Abstracts
PURPOSE: To study if the pre-radiotherapy physical activity has radio-protective elements, by measuring the radio-induced activation of pro-inflammatory cytokines as interleukin-6 (il-6), transforming growth factor -β (tgf -β), tumor necrosis factor -α (tnf-α) and protein beta kinase β (ikkβ), through western blotting analysis. METHODS: A randomized study with 28 Wistar hannover rats, males, with a mean age of 90 days and weighing about 200 grams. The animals were divided into three groups: (GI, GII and GIII). GIII group were submitted to swimming for eight weeks (zero load, three times a week, about 30 minutes). Then, the groups (except the control group) were submitted to irradiation by cobalt therapy, single dose of 3.5 gray in the whole body. All animals were sacrificed by overdose of pentobarbital, according to the time for analysis of cytokines, and then a fragment of the lower lobe of the right lung went to western blotting analysis. RESULTS: The cytokines IKK β, TNF-α and IL-6 induced by radiation in the lung were lower in the exercised animals. However, exercise did not alter the radiation-induced increase in tgf-β. CONCLUSION: The results show a lower response in relation to inflammatory cytokines in the group that practiced the exercise pre-radiotherapy, showing that exercise can protect tissues from tissue damage due to irradiation.
Radiotherapy; Radiation Pneumonitis; Pulmonary Fibrosis; Physical Therapy Modalities; Rats
OBJETIVO: Verificar se a radioterapia pré-atividade física tem elementos de rádio-proteção, medindo-se a ativação de citocinas pró-inflamatórias como a interleucina-6 (IL-6), fator transformador de crescimento - β (TGF - β), fator de necrose tumoral - α (TNF-α) e quinase de proteína beta β (IKK β), por meio da análise blotting ocidental. MÉTODOS: Um estudo randomizado empregando 28 ratos Wistar Hannover, machos, com idade média de 90 dias e pesando cerca de 200 gramas. Os animais foram divididos em três grupos: (GI, GII e GIII). Os animais do grupo GIII foram submetidos à natação durante oito semanas (carga zero, três vezes por semana, cerca de 30 minutos). Então, os grupos (exceto o grupo controle) foram submetidos à irradiação por cobalto terapia, dose única de 3,5 cinza em todo o corpo. Todos os animais foram sacrificados por overdose de pentobarbital, de acordo com o tempo de análise de citocinas, em seguida, um fragmento do lobo inferior do pulmão direito foi a análise de mata-borrão ocidental. RESULTADOS: As citocinas IKK β, TNF-α e IL-6 induzidas por radiação no pulmão foram menores nos animais que se exercitaram. No entanto, o exercício não alterou o aumento induzido pela radiação na TGF-β. CONCLUSÃO: Os resultados mostraram uma menor resposta em relação às citocinas inflamatórias no grupo que praticou o exercício físico pré-radioterapia, evidenciando que o exercício pode proteger os tecidos das lesões teciduais decorrentes da irradiação.
Radioterapia; Pneumonite por Radiação; Fibrose Pulmonar; Modalidades de Fisioterapia; Ratos
4 - ORIGINAL ARTICLE
MODELS, BIOLOGICAL
Analysis of the physical activity effects and measurement of pro-inflammatory cytokines in irradiated lungs in rats1 Correspondence: Renata Cristiane Gennari Bianchi Curso de Pós-Graduação em Cirurgia - UNICAMP Cidade Universitária "Zeferino Vaz" Distrito de Barão Geraldo 13083-970 Campinas - SP - Brasil Tel.: (55 19)3521-8863 renatagennari@gmail.com
Análise dos efeitos da atividade física e mensuração de citocinas pró-inflamatórias em pulmões irradiados em ratos
Renata Cristiane Gennari BianchiI; Eduardo Rochete RopelleII; Carlos Kiyoshi KatashimaIII; José Barreto Campello CarvalheiraIV; Luiz Roberto LopesV; Nelson Adami AndreolloVI
IFellow PhD degree in Sciences of Surgery, Faculty of Medical Sciences, UNICAMP, Campinas-SP, Brazil. Technical procedures, acquisition and interpretation of data
IIAssociate Professor, School of Applied Sciences, University of Campinas-UNICAMP, Limeira-SP, Brazil. Technical procedures and interpretation of data
IIIFellow PhD degree in Medical Physiopathology, Faculty of Medical Sciences, UNICAMP, Campinas-SP, Brazil. Technical procedures and interpretation of data
IVAssociate Professor, Department of Internal Medicine, UNICAMP, Campinas-SP, Brazil. Technical procedures and interpretation of data
VAssociate Professor, Department of Surgery, UNICAMP, Campinas-SP, Brazil. Tutor, responsible for conception, design, intellectual and scientific content of the study, critical analysis, final approval of manuscript
VIFull Professor, Department of Surgery, UNICAMP, Campinas-SP, Brazil. Critical revision and analysis of data
Correspondence Correspondence: Renata Cristiane Gennari Bianchi Curso de Pós-Graduação em Cirurgia - UNICAMP Cidade Universitária "Zeferino Vaz" Distrito de Barão Geraldo 13083-970 Campinas - SP - Brasil Tel.: (55 19)3521-8863 renatagennari@gmail.com
ABSTRACT
PURPOSE: To study if the pre-radiotherapy physical activity has radio-protective elements, by measuring the radio-induced activation of pro-inflammatory cytokines as interleukin-6 (il-6), transforming growth factor -β (tgf -β), tumor necrosis factor -α (tnf-α) and protein beta kinase β (ikkβ), through western blotting analysis.
