Abstracts
PURPOSE: To develop an easily reproducible model of acute lung injury due to experimental muscle trauma in healthy rats. METHODS: Eighteen adult Wistar rats were randomized in 3 groups (n=6): G-1- control, G-2 - saline+trauma and G-3 - dexamethasone+trauma. Groups G-1 and G-2 were treated with saline 2,0ml i.p; G-3 rats were treated with dexamethasone (DE) (2 mg/kg body weight i.p.). Saline and DE were applied 2h before trauma and 12h later. Trauma was induced in G-2 and G-3 anesthetized (tribromoethanol 97% 100 ml/kg i.p.) rats by sharp section of anterior thigh muscles just above the knee, preserving major vessels and nerves. Tissue samples (lung) were collected for myeloperoxidase (MPO) assay and histopathological evaluation. RESULTS: Twenty-four hours after muscle injury there was a significant increase in lung neutrophil infiltration, myeloperoxidase activity and edema, all reversed by dexamethasone in G-3. CONCLUSION: Trauma by severance of thigh muscles in healthy rats is a simple and efficient model to induce distant lung lesions.
Models; Muscle; Acute Lung Injury; Dexamethasone; Peroxidase; Rats
OBJETIVO: Desenvolver um modelo facilmente reprodutível de lesão pulmonar aguda decorrente de trauma muscular experimental em ratos sadios. MÉTODOS: Dezoito ratos Wistar adultos foram randomizados em 3 grupos (n=6): G-1-controle, G-2 - trauma+salina e G-3 - trauma+dexametasona. Grupos G-1 e G-2 foram tratados com salina 2,0 ml ip, G-3 ratos foram tratados com dexametasona (DE) (2 mg/kg peso corporal ip). Salina e DE foram aplicadas 2h antes e 12h depois do trauma. Trauma foi induzido em ratos G-2 e G-3 anestesiados (tribromoetanol 97% de 100 ml/kg, i.p.) por secção da musculatura anterior da coxa logo acima da articulação do joelho, preservando os grandes vasos e nervos. Amostras de tecido (pulmão) foram coletadas para avaliação da mieloperoxidase (MPO), e exames histopatológicos. RESULTADOS: Vinte e quatro horas após a indução da lesão muscular houve um aumento significativo na infiltração de neutrófilos pulmonares, atividade da mieloperoxidase e edema, todos revertidos por dexametasona, no G-3. CONCLUSÃO: O trauma decorrente da secção dos músculos da coxa em ratos saudáveis é um modelo simples e eficaz para induzir lesões pulmonares à distância.
Modelos Animais; Músculo Esquelético; Lesão Pulmonar Aguda; Dexametasona; Peroxidase; Ratos
9 - ORIGINAL ARTICLE
MODELS, BIOLOGICAL
Acute pulmonary injury induced by experimental muscle trauma1 Correspondence: Paulo Roberto Leitão de Vasconcelos Rua Professor Costa Mendes, 1608/3º andar, Bloco Didático 60430-140 Fortaleza - CE Brasil Tel.: (55 85)3366-808 Fax: (55 85)3366-8064 paulo.vasconcelos@ufc.br
Lesão pulmonar aguda induzida por trauma muscular experimental
Márcia Andréa da Silva Carvalho SombraI; Marcelo Pinho Pessoa de VasconcelosII; Sergio Botelho GuimarãesIII; Rodrigo Dornfeld EscalanteIV; José Huygens Parente GarciaV; Paulo Roberto Leitão de VasconcelosVI
IFellow PhD Degree, Department of Surgery, Postgraduate Program, UFC, Ceara, Brazil. Technical procedures, acquisition and interpretation of data. The article is part of a doctorate degree thesis
IIGraduate student, UFC, Ceara, Brazil. Helped with technical procedures, acquisition of data
IIIPhD, Associate Professor, Department of Surgery, Head, LABCEX, UFC, Ceara, Brazil. Manuscript writing, statistical analysis, graphics design
IVPhD, Associate Professor, Faculty of Medicine, University de Fortaleza (UNIFOR). Critical revision and analysis of data
VPhD, Associate Professor, Head, Department of Surgery, UFC, Ceara, Brazil. Critical revision and analysis of data
VIPhD, Associate Professor, Coordinator, Postgraduate Program, Department of Surgery, UFC, Ceara, Brazil. Tutor, responsible for conception, design, intellectual and scientific content of the study, critical analysis, final approval of manuscript
Correspondence Correspondence: Paulo Roberto Leitão de Vasconcelos Rua Professor Costa Mendes, 1608/3º andar, Bloco Didático 60430-140 Fortaleza - CE Brasil Tel.: (55 85)3366-808 Fax: (55 85)3366-8064 paulo.vasconcelos@ufc.br
ABSTRACT
PURPOSE: To develop an easily reproducible model of acute lung injury due to experimental muscle trauma in healthy rats.
