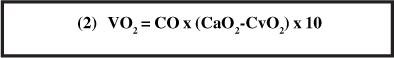Abstracts
PURPOSE: The purpose of this study is to compare the resting energy expenditure (REE) obtained by indirect calorimetry (IC) to the REE calculated by predictive equations (Brandi and Liggett) using the oxygen consumption (VO2) obtained by Fick's method in septic patients. METHODS: Prospective study in septic patients admitted in an intensive care unit of a university hospital. Fifteen adult patients (11 men and four women) were studied. VO2 measurements were made using a portable metabolic cart connected to a respirator and a pulmonary artery catheter. RESULTS: The APACHE II at admission was 22.6±7.2 with a mortality risk of 46.1±27.7%. The mean REE obtained by IC and by the Brandi and Liggett equations were 1699±271, 1815±355 and 1361±277 kcal.day-1 respectively. There were no statistically significant differences between the two methods, with the two means showing a difference of 8.7%. REE values were close for IC x BRANDI (r=0.80), but the IC x LIGGETT correlation was only 0.58. CONCLUSIONS: The results suggest that REE can be easily calculated by obtaining VO2 with the Swan-Ganz catheter and using the Brandi equation even for septic patients under mechanical ventilation.
Oxygen Consumption; Resting Energy Expenditure; Indirect Calorimetry; Sepsis
OBJETIVO: O objetivo desse estudo foi o de comparar o consumo de oxigênio em repouso (COR) obtido por calorimetria indireta (CI) com valores do COR calculados por equações preditivas (Brandi e Ligget) que utilizam o consumo de oxigênio (VO2) obtido pelo método de Fick em pacientes sépticos. MÉTODOS. Estudo prospectivo em pacientes sépticos internados em unidade de terapia intensiva de um hospital universitário. Quinze pacientes adultos (onze homens e quatro mulheres). As medidas de VO2 foram realizadas usando um aparelho metabólico portátil conectado ao respirador e por cateter em artéria pulmonar. RESULTADOS: O índice APACHE II na admissão foi 22.6±7.2 com risco de mortalidade de 46.1±27.7%. As médias do COR obtidas por CI e pelas equações de Brandi and Liggett foram, respectivamente, 1699±271, 1815±355 e 1361±277 kcal.day-1 Não se observaram diferenças estatisticamente significantes entre os dois métodos com as duas médias mostrando uma diferenças de 8.7%. Os valores de COR Também foram próximo dos valores de CI x BRANDI (r=0.80), mas a correlação com CI x LIGGETT apenas 0.58. CONCLUSÕES: Os resultados sugerem que o COR pode ser facilmente calculado para obter-se o VO2 com o cateter de Swan-Ganz e pelo uso da equação de Brandi para pacientes sépticos em ventilação mecânica.
Consumo de Oxigênio; Consumo de Energia em Repouso; Calorimetria Indireta; Sepse
19CLINICAL RESEARCH
ORIGINAL ARTICLE
PRÉ AND POSTOPERATIVE CARE
An easy way to estimate energy expenditure from hemodynamic data in septic patients1 1 . Research performed at the Intensive Care Unit of the Ribeirão Preto Hospital of Clinics, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brasil.
Um modo simples de estimar o consumo de energia a partir de dados hemodinâmicos em pacientes sépticos
Anibal BasileFilhoI; Maria Auxiliadora MartinsII; Flavio MarsonIII; Paulo Roberto Barbosa EvoraIV
IAssociate Professor, Division of Intensive Care Unit, Department of Surgery and Anatomy, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brazil
IIMD, PhD and SurgeoninChief, Division of Intensive Care Unit, Department of Surgery and Anatomy, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brasil
IIIMD, PhD, Division of Intensive Care Unit, Department of Surgery and Anatomy, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brasil
IVFull Professor and Head, Division Thoracic and Cardiovascular Surgery, Department of Surgery and Anatomy, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brazil
Correspondence Correspondence: Paulo Roberto Barbosa Évora Rua Rui Barbosa, 367, Ap. 15 14015120 Ribeirão Preto, SP, Brazil Phone/Fax: (+55) 16 36022497 prbevora@netsite.com.br
ABSTRACT
PURPOSE: The purpose of this study is to compare the resting energy expenditure (REE) obtained by indirect calorimetry (IC) to the REE calculated by predictive equations (Brandi and Liggett) using the oxygen consumption (VO2) obtained by Fick's method in septic patients.