METHODS: A randomized study with 28 Wistar hannover rats, males, with a mean age of 90 days and weighing about 200 grams. The animals were divided into three groups: (GI, GII and GIII). GIII group were submitted to swimming for eight weeks (zero load, three times a week, about 30 minutes). Then, the groups (except the control group) were submitted to irradiation by cobalt therapy, single dose of 3.5 gray in the whole body. All animals were sacrificed by overdose of pentobarbital, according to the time for analysis of cytokines, and then a fragment of the lower lobe of the right lung went to western blotting analysis.
RESULTS: The cytokines IKK β, TNF-α and IL-6 induced by radiation in the lung were lower in the exercised animals. However, exercise did not alter the radiation-induced increase in tgf-β.
CONCLUSION: The results show a lower response in relation to inflammatory cytokines in the group that practiced the exercise pre-radiotherapy, showing that exercise can protect tissues from tissue damage due to irradiation.
Key words: Radiotherapy. Radiation Pneumonitis. Pulmonary Fibrosis. Physical Therapy Modalities. Rats.
RESUMO
OBJETIVO: Verificar se a radioterapia pré-atividade física tem elementos de rádio-proteção, medindo-se a ativação de citocinas pró-inflamatórias como a interleucina-6 (IL-6), fator transformador de crescimento - β (TGF - β), fator de necrose tumoral - α (TNF-α) e quinase de proteína beta β (IKK β), por meio da análise blotting ocidental.
MÉTODOS: Um estudo randomizado empregando 28 ratos Wistar Hannover, machos, com idade média de 90 dias e pesando cerca de 200 gramas. Os animais foram divididos em três grupos: (GI, GII e GIII). Os animais do grupo GIII foram submetidos à natação durante oito semanas (carga zero, três vezes por semana, cerca de 30 minutos). Então, os grupos (exceto o grupo controle) foram submetidos à irradiação por cobalto terapia, dose única de 3,5 cinza em todo o corpo. Todos os animais foram sacrificados por overdose de pentobarbital, de acordo com o tempo de análise de citocinas, em seguida, um fragmento do lobo inferior do pulmão direito foi a análise de mata-borrão ocidental.
RESULTADOS: As citocinas IKK β, TNF-α e IL-6 induzidas por radiação no pulmão foram menores nos animais que se exercitaram. No entanto, o exercício não alterou o aumento induzido pela radiação na TGF-β.
CONCLUSÃO: Os resultados mostraram uma menor resposta em relação às citocinas inflamatórias no grupo que praticou o exercício físico pré-radioterapia, evidenciando que o exercício pode proteger os tecidos das lesões teciduais decorrentes da irradiação.
Descritores: Radioterapia. Pneumonite por Radiação. Fibrose Pulmonar. Modalidades de Fisioterapia. Ratos.
Introduction
Esophageal cancer still remains a health threat. This type of cancer has had a special attention in the last three decades because of the changing of the epidemiological profile and increase of the treatment options. Worldwide, the esophagus cancer is the sixth leading cause of cancer death, and the survival time employing surgical and oncological treatments is very low in five years1,2.
Recently, the therapeutics for such subjects has been well studied, including the neoadjuvant therapies3. Esophagectomy was the only and the best option for the esophageal cancer for decades4. In order to improve the prognosis of esophageal cancer, multimodality therapy including surgery, radiotherapy and chemotherapy, has been introduced3,4. Advances in radiotherapy technique in particular, according to Liao et al.1, allow to reach more precisely the tumor volume and preserve the normal tissue, with a consequent improvement of the results presented. Radiation pneumonitis is a major complication in this type of cancer in subjects treated with a definitive radiochemotherapy5. In addition, other malignant tumors are inoperable and the radiotherapy is the most important treatment modality, such as, for instance, for the non-small cells lung cancer (NSCLC) (advanced, located and non-operable)6. The lung is the organ with a higher limit dose for cancer within the radiotherapy of the chest area7.
As the lung is a very radiosensitive organ, the radiotherapy for both esophagus and the lung itself can lead to serious complications over 50% of the subjects. The sequel typically occurs in two phases: acute inflammatory pneumonitis and later pulmonary fibrosis8. Inflammatory cytokines are the key to improve and optimize the effects of pneumonitis caused by pulmonary radiation7. The clinical symptoms of this pneumonitis include dispnea, cough, fever and even death from respiratory failure. These symptoms occur about one month after the completion of radiotherapy6.
The pro-inflammatory cytokines as interleukin-6 (IL-6), transforming growth factor β1 (TGF-β1) and tumor necrosis factor α (TNF-α) perform a very important role, both directly and indirectly, in the pulmonary injury induced by radiation and the consequently process of fibrosis9. Schmid et al.10 demonstrated that the beta kinase protein (IKK) also plays a role in the inflammation and changes in activity and/or regulation of IKKβ are found in many diseases associated to the acute or chronic inflammation.
Physical exercise reduces the deleterious effects of cardiovascular and inflammatory disorders. Currently, the healthy lifestyle has been associated with the regular practice of a physical activity. Evidence shows that this practice causes the individual to have more longevity, in addition to decrease the morbidity and mortality grade. Therefore, evidence has shown that a regular physical activity exerts protective effects against diseases associated to systemic inflammation11.