METHODS: Eighteen adult Wistar rats were randomized in 3 groups (n=6): G-1- control, G-2 - saline+trauma and G-3 - dexamethasone+trauma. Groups G-1 and G-2 were treated with saline 2,0ml i.p; G-3 rats were treated with dexamethasone (DE) (2 mg/kg body weight i.p.). Saline and DE were applied 2h before trauma and 12h later. Trauma was induced in G-2 and G-3 anesthetized (tribromoethanol 97% 100 ml/kg i.p.) rats by sharp section of anterior thigh muscles just above the knee, preserving major vessels and nerves. Tissue samples (lung) were collected for myeloperoxidase (MPO) assay and histopathological evaluation.
RESULTS: Twenty-four hours after muscle injury there was a significant increase in lung neutrophil infiltration, myeloperoxidase activity and edema, all reversed by dexamethasone in G-3.
CONCLUSION: Trauma by severance of thigh muscles in healthy rats is a simple and efficient model to induce distant lung lesions.
Keywords: Models, Animal. Muscle, Skeletal. Acute Lung Injury. Dexamethasone. Peroxidase. Rats.
RESUMO
OBJETIVO: Desenvolver um modelo facilmente reprodutível de lesão pulmonar aguda decorrente de trauma muscular experimental em ratos sadios.
MÉTODOS: Dezoito ratos Wistar adultos foram randomizados em 3 grupos (n=6): G-1-controle, G-2 - trauma+salina e G-3 - trauma+dexametasona. Grupos G-1 e G-2 foram tratados com salina 2,0 ml ip, G-3 ratos foram tratados com dexametasona (DE) (2 mg/kg peso corporal ip). Salina e DE foram aplicadas 2h antes e 12h depois do trauma. Trauma foi induzido em ratos G-2 e G-3 anestesiados (tribromoetanol 97% de 100 ml/kg, i.p.) por secção da musculatura anterior da coxa logo acima da articulação do joelho, preservando os grandes vasos e nervos. Amostras de tecido (pulmão) foram coletadas para avaliação da mieloperoxidase (MPO), e exames histopatológicos.
RESULTADOS: Vinte e quatro horas após a indução da lesão muscular houve um aumento significativo na infiltração de neutrófilos pulmonares, atividade da mieloperoxidase e edema, todos revertidos por dexametasona, no G-3.
CONCLUSÃO: O trauma decorrente da secção dos músculos da coxa em ratos saudáveis é um modelo simples e eficaz para induzir lesões pulmonares à distância.
Descritores: Modelos Animais. Músculo Esquelético. Lesão Pulmonar Aguda. Dexametasona. Peroxidase. Ratos.
Introduction
Acute lung injury (ALI) following musculoskeletal trauma is an important cause of morbidity and mortality in trauma patients1.
Animal experiments represent a very useful tool to explore the pathogenesis of different diseases and to evaluate novel treatments. Hypotheses generated in human studies can be tested directly in animal models. Different animal models of experimental lung injury have been used to investigate the mechanisms of lung injury2. Studies in primates are relevant for humans but are beyond the reach of most post-graduate students, not only the difficulty of obtaining these animals but also by the elevated cost of acquisition. On other hand, studies with mice or lower animals are relatively simple and rapid to do, but the relevance for humans is limited2. Most models that study ALI rely on clinical disorders commonly associated with this syndrome in humans, namely sepsis, multiple transfusions, multiple trauma, aspiration of gastric contents, and reperfusion of ischemic tissues3,4. Whereas trauma can trigger the onset of acute lung injury we decided to develop a model using muscle trauma as a causative agent of ALI.
