Abstracts
PURPOSE: Laparoscopic surgery is associated with reduced surgical trauma, and less acute phase response, as compared with open surgery. Cytokines are important regulators of the biological response to surgical and anesthetic stress. The aim of this study was to determine if CO2 pneumoperitoneum would change cytokine expression, gas parameters and leukocyte count in septic rats. METHODS: Wistar rats were randomly assigned to five groups: control (anesthesia only), laparotomy, CO2 pneumoperitoneum, cecum ligation and puncture by laparotomy, and laparoscopic cecum ligation and puncture. After 30 min of the procedures, arterial blood samples were obtained to determine leukocytes subpopulations by hemocytometer. TNFalpha, IL-1beta, IL-6 were determined in intraperitoneal fluid (by ELISA). Gas parameters were measured on arterial blood, intraperitoneal and subperitoneal exsudates. RESULTS: Peritoneal TNFalpha, IL-1beta and IL-6 concentrations were lower in pneumoperitoneum rats than in all other groups (p<0.05). TNFalpha, IL-1beta and IL-6 expression was lower in the laparoscopic than in laparotomic sepsis (p<0.05). Rats from laparoscopic cecum ligation and puncture group developed significant hypercarbic acidosis in blood and subperitoneal fluid when compared to open procedure group. Total white blood cells and lymphocytes were significantly lower in laparoscopic cecum ligation and puncture rats than in the laparotomic (p<0.01). Nevertheless, the laparotomic cecum ligation rats had a significant increase in blood neutrophils and eosinophils when compared with controls (p<0.05). CONCLUSIONS: This study demonstrates that the CO2 pneumoperitoneum reduced the inflammatory response in an animal model of peritonitis with respect to intraperitoneal cytokines, white blood cell count and clinical correlates of sepsis. The pneumoperitoneum produced hypercarbic acidosis in septic animals.
Pneumoperitoneum; Carbon Dioxide; Sepsis; Acidosis; Leucocytes
OBJETIVO: A cirurgia laparoscópica está associada com trauma reduzido e baixa resposta na fase aguda do trauma, quando comparada com a cirurgia aberta. As citocinas e o balanço ácido-base são fatores importantes da resposta biológica ao trauma cirúrgico-anestésico. O objetivo deste estudo foi determinar se o pneumoperitôneo com CO2 altera a expressão das citocinas, a gasometria e a contagem diferencial de leucócitos em ratos com sepse abdominal. MÉTODOS: Ratos Wistar foram aleatoriamente distribuídos em 5 grupos: controle (somente anestesia), laparotomia, pneumoperitôneo com CO2, ligadura e punção do ceco por laparotomia, ligadura e punção do ceco por laparoscopia. Após 30 minutos dos procedimentos, sangue arterial foi colhido para leucometria diferencial em hemocitômetro. TNFalfa, IL-1beta e IL-6 foram dosadas no líquido intraperitoneal (por ELISA). Os parâmetros gasosos foram medidos no sangue arterial e nos exsudatos intraperitoneal e subperitoneal. RESULTADOS: Os valores de TNFalfa, IL-1beta e IL-6 foram significantemente menores nos ratos submetidos ao pneumoperitôneo do que em todos os outros grupos (p<0.05). Expressão de TNFalfa, IL-1beta e IL-6 foi menor no grupo sepse induzida por laparoscopia do que por laparotomia (p<0.05). Os ratos submetidos a ligadura e punção do ceco via laparoscópica desenvolveram acidose hipercárbica no sangue arterial e exsudato subperitoneal, mais intensa do que no grupo sepse laparotômica. Leucopenia e linfopenia foram mais acentuadas no grupo sepse laparoscópica (p<0.01). Entretanto, os animais submetidos a sepse laparotômica desenvolveram significante aumento de neutrófilos e eosinófilos quando comparados com os controles (p<0.05). CONCLUSÕES: Este estudo demonstrou que o pneumoperitôneo com CO2 contribuiu para reduzir a resposta inflamatória em ratos submetidos a modelo de sepse abdominal, no que diz respeito à expressão de citocinas intraperitoneais e leucometria diferencial. O pneumoperitôneo também contribuiu para instalação de acidose hipercárbica nos ratos sépticos.
Pneumoperitônio; Dióxido de Carbono; Sepse; Acidose; Leucócitos
ORIGINAL ARTICLE
Influence of laparoscopy and laparotomy on gasometry, leukocytes and cytokines in a rat abdominal sepsis model1 1 . Article from Nucleus of Experimental Surgery, Postgraduate Program in Health Sciences, Federal University of Rio Grande do Norte (UFRN), Brazil.