Exercises that induce the muscle cell injury cause a sequence of pro-inflammatory cytokines release, such as: TNF-α, IL-1β E IL-6, followed very little by anti-inflammatory cytokines, such as IL-10 and IL-111. TNF-α and IL-1β stimulate the production of IL-6, which induces the acute phase of the inflammatory response11-13.
The objective of this study is to associate the aerobic physical activity of Wistar rats to the possible increase in cardiopulmonary fitness, in order to verify the potential decrease of post-radiation pulmonary injury.
Methods
After the approval by the Ethics Committee on Animal Experiments, from the Biology Institute (UNICAMP - Protocol number 1349-1), the experimental study included 28 Wistar-Hannover rats, C3H/HeJ, male, with a mean age of 90 days and weighing about 200 grams. The animals origined from the Multidisciplinary Laboratory Animals (CEMIB - UNICAMP) were kept in the Bioterium of the Associated Colleges of Education (UNIFAE - Sao Joao da Boa Vista, Sao Paulo, Brazil) in a number of 5 per cage, on sawdust bedding type, with filtered and chlorinated water ad libitum, receiving an industrial food (Biobase®, Santa Catarina, Brazil), three times a day.
The animals were randomized in three groups, with the first group being the control (GI) with 4 animals.
The GII group was subdivided into subgroup GIIa, which was subjected to irradiation and was sacrificed on day 1 after the radiotherapy (6 animals) and subgroup GIIb, which was subjected to irradiation and was sacrificed on day 7 after the radiotherapy (six animals).
The GIII group was subdivided into subgroup GIIIa, who performed an aerobic physical activity prior to irradiation and was sacrificed on day 1 after the radiotherapy (six animals) and subgroup GIIIb, who performed an aerobic physical activity prior to irradiation and was sacrificed on day 7 after the radiotherapy (six animals).
Physical activity
The animals of GIII were submitted to swimming for 8 weeks, divided into 3 sessions each, zero load, lasting 30 minutes each session in the first week, progressing to 2 minutes each week, and the last one reached 44 minutes. The animals remained in glass tanks, with 120 x 60 x 120 centimeters (cm) - length, width and height, respectively - in size, and containing water supply of from Bioterium of the Associated Colleges of Education (UNIFAE), at a room temperature.
Irradiation
After the physical training, the animals of group GIII and GII, were submitted to the gamma radiation in a telecobalt therapy device, from the brand Alcyon II®, with an energy of 1.25 MeV (1 million of electrons-volts), having a source of 60CO with a yield of 67.77 cGy/min (centigray by minute), in a field of 10 x 10 cm, at a distance of 80 cm from the source, used an isocenter radiation. At the time of the exposition to the radiation, the animals were placed in wood boxes of 10 x 10 x 10 cm (height, length and deep, respectively), with a lid also in wood and internal divisions that make possible the immobilization of them and, therefore, with no need to anesthesia. They were radiated with the dosage of 3.5 Gy (Gray) in the whole body, calculated in the whole body volume, with the technique of opposed parallel, in a field of 10 x 10 cm, with a source-surface distance of 75 cm and source-point distance of calculation of 80 cm.
Western blotting
After the irradiation, all animals were sacrificed by overdose of pentobarbital, according to the time for analysis of cytokines: within 1 day (GI, n = 2; GIIa, n = 6 and GIIIa, n = 6) or 7 days (GI, n = 2; GIIb, n = 6 and GIIIb, n = 6) and the lungs were removed. A fragment of the lower right lung lobes of all animals were removed and were preserved by cooling to -80°C for immunoblotting analysis of their respective pro-inflammatory cytokines: IL-6, TGF-β and TNF-α and IKKβ.
Extraction of tissue and proteic analysis by immunoblotting:
The lung fragments from the rats with and without radiotherapy were homogenized in extraction buffer, with a polytron PTA 20S (Brinkmann Instruments model PT 10/35). This buffer is composed of Triton-X 100 1%, TRIS (PH 7.4) 100 mM, sodium pyrophosphate 100mM, sodium fluoride 100mM, EDTA 10mM, sodium orthovanadate 10mM, PMSF 2mM and aprotinin 0.1 mg/ml, at 4°C.
The samples were subjected to centrifugation (12,000 rpm at 4°C for 20 minutes) to remove the insoluble material. The protein concentration was determined using a spectrophotometer reading. After that, the samples received a buffer having DTT 100mM and heated in boiling water for 5 minutes and were subjected to polyacrylamide gel electrophoresis (SDS-PAGE 6.5%), with the gel being marked by a high-molecular weight marker from Bio Rad® (California, USA), containing: myosin (206 kDa), ß-galactosidase (116 kDa), bovine serum albumin (80 kDa) and ovalbumin (49.5 kDa). Electrophoresis was performed in a tank of Bio Rad® minigel, with a buffer solution for the electrophoresis previously diluted. The SDS-PAGE always subjected to 20 volts initially up to the passage through the stacking phase and 100 volts until the end of the resolving gel.
The transfer of separated proteins in the gel was electrically done to a nitrocellulose membrane, through a device also from Bio Rad® for 90 minutes at 120 volts. The membranes with transferred proteins were incubated in a blocking solution (skimmed milk Molico® - Sao Paulo, Brazil) 5%, Tris 10mM, NaCl 150mM and Tween 20 0.02%) for two hours at room temperature. After washed in a baseline solution, these membranes were then incubated with specific antibodies. Kept at 4°C overnight under a continuous stirring, the membranes were again washed with the baseline solution. Then, the membranes were incubated with 60 μl of protein A labeled with 125I for two hours at room temperature and washed again.