METHODS: Prospective study in septic patients admitted in an intensive care unit of a university hospital. Fifteen adult patients (11 men and four women) were studied. VO2 measurements were made using a portable metabolic cart connected to a respirator and a pulmonary artery catheter.
RESULTS: The APACHE II at admission was 22.6±7.2 with a mortality risk of 46.1±27.7%. The mean REE obtained by IC and by the Brandi and Liggett equations were 1699±271, 1815±355 and 1361±277 kcal.day1 respectively. There were no statistically significant differences between the two methods, with the two means showing a difference of 8.7%. REE values were close for IC x BRANDI (r=0.80), but the IC x LIGGETT correlation was only 0.58.
CONCLUSIONS: The results suggest that REE can be easily calculated by obtaining VO2 with the SwanGanz catheter and using the Brandi equation even for septic patients under mechanical ventilation.
Keywords: Oxygen Consumption. Resting Energy Expenditure. Indirect Calorimetry. Sepsis.
RESUMO
OBJETIVO: O objetivo desse estudo foi o de comparar o consumo de oxigênio em repouso (COR) obtido por calorimetria indireta (CI) com valores do COR calculados por equações preditivas (Brandi e Ligget) que utilizam o consumo de oxigênio (VO2) obtido pelo método de Fick em pacientes sépticos.
MÉTODOS. Estudo prospectivo em pacientes sépticos internados em unidade de terapia intensiva de um hospital universitário. Quinze pacientes adultos (onze homens e quatro mulheres). As medidas de VO2 foram realizadas usando um aparelho metabólico portátil conectado ao respirador e por cateter em artéria pulmonar.
RESULTADOS: O índice APACHE II na admissão foi 22.6±7.2 com risco de mortalidade de 46.1±27.7%. As médias do COR obtidas por CI e pelas equações de Brandi and Liggett foram, respectivamente, 1699±271, 1815±355 e 1361±277 kcal.day1 Não se observaram diferenças estatisticamente significantes entre os dois métodos com as duas médias mostrando uma diferenças de 8.7%. Os valores de COR Também foram próximo dos valores de CI x BRANDI (r=0.80), mas a correlação com CI x LIGGETT apenas 0.58.
CONCLUSÕES: Os resultados sugerem que o COR pode ser facilmente calculado para obterse o VO2 com o cateter de SwanGanz e pelo uso da equação de Brandi para pacientes sépticos em ventilação mecânica.
Descritores: Consumo de Oxigênio. Consumo de Energia em Repouso. Calorimetria Indireta. Sepse.
Introduction
Acute disease and its treatment invariably alter the metabolism of critically ill patients, causing important changes in resting energy expenditure (REE). For this reason, an accurate determination of the REE of these patients is imperative, especially for patients in sepsis or septic shock. This insures their energy requirements, preventing the complications associated with hyper or undernutrition whose deleterious consequences have been well documented. 1,2 Many methods to assess the REE in critically ill patients have been described, but all of them have limitations. Indirect calorimetry, although being currently considered to be the "gold standard", has technical limitations such as requiring trained personnel with available time, the need for an inspired oxygen fraction of less than 60% and equipment of high cost. 3,4 On the other hand, other existing methods involve predictive equations such as the classical equation of Harris & Benedict. 5, plus various stress factors, whose purpose is to predict the energy cost of metabolic changes in this patient population. 6 These equations are extensively used because of their easy calculation and zero cost. However, these equations have proved to be imprecise in a wide variety of welldefined clinical situations, showing important inter and intraindividual variations. 7,8,9,10
Other methods developed some time ago and extensively used to calculate the REE are based on the Fick equation, which uses hemodynamic data such as cardiac output, serum hemoglobin concentrations, and mixed arterial and venous oxygen saturation obtained through a threelumen catheter inserted into the pulmonary artery. 11,12,13
On the basis of information related above, the objective of the present study was to assess the REE of septic patients through the correlation between indirect calorimetry and two predictive equations obtained from hemodynamic data.