Influência da laparoscopia e laparotomia na gasometria, leucócitos e citocinas em modelo de sepse abdominal em ratos
Irami Araújo FilhoI; Abraão Allen Honorato SobrinhoII; Amália Cinthia Meneses do RegoII; Ana Claudia M. de Amorim GarciaII; Daniele Pimentel FernandesII; Thaís Medeiros CruzII; Ticiana Cabral da CostaII; Aldo Cunha MedeirosIII
IPostgraduate Fellow, Postgraduate Program in Health Sciences, UFRN, Brazil
IIMedical Student, Scientific Initiation Program, UFRN, Brazil
IIIPhD, Chief, Nucleus of Experimental Surgery, UFRN, Brazil
Correspondência Correspondência: Aldo da Cunha Medeiros Av. Miguel Alcides Araújo, 1889 59078-270 Natal-RN Brazil. aldo@ufrnet.br
ABSTRACT
PURPOSE: Laparoscopic surgery is associated with reduced surgical trauma, and less acute phase response, as compared with open surgery. Cytokines are important regulators of the biological response to surgical and anesthetic stress. The aim of this study was to determine if CO2 pneumoperitoneum would change cytokine expression, gas parameters and leukocyte count in septic rats.
METHODS: Wistar rats were randomly assigned to five groups: control (anesthesia only), laparotomy, CO2 pneumoperitoneum, cecum ligation and puncture by laparotomy, and laparoscopic cecum ligation and puncture. After 30 min of the procedures, arterial blood samples were obtained to determine leukocytes subpopulations by hemocytometer. TNFa, IL-1b, IL-6 were determined in intraperitoneal fluid (by ELISA). Gas parameters were measured on arterial blood, intraperitoneal and subperitoneal exsudates.
RESULTS: Peritoneal TNFa, IL-1b and IL-6 concentrations were lower in pneumoperitoneum rats than in all other groups (p<0.05). TNFa, IL-1b and IL-6 expression was lower in the laparoscopic than in laparotomic sepsis (p<0.05). Rats from laparoscopic cecum ligation and puncture group developed significant hypercarbic acidosis in blood and subperitoneal fluid when compared to open procedure group. Total white blood cells and lymphocytes were significantly lower in laparoscopic cecum ligation and puncture rats than in the laparotomic (p<0.01). Nevertheless, the laparotomic cecum ligation rats had a significant increase in blood neutrophils and eosinophils when compared with controls (p<0.05).
CONCLUSIONS: This study demonstrates that the CO2 pneumoperitoneum reduced the inflammatory response in an animal model of peritonitis with respect to intraperitoneal cytokines, white blood cell count and clinical correlates of sepsis. The pneumoperitoneum produced hypercarbic acidosis in septic animals.
Key words: Pneumoperitoneum. Carbon Dioxide. Sepsis. Acidosis. Leucocytes.
RESUMO
OBJETIVO: A cirurgia laparoscópica está associada com trauma reduzido e baixa resposta na fase aguda do trauma, quando comparada com a cirurgia aberta. As citocinas e o balanço ácido-base são fatores importantes da resposta biológica ao trauma cirúrgico-anestésico. O objetivo deste estudo foi determinar se o pneumoperitôneo com CO2 altera a expressão das citocinas, a gasometria e a contagem diferencial de leucócitos em ratos com sepse abdominal.
MÉTODOS: Ratos Wistar foram aleatoriamente distribuídos em 5 grupos: controle (somente anestesia), laparotomia, pneumoperitôneo com CO2, ligadura e punção do ceco por laparotomia, ligadura e punção do ceco por laparoscopia. Após 30 minutos dos procedimentos, sangue arterial foi colhido para leucometria diferencial em hemocitômetro. TNFa, IL-1b e IL-6 foram dosadas no líquido intraperitoneal (por ELISA). Os parâmetros gasosos foram medidos no sangue arterial e nos exsudatos intraperitoneal e subperitoneal.
RESULTADOS: Os valores de TNFa, IL-1b e IL-6 foram significantemente menores nos ratos submetidos ao pneumoperitôneo do que em todos os outros grupos (p<0.05). Expressão de TNFa, IL-1b e IL-6 foi menor no grupo sepse induzida por laparoscopia do que por laparotomia (p<0.05). Os ratos submetidos a ligadura e punção do ceco via laparoscópica desenvolveram acidose hipercárbica no sangue arterial e exsudato subperitoneal, mais intensa do que no grupo sepse laparotômica. Leucopenia e linfopenia foram mais acentuadas no grupo sepse laparoscópica (p<0.01). Entretanto, os animais submetidos a sepse laparotômica desenvolveram significante aumento de neutrófilos e eosinófilos quando comparados com os controles (p<0.05).