The protein A labeled with radioactive iodine binds to antibodies and can be detected and viewed in autoradiographs using intensifiers, and kept at -80°C for 12 to 48 hours. Once identified, the bands of autoradiographs proceeded to the reading by optical densitometry. Since then, it was executed the analysis of data, by comparing the control animal tissue to the other under experiment, on such a way that there always were an intra-experiment control.
Statistical analysis
All numeric data were expressed as mean + SEM of the indicated number of experiments, except the control data (control group). The results of the blots were expressed as direct comparisons of bands or spots in autoradiographs and quantified by optical densitometry (Scion Image). The statistical analysis was performed through the ANOVA test with Bonferroni post test, and the level of significance was pre-fixed at p<0.05.
Results
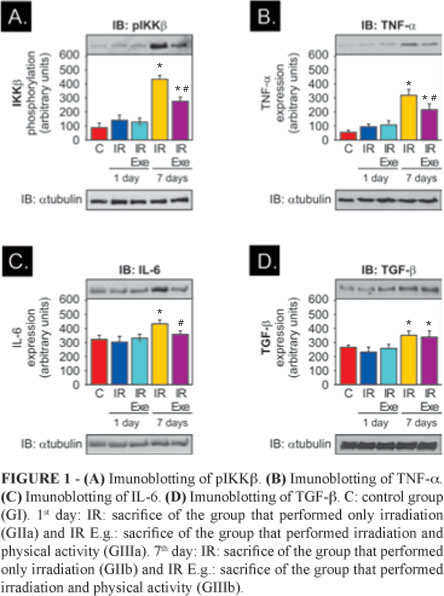
For all cytokines studied, no significant difference occurred between the groups GIIa and GIIIa, i.e., in the first day post-irradiation, in relation to the group GI (control).
Regarding IKKβ, there was a greater expression of such protein in GIIb, followed by GIIIb. There were a statistically significance (p<0.05) of GIIb in relation to GI, as well as the GIIIb in relation to GI and also of GIIIb in relation to GIIb. This means that the IKKβ showed a greater response in the 7th day post-irradiation, regardless the physical activity or otherwise, in relation to the 1st day. However, the increase in IKKβ in the 7th day post-irradiation in Group IIIb was significantly lower in relation to the Group IIb.
The expression of TNF-α showed the same pattern of cytokine IKKβ, i.e., the expression of TNF-α in the GIIb and GIIIb had similar statistical significance both in relation to the GI and in the GIIb.
There was a statistical significance in relation to IL-6 (p<0.05) of GIIb compared to GI, as well as GIIIb compared to GIIb. It means that there was a greater expression statistically significant of IL-6 in the 7th day post-irradiation, in addition to the group that only performed irradiation compared to the control group, as the group in the 7th day that performed irradiation + physical activity in relation to group of the 7th day post-irradiation only.
Regarding TGF-β, there was a statistical significance of GIIb and GIIIb in relation to GI, but there was no statistical significance between the groups GIIb and GIIIb, i.e., there was no difference between them as there was to the other proteins.
The records of the tissue expression of cytokines studied can be seen in Figure 1.
Discussion
Triboulet and Mariette14 emphasized that patients with advanced esophageal carcinoma should be given the combination therapy in order to extend the survival time. Hermann et al.15 concluded that neoadjuvant radiochemotherapy may resolve the clinical stage of the tumor and increase the rate of resection. Other studies showed that the radiochemotherapy improves the quality of life postoperative and extend the survival time, but also increases the risk of complication and mortality in the medium term16.
This study examined the expression of some pro-inflammatory cytokines in the pulmonary tissue, such as IL-6, TNF-α, TGF-β and IKK in the Wistar rat lungs after the aerobic physical training in a group, followed by a single dose irradiation of the whole body, with a subsequent sacrifice at 1 and 7 days, respectively.
The elevation of pro-inflammatories cytokines TNF-α and IL-6 showed the same pattern in order to irradiation and physical exercise, in the animals sacrificed at the 7th day post-irradiation, as well as IKKβ. Such expression increased was lower in all cytokines, including TGF-β, in relation to the groups that also performed physical activity in the 7th day post-irradiation. IKKβ showed the same behavior. Rübe et al.17, in similar studies on the exposure of mice to 12 Gy irradiation, showed a significant increase in radiation-induced TNF-α in the 1th day post-irradiation, in contrast to our study. However, must be considered the high dose of irradiation applied to animals, which had about 20 grams of weight.
The results of increased IL-6 suggests a correlation between the development of pneumonitis after irradiation in lung inflammation as it becomes increasingly pronounced in a few days after lung injury, since that IL-6 is synthesized by a variety of cells in the lung parenchyma, including alveolar macrophages, type II pneumocytes, T lymphocytes and lung fibroblasts18. Despite the post-irradiation pneumonitis, Chen et al.19 demonstrated the exclusion of much evidence contained in several studies of such lung injury, reported between 45 and 75% of cases.
Sriwijitkamol et al.20 related that acute physical activity in experiments with humans was efficient in blocking via IKK/NFkappaβ. In addition, this lock prevents NFkappaβ start the transcription of proinflammatory proteins such as TNF-α. In our study a similar result in relation to the IKK in the 7th day post-irradiation, with increasing evidence of its lower expression, when performed physical activity, even if it is chronic.
