Abstracts
PURPOSE: To investigate the effect of L-alanyl-L-glutamine (L-Ala-Gln) preconditioning in an acute cerebral ischemia/reperfusion (I/R) model in gerbils. METHODS: Thirty-six Mongolian gerbils (Meriones unguiculatus), (60-100g), were randomized in 2 groups (n=18) and preconditioned with saline 2.0 ml (Group-S) or 0.75g/Kg of L-Ala-Gln, (Group-G) administered into the femoral vein 30 minutes prior to I/R. Each group was divided into three subgroups (n=6). Anesthetized animals (urethane, 1.5g/Kg, i.p.) were submitted to bilateral occlusion of common carotid arteries during 15 minutes. Samples (brain tissue and arterial blood) were collected at the end of ischemia (T0) and after 30 (T30) and 60 minutes (T60) for glucose, lactate, myeloperoxidase (MPO), thiobarbituric acid reactive substances (TBARS), glutathione (GSH) assays and histopathological evaluation. RESULTS: Glucose and lactate levels were not different in studied groups. However glycemia increased significantly in saline groups at the end of the reperfusion period. TBARS levels were significantly different, comparing treated (Group-G) and control group after 30 minutes of reperfusion (p<0.05) in cerebral tissue. Pretreatment with L-Ala-Gln promoted a significant increase in cerebral GSH contents in Group-G at T30 (p<0.001) time-point compared with Group-S. At T30 and T60, increased levels of GSH occurred in both time-points. There were no group differences regarding MPO levels. Pyknosis, presence of red neurons and intracellular edema were significantly smaller in Group-G. CONCLUSION: Preconditioning with L-Ala-Gln in gerbils submitted to cerebral ischemia/reperfusion reduces oxidative stress and degeneration of the nucleus (pyknosis) and cell death (red neurons) in the cerebral tissue.
Brain Ischemia; Reperfusion; Metabolism; Drug effects; Glutamine; Comparative Study; Gerbillinae
OBJETIVO: Investigar o efeito do pré-condicionamento com L-alanil-L-glutamina (L-Ala-Gln) em gerbils submetidos à isquemia/reperfusão (I/R) cerebral aguda. MÉTODOS: Trinta e seis gerbils (Meriones unguiculatus) (60-100g) foram divididos em dois grupos (n=18) e pré-condicionados com 2,0 ml de soro fisiológico (Grupo-S) ou 0.75g/kg de L-Ala-Gln, (Grupo-G), administrados na veia femoral 30 minutos antes da I / R. Cada grupo foi dividido em três subgrupos (n=6).Animais anestesiados com uretano, 1.5g/kg, ip, foram submetidos à oclusão bilateral das artérias carótidas comuns, durante 15 minutos. Amostras (tecido cerebral e sangue arterial) foram coletadas no final da isquemia (T0) e após 30 (T30) e 60 minutos (T60) para a aferição das concentrações de glicose, lactato, mieloperoxidase (MPO), substâncias reagentes ao ácido tiobarbitúrico (TBARS), glutationa (GSH) e avaliação histopatológica. RESULTADOS: As concentrações de glicose e lactato não foram diferentes nos grupos estudados; a glicemia aumentou significativamente no Grupo-S ao final da reperfusão. Concentrações de TBARS no tecido cerebral foram significativamente diferentes, comparando os Grupos G e S, no T30 (p <0,05). O pré-tratamento com L-Ala-Gln promoveu um aumento significativo de GSH cerebral no Grupo-G comparado ao Grupo-S no T30 (p <0,001). Houve aumento das concentrações de GSH no T30 e T60 no Grupo-G. Não houve diferenças quanto as concentrações de MPO. Picnose, presença de neurônios vermelhos e edema intracelular foram significativamente menores no Grupo-G. CONCLUSÃO: O pré-condicionamento com L-Ala-Gln em gerbils submetidos à isquemia/reperfusão cerebral reduz o estresse oxidativo, a degeneração nuclear (picnose) e morte celular (neurônios vermelhos) no tecido cerebral.