CONCLUSÕES: Este estudo demonstrou que o pneumoperitôneo com CO2 contribuiu para reduzir a resposta inflamatória em ratos submetidos a modelo de sepse abdominal, no que diz respeito à expressão de citocinas intraperitoneais e leucometria diferencial. O pneumoperitôneo também contribuiu para instalação de acidose hipercárbica nos ratos sépticos.
Descritores: Pneumoperitônio. Dióxido de Carbono. Sepse. Acidose. Leucócitos.
Introduction
Operative laparoscopy brought a new dimension to surgical practice, and many experimental and clinical studies have demonstrated feasibility, safety, cost-benefit, and pathophysiologic occurrences. The intraabdominal insufflation of carbon dioxide (CO2) is the most widely used technique for the creation of a pneumoperitoneum. Insufflation under a continuous monitoring of intraabdominal pressure throughout the surgical procedure provides adequate exposure of the operating field. As alternatives, different gases (e.g., helium, argon, nitrous oxide, air) may be used, but they have not been adopted clinically1,2,3,4,5,6. The actual knowledge concerning pneumoperitoneum are sometimes inconsistent about the physiologic consequences induced by insufflation of the peritoneal cavity with CO2. Results from animal models about the effects of a pneumoperitoneum in certain pathologic conditions are often alarming7. However, the clinical impact of these changes is unknown, since the majority of patients who undergo laparoscopic procedures do not exhibit any adverse clinical effects either in the short or the long-term course. Laparoscopíc surgery is applied increasingly to abdominal diseases complicated by diffuse or localized peritonitis such as appendicitis, perforated peptic ulcers and diverticulitis8,9. A specific paper has reported the use of laparoscopy in diverticulitis complicated by localized peritonitis with intra-abdominal abscess formation10, and diagnostic laparoscopy is being advocated in diffuse peritonitis after blunt abdominal trauma11. However, a theoretical concern with the use of laparoscopic techniques in clinical cases complicated by intra-abdominal infection and peritonitis, is that carbon dioxide pneumoperitoneum may increase the risk of bacteraemia and sepsis by increasing intra-abdominal pressure. Some studies have demonstrated immunossuppressive effects of carbon dioxide on neutrophil and macrophage function. In one study, CO2 blocked superoxide release from activated polimorphonuclear leukocytes and significantly reduced the secretion of IL-1 from human peritoneal macrophages12. Whereas these effects might be considered beneficial from the standpoint of inflammation following elective surgery, experimental evidence suggests that the CO2 induced immunossuppression might be deleterious in the setting of infection13. This may have an adverse effect on clinical outcome when compared with open procedures. Although some evidences, few data exist regarding the effect of pneumoperitoneum and increased intra-abdominal pressure on sepsis and physiological outcome. The aims of the current study were to investigate the influence of laparoscopic procedures, in particular CO2 insufflation, on the response to sepsis in an animal modelcecal ligation and puncture (CLP) in the rat. Clinical evolution, gasometry, pro-inflammatory cytokines and leukocytes were analyzed.
Methods
Male Wistar rats (Animal Colony from Nucleus of Experimental Surgery, Federal University of Rio Grande do Norte, Brazil), 12 to 13 weeks old, were housed in cages where standard chow and water were available ad libitum. The rats were acclimatized to the laboratory environment for 5 days on arrival and then fasted for 12 hours before any procedures. Anesthesia was obtained using pentobarbital 20 mg/Kg intraperitoneal and ketamine 50 mg/Kg intramuscular. All surgical procedures were performed under aseptic conditions. The animals were allowed to breathe spontaneously for the duration of the experiment. The group C (control) rats were subjected to anesthesia only (n=6). In the LAP (laparotomy) group (n=7) the following procedures were performed: after anesthesia and antisepsis with povidone, a 5cm laparotomy kept the peritoneal cavity exposed to the room air during 30 minutes and the abdominal wall was sutured with nylon 4-0. The PNP rats (n=7) were subjected to CO2 pneumoperitoneum using a Veress needle under 3 mmHg for 30 minutes. On the CLP/LAP (n=6) the CLP was performed after laparotomy. A cecum ligation and puncture (CLP) by laparoscopy under pneumoperitoneum were performed on the CLP/PNP rats (n=6).