Increased expression of TGF-β was only statistically significant groups of 7th day post-irradiation compared the control group. This may be explained by the fact that TGF-β participate in the process of fibrosis, which occurs at a later stage (beyond seven days) tissue repair. The predictive value of TGF-β1 in pulmonary toxicity was first reported by Anscher et al.21 in patients with advanced breast cancer treated with high doses of chemotherapy and the bone marrow transplant. Fu et al.22 also concluded that elevated plasma levels of TGF-β1 to the end of radiation therapy in humans is an independent risk factor for lung injury.
There was no statistically significant difference in the expression of cytokines measured in the study on the 1st day post-irradiation. This fact may be justified due to the short time for the change of the expression of such proteins, which become changed quantitatively in a period greater than 24 hours, mainly TGF-β, in addition to the irradiation dose not being significantly high. These findings are similar to those of Mattos et al.23 and contradict others, such as Rübe et al.17.
The NF-kappa β is an essential factor in chronic and acute inflammation, according to Schmid and et al.10 and Lixuan et al.24, and plays an important role in the inflammatory response, reinforcing the findings already cited on the protein.
The literature shows several studies about physical activity. However, some of them correlate the benefits of the physical practice to the pulmonary and systemic deleterious effects of radiotherapy.
According to Colbert et al.25, many researchers have examined the effects of exercise in the blood flow in humans, by quantifying the inflammatory cytokines. Typically, the concentration of plasma IL-1β does not tend to increase with exercise. However, a prolonged or a high-intensity exercise has increased the plasma concentration of TNF-α and IL-6, with peaks reached up to 2 hours after the exercise. Particularly, these other studies have already demonstrated an association between the eccentric exercise and exhaustion and induction of IL-626. Studies involving moderate exercise suggesting that the inflammatory response is dependent on other variables27.
Although many inflammatory cytokines were evaluated through the circulation, the expression of these proteins in tissues require further studies, especially in low-intensity physical exercises. In addition, it is speculates about other potential ways of cytokine production in response to this practice, which are not directly associated to skeletal muscle injury25.
It is speculated that the physical activity may have contributed significantly to what was the increase of cytokines TNF-α, IKKβ and IL-6 in the lung, as a response to the aerobic physical conditioning and pulmonary adaptation to the new ventilatory condition.
Oliveira et al.28 studied the effects of acute and chronic exercise on insulin sensitivity in obese mice. The exercise protocol included chronic swimming for eight weeks, with progressively increasing load greater than 5% of body weight. In our study the objective was mainly to improve cardiorespiratory fitness. So, progressive loads were not used. Moreover, an analogy must be made of training animals to impaired patients with thoracic tumor, for the purpose of the study. Some studies also show that the relationship of the effects of physical activity with an overload and short recovery period on the pro-inflammatory cytokines is still unknown29.
Ropelle et al.30 demonstrated that the physical activity suppresses the excess of appetite and the hypothalamic activation of IKKβ/NF-kappaβ by a mechanism dependent on the proinflammatory cytokine IL-6. Excessive food intake leads to hypothalamic activation of IKKβ and endoplasmic reticulum stress, leading to the central resistance to insulin and leptin, excessive appetite and obesity. The physical activity performed in an experimental model with rats C3H/HeJ, increased the central anti-inflammatory response, with an increased expression of IL-6 and IL-10. It was postulated that there is a reduction of the hypothalamic activation of IKKβ and the endoplasmic reticulum stress, restoring the production of insulin and leptin, and rearranging the nutritional balance.
The usage of the immunohistochemistry method for the assessment of the pulmonary proinflammatory cytokines, as well as the method used in this study (Western blotting), has been used for a long time successfully31-33. The usage of peripheral blood to evaluate the expression of such cytokines was not effective, such as, for instance, the studies performed by Morita et al.34 and Sakamoto et al.35.
In our study, it was used a cobalt therapy for the execution of the radiotherapy. Due to the fact that we deal about small animals and the need to irradiate tissue not too deep, Co-60 appears to be more indicated for the usage of the linear accelerator machine.
Studies show that the simplicity of the cobalt units gives them the advantage of an easy maintenance, reduced and with a low cost, when compared to the linear accelerators36.
The advantages of the cobalt therapy, as its low cost and maintenance, are well known in the community of radiotherapy. It is considered one of the basis of the health care, because it is simple and economical and, despite complex techniques are being explored and some departments are abandoning the usage of cobalt therapy for these more sophisticated techniques, their advantages prevail and still makes it exceptional for the rehabilitation36.
The graduation of the radiation dose is frequently limited by the tolerance to radiation of normal tissues. The pulmonary parenchyma itself is the most important dose-limit for radiotherapy in tumors in the thoracic region37. The graduation of the dose, however, also leads to an excessive exposure of organs at higher risk, such as lung, heart, esophagus and spinal cord. Esophagitis that occurs in less than 90 days after initiation of treatment is a common side effect of patients undergoing radiotherapy for thoracic tumors38.
Clinical studies have shown that the irradiation dose in the lower pulmonary regions is more associated to the pulmonary injury than the dose of irradiation applied to the upper pulmonary regions39,40. Other studies, such as Hope et al.41, found that the incidence of pneumonitis is higher in subjects with tumors of the lower lobes. However, Kocak et al.39, in a study, conclude that this pulmonary regional variable deserves further investigations. A number of researches performed by other groups did not find different rates of pneumonitis based on the tumor location42,43.