Isquemia Encefálica; Reperfusão; Metabolismo; Efeitos de drogas; Glutamina; Estudo Comparativo; Gerbillinae
4 - ORIGINAL ARTICLE
ISCHEMIA-REPERFUSION
Preconditioning with L-alanyl-L-glutamine in a Mongolian Gerbil model of acute cerebral ischemia/reperfusion injury1 Correspondence: Paulo Roberto Leitão de Vasconcelos Rua Professor Costa Mendes, 1608/3º andar, Bloco Didático 60430-140 Fortaleza - CE Brasil Tel.: (55 85)3366-8083 Fax: (55 85)3366-8064 paulo.vasconcelos@ufc.br
Pré-condicionamento com L-alanil-L-glutamina em modelo de isquemia/reperfusão cerebral aguda em Gerbils da Mongólia
Vilma Leite de Sousa PiresI; José Reniclebson Feitosa de SouzaII; Sergio Botelho GuimarãesIII; Antonio Ribeiro da Silva FilhoIV; José Huygens Parente GarciaV; Paulo Roberto Leitão de VasconcelosVI
IFellow PhD Degree, Department of Surgery, Postgraduate Program, UFC, Ceara, Brazil. Technical procedures, acquisition and interpretation of data. The article is part of a PhD Degree thesis
IIGraduate student, Christus Faculty, Fortaleza-CE, Brazil. Helped with technical procedures, acquisition of data
IIIPhD, Associate Professor, Department of Surgery, Head, LABCEX. UFC, Ceara, Brazil. Manuscript writing, statistical analysis, graphics design
IVPhD, Full Professor and Dean, Christus Faculty, Fortaleza-CE, Brazil. Critical revision and analysis of data
VPhD, Associate Professor, Head, Department of Surgery, UFC, Ceara. Critical revision and analysis of data
VIPhD, Associate Professor, Coordinator, Postgraduate Program, Department of Surgery, UFC, Ceara, Brazil. Tutor, responsible for conception, design, intellectual and scientific content of the study, critical analysis, final approval of manuscript
Correspondence Correspondence: Paulo Roberto Leitão de Vasconcelos Rua Professor Costa Mendes, 1608/3º andar, Bloco Didático 60430-140 Fortaleza - CE Brasil Tel.: (55 85)3366-8083 Fax: (55 85)3366-8064 paulo.vasconcelos@ufc.br
ABSTRACT
PURPOSE: To investigate the effect of L-alanyl-L-glutamine (L-Ala-Gln) preconditioning in an acute cerebral ischemia/reperfusion (I/R) model in gerbils.
METHODS: Thirty-six Mongolian gerbils (Meriones unguiculatus), (60-100g), were randomized in 2 groups (n=18) and preconditioned with saline 2.0 ml (Group-S) or 0.75g/Kg of L-Ala-Gln, (Group-G) administered into the femoral vein 30 minutes prior to I/R. Each group was divided into three subgroups (n=6). Anesthetized animals (urethane, 1.5g/Kg, i.p.) were submitted to bilateral occlusion of common carotid arteries during 15 minutes. Samples (brain tissue and arterial blood) were collected at the end of ischemia (T0) and after 30 (T30) and 60 minutes (T60) for glucose, lactate, myeloperoxidase (MPO), thiobarbituric acid reactive substances (TBARS), glutathione (GSH) assays and histopathological evaluation.
RESULTS: Glucose and lactate levels were not different in studied groups. However glycemia increased significantly in saline groups at the end of the reperfusion period. TBARS levels were significantly different, comparing treated (Group-G) and control group after 30 minutes of reperfusion (p<0.05) in cerebral tissue. Pretreatment with L-Ala-Gln promoted a significant increase in cerebral GSH contents in Group-G at T30 (p<0.001) time-point compared with Group-S. At T30 and T60, increased levels of GSH occurred in both time-points. There were no group differences regarding MPO levels. Pyknosis, presence of red neurons and intracellular edema were significantly smaller in Group-G.
CONCLUSION: Preconditioning with L-Ala-Gln in gerbils submitted to cerebral ischemia/reperfusion reduces oxidative stress and degeneration of the nucleus (pyknosis) and cell death (red neurons) in the cerebral tissue.
Keywords: Brain Ischemia. Reperfusion. Metabolism. Drug effects. Glutamine. Comparative Study. Gerbillinae.