Cecal ligation and puncture - Pneumoperitoneum was achieved by introducing a Veress needle into the peritoneal cavity and insufflating (Endomed insufflator) the abdomen with 3 mmHg CO2. Laparoscopic procedures were performed using 3-mm instruments (Henke-Sass, WolfTM) introduced into the abdomen. Cecal ligation and puncture (CLP) consisted of dissection of the cecum, ligation midway between the ileocecal valve and the terminal cecum using a 3-0 chromic catgut tie, and 8-punctures of the isolated cecum with a hollow 25-gauge needle introduced through the abdominal wall. Laparotomy, for the open CLP group, consisted of a 5-cm midline abdominal incision. The duration of the total procedure, and therefore the duration of anesthesia, pneumoperitoneum, and laparotomy, was standardized to 30 minutes for all groups. Postoperatively, animals were resuscitated with a subcutaneous injection of lactated Ringer's (30 mL/kg) and were again housed in cages where water was available ad libitum. The experimental protocol was approved by the Research Ethics Committee of the Federal University of Rio Grande do Norte, Brazil, and adhered to the Guide for the Care and Use of Laboratory Animals, US National Research Council, 1996.
Gasometry and cytokines dosage - After the surgical procedures, 5mL of buffered saline were injected in peritoneal cavity and the abdomen was softly massaged for 1 minute. Thirty minutes later, whole blood was collected by cardiac puncture and liquid exsudate was collected from peritoneal cavity and from subperitoneal space, using heparinized capilar tube, for determination of pH, pCO2 and pO2. An automatic AVL (Roche®) equipment was used. TNFa, IL-1b and IL-6 were determined in the intraperitoneal exsudate, by enzyme-linked immunosorbent assay, using cytokine-kits from PeproTec (Rocky Hill, NJ,USA).
Leukometry - Animals were evaluated 24 hours postoperatively for clinical signs of CLP-induced sepsis (i.e., dark halo around the eyes, piloerection and lethargy). Whole blood was collected by cardiac puncture for leukocyte cell counts and the rats were then killed via anesthetic overdose. The determination of leukocyte cell counts was preformed using a commercially available automated cell counter (Abbott Cell-Dyn 3500R- CD 3500 5L, USA).Data were expressed as mean±standard deviation. Statistical significance was established using the one way analysis of variance ANOVA followed by Newman-Keuls test. Probabilities less than 0.05 were considered significant.
Results
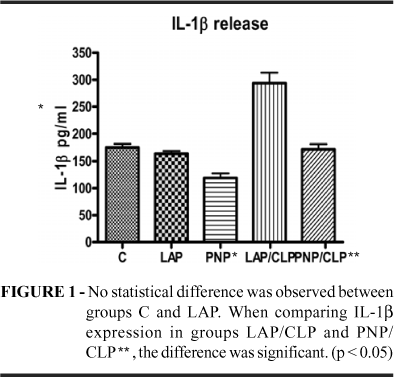
Cytokines release in peritoneal exsudate - Rats in the control, LAP and PNP groups exhibited normal activity and had no piloerection during the 24 hours after interventions. In contrast, all rats that underwent CLP exhibited decreased activity and significant piloerection. The cytokine levels on LAP rats were higher than PNP ones; however, the difference between these groups was not significant (p<0.05). Both surgical procedures, PNP/CLP and LAP/CLP, induced higher TNFa, IL-1b and IL-6 in the peritoneal fluid than were found in the control group. In contrast, the peritoneal fluid TNFa, IL-1b and IL-6 levels in the pneumoperitoneum (PNP) group were significantly lower than in the other groups (p<0.05). The PNP/CLP rats had a significantly lower elevation of TNFa, IL-1b and IL-6 expression in the peritoneal fluid than LAP/CLP rats (p<0.05) (Figures 1, 2 and 3).
Gasometry - Arterial blood gas parameters (pH, pO2) in rats from the control group (C) remained significantly higher than in rats from LAP, PNP and PNP/CLP groups (p<0.01). Rats from PNP/CLP group developed significant hypercarbic acidosis with mean pH of 7.18±0.05 and pCO2 60.7±10.2 when compared to LAP/CLP group. The LAP/CLP rats acidosis was not hypercarbic (Table 1). The pCO2 was significantly higher on PNP/CLP rats than controls (p<0.01). Significantly reduced pCO2 was observed following LAP/CLP, compared to LAP, PNP and PNP/CLP (p<0.01). Intraperitoneal exsudate gas analysis revealed acidosis in the PNP group, and the difference was significant when compared with C and LAP groups (p<0.01). However, the septic rats subjected to CO2 pneumoperitoneum (PNP/CLP) developed a profound intraperitoneal acidosis, as a consequence of pCO2 significantly higher (p<0.01) than in all other groups (Table 3). In contrast, the pO2 was significantly lower than in C and LAP/CLP groups (p<0.05). While subperitoneal exsudate pH (6.5±0.05) following laparoscopic CLP using CO2 (PNP/CLP) was significantly lower (p<0.01) than in C, LAP and LAP/CLP rats, the difference did not reach statistical significance when compared with the acidotic pH (6.7±0.08) of PNP group (Table 3). The CO2 pneumoperitoneum produced a significant increase in pCO2 in the subperitoneal exsudate, when compared with all the other groups (p<0.01).