Methods
The present prospective clinical study was conducted in the Intensive Care Unit of the University Hospital of the Ribeirão Preto Medical School, University of São Paulo, Brazil. The project was approved by the Human Research Ethics Committee of the same Hospital and free informed consent was obtained in all cases. The study was conducted on 15 patients (11 males and four females) with septic shock after blood volume restoration and hemodynamic stabilization. The criteria used for the diagnosis of septic shock were those established by the consensus conference held by the American College of Chest Physicians/Society of Critical Care Medicine. 14 Inclusion criteria were patients victims of septic shock from the third to the fifth day of evolution with an indication of invasive hemodynamic monitoring. Exclusion criteria were age > 85 and < 15 years, need for FiO2 (oxygen fraction in inspired air) > 60%, mean arterial pressure (MAP) < 50 mmHg, and diuresis < 50 mL.h1.
Upon admission to the Intensive Care Unit, all patients were stratified according to the APACHE II severity index 15 and by the system of sepsis grading.16 Invasive and noninvasive hemodynamic monitoring, blood volume replacement, mechanical ventilation (Servo 900C, Siemens, Sweden), antibiotic treatment, administration of vasoactive drugs and nutritional therapy according to the REE calculated by indirect calorimetry were performed in all cases. The patients were sedated with benzodiazepines and/or opiates and a neuromuscular blocker was used when necessary. Current patient weight was measured with a portable electronic scale (Slingscale 2002, HillRom, Hillenbrand Industries, USA). The mean duration of the protocol was three hours, during which three series of hourly and simultaneous measurements of VO2 and of the REE were performed by the method of Fick and by indirect calorimetry. All patients were submitted to invasive hemodynamic monitoring and to indirect calorimetry by the bedside using a portable calorimeter. In parallel, venous blood specimens were collected from the proximal orifice (right atrium) and from the distal orifice (pulmonary artery) of the SwanGanz catheter and arterial blood was obtained from the radial artery for blood gas analysis. The samples were used for the determination of hemoglobin (Hb) concentration, arterial blood oxygen saturation (SaO2), venous blood oxygen saturation (SvO2), and arterial and venous blood partial oxygen pressure (PaO2 and PvO2) respectively. After obtaining VO2 (mL.min1) by the method of Fick and oxygen content in arterial and venous blood, it was possible to calculate REE (kcal.day1) according to the equations described below:
Where 4.838 kcal.L1 (20.2 kJ.L1) is the caloric value of oxygen of a respiratory quotient (RQ) of approximately 0.824 (the mean value obtained for the present population as measured by indirect calorimetry), assuming a nitrogen excretion of 13 to 15 g.day1, and 1.44 is the conversion factor for kcal.day1. The method of Fick uses the following equations for VO2 and REE calculations: 12,13
Where:
CO = Cardiac output (L.min1)
CaO2 = Arterial O2 content (1.38 x Hb x SaO2) + (0.00301 x PaO2)
CvO2 = Venous O2 content (1.38 x Hb x SvO2) + (0.00301 x PvO2)
By replacing the above values in equation (2), we obtain:
By replacing the values of equation (3) in equation (1), we obtain the equation below:
However, the addition of the value of 0.211 x (PaO2PvO2) from the above equation represents less than 1% of the final result of the calculation. On this basis, the final equation summarized for practical purposes is as follows:
Indirect calorimetry was performed in all patients using the DELTATRAC II Metabolic Monitor portable calorimeter (DatexOhmeda, Finland). Barometric and gas pressure was calibrated before each protocol. Each patient was submitted to three hourly series of consecutive VO2 measurements lasting 30 minutes each, for a total of 45 measurements. The two assessment methods were compared by the nonparametric sign Wilcoxon test for two correlated samples. The level of significance was set at p < 0.05 for all comparisons. The Pearson correlation coefficient was also used to determine the correlation between the REE values obtained by the three methods
Results
The study was conducted on 15 hemodynamically stable patients (11 males and four females) with a diagnosis of septic shock admitted to the intensive care unit between the third and fifth day of evolution. Mean age was 41.3 ± 18.9 years. The mean prognostic APACHE II index was 22.6 ± 7.2, with a calculated death risk of 46.1 ± 27.7%. The mean sepsis score (SS) was 20.3 ± 9.5. The overall mortality rate for the population studied was 20% (n = 3). The main clinical and demographic data of the patients are presented in Table 1. Most patients (60%; n = 9) had pneumonia with acute respiratory insufficiency. No complication occurred due to the procedures performed during the study (venous catheterization, arterial puncture and indirect calorimetry). The mean REE obtained by indirect calorimetry (IC) was 1669 ± 271 kcal.day1 and the values calculated by the equations of BRANDI and LIGGETT were 1815 ± 355 and 1361 ± 277, respectively (Table 2). The differences in the values obtained by indirect calorimetry and in the predictive equations was +8.7% (IC x BRANDI) (p<0.5) and 22.7% (IC x LIGGETT) (p>0.5). The correlation coefficient between indirect calorimetry and the BRANDI equation was 0.80 (Figure 1), and the correlation between indirect calorimetry and the LIGGETT equation was 0.58 (Figure 2).