RESUMO
OBJETIVO: Investigar o efeito do pré-condicionamento com L-alanil-L-glutamina (L-Ala-Gln) em gerbils submetidos à isquemia/reperfusão (I/R) cerebral aguda.
MÉTODOS: Trinta e seis gerbils (Meriones unguiculatus) (60-100g) foram divididos em dois grupos (n=18) e pré-condicionados com 2,0 ml de soro fisiológico (Grupo-S) ou 0.75g/kg de L-Ala-Gln, (Grupo-G), administrados na veia femoral 30 minutos antes da I / R. Cada grupo foi dividido em três subgrupos (n=6).Animais anestesiados com uretano, 1.5g/kg, ip, foram submetidos à oclusão bilateral das artérias carótidas comuns, durante 15 minutos. Amostras (tecido cerebral e sangue arterial) foram coletadas no final da isquemia (T0) e após 30 (T30) e 60 minutos (T60) para a aferição das concentrações de glicose, lactato, mieloperoxidase (MPO), substâncias reagentes ao ácido tiobarbitúrico (TBARS), glutationa (GSH) e avaliação histopatológica.
RESULTADOS: As concentrações de glicose e lactato não foram diferentes nos grupos estudados; a glicemia aumentou significativamente no Grupo-S ao final da reperfusão. Concentrações de TBARS no tecido cerebral foram significativamente diferentes, comparando os Grupos G e S, no T30 (p <0,05). O pré-tratamento com L-Ala-Gln promoveu um aumento significativo de GSH cerebral no Grupo-G comparado ao Grupo-S no T30 (p <0,001). Houve aumento das concentrações de GSH no T30 e T60 no Grupo-G. Não houve diferenças quanto as concentrações de MPO. Picnose, presença de neurônios vermelhos e edema intracelular foram significativamente menores no Grupo-G.
CONCLUSÃO: O pré-condicionamento com L-Ala-Gln em gerbils submetidos à isquemia/reperfusão cerebral reduz o estresse oxidativo, a degeneração nuclear (picnose) e morte celular (neurônios vermelhos) no tecido cerebral.
Descritores: Isquemia Encefálica. Reperfusão. Metabolismo. Efeitos de drogas. Glutamina. Estudo Comparativo. Gerbillinae.
Introduction
The use of animal models for studying the effects of cerebral ischemia has been frequent in recent decades. Studies dealing with the understanding of pathological mechanisms associated with stroke-like events have partly elucidated the complex molecular mechanisms associated with cerebral ischemia/reperfusion cell death1,2.
Studies in the Mongolian gerbil (Meriones unguiculatus) showed that these animals are very susceptible to experimental cerebral ischemia by unilateral occlusion (permanent) or bilateral (transient) of the common carotid artery3.
Histological studies revealed absence of posterior communicating artery in the gerbil, different from other rodents, where there is a communication connecting the carotid and vertebrobasilar arterial systems4. The lack of collateral communications in gerbils makes them more likely to succumb after severe ischemic events5. Somova et al.6 studied the effects of cerebral ischemia after 6, 12 and 30 days in a model of unilateral cerebral ischemia in gerbils. In that experiment 50% of the animals died after three days of ischemia6.
Reperfusion following cerebral ischemia leads to the generation of pro-oxidant species in the brain tissue which cause neuronal damage by acting directly on macromolecules, including proteins, lipids and DNA, or indirectly by interfering with cell signaling pathways and gene expression regulation7. Cerebral tissue is probably particularly susceptible to the action of oxidating agents and therefore to ischemia-reperfusion injury8.
The models of coronary artery occlusion whether unilateral or bilateral, transient or permanent in gerbils have the following advantages: low maintenance cost, relatively homogeneous samples, strong cerebral anatomical and physiological similarity with other species, and an uncomplicated surgical procedure, thus providing a large number of ischemic animals to evaluate the efficacy of cerebral protective agents9.
Cerebral ischemia is the consequence of the reduction in blood flow below a critical threshold and the concomitant limitation in the supplies of primary cerebral substrates like glucose and oxygen. The amino acids glutamate, GABA, and glutamine are available as substrates in the extracellular fluid, and may be used as alternative fuels by astrocytes or neurons under conditions of glucose deprivation10.