Leukocytes - White blood cell count in pneumoperitoneum and laparoscopic CLP was similar to those of controls (Table 4). Significantly reduced white cell counts were observed following laparoscopic CLP compared to open CLP (p<0.01). No significant difference in blood neutrophil and lymphocyte count was found among control, LAP, PNP and PNP/CLP rats. Nevertheless, the LAP/CLP rats had a significant increase in blood neutrophils when compared with controls (p<0.05), as can be observed in Table 4.
The PNP/CLP and LAP/CLP procedures produced a significant reduction in lymphocyte count, compared with controls (p<0.05). Additionally, the reduction in lymphocytes was significantly higher in open CLP (LAP/CLP) rats than in PNP/CLP (p<0.05). There was a significant reduction in eosinophil count in PNP, LAP/CLP and PNP/CLP, compared with controls (p<0.05). The decrease in LAP/CLP eosinophil was greater than in PNP/CLP rats.
Discussion
Because laparoscopic surgery is increasingly used for treating peritonitis and other septic states, a theoretic concern is related to the hypothesis that CO2 pneumoperitoneum may increase bacteremia with adverse effects for the patient. Some studies have reported technical feasibility of laparoscopic appendectomy, perforated peptic ulcer, perforated diverticulitis and other septic surgical situations14,15,16. They are small studies with low numbers of included patients, that can not give strong evidence about improved safety of laparoscopic surgery regarding septicemia. Study in rats showed that pneumoperitoneum causes intestinal ischemia with oxygen free radical production and bacterial translocation, related to mechanical pressure of CO217. Other studies focused on whether a pneumoperitoneum amplifies the extent and severity of peritonitis or of bacteremia in various animal models18,19,20,21. Findings from these investigations are controversial. Whereas some authors reported no increase in bacteremia, intraperitoneal abscess formation, or correlates of sepsis, others reported increased bacterial translocation and severity of peritonitis and sepsis. Laparoscopic surgical technique requires the maintenance of a continuous positive intraperitoneal pressure in patients (approximately 10-15 mmHg) for visualization and manipulation of the viscera. In cases of peritonitis, viable bacteria and bacterial byproducts (including endotoxin) exist free in the peritoneal cavity. Positive intraperitoneal pressure may increase bacteraemia and endotoxaemia, and thus may worsen clinical sepsis. As experience with laparoscopic surgery increases, its use in more debilitated and critically ill patients is being reported22. These patients often have sepsis and suffer from diffuse peritonitis of unclear aetiology, usually the result of a perforated viscus. It has not been clear whether a laparoscopic approach worsens the septic state or whether minimally invasive surgery is beneficial in these critically ill patients. Experimental model - Several animal models of peritoneal sepsis have been described varying in many respects, including the specific animal utilized and the method of sepsis20,23,24,25. The model of creating sepsis used in this study was adapted from the work of Hanly et al26. The convenience of the model of laparoscopic CLP in rats, used in the present experiment, is two-fold. First, the use of a septic animal model magnifies the stress induced by a surgical procedure to more clearly delineate the modifying effects of laparoscopy on the inflammatory response. Second, the combined stressors of bacterial contamination of the peritoneal cavity and bowel ischemia present following laparoscopic CLP provide an environment analogous to clinical situations in which laparoscopy is used to aid in the diagnosis and treatment of patients with peritonitis. The model used in this study presupposes that CLP caused sepsis and that the injury caused by CLP was equivalent between groups. We established the presence or absence of sepsis in rats by evaluating each rat for the presence or absence of periorbital dark halo, the presence or absence of piloerection and normal or decreased activity. All 12 rats that were subjected to CLP were identified as having clinical sepsis, and all 20 rats considered control, or that had received other procedures, were identified as not having sepsis.
Cytokines - In the present study we observed a significant decrease of TNFá, IL-1â and IL-6 in rats subjected to CO2 pneumoperitoneum with and without sepsis, and these findings coincided with acidosis in arterial blood, in intraperitoneal and subperitoneal exsudates. These data are in agreement with other investigators, who showed inhibition of human peritoneal macrophage cytokine production when these cells were incubated in an acidic extracellular environment, lowered the intracellular pH and attenuated cytokine release27. Similarly, Carozzi et al.28 showed decreased spontaneous release of IL-1, IL-6, IL-8, and TNF when incubations were performed in pH 5.5 medium compared to much higher cytokine levels from cells incubated in medium with a pH of 7.4. In the present study the PNP and PNP/CLP rats exhibited arterial, intraperitoneal and subperitoneal acidosis, suggesting intracellular acidosis. West et al.32 have proposed relative intracellular acidosis as the mechanism by which the decrease in cytokines is exerted29. Redmond et al.30 showed that circulating monocytes obtained from patients after laparoscopic cholecystectomy exhibited reduced TNF release compared to those from patients who had open cholecystectomy. They also reported that peritoneal macrophages derived from animals undergoing CO2 laparoscopy released less TNF in response to lipopolyssacharides (LPS) than those undergoing air laparoscopy31. West et al.32 have shown that murine peritoneal macrophages exposed to CO2 in vitro exhibit inhibition of LPS-stimulated IL-1 and TNF cytokine release, suggesting that this effect is related to the influence of the CO2 environment. These findings contribute to explain the reduction of cytokine release when animals and patients are operated under effect of CO2 pneumoperitoneum. Whereas these effects might be considered beneficial from the standpoint of inflammation following elective surgery, one experimental study suggests that CO2 induced immunossuppression might be deleterious in the setting of infection13. The clinical significance of these findings remains unknown. In studies where laparoscopy was compared to open surgery for peritoneal infection such as appendicitis, there was no clear augmentation of infectious complications associated with the use of CO2 pneumoperitoneum33.
Gas analysis - The effect of pneumoperitoneum on hemodynamics and blood gas variables has been studied extensively in nonseptic animals34, and to a lesser degree in acute models of sepsis. In the present study, pneumoperitoneum alone and associated with CLP sepsis induced alterations of the acid-base balance, such as fall of pH, and elevation of pCO2 , in arterial blood, intraperitoneal and subperitoneal exsudate, without correlation to pO2. The decrease in pH, that was more accentuated with CLP, was similar to what has been reported25. These findings were substantiated by other investigators, who found that intraabdominal pH diminishes with application of a CO2 pneumoperitoneum. CO2 used as an insufflation gas appeared to lower peritoneal, blood, and subcutaneous pH more than helium, which induced smaller changes35. Gandara et al36 stated that, in addition to CO2 absorption, this might be a phenomenon of tissue hypoperfusion. Nevertheless, in our study pO2 was not affected by pneumoperitoneum in septic and non septic rats. This finding can be explained by the fact that pneumoperitoneum was performed with 3 mmHg pressure, sufficient to keep a normal and spontaneous respiration in rats. This pressure was used in rats by other investigators37. By the way, Kuntz et al35. showed that, after insufflation with CO2, intraperitoneal pH was inversely related to the intraabdominal pressure.
Leukocytes - All rats subjected to CLP were found at autopsy having darkish, foul-smelling peritoneal fluid consistent with gross fecal contamination of the abdominal cavity. In the present study the total white cell count and circulating neutrophil were significantly lower following laparoscopic CLP using CO2 than following LAP/CLP. Laparotomic CLP produced a significant reduction in lymphocyte count compared to laparoscopic CLP using CO2, corroborating with data from other authors37. Another work has shown a profound drop in white blood cells in animals subjected to laparotomy or pneumoperitoneum under intraperitoneal inoculation of Escherichia coli, without difference between them. These data suggest that some results are conflicting, but there is a tendency to leucopenia and lymphopenia after laparoscopic CLP.
Conclusions
The CO2 pneumoperitoneum reduced the inflammatory response in an animal model of peritonitis with respect to intraperitoneal cytokines, white blood cell count and clinical correlates of sepsis. The pneumoperitoneum produced hypercarbic acidosis in septic rats.
Received: October 10, 2005
Review: November 21, 2005
Accepted: December 19, 2005
Conflict of interest: none
Financial source: none
How to cite this article: Araújo Filho I, Honorato Sobrinho AA, Rego ACM, Garcia ACMA, Fernandes DP, Cruz TM, Costa TC, Medeiros AC. Influence of laparoscopy and laparotomy on gasometry, leukocytes and cytokines in a rat abdominal sepsis model. Acta Cir Bras. [serial on the Internet] 2006 Mar-Apr;21(2). Available from URL: http://www.scielo.br/acb
- 1. Aneman A, Svensson M, Stenqvist O, Dalenback J, Lonnroth H. Intestinal perfusion during pneumoperitoneum with carbon dioxide, nitrogen, and nitric oxide during laparoscopic surgery. Eur J Surg. 2000;166:706.