Methods
Approval for experimental use of laboratory animals was obtained on September, 2007 (Protocol #083/07) from the Ethics Committee on Animal Research (CEPA) of the Federal University of Ceara, now Ethics Committee on the Use of Animals (CEUA), in view of the Federal Law No. 11794 of October 8, 2008, http://www.planalto.gov.br/ccivil_03/_Ato2007-2010/2008/Lei/L11794.htm and Decree No. 6689 of July 15, 2009 that regulated the Law 11794, available at: http://www.planalto.gov.br/ccivil_03/_Ato2007-2010/2009/Decreto/D6899.htm.
The study was designed so as to minimize the number of animals required for the experiments. Eighteen adult Wistar rats weighing 250-300g, were randomized in 3 groups (n=6): G-1- control, G-2 - saline+trauma and G-3 - dexamethasone+trauma. Groups G-1 and G-2 were treated with saline 2,0ml i.p; G-3 rats were treated with dexamethasone (DE) (2 mg/kg body weight i.p.). Saline and DE were applied 2h before trauma and 12h later. All Rats were anestethized by tribromoethanol 97% 100 ml/kg i.p., Trauma was induced in anesthetized G-2 and G-3 using sharp section of anterior thigh muscles just above the knee, preserving major vessels and nerves. Tissue samples (lung) were collected for myeloperoxidase (MPO) assay and histopathological evaluation. Dexamethasone dosage (2 mg/kg body weight) was based in a published study5. Dexamethasone and saline were purchased from standard commercial sources and were of the highest quality available.
Surgical procedure
Under adequate anesthesia, a 2 centimeter longitudinal incision of the skin extending into the subcutaneous fat and muscular fascia of the right pelvic limb was carried out in G-2 and G-3 rats. Following careful dissection of the anterior muscles of the thigh, a hemostatic Kelly clamp was passed between the muscles and the femur. Next, the anterior muscles of the right thigh were sectioned with a surgical scalp just above the knee. Care was taken to preserve the major vessels and nerves. There was minimal bleeding which stopped spontaneously. The skin was sutured with nylon 3-0. The mean time of the procedure was 7 minutes. Group G1 served as control; G-1 animals were also anesthetized but no surgical procedure was performed. Twenty-four hours later, all animals were killed by cervical dislocation and fragments of lung tissue were removed, collected and stored in a -80ºC freezer until processed for determination of myeloperoxidase activity and for histopathological evaluation of edema and neutrophilic infiltration by methods described previously6,7.
Statistical methods
Graphpad Prism 5.0 (GraphPad Software, www.graphpad.com) was used for statistical analysis and graphics design. All data were tested for distribution (Kolmorogov-Smirnov test with Dallal-Wilkinson-Lilliefor P value). Results were expressed as mean+SD.All data were tested for distribution by applying the Kolmogorov-Smirnov test. A Chi-square test was used to evaluate the edema. ANOVA with Boferroni post-test was used to evaluate MPO activity and neutrophil infiltration.
Results
Results are presented in tables and graphs. Lung histological findings and values of MPO activation are presented in Table 1.
Neutrophil count increased significantly (p<0,001) in trauma group 24h after muscle sectioning. Values did not increase in dexamethasone treated rats (Figure 1).
MPO activity (MPO/100 mg tissue) increased significantly (p<0,001) in Trauma group 24h after muscle sectioning. Values did not increase in dexamethasone treated rats (Figure 2).
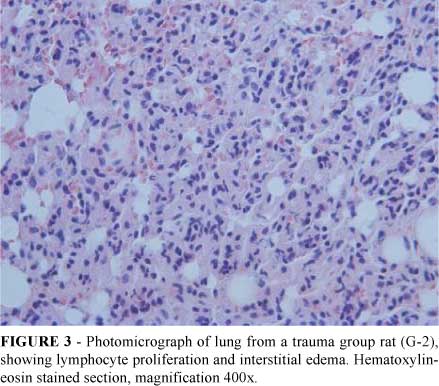
Histological studies showed neutrophil infiltration and edema of lung in rats submitted to trauma (Figure 3, group G-2). This finding was not present in rats treated with dexametasone (Figure 4, G-3).