Glutamine (GLN) is a conditionally essential nutrient during sepsis or trauma11. GLN is the most abundant amino acid in plasma and skeletal muscle. However, GLN levels fall dramatically after major injury or infection12. L-alanyl-glutamine (Ala-Gln) is a highly stable dipeptide, can be heat-sterilized and when infused intravenously is promptly hydrolyzed to glutamine and alanine13. Recently, glutamine has been demonstrated to protect against ischemia/reperfusion (I/R) injury of the gut, heart, liver and skeletal muscle14. The mechanism is still incompletely understood and may be partly related to the preservation of GSH content15,16. Another study has demonstrated that GLN preconditioning protects effectively against hepatic ischemia-reperfusion injury17.
Considering the known protective effects of GLN on ischemia/reperfusion, the aim of this study is therefore to evaluate the effect L-Ala-Gln preconditioning on metabolic and histopathological parameters in a gerbil model of acute cerebral ischemia/reperfusion injury.
Methods
Approval for experimental use of laboratory animals was obtained from the local Ethics Committee on Animal Use (CEUA, former CEPA) (protocol #127/07, February, 2008). All surgical procedures and animal handling were conducted in accordance with the Brazilian Federal Law No. 11794 of October 8, 2008 (http://www.planalto.gov.br/ccivil_03/_Ato2007-2010/2008/Lei/L11794.htm). The study was designed so as to minimize the number of animals required for the experiments. The animals were housed in polypropylene cages at ambient temperature of 24ºC on a 12 h light-dark cycle. Gerbils were allowed free access to food (Purina chow) and tap water until the beginning of the experiment.
Study design
In this controlled experimental study, 36 Mongolian gerbils (Meriones unguiculatus) aged 8-16 months, weighing 60-100 grams, were randomly distributed in two groups: 18 gerbils preconditioned with saline solution prior to ischemia-reperfusion (Group S), and 18 gerbils preconditioned with L-Ala-Gln prior to ischemia-reperfusion (Group G). Each group was divided into three subgroups (n=6) based on the time of reperfusion following ischemia: 0 min (T0), 30 min (T30) and 60 min (T60). Thirty minutes before induction of ischemia, 2 mL 0.9% saline solution or 0.75g/Kg Ala-Gln (completed to 2 mL with saline solution) was administered i.v. into the femoral vein. Anesthetized animals (urethane, 1.5g/Kg, i.p.), were submitted to surgery with bilateral occlusion of the common carotid arteries (CCAs) during 15 minutes using vascular bulldog clamps. The clamps were removed simultaneously and brain tissue and arterial blood was sampled at T0, T30 and T60 for the determination of tissue and blood levels of metabolites and for histopathological evaluation of the internal pyramidal layer and the internal granular layer of the cerebral cortex.
Cerebral ischemia induction technique
Under satisfactory level of anesthesia18 the CCAs were isolated bilaterally at 0.5cm from the internal/external artery bifurcation (clamping site) and occluded during 15 minutes with vascular bulldog clamps19. Ischemia was followed by reperfusion for 30 or 60 minutes. Removal of cerebral hemispheres was performed using a technique published elsewhere19. Blood samples were collected from the abdominal aorta. Samples were snap-frozen in liquid nitrogen, placed in test tubes with 10% perchloric acid and stored at -4ºC for posterior enzyme analysis. Tissues for malondialdehyde (MDA), reduced glutathione (GSH) and Myeloperoxidase (MPO) assays were frozen and stored at -4ºC and preserved.
Chemicals and drugs
L-Ala-Gln (DipeptivenTM) was purchased from Frenesius Kabi Áustria GmbH Graz/Áustria. All other chemicals were purchased from standard commercial sources and were of the highest quality available.
Histopathological evaluation
The histopathological evaluation was performed by two independent pathologists blinded to clinical and pathological data. Tissue samples were fixated in 10% formaldehyde and embedded in paraffin, following standard histology procedures. Cerebral tissue sections (4 µm) were prepared, deparaffinized, hydrated and stained with hematoxylin and eosin. Histological parameters (pyknosis, red neurons, congestion and intracellular edema) were scored on a 4-point scale (0, 1, 2, 3) from absent to severe, as described by Greca et al.20.