- 2. Crabtree JH, Fishman A. Videoscopic surgery under local and regional anesthesia with helium abdominal insufflation. Surg Endosc. 1999;13:10359.
- 3. Eisenhauer DM, Saunders CJ, Ho HS, Wolfe BM. Hemodynamic effects of argon pneumoperitoneum. Surg Endosc. 1994; 8:315320.
- 4. Fernandez-Cruz L, Saenz A, Taura P, Sabater L, Astudillo E, Fontanals J. Helium and carbon dioxide pneumoperitoneum in patients with pheochromocytoma undergoing laparoscopic adrenalectomy. World J Surg. 1998;22:12505.
- 5. Mann C, Boccara G, Grevy V, Navarro F, Fabre JM, Colson P. Argon pneumoperitoneum is more dangerous than CO2 pneumoperitoneum during venous gas embolism. Anesth Analg. 1997;85:136771.
- 6. Vezakis A, Davides D, Gibson JS, Moore MR, Shah H, Larvin M, McMahon MJ. Randomized comparison between low-pressure laparoscopic cholecystectomy and gasless laparoscopic cholecystectomy. Surg Endosc. 1999;13: 8903.
- 7. Gurtner GC, Robertson CS, Chung SCS, Ling TKW, Ip SM, Li AKC. Effect of carcon dioxide pneumoperitoneum on bacteremia and endotoxemia in an animal model of peritonitis. Br J Surg. 1995;82:844-8.
- 8. Tate JJT, Dawson JW, Chung SCS, Lau WY, Li AKC. Laparoscopic versus open appendicectomy - prospective randomised trial. Lancet. 1993;342:633-7.
- 9. Lau JY, Lo SY, Ng EK, Lee DW, Lam YH, Chung SC. A randomized comparison of acute phase response and endotoxemia in patients with perforated peptic ulcers receiving laparoscopic or open patch repair. Am J Surg. 1998;175:325-7.
- 10. Phillips EH, Franklin M, Carroll BJ, Fallas MJ, Ramos R, Rosenthal D. Laparoscopic colectomy. Ann Surg. 1992; 216: 703-7.
- 11. Basso N, Chang TM, Howard TJ, Passaro E Jr. Laparoscopic surgery. A difference. Arch Surg. 1992;127:1269-71.
- 12. Kopernik G, Avinoach E, Grossman Y, Levy R, Yulzari R, Rogachev B, Douvdevani A. The effect of a high partial pressure of carbon dioxide environment on metabolism and immune functions of human peritoneal cells-relevance of carbon dioxide pneumoperitoneum. Am J Obstet Gynecol. 1998;179:1503-10.
- 13. Chekan EG, Nataraj C, Clary EM, Hayward TZ, Brody FJ, Stamat JC, Fina MC, Eubanks WS, Westcott CJ. Intraperitoneal immunity and pneumoperitoneum. Surg Endosc. 1999;13:1135-8.
- 14. Miserez M, Eypasch E, Spangenberger W, Lefering R, Troidl H. Laparoscopic and conventional closure of perforated peptic ulcer: a comparison. Surg Endosc. 1996;10:831-6.
- 15. Druart ML, Van Hee R, Etienne J, Cadiere GB, Gigot JF, Legrand M, Limbosch JM, Navez B, Tugilimana M, Van Vyve E, Vereecken L, Wibin E, Yvergneaux JP. Laparoscopic repair of perforated duodenal ulcer: a prospective multicenter clinical trial. Surg Endosc. 1997;11:1017-20.
- 16. Benoit J, Cruaud P, Lauroy J, Boutelier P, Champault G. Does laparoscopic treatment of abdominal infections generate bacteremias? Prospective study: 75 cases. J Chir. 1995;132:472-7.
- 17. Eleftheriadis E, Kotzampassi K, Papanotas K, Heliadis N, Sarris K.. Gut ischemia, oxidative stress, and bacterial translocation in elevated abdominal pressure in rats. World J Surg. 1996;20:11-6.
- 18. Erenoglu C, Akin ML, Kayaoglu H, Celenk T, Batkin A. Is helium insufflation superior to carbon dioxide insufflation in bacteremia and bacterial translocation with peritonitis? J Laparoendosc Adv Surg Tech A. 2001;11:69-72.
- 19. Collet e Silva FD, Ramos RC, Zantut LF, Poggetti RS, Fontes B, Birolini D. Laparoscopic pneumoperitoneum in acute peritonitis does not increase bacteremia or aggravate metabolic or hemodynamic disturbances. Surg Laparosc Endosc Percutan Tech. 2000;10:305-10.