It is necessary to establish the relationship between the dose of radiation used in animals and humans. Although there are few studies on the issue44, in a study where mice received higher doses (10 to 18 Gy) radiotherapy than that used in the present study, it was found increased lung density by computed tomography besides the death of animals after a dose of 15 Gy. It was not the purpose of this study lead to organic weakness or even death in animals in question, so the use of lower doses (3,5 Gy). Typically, the dose of irradiation in the thoracic region in humans is about 40 to 65 Gy3,45.
Ysayama et al.46 performed a study with the purpose to assess the benefits of the pre-operative respiratory muscle training in patients undergoing esophageal surgery. The results obtained suggest that the pre-operative conditioning is of great importance in the obtaining of a pulmonary improvement, increasing the respiratory muscle strength and endurance and, consequently, a shorter hospital stay.
The respiratory physiotherapy has been mentioned as a conduct that reduces the incidence of postoperative pulmonary complications of open surgery47. In the study, such authors had as a purpose to assess the effects of respiratory physical therapy, among other effects, in subjects who underwent a subtotal right transthoracic esophagectomy for esophageal carcinoma of squamous cells and concluded that postoperative respiratory physical therapy plays an important role to reduce such complications. They also concluded that, in addition to physical therapy, systemic exercises also contributed to the satisfactory results. It is important to emphasize the lack of studies that mention the importance of respiratory physical therapy associated to esophageal and lung resection, and even the lack of studies in relation to the lung injury caused by radiotherapy (either pulmonary or esophageal cancer)47.
Conclusion
The pre-radiotherapy physical activity has radio-protective elements, by measuring the radio-induced activation of pro-inflammatory cytokines as IKKβ, TNF-α and IL-6.
Acknowledgements
To the Laboratory of the Internal Medicine of UNICAMP, Professor Valdemir Samonetto and employees of the Animal Colony of UNIFAE, Doctor Eduardo Baldon, Hugo Evangelista Brandino and Renata Rodrigues dos Santos Lixandrão, for their aid in the execution of this research.
Received: October 18, 2011
Review: December 14, 2011
Accepted: January 20, 2012
Conflict of interest: none
Financial source: FAEPEX-UNICAMP
1 Research performed at Laboratory of Molecular Oncology and Science Surgery Area, Department of Surgery, Faculty of Medical Sciences, State University of Campinas (UNICAMP), SP, Brazil. Part of Fellow PhD degree thesis of Renata Cristiane Gennari Bianchi. Tutor: Luiz Roberto Lopes. Postgraduate Program in Science Surgery.
- 1. Liao Z, Cox JD, Komaki R. Radiochemotherapy of esofaphageal cancer. J Thorac Oncol. 2007;2(6):553-68.
- 2. D'amico TA. Outcomes after surgery for esophageal cancer. Gastrointest Cancer Res. 2007;1(5):188-96.
- 3. Tercioti Junior V, Lopes LR, Coelho Neto JD, Carvalheira JB, Andreollo NA. Local effectiveness and complications of neoadjuvant therapy in esophageal squamous cell carcinoma: radiotherapy versus chemoradiotherapy. Rev Col Bras Cir. 2011;38(4):227-34.
- 4. Miyata H, Yamasaki M, Takiguchi S, Nakajima K, Fujiwara Y, Nishida T, Mori, M, Doki, Y. Salvage esophagectomy after definitive chemoradiotherapy for thoracic esophageal cancer. J Surg Oncol. 2009;100:442-6.
- 5. Asakura H, Hashimoto T, Zenda S, Harada H, Hirakawa K, Mizumoto M, Furutani, K, Hironaka S, Fuji H, Murayama S, Boku N, Nishimura T. Analysis of dose-volume histogram parameters for radiation pneumonitis after definitive concurrent chemoradiotherapy for esophageal cancer. Radiother Oncol. 2010;95:240-4.
- 6. Yu HM, Liu YF, Cheng YF, Hu LK, Hou M. Effects of rhubarb extract on radiation induced lung toxicity via decreasing transforming growth factor-beta-1 and interleukin-6 in lung cancer patients treated with radiotherapy. Lung Cancer. 2008;59:219-26.
- 7. Sakai M, Iwakawa M, Iwakura Y, Ohta T, Tsujii H, Imai T. CD44 and bak expression in IL-6 or TNF-alpha gene knockout mice after whole lung irradiation. J Radiat Res. 2008;49:409-16.
- 8. Novakova-Jiresova A, Luijk PV, Goor HV, Kampinga HH, Coppes RP. Pulmonary radiation injury: identification of risk factors associated with regional hypersensitivity. Cancer Res. 2005;65:3568-76.
- 9. Bao P, Gao W, Li S, Zhang L, Qu S, Wu C, Qi H. Effect of pretreatment with high-dose ulinastatin in preventing radiation-induced pulmonary injury in rats. Eur J Pharmacol. 2009;603:114-9.
- 10. Schmid JA, Birbach A. IkB kinase β (Ikkβ/Ikk2/IkBkB)- a key molecule in signaling to the transcription factor NF-kB. Cytokine Growth Factor Rev. 2008;19:157-65.