Discussion
The objective of the present study was to compare the REE obtained by indirect calorimetry to the REE calculated by the predictive equations of Brandi and Liggett using hemodynamic VO2 data obtained by the method of Fick in patients with septic shock. Indeed, the REE of critically ill patients can be estimated by a wide variety of methods. Most of the formulas proposed in the literature are derivations of the equation of Harris & Benedict, which was developed, in principle, for healthy individuals. Since, by definition, basal energy expenditure is the minimum energy requirement of healthy individuals at rest, a problem appears when this energy must be calculated for patients in special situations such as sepsis, trauma and extensive burns. Efforts performed by some authors to improve the precision of the equation of Harris & Benedict for these situations by adding stress factors to the original equation have introduced an even greater source of error and perhaps of cost in the planning of nutritional therapy. Accurate administration of nutritional therapy is important for this patient population since it is well known that insufficient nutrient administration depresses the ventilatory drive17, reduces the strength of respiratory muscles18, and generates immunodepression19. On the other hand, overnutrition causes hyperglycemia and fatty liver infiltration and may favor the onset of respiratory acidosis and delay of patient weaning from the respirator. 20 On this basis, since over or undernutrition is deleterious, indirect calorimetry became the gold standard for the assessment of energy requirements.
We attempted in the present study to asses two equations based on hemodynamic data and to correlate them with calorimetry, especially considering that in most intensive care units a threelumen catheter is routinely introduced into the pulmonary artery for purposes of hemodynamic exploration. We opted for the assessment of the two most commonly used equations, i.e., the equations of Brandi and of Liggett. Originally, Liggett et al 11, in 1987, suggested a new method for determining the REE using a catheter positioned in the pulmonary artery. These authors developed an equation based on hemodynamic data and correlated it with classical calorimetry. According to this initial study, there was a good correlation between the two methods (r = 0.90, p<0.0001).