- 20. Ipek T, Paksoy M, Colak T, Polat E, Uygun N. Effect of carbon dioxide pneumoperitoneum on bacteremia and severity of peritonitis in an experimental model. Surg Endosc. 1998;12:432-5.
- 21. Ozguc H, Yilmazlar T, Zorluoglu A, Gedikoglu S, Kaya E . Effect of CO2 pneumoperitoneum on bacteremia in experimental peritonitis. Eur Surg Res. 1996;28:124-9.
- 22. Nordentoft T, Bringstrup FA, Bremmelgaard A, Stage JG. Effect of laparoscopy on bacteremia in acute appendicitis: a randomized controlled study. Surg Laparosc Endosc Percutan Tech. 2000;10:302-4.
- 23. Aguiar JLA, Moreira IEG, Chaves MM, Lopes SL, Santana V. Peritonite experimental: Modificação técnica do modelo de ligadura do ceco em ratos. An Fac Med Univ Fed Pernamb. 1996; 41: 59-62.
- 24. Kreimer F , Aguiar JLA, Castro C M M B, Lacerda CM, Reis T, Lisboa Jr F. Resposta terapêutica e inflamatória de ratos com peritonite secundária submetidos ao uso tópico de ampicilina/sulbactam. Acta Cir Bras. 2005;20(suppl 1):31-9.
- 25. Clary DVM, Bruch SM, Lau CL, Ali A, Chekan EG, Garcia-Oria MJ, Eubanks S. Effects of pneumoperitoneum on hemodynamic and systemic immunologic responses to peritonitis in pigs. J Surg Res. 2002;108:32-8.
- 26. Hanly EJ, Bachman SL, Marohn MR, Boden JH, Herring AE, DeMaio A, Talamini MA. Carbon dioxide pneumoperitoneum-mediated attenuation of the inflammatory response is independent of systemic acidosis. Surgery. 2005;137:559-66.
- 27. Douvdevani A, Rapaport J, Konforty A. Intracellular acidification mediates the inhibitory effect of peritoneal dialysate on peritoneal macrophages. J Am Soc Nephrol. 1995; 6:207-.13.
- 28. Carozzi S, Caviglia PM, Nasini MG, Schelotto C, Santoni O, Pietrucci A. Peritoneal dialysis solution pH and Ca2+ concentration regulate peritoneal macrophage and mesothelial cell activation. ASAIO J. 1994; 40:20-3.
- 29. West MA, Hackam DJ, Baker J, Rodriguez JL, Bellingham J, Rotstein OD. Mechanism of decreased in vitro murine macrophage cytokine release after exposure to carbon dioxide. Ann Surg. 1997;226:17990.
- 30. Redmond HP, Watson RWG, Houghton TO, Condron C, Watson RG, Bouchier-Hayes D. Immune function in patients undergoing open vs laparoscopic cholecystectomy. Arch Surg. 1994;129:1240-6.
- 31. Watson RWG, Redmond HP, McCarthy J, Burke PE, Bouchier-Hayes D. Exposure of the peritoneal cavity to air regulates early inflammatory responses to surgery in a murine model. Br J Surg. 1995; 82:1060-5.
- 32. West MA, Baker J, Bellingham J. Kinetics of decreased LPS-stimulated cytokine release by macrophages exposed to CO2. J Surg Res. 1996;63:26974
- 33. Temple LK, Litwin DE, McLeod RS. A meta-analysis of laparoscopic versus open appencectomy in patients suspected of having acute appendicitis. Can J Surg. 1999;42:377-83.
- 34. Kheirabadi BS, Tuthill D, Pearson R, MacPhee M, Drohan W, Tuthill D. Metabolic and hemodynamic effects of CO2 pneumoperitoneum in a controlled hemorrhage model. J Trauma. 2001;50:1031-43.
- 35. Kuntz C, Wunsch A, Bodeker C, Bay F, Rosch R, Winderer J, Herfarth C. Effect of pressure and gas type on intraabdominal, subcutaneous, and blood pH in laparoscopy. Surg Endosc. 2000;14:367-71.
- 36. Gandara V, De Vega DS, Escrin N, Zorrilla IG. Acid-base balance alterations in laparoscopic cholecystectomy. Surg Endosc. 1997;11:707-10.
- 37. Hanly EJ, Mendoza-Sagaon M, Murata K, Hardacre J, De Maio A, Talamini MA. CO2 pneumoperitoneum modifies inflammation response to sepsis. Ann Surg. 2003;237:343-50.
Publication Dates
-
Publication in this collection
21 Mar 2006 -
Date of issue
Apr 2006
History
-
Accepted
19 Dec 2005 -
Reviewed
21 Nov 2005 -
Received
10 Oct 2005