- 11. Mussi RK, Camargo EA, Ferreira T, De Moraes C, Delbin MA, Toro, IF, Brancher S, Landucci EC, Zanesco A, Antunes E. Exercise training reduces pulmonary ischaemia-reperfusion-induced inflammatory responses. Eur Respir J. 2008;31(3):645-9.
- 12. Gannon GA, Rhind SG, Suzui M, Shek PN, Shephard, RJ. Circulating levels of peripheral blood leucocytes and cytokines following competitive cycling. Can J Appl Physiol. 1997;22:133-47.
- 13. Nieman DC. Special feature for the Olympics: exercise effects on systemic immunity. Immunol Cell Biol. 2000;78:496-501.
- 14. Triboulet JP, Mariette C. Esophageal squamous cell carcinoma stage III: state of surgery after radiochemotherapy. Cancer Radiother. 2006;10:456-61.
- 15. Hermann RM, Horstmann O, Haller F, Perske C, Christiansen H, Hille A, Schimidberger H, Füzesi L. Histomorphological tumor regression grading esophageal carcinoma after neoadjuvante radiochemotherapy: with score to use? Dis Esophagus. 2006;5:329-34.
- 16. Cagol M, Ruol A, Sileni VC, De Salvo Gl, Corti L, Alfieri R, Innocente R, Fumagalli U, Rosati R, Ancona E. Multinodal treatment in locally advanced esophageal cancer: a multicenter phase II study with neoadjuvant oxaliplatin, 5-fluorouracil, leucovorin and neoadjuvante radiotherapy: prelimirary results. Chir Ital. 2006;4:433-9.
- 17. Rübe CE, Wilfert F, Uthe D, Schmid KW, Knoop R, Willich N, Schuck A, Rübe C. Modulation of radiation-induced tumor necrosis factor α (TNF-α) expression in the lung tissue by pentoxifyline. Radiother Oncol. 2002;64:177-87.
- 18. Arnold R, Humbert B, Werchau H, Gallati H, Königg W. Interleukin-8, interleukin-6, and soluble tumor necrosis factor receptor type I release from a human pulmonary epithelial cell line (A549) exposed to respiratory syncytial virus. Immunology. 1994;82:126-33.
- 19. Chen Y, Rubin P, Williams J, Hernady E, Smudzin T, Okunieff P. Int J Radiat Oncol Biol Phys. 2001;49(3):641-8.
- 20. Sriwijitkamol A, Coletta DK, Wajcberg E, Balbontin GB, Reyna SM, Barrientes J, Eagan PA, Jenkinson CP, Cersosimo E, Defronzo RA, Sakamoto K, Musi N. Effect of acute exercise on AMPK signiling in skeletal muscle of subjects with type 2 diabetes: a time-course and dose-response study. Diabetes. 2007;56(3):836-48.
- 21. Anscher MS, Peters WP, Reisenbichler H, Petros WP, Jirtle RL. Transforming growth factor beta as predictor of liver and lung fibrosis after autologous bone marrow transplantation for advanced breast cancer. N Engl J Med. 1993;328:1592-8.
- 22. Fu XL, Huang H, Bentel G, Clough R, Jirtle RL, Kong FM, Marks LB, Anscher MS. Predicting the risk of symptomatic radiation-induced lung injury using both the physical and biologic parameters V30 and transforming growth factor β. Int J Radiat Oncol Biol Phys. 2001;50(4):899-908.
- 23. Mattos MD, Kimura ET, Silva MRR, Egami MI, Segreto RA, Segreto HRC. Ativação da proteína TGFβ1 latente em pulmão irradiado in vivo. Rev Assoc Med Bras. 2002;48(4):329-34.
- 24. Lixuan Z, Jingcheng D, Wenqin Y, Jianhua H, Baojun L, Xiaotao F. Baicalin attenuates inflammation by inhibiting NF-kappaβ activation in cigarette smoke induced inflammatory models. Pulm Pharmacol Ther. 2010;23(5):411-9.
- 25. Colbert LH, Davis JM, Essig DA, Ghaffar A, Mayer EP. Tissue expression and plasma concentrations of TNF-α, IL-1β, and IL-6 following treadmill exercise in mice. Int J Sports Med. 2001;22:261-7.
- 26. Ostrowski K, Rohde T, Zacho M, Asp S, Pedersen BK. Evidence that interleukin-6 is produced in human skeletal muscle during prolonged running. J Physiol. 1998;508:949-53.
- 27. Ullum H, Haahr PM, Diamant M, Palmo J, Halkjaer-Kristensen J, Pedersen BK. Bicycle exercise enhances plasma IL-6 but does not change IL-1α, IL-1β, IL-6 or TNFα pre-mRNA in BMNC. J Appl Physiol. 1994;77:93-7.
- 28. Oliveira AG, Carvalho BM, Tobar N, Ropelle ER, Pauli JR, Bagarolli RA, Guadagnini D, Carvalheira JB, Saad MJ. Physical exercise reduces circulating lipopolysaccharide and TLR4 activation and improves insulin signaling in tissues of DIO rats. Diabetes. 2011;60:784-96.
- 29. Lira FS, Rosa JC, Pimentel GD, Tarini VAF, Arida RM, Faloppa F, Alves ES, Do Nascimento CO, Oyama LM, Seelaender M, De Mello MT, Santos RV. Inflammation and adipose tissue: effects of progressive load training in rats. Lipids Health Dis. 2010;9:109.