On the other hand, Flancbaum et al.9, in 1999, studied 36 critically ill and mechanically ventilated patients and detected a weak correlation between the REE measured by indirect calorimetry and by the Liggett equation (r = 0.28). The authors attributed these findings to the fact that any variation in one of the components of the Fick equation may introduce errors in the calculation of the REE. According to the cited authors, SvO2 may fluctuate during the study period because a fall in its value below 60% may indicate increased tissue oxygen consumption or the impairment of one of the variables related to oxygen transport. 21 Although this information has a certain logic, the present patient series showed SvO2 and SaO2 within normal limits (SvO2 = 80 ± 5%; range = 6588%; SaO2 = 99 ± 1%; range = 96100%), and the correlation detected (r = 0.58) is unlikely to be responsible for the small variations of these variables. Another potential explanation for the weak correlation between the two methods may be due to the relation between oxygen supply and demand, a situation described for sepsis. 22 However, the most plausible reason, which was also the major challenge for the validation of this initial concept, is related to the mathematical coupling of the different hemodynamic variables involved in the calculation. 23
The present patients showed an energy consumption of 1669 ± 271 kcal and an APACHE II score of 22.6 ± 7.2. These data are indicative of metabolic stress. Interestingly, the disease severity scores have been used to predict the REE of critically ill patients. Swinamer et al. 24 demonstrated that patients with an APACHE II score > 20 presented a very poor correlation between calorimetric data and data obtained by the method of Fick, with differences of up to 50% being detected between the two methods. Technical errors or misinterpretation of the results may also be introduced in both methods for the calculation of VO2. It should be pointed out that the calculation of VO2 by the method of Fick has an inherent limitation represented by the fact that this procedure does not take into account pulmonary O2 consumption. Total O2 consumption may be underestimated by as much as 25% in the presence of pulmonary infections in patients on mechanical ventilation. 25
With respect to the proposed calculation of the REE by the equation REE = VO2 x 6.996 using the VO2 obtained by the method of Fick, Brandi et al. 12 compared the calculation of energy expenditure by the method of Fick and by indirect calorimetry in a group of spontaneously ventilating 26 patients during the postoperative phase of interventions due to various types of trauma. These authors detected an excellent correlation between the two methods (r = 0.96, p<0.001). It should be pointed out that all the patients studied here were submitted to mechanical ventilation and, even so, the correlation between the two methods was 0.80. We used the same equation because the 6.996 variable (equation 1) was obtained from a mean RQ of 0.827, extremely close to the mean value of 0.824 detected in the present patient series (Table 2). In this respect, it should be emphasized that the introduction of a fixed RQ value (0.824) in this equation and a nitrogen excretion of 13 to 15 g.day1 introduces an error of approximately 3.6% because the caloric value of oxygen is 4.686 to 5.047 for an RQ with a physiological variation of 0.70 to 1.00, respectively. For the calculation of energy expenditure this margin of error can be disregarded.
Conclusion
Although indirect calorimetry is the most reliable method for the determination of the energy needs of a given patient, the satisfactory correlation detected between calorimetry and the Brandi equation, and the small difference of only 8.7% between the two methods permit us to propose the use of the equation REE = VO2 x 6.996, from the VO2 obtained in the method of Fick, even for septic patients under mechanical ventilation. The present study suggests that the prediction of energy expenditure by this simple procedure can be used for clinical purposes, especially in intensive care units where only the technique of insertion of a threelumen catheter into the pulmonary artery is available. In addition, the equation uses only VO2 as the variable to be estimated, which is about 75% of the calculation of total energy expenditure.13
Acknowledgements
FAPESP Fundação de Amparo À Pesquisa do Estado de São Paulo and FAEPA Fundação de Apoio ao Ensino, Pesquisa e Assistência do HCFMRPUSP
Conflict of interest: none
Financial source: FAPESP and FAEPA
Comments:
Accurate nutritional therapy is important for critically ill patients such as the subjects studied in the present series; and, to determine the individual nutritional needs the measurement of REE is mandatory. The gold standard and the most reliable method for REE calculation is the indirect calorimetry. However, such method has some limitations because it is time consuming and requires an equipment of high cost. As stressed by the authors, the estimate of REE by using easier methods based on predictive equations such as the classical equation of Harris & Benedict. 5 have shown imprecise results in a wide variety of clinical situations. Thus, the authors have compared REE obtained by indirect calorimetry (IC) to the REE calculated by predictive equations (Brandi and Liggett) using the oxygen consumption (VO2) obtained by Fick's method in 15 septic patients. A good correlation was found between the IC method and Brandi equation. The results support the authors´ proposal of using the equation REE= VO2 x 6.996 to calculate the diary caloric requirement for septic patients under mechanical ventilation.
Antonio Carlos Pereira Martins
Full Professor and Head, Division of Urology, Department of Surgery and Anatomy, Ribeirão Preto Faculty of Ribeirão Preto, University of São Paulo, Brazil.
- 1. Kiney JM. Metabolic responses of the critically ill patient. Crit Care Clin. 1995;11:56986.
- 2. Hill AG, Hill GL. Metabolic response to severe injury. Brit J Surg. 1998;85:88490
- 3. Ultman JS, Bursztein S. Analysis of error in the determination of respiratory gas exchange at varying FiO2. J Appl Physiol. 1981;50:2106.