Discussion
Because corticosteroids may inhibit the action of inflammatory mediators and improve morbidity and mortality in experimental models that induce acute lung injury5,8 dexamethasone was used in this study as a positive control.
In the experimental model described here, muscular injury induced pulmonary lesions similar to those found in acute lung injury within 24 hours, as confirmed by the presence of leukocyte infiltration, neutrophil activation (indirectly measured by myeloperoxidase assay) and pulmonary interstitial edema. It was also demonstrated that these lesions could be reduced by dexamethasone administration. There was a statistically significant increase in lung neutrophil infiltration (Figure 1), myeloperoxidase activity (Figure 2) and interstitial edema intensity (Table 1) in rats submitted to trauma. Histopathological findings legitimized, visually, these findings (Figures 3 and 4).
Lung injuries following musculoskeletal trauma are important causes of morbidity and mortality in trauma patients1. Lung injuries are consequences of systemic inflammation. The main key of this process are pro-inflammatory cytokines and subsequent leucocyte endothelial cell interactions1,9,10. Activation of polymorphonuclear leukocytes, with lung tissue accumulation of capillaries and venules is also a potential source production and release of oxygen free radicals mediating tissue damage10,11.
Conclusion
The present experimental model is quite simple and easily reproducible. Bearing in mind that pulmonary lesions are present within 24 hours after the traumatic event, it can be potentially used to study the early events involved in the systemic inflammatory response following trauma injuries.
Conflict of interest: none
Financial source: CNPq
1 Research performed at the Experimental Surgery Research Laboratory (LABCEX), Department of Surgery, Federal University of Ceara (UFC), Brazil.
- 1. Sgarbi MWM, Silva BAJ, Neto JSH. The importance of systemic inflammatory response (SIRS) in the prognosis of polytraumatized patients. Rev Bras Ortop. 2006;41(1/2):1-6.
- 2. Matule-Bello G, Frevert CW, Martins TR. Animal models of acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2008;295(3):L379-99.
- 3. Pepe PE, Potkin RT, Reus DH, Hudson LD, Carrico CJ. Clinical predictors of the adult respiratory distress syndrome. Am J Surg. 1982;144(1):124-30.
- 4. Hudson LD, Milberg JA, Anardi D, Maunder RJ. Clinical risks for development of the acute respiratory distress syndrome. Am J Respir Crit Care Med. 1995;151(2 Pt 1):293-301.
- 5. Ito A, Miyake M, Morishita M, Ito K, Torii S, Sakamoto T. Dexamethasone reduces lung eosinophilia, and VCAM-1 and ICAM-1 expression induced by Sephadex beads in rats. Eur J Pharmacol. 2003;468(1):59-66.
- 6. Bradley PP, Christensen RD, Rothstein G. Cellular and extracellular myeloperoxidase in pyogenic inflammation. Blood. 1982;60(3):618-22.
- 7. Greca FH, Gonçalves NM, Souza Filho ZA, Silva APG, Mima WH, Mima HH. A influência do azul de metileno na prevenção da lesão pulmonar após isquemia-reperfusão intestinal. Acta Cir Bras. 2004;19(4). Available from URL: http://www.scielo.br/acb
- 8. Fernandes ABS, Zin WAm, Rocco PRM. Corticosteroids in acute respiratory distress syndrome. Braz J Med Biol Res. 2005;38(2):147-59.
- 9. Wang HM, Bodenstein M, Markstaller K. Overview of the pathology of three widely used animal models of acute lung injury. Eur Surg Res. 2008;40(4):305-16.
- 10. Giannoudis PV. Current concepts of the inflammatory response after major trauma: an update. Injury. 2003;34(6):397-404.
- 11. ten Raa S, van den Tol MP, Sluiter W, Hofland LJ, van Eijck CH, Jeekel H. The role of neutrophils and oxygen free radicals in post-operative adhesions. J Surg Res. 2006;136(1):45-52.
Publication Dates
-
Publication in this collection
23 Sept 2011 -
Date of issue
2011