- 30. Ropelle ER, Flores MB, Cintra DE, Rocha GZ, Pauli JR, Morari J, De Souza CT, Moraes JC, Prada PO, Guadagnini D, Marin RM, Oliveira AG, Augusto TM, Carvalho HF, Velloso LA, Saad MJ, Carvalheira JB. IL-6 and IL-10 anti-inflammatory activity links exercise to hypothalamic insulin and leptin sensitivity through IKKbeta and ER stress inhibition. PLoS Biol. 2010;8(8). pii: e1000465.
- 31. Chen L, Wang T, Wang X, Sun BB, Li JQ, Liu DS, Zhang SF, Liu L, Xu D, Chen YJ, Wen FQ. Blockade of advanced glycation end product formation attenuates bleomycin-induced pulmonary fibrosis in rats. Respir Res. 2009;10:55.
- 32. Kim JY, Kim YS, Kim YK, Park HJ, Kim SJ, Kang JH, Wang YP, Jang HS, Lee SN, Yoon SC. TGF-β1 dynamics during radiation therapy and its correlation to symptomatic radiation pneumonitis in lung cancer patients. Radiat Oncol. 2009;4:59.
- 33. Moon C, Lee YJ, Park HJ, Chong YH, Kang JL. N-acetylcysteine inhibits RhoA and promotes apoptotic cell clearance during intense lung inflammation. Am J Respir Crit Care Med. 2010;181:374-87.
- 34. Morita M, Yoshida R, Ikeda K, Egashira A, Oki E, Sadanaga N, Kakeji Y, Ichiki Y, Sugio K, Yasumoto K, Maehara Y. Acute lung injury following an esophagectomy for esophageal cancer, with special reference to the clinical factors and cytokine levels of peripheral blood and pleural drainage fluid. Dis Esophagus. 2008;21(1):30-6.
- 35. Sakamoto K, Arakawa H, Mita S, Ishiko T, Ikei S, Egami H, Hisano S, Ogawa M. Elevation of circulating interleukin-6 after surgery: factors influencing serum level. Cytokine. 1994;6:181-6.
- 36. Adams EJ, Warrington AP. A comparison between cobalt and linear accelerator-based treatment plans for conformal and intensity-modulated radiotherapy. Br J Radiol. 2008;81:304-10.
- 37. Jin H, Tucker SL, Liu HH, Wei X, Yom SS, Wang S, Komaki R, Chen Y, Martel MK, Mohan R, Cox JD, Liao Z. Dose-volume thresholds and smoking status for the risk of treatment-related pneumonitis in inoperable non-small cell lung cancer treated with definitive radiotherapy. Radiother Oncol. 2009;91:427-32.
- 38. Werner-Wasik M, Yorke E, Deasy J, Nam J, Marks LB. Radiation dose-volume effects in the esophagus. Int J Radiat Oncol Biol Phys. 2010;76(3):S86-93.
- 39. Kocak Z, Borst GR, Zeng J, Zhou S, Hollis R, Zhang J, Evans ES, Folz RJ, Wong T, Kahn D, Belderbos JS, Lebesque JV, Marks LB. Prospective assessment of dosimetric/physiologic-based models for predicting radiation pneumonitis. Int J Radiat Oncol Biol Phys. 2007;67(1):178-86.
- 40. Yorke ED, Jackson A, Rosenzweig KE, Braban L, Leibel SA, Ling CC. Correlation of dosimetric factors and radiation pneumonitis for non-small-cell lung cancer patients in a recently completed dose escalation study. Int J Radiat Oncol Biol Phys. 2005;63(3):672-82.
- 41. Hope AJ, Lindsay PE, El Naga I, Alaly JR, Vicic M, Bradley JD, Deasy GO. Modeling radiation pneumonitis risk with clinical, dosimetric, and spatial parameters. Int J Radiat Oncol Biol Phys. 2006;65(1):112-24.
- 42. Tsujino K, Hirota S, Endo M, Obayashi K, Kotani Y, Satouchi M, Kado T, Takada Y. Predictive value of dose-volume histogram parameters for predicting radiation pneumonitis after concurrent chemoradiation for lung cancer. Int J Radiat Oncol Biol Phys. 2003;55(1):110-5.
- 43. Fay M, Tan A, Fisher R, Mac Manus M, Wirth A, Ball D. Dose-volume histogram analysis as predictor of radiation pneumonitis in primary lung cancer patients treated with radiotherapy. Int J Radiat Oncol Biol Phys. 2005;61(5):1355-63.
- 44. Szabo S, Ghosh SN, Fish BL, Bodiga S, Tomic R, Kumar G, Morrow NV, Moulder JE, Jacobs ER, Medhora M. Cellular inflammatory infiltrate in pneumonitis induced by a single moderate dose of thoracic versus radiation in rats. Radiat Res. 2010;173(4):545-56.
- 45. Dähn D, Martell J, Vorwerk H, Hess CF, Becker H, Jung K, Hilgers R, Wolff HA, Hermann RM, Christiansen H. Influence of irradiated lung volumes on perioperative morbidity and mortality in patients after neoadjuvante radiochemotherapy for esophageal cancer. Int J Radiat Oncol Biol Phys. 2010;77(1):44-52.
- 46. Ysayama L, Lopes LR, Silva AMO, Andreollo NA. The influence of the respiratory muscular training in the recovery of esophagectomies. ABCD Arq Bras Cir Dig. 2008;21(2):61-4.
- 47. Nakamura M, Iwahashi M, Nakamori M, