For activating the photosensitizer in photodynamic process, it is necessary to use light excitation in the resonant frequency with the optical absorption level of the substance. Generally, lasers are the most useful equipment in this process, thank to their very high collimation and power density, joined with their excellent monochromaticity. Fast laser development and their prices decreasing discourage the use of lamps for PDT. A few works use non laser light sources for activating photodynamic process10,11 remaining unknown, until now, some particularities concerning possible difference in the final effect when the excitation is laser or non laser light. This lack in experimental data encourages the development of non laser light sources for PDT studies.
Methods
With a commercial 500 W tungsten lamp, optical filters and lenses, it has been projected and developed a non-laser light source as sketched in Figure 1 and photographically showed in Figure 2.
The emission spectrum of the tungsten lamp is shown in Figure 3.
This spectrum shows that the light source of the system emits photons from the visible to the near infrared region, in a continuous way.
A fraction of light rays emitted from the lamp is absorbed by the walls or transmitted outside the equipment, causing some losses and converting energy into heat. It was necessary to use micro ventilators in order to cool the developed system. A fraction of the remaining rays go through the sequence of filters and lenses. For this system, it has been used an infrared absorption filter and an interference filter, band-pass centered at 630 nm. We have used two lenses for converging the light beam and another absorption filter in blue and near ultraviolet region (high frequency). The output light beam enters into another lens with variable focus in order to collimate it. All system components were fixed with movable supports in order to permit an optimization of light concentration at the end.
To analyze photodynamic effect produced by the non-laser system we used the Harderian gland of Wistar rats model9. A group of 20 Specific Pathogen Free (SPF) Wistar rats, all males, 7 weeks old and body mass from 150 to 200 g, has been submitted to an anesthetic procedure according to ethical international protocols, with intravenous sodium hypinol. Each animal had its two Harderian glands surgically exposed by means of microsurgery. One of the glands was kept as control while the other was treated with the system developed in the present work for 30 minute.
During the treatment period, fluorescence spectrum of both glands (control and treated one) was recorded with a plug-in spectrometer (OceanOptica PC2000). The activation of this fluorescence was done with ultraviolet light (Hg lamp, 20 W, 365 nm).
Immediately after treatment both glands have been removed for histological analysis by optical microscopy. The animals were sacrificed according to established bioethics protocols.
Results
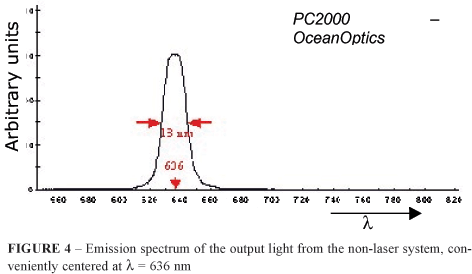
The resultant emission spectrum of the constructed non laser system, recorded with the PC2000 spectrometer, can be seen in Figure 4, and it displays the characteristic gaussian shape of interference filters, centered at 636 nm with a measured width at half height of 13 nm (636 ± 6.5) nm.
At a convenient distance of 27 cm from the last lens of the equipment, almost all output energy is concentrated in an area of 0.93 cm2. At this position, with aid of a power meter, it was possible to establish the output power of the system, which was 10.5 mW, giving a power density of 11.3 mW/cm2. Since the system is a non laser light source, the beam can not be focused to small areas so, this is the highest power density achieved by the system. By positioning the gland at this focal plane the received energy density (fluence) during a convenient time of 30 minutes was 20.34 J/cm2.
Figure 5 show histological aspect of a typical healthy Harderian gland (control gland).
The analysis characterizes a typical acinar pattern arrangement of the tissue, round nuclei, with clearly defined limits. It is possible to identify the tubules of a normal rat Harderian gland, composed of a single layer of columnar epithelial cells
Figure 6 shows the fluorescence spectrum of a control gland, displaying the two characteristic optical band of Pp IX, centered at 636 nm and 708 nm, respectively12,13.
After a 30 minutes period of treatment with the non laser light source, and immediately after exposure to light, necrosis of the tubular cells characterized by fragments of the epithelial cells can be observed (Figure 7) in the treated gland.
Fluorescence measurements also show modifications after non laser light exposure in treated Harderian gland (Figure 8).
The origin of the new fluorescence at 675 nm band is known to be due to formation of fluorescent photoproducts14, and indicates Photodynamic activity.
The contra lateral operated Harderian gland, kept as control for 30 minutes, showed neither necrosis nor inflammatory infiltrate. These standard behaviors of both glands have been observed for all animals of the sample. The fluorescence spectrum of this control gland stays unchangeable during the experiment.
Discussion
One important property of laser beam is its high collimation. For instance, a HeNe laser beam can be focused in areas smaller than 0.1 mm2, leading to high power densities, even for small lasers. Comparing the final power density of our non laser light source with, for example, the achieved power density of a HeNe laser of nominal power of 7 mW our system is in great disadvantage but, for PDT application in larger areas, the non-laser system can be preferable. For instance, the power density of the HeNe laser, if the beam is expanded to cover an area of 0.93 cm2, is 7.53 mW/cm2, which is only 67 % of the power density achieved by our system. As a consequence, the required time to irradiate tumor areas of 0.93 cm2, to achieve a fixed fluence with the non laser system, will be 23% smaller than the time required by the mentioned HeNe laser.
Other important difference between both systems is the difference in the emitted wavelengths (l ). The HeNe emits a narrow band of light centered at l = 632.8 nm while the non laser system emits a relatively wide band centered at l = 638 nm. This affects positively the efficiency of absorption of the light by Pp IX which has a maximum absorption near 638 nm.