- 4. Matarese L. Indirect calorimetry: Technical aspects. J Am Diet Assoc. 1997;97:S15460.
- 5. Harris JA, Benedict FG. A biometric study of basal metabolism in man. Washington, CO, Carnegie Institute of Washington. 1919, Publ. nş 279.
- 6. Long CL, Schaffel N, Geiger JW, Schiller WR, Blakemore WS. Metabolic response to injury and illness: estimation of energy and protein needs from indirect calorimetry and nitrogen balance. JPEN. 1979;3:4526.
- 7. Daly JM, Heymsfield SB, Head CA, Harvey LP, Nixon DW, Katzeff H, Grossman GD. Human energy requirements: overestimation by widely used prediction equation. Am J Clin Nutr. 1985;42:11704.
- 8. Van Lanschot JJ, Feenstra BWA, Vermeij Cg, Bruining HA. Calculations versus measurement of total energy expenditure. Crit Care Med. 1986;14:9815.
- 9. Flancbaum L, Choban PS, Sambucco S, Verducci J, Burge JC. Comparison of indirect calorimetry, the Fick method, and prediction equations in estimating the energy requirements of critically ill patients. Am J Clin Nutr. 1999;69:4616.
- 10. Zauner C, Schuster B, Schneeweiss B: Similar metabolic responses to standardized total parenteral nutrition of septic and nonseptic critically ill patients. Am J Clin Nutr. 2001;74:26570.
- 11. Liggett SB, St Jonhn RE, Lefrak SS. Determination of resting energy expenditure utilizing the thermodilution pulmonary artery catheter. Chest. 1987;91:5626.
- 12. Brandi LS, Grana M, Mazzanti T, Giunta F, Natali A, Ferrannini E. Energy expenditure and gas exchange measurements in postoperative patients: Thermodilution versus indirect calorimetry. Crit Care Med. 1992;20:127383.
- 13. Brandi LS, Bertolini R, Calafa M. Indirect calorimetry in critically ill patients: Clinical applications and practical advice. Nutrition. 1997;13:34958.
- 14. Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA, Schein RM, Sibbald WJ. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 1992;101:164455.
- 15. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. Apache II: a severity of disease classification system. Crit Care Med. 1985;13:81829.
- 16. Elebute EA, Stoner HB. The grading of sepsis. Br J Surg. 1983;70:2931.
- 17. Doekel RC, Zwillich CW, Scoggin GH, Kryger M, Weil JV. Clinical semi starvation: Depression of hypoxic ventilatory response. N Engl J Med. 1976;295:35861.
- 18. Kelly SM, Rosa A, Field S, Coughlin M, Shizgal HM, Macklem PT. Inspiratory muscle strength and body composition in patients receiving total parenteral nutrition therapy. Am Rev Respir Disease. 1984;130:337.
- 19. Good RA, Lorenz E. Influence of energy levels and trace metals on health and life span. JPEN. 1990;14:2306.
- 20. Laaban JP, Lemarie F, Baron JF, Trunet P, Harf A, Bonnet JL, Teisseire B. Influence of caloric intake on respiratory mode during mandatory minute volume ventilation. Chest. 1985;87:6772.
- 21. Ermakov S, Hoyt JW. Pulmonary artery catheterization. Crit Care Clin. 1992;8:773806.
- 22. Tuchschmidt J, Oblitas D, Fried JC. Oxygen consumption in sepsis and septic shock. Crit Care Med. 1991;19:66471.
- 23. Archie JP. Mathematical coupling of data: a common source of error. Ann Surg. 1981; 193:296303.
- 24. Swinamer DL, Phang PT, Jones RL, Grace M, King EG. Twentyfour hour energy expenditure in critically ill patients. Crit Care Med. 1987;60:63743.
- 25. Hensel M, Kox WJ. Increased intrapulmonary oxygen consumption in mechanically ventilated patients with pneumonia. Am J Respir Crit Care Med. 1999;160:13743.
Publication Dates
-
Publication in this collection
20 May 2008 -
Date of issue
2008