Laboratory parameters
Cerebral tissue and blood metabolites (glucose21, lactate22), oxidative stress (malondialdehyde23 - MDA, reduced glutathione24 - GSH) and inflammation (myeloperoxidase25 - MPO) were evaluated by methods described in the literature. Internal pyramidal layer and the internal granular layer of the cerebral cortex were submitted to histopathological examination26.
Calculations were based on different optical densities read with a spectrophotometer at 412 nm (Beckman Du-640). Differences in absorbance before and after the biochemical reactions were used to estimate GSH levels, expressed in µmol/g fresh tissue.
Statistical analysis
Graphpad Prism 5.0 (GraphPad Software, www.graphpad.com) was used for statistical analysis and graphics design. All data were tested for distribution (Kolmorogov-Smirnov test with Dallal-Wilkinson-Lilliefor P value). Group S and Group G were compared at different reperfusion times (T0, T30 and T60) using Student's t test for unpaired variables. Intragroup reperfusion times were compared with ANOVA associated with the Tukey multiple comparison test (pairwise comparisons) or with the Kruskal-Wallis test combined with Dunn's test (multiple comparisons). Median values, interquartile ranges and minimum and maximum values were determined for ordinal variables (histopathological scores), followed by intergroup comparisons of different reperfusion times with the Mann-Whitney test. The level of statistical significance was set at 5% (p<0.05).
Results
Metabolites
Results are presented in tables and figures. Glucose values were not different, comparing groups. However glycemia levels increased significantly in saline groups at the end of the reperfusion period (Table 1). Lactate concentrations were not different in studied groups (Table 2).
Oxidative stress
TBARS levels (µmol MDA/g of wet tissue) in cerebral tissue were significantly different, comparing treated (Group G) and control group (saline), after 30 minutes of reperfusion (p<0.05). No significant intragroup variations were observed (Figure 1).
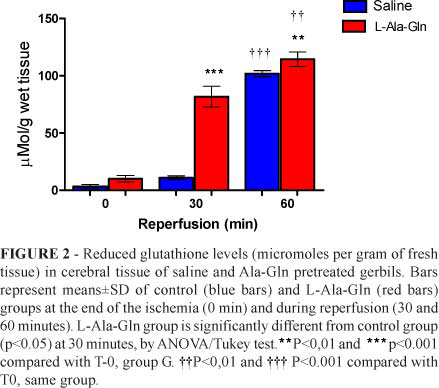
The pretreatment with L-Ala-Gln promoted a significant increase in cerebral GSH contents in T30 (P<0.001) time-point compared with saline pretreated gerbils. After 30 and 60 minutes of reperfusion, increased levels of GSH occurred in both time-points (compared with T0 in group G animals (Figure 2).
Figure 3 shows the mean MPO activity (MPO units/mg tissue) measured in cerebral tissue for Group S and Group G. The groups did not differ significantly, but within both groups MPO levels were significantly greater in T60 than in T0 or T30 (p<0.001).
Histological studies
The degree of pyknosis in the internal pyramidal layer was significantly smaller in Group G than in Group S at T30 (p=0.009) and T60 (p=0.037). Within each group, the degree of pyknosis in this layer was significantly greater in T60 than in T0 (p<0.01 for Group S; p<0.05 for Group G). Likewise, the degree of pyknosis in the internal granular layer was significantly smaller in Group G than in Group S at T30 (p=0.021) and T60 (p=0.041). However, in this layer pyknosis did not vary significantly between the different perfusion times (Table 3).
The number of red neurons in the internal pyramidal layer was significantly smaller in Group G than in Group S at T30 (p=0.01) and T60 (p=0.04). Within each group, the number of red neurons in this layer was significantly greater in T60 than in T0 (p<0.01 for Group S; p<0.05 for Group G). Likewise, the number of red neurons in the internal granular layer was significantly smaller in Group G than in Group S at T30 (p=0.0165) and T60 (p=0.0475). In this layer, no significant variation was observed between the different perfusion times (Table 4).
Table 5 shows mean extent of intracellular edema observed upon histological evaluation of the internal pyramidal layer and the internal granual layer of the cerebral cortex for Group S (saline solution) and Group G (L-alanyl-L-glutamine) before (T0) and after 30 minutes (T30) and 60 minutes (T60) of reperfusion. No intergroup or intragroup differences were observed for the internal pyramidal layer. However, at T30 edema in the internal granular layer was significantly less severe in Group G than in Group S (p=0.0225).
Discussion
Gerbils are a widely employed model for inducing selective ischemia partly due to the absence of a cerebral arterial circle. In fact, in this species, the anterior and posterior vascular beds are not integrated by posterior communicating arteries. Since the absence of blood flow in the CCAs is not compensated by supply from the vertebral arteries, occlusion of these arteries induces extensive ischemia. Studies conducted by Somova et al.6 on cerebral infarction induced by clamping of the left CCA in gerbils have shown that the model is appropriate for evaluating drug efficacy and for recreating the circumstances surrounding ischemic cerebral vascular accidents in humans.
Blood and tissue glucose levels were significantly increased in Group G at T0 (p=0.0375) (Table 1). It is possible that in response to recent cerebral ischemic injury a greater amount of glucose (the primary source of energy in the human brain) was made available from liver and muscle reserves through glycogenolysis19. On the other hand, the observed decrease in glucose concentrations in Group G at T30 (p=0.0132) and T60 (p=0.0410) (Table 1) may be explained by a greater consumption of glucose (increased glycolysis) in peripheral tissues. The groups did not differ significantly with regard to tissue glucose levels. These findings support the results of a study by Yang et al.27 in which gerbils were submitted to focal ischemia by occlusion of the right CCA and middle right cerebral artery for 60 minutes with microdialysis probes inserted on each side of the cerebral cortex, resulting in decreased glucose and pyruvate concentrations and increased lactate and glutamate concentrations in the ipsilateral cortex.
In our study, the fact that blood lactate concentrations were lower in Group G than in Group S at T0 (p=0.0321), T30 (p=0.0490) and T60 (p=0.0290) (Table 1) suggests that Ala-Gln may have a protective effect on cerebral ischemia-reperfusion injury. The mechanism proposed to explain this feature involves activation of the malate-aspartate shuttle and increased conversion of pyruvate into acetyl-CoA in detriment to lactate, with prevalence of aerobic glycolysis28 In Group G, tissue lactate concentrations were also reduced at T0 (p=0.0228), confirming the prevalence of aerobic glycolytic activity.
MDA levels were not significantly different at T0, (Group G vs. Group S) indicating that Ala-Gln did not protect the cerebral tissue of gerbils against lipoperoxidation during ischemia, in this experiment. However, MDA levels decreased significantly on L-Ala-Gln group compared with control (p<0.05) at 30 minutes (Figure 1). Studies on cerebral ischemia-reperfusion injury in gerbil models have shown significant increases in MDA levels in brain tissue, especially the hippocampus, 3-6 hours after induction of ischemia. One to three days after occlusion, levels returned to baseline29. In our study, the significant decrease of MDA levels in Ala-Gln treated animals 30 minutes after the onset of reperfusion may be considered an isolated finding, not allowing definitive conclusions of the possible protective effect of ALA-Gln against lipid peroxidation in brain tissue.
It is known that GSH, a tripeptide relatively abundant in the brain exerts a protective action, promoting the reduction of free radicals and oxidative metabolites30. In the present study, GSH was significantly higher in Group G than in Group S at T0 (p=0.0006), T30 (p<0.0001) and T60 (p=0.0011) (Figure 2). These findings show that pre-treatment with Ala has a protective effect on cerebral ischemia / reperfusion in a rodent model.
A marker of activated neutrophils, MPO is commonly used to evaluate acute inflammatory response in experimental models. In this study, no significant intergroup differences were observed in cerebral tissue with regard to MPO. Thus, based on the similar levels of MPO observed in Group S and Group G, it may be concluded that Ala-Gln provides no anti-inflammatory protection against cerebral ischemia-reperfusion injury in gerbils (Figure 3).
The histological examination revealed a significant reduction in pyknosis in Group G at T30 (p=0.0086) and T60 (p=0.0379) and in red neurons in the internal pyramidal layer of the cerebral cortex at T30 (p=0.0159) and T60 (p=0.0493), when compared to Group S (Table 3 and 4). As for the internal granular layer, reductions were observed in pyknosis at T30 (p=0.0208) and T60 (p=0.0412) and in red neurons at T30 (p=0.0208) and T60 (p=0.0412) (Tables 3 and 4).
Intracellular edema in the internal granular layer of the cerebral cortex at T30 was significantly smaller in Group G than in Group S (p=0.0225) (Table 5). Ischemic cerebral edema starts minutes after trauma and peaks on the third or fourth day. It results from tissue necrosis and extensive cell membrane disruption due to fluid transudation from the vessels to the cerebral tissue. In a study by Tomimoto and Yanagihara31 in which the CCAs of gerbils were occluded bilaterally for 5 minutes, edema of the dendrites was observed along with mitochondrial swelling, cytoplasmatic vacuolization of microtubules and disintegration of layer I (spread to layer III after ischemia for 20 minutes of ischemia).
Analyzing all results it can be inferred that additional studies are needed, using different times of ischemia and reperfusion to validate, in a definitive manner, the protective effect of preconditioning with GLN in gerbil cerebral ischemia / reperfusion injuries.
Conclusion
Preconditioning with L-Ala-Gln in gerbils submitted to cerebral ischemia/reperfusion reduces oxidative stress and degeneration of the nucleus (pyknosis) and cell death (red neurons) in the cerebral tissue.
Conflict of interest: none
Financial source: CNPq
1 Research performed at Experimental Surgery Laboratory (LABCEX), Faculty of Medicine, Federal University of Ceara (UFC), Fortaleza-CE, Brazil.
- 1. Lipton P. Ischemic cell death in brain neurons. Physiol Rev. 1999;79(4):1431-568.
- 2. Dirnagl U, Iadecola C, Moskowitz MA. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci. 1999; 22(9):391-7.
- 3. Levine S, Payan H. Effects of ischemia and other procedures on the brain and retina of the gerbil (Meriones unguiculatus). Exp Neurol. 1966; 16(3):255-62.
- 4. Levine S, Shon D. Cerebral ischemia in infant and adult gerbils. Arch Pathol. 1969;87(3):315-7.
- 5. Harrison MJ, Brownbill D, Lewis PD, Russell RW. Cerebral edema following carotid artery ligation in gerbil. Arch Neurol. 1973;28(6):389-91.
- 6. Somova LI, Gregory MA, Nxumalo EN. Mongolian gerbil (Meriones unguiculatus) as a model of cerebral infarction for testing new therapeutic agents. Methods Find Exp Clin Pharmacol. 2000;22(4):203-10.
- 7. Moro MA, Almeida A, Bolaños JP, Lizasoain I. Mitochondrial respiratory chain and free radical generation in stroke. Free Radic Biol Med. 2005;39(10):1291-304.
- 8. Fatokun AA, Stone TW, Smith RA. Adenosine receptor ligands protect against a combination of apoptotic and necrotic cell death in cerebellar granule neurons. Exp Brain Res. 2008;186(1):151-60.
- 9. Garcia JH. Experimental ischemic stroke: a review. Stroke. 1984;15(1):5-14.
- 10. McKenna MC, Sonnewald U, Huang X, Stevenson J, Zielke HR. Exogenous glutamate concentration regulates the metabolic fate of glutamate in astrocytes. J Neurochem. 1996;66:386-93.
- 11. Lacey JM, Crouch JB, Benfell K, Ringer SA, Wilmore CK, Maguire D, Wilmore DW. The effects of glutamine-supplemented parenteral nutrition in premature infants. JPEN J Parenter Enteral Nutr. 1996;20(1):74-80.
- 12. Bergström J, Fürst P, Norée LO, Vinnars E. Intracellular free amino acid concentration in human muscle tissue. J Appl Physiol. 1974;36(6):693-7.
- 13. Albers S, Wernerman J, Stehle P, Vinnars E, Fürst P. Availability of amino acid supplied by constant intravenous infusion of synthetic dipeptide in healthy man. Clin Sci. 1989;76(6):643-8.
- 14. Jia CJ, Dai CL, Zhang X, Cui K, Xu F, Xu YQ. Alanylglutamine dipeptide inhibits hepatic ischemia-reperfusion injury in rats. World J Gastroenterol 2006;12:1373-8.
- 15. Schauer RJ, Kalmuk S, Gerbes AL, Leiderer R, Meissner H, Schildberg FW, Messmer K, Bilzer M. Intravenous administration of glutathione protects parenchymal and non-parenchymal liver cells against reperfusion injury following rat liver transplantation. World J Gastroenterol 2004;10:864-70.
- 16. Armeni T, Ghiselli R, Balercia G, Goffi L, Jassem W, Saba V, Principato G. Glutathione and ultrastructural changes in inflow occlusion of rat liver. J Surg Res. 2000;88:207-14.
- 17. Zhang WX, Zhou LF, Zhang L, Bao L, Wang CC, Meng HY, Yin W. Protective effects of glutamine preconditioning on ischemia-reperfusion injury in rats. Hepatobiliary Pancreat Dis Int. 2011;10(1):78-82.
- 18. Weinberger J, Cohen G. The differential effect of ischemia on the active uptake of dopamine, gamma-aminobutyric acid, and glutamate by brain synaptosomes. J Neurochem. 1982;38(4):963-8.
- 19. Muniz LRF, Faria MHG, Vasconcelos PRL. Avaliação metabólica das lesões de isquemia e reperfusão cerebrais após oclusão bilateral das artérias carótidas comuns: estudo experimental em ratos. Acta Cir Bras. 2004;19(5):529-34.
- 20. Greca FH, Gonçalves NMFM, Souza ZAF, Silva APG, Mima WH, Mima HH. O papel do azul de metileno como um protetor de pulmão após isquemia e reperfusão intestinal. Acta Cir Bras. 1994;19(4):431-40.
- 21. Slein MW. Determination with hexokinase and glucose-6-phosphate dehydrogenase. In: Methods of enzymatic analysis. London: Weinheim/Academic Press; 1963. p.117-23.
- 22. Hohorst HJD. Glucose-6-phosphate and D-fructose-6-phosphate. Determination with glucose-6-phosphate dehydrogenase and phosphoglucose isomerase. In: Bergmeyer HU. Methods of enzymatic analysis. London: Weinheim/Academic Press; 1963. p.134-8.
- 23. Uchiyama M, Mihara M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem. 1978;86(1):271-8.
- 24. Sedlak J, Lindsay RH. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem. 1968;25(1):192-205.
- 25. Bradley PP, Christensen RD, Rothstein G. Celullar and extracellular mieloperoxidase in pyogenic inflammation. Blood. 1982;60(3):618-22.
- 26. Bradley JR, Thiru S, Pober JS. Disparate localization of 55-kd and 75-kd tumor necrosis factor receptors in human endothelial cells. Am J Pathol.1995;146(1):27-32.
- 27. Yang DY, Tsai TH, Cheng CH, Lee CW, Chen SH, Cheng FC. Simultaneous monitoring of extracellular glucose, pyruvate, lactate and glutamate in gerbil cortex during focal cerebral ischemia by dual probe microdialysis. J Chromatogr A. 2001;913(1-2):349-54.
- 28. Alves WF, Guimarães SB, Vasconcelos PRC, Vasconcelos PRL. Repercussão da L-alanil- glutamina sobre as concentrações de lactato e lactato desidrogenase (LDH) em pacientes com isquemia crítica dos membros inferiores submetidos a revascularização distal. Acta Cir Bras. 2003;18(3):209-15.
- 29. Mollace V, Iannone M, Muscoli C, Palma E, Granato T, Modesti A, Nisticò R, Rotiroti D, Salvemini D. The protective effect of M40401, a superoxide dismutase mimetic, on post-ischemic brain damage in Mongolian gerbils. BMC Pharmacol. 2003;3:8.
- 30. Gupta A, Gupta A, Datta M, Shukla GS. Cerebral antioxidant status and free radical generation following glutathione depletion and subsequent recovery. Mol Cell Biochem. 2000;209(1-2):55-61.
- 31. Tomimoto H, Yanagihara T. Electron microscopic investigation of the cerebral cortex after cerebral ischemia and reperfusion in the gerbil. Brain Res. 1992;598(1-2):87-97.
Publication Dates
-
Publication in this collection
23 Sept 2011 -
Date of issue
2011