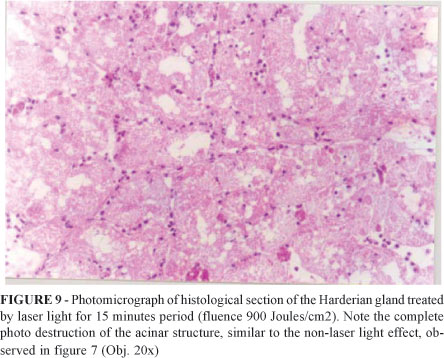
The results obtained by Reis et al9, using HeNe (l =632.8 nm) laser light in similar experiment with Harderian gland of Wistar rats were achieved with fluence about 900 J/mm2, which is 44.24 times grater than the fluence used in the present experiment. Figure 9 is a photomicrograph of histological section of a treated Harderian gland, after 15 minutes period of HeNe light irradiation.
Figure 10 is the correspondent spectrum of fluorescence (showing here by permission of the authors of reference 9).
A simple comparison of these results with the ones of the present work, as depict in Figures 7 and 8, evinces our prior statement that both light sources bring similar photodynamic results in Harderian gland of Wistar rats, assisted by endogenous PpIX.
The new equipment developed has an effective power density for larger areas (covering surfaces of about 1 cm2), on the contrary of a 7 mW HeNe laser. This is an excellent advance, principally for photodynamic therapy applications in superficial lesions, like skin cancer. Additionally, the emission frequency (correspondent to l = 636 nm) of the non-laser system is almost resonant with the absorption frequency of PpIX, providing better absorption and, consequently, observable photodynamic effect even at low fluence.
Conclusion
The construction of a suitable non-laser light source for photodynamic application, where low fluence is required, is relatively simple. Despite the fact that the light beam from the non-laser source can not be focused to the same limits of laser beam, it can be used in situations where large areas of superficial lesions must be covered.
The use of the non-laser light source in a simple experiment with Harderian gland of Wistar rats, which results achieved with HeNe light is previously know, was fundamental to validated the developed device.
Received: August 15, 2004
Review: September 21, 2004
Accepted: October 25, 2004
Conflict of interest: none
Financial source: FAPESP and FAEP UNICAMP
How to cite this article:
Mesquita RC, Reis ER, Nicola JH, Nicola EMD. Construction and evaluation of a non-laser optical system for photodynamic process excitation. Acta Cir Bras [serial online] 2004 Nov-Dec;19(6). Available from URL: http://www.scielo.br/acb [also in CD-ROM].
*Color figures available from www.scielo.br/acb
- 1. Schuitmaker JJ. Photodynamic therapy: a promising new modality for the treatment for cancer. J Photochem Photobiol B Biol. 1996;34:3-12.
- 2. Dougherty TJ. Photodynamic therapy: yearly review. Photochem Photobiol. 1993;58(6):895-900.
- 3. Henderson BW, Dougherty TJ. How does photodynamic therapy work? Photochem Photobiol. 1992;55:145-57.
- 4. Nicola JH, Colussi VC, Nicola EMD, Metze K. Enhancement of Photodynamic Therapy due to Hyperbaric Hyperoxia: an experimental study of Walker 256 tumors in rats. Optical Methods for Tumor Treatment and Detection Mechanisms and Techniques in Photodynamic Therapy VI, Thomas J Dougherty Editor:1997; Vol. 2972: p. 88-94.
- 5. Colussi VC, Nicola ED, Nicola JH. Fototerapia, Fotoquimioterapia e alguns fotossensibilizadores. Rev Assoc Med Bras. 1996;42(4):229-36.
- 6. Payne AP. The harderian gland: a tercentennial review. J Anat. 1994;185: 1-49.
- 7. Chieffi G, Chieffi BG, Di Matteo L, Distria M, Minucci S, Varriale B. Cell biology of the harderian gland. Int Rev Cytol. 1996;168:1-80.
- 8. Chieffi B. Organogenisis of the Harderian gland: a comparative survey. Microsc Res Tech. 1996;34:6-15.
- 9. Reis ER, Nicola EMD, Metze K, Nicola JH. Photodynamic effect produced by HeNe radiation in Harderian glands of Wistar rats: an experimental model for PDT studies. In: Duncan DD, Hollinger JO, Jacques SL (editors). Laser-tissue interaction XI: photochemical, photothermal, and photomechanical. 2000; p. 40-5.
- 10. McCaughan JS, Guy JT, Hawley P, Hicks W, Inglis W, Laufman L, May E, Nims TA, Sherman R. Hematoporphyrin-Derivative and photoradiation therapy of malignant tumors. Lasers Surg Med. 1983;3:199-209.
- 11. Beyer W. Systems for light application and dosimetry in photodynamic therapy. J Photochem Photobiol B. 1996;36: 153-6.
- 12. König K, Schneckenburger H, Rück A. In vivo photoproduct formation during PDT with ALA-induced endogenous porphyrins. J Photochem Photobiol B. 1993;18:287-90.
- 13. Klintberg C, Enejder AMK, Wang I, Anderson-Engels S, Svanberg S, Svanberg K. Kinetc fluorescence studies of 5-aminolevulinic acid-induced protoporphyrin IX accumulation in basal cell carcinomas. J Photochem Photobiol B. 1999;49:120-8.
- 14. Gudgin Dickson EF, Pottier RH. On the role of protoporphyrin IX photoproducts in photodynamic therapy. J Photochem Photobiol B. 1995;29:91-3.
Publication Dates
-
Publication in this collection
08 Mar 2005 -
Date of issue
Dec 2004
History
-
Received
15 Aug 2004 -
Reviewed
21 Sept 2004 -
Accepted
25 Oct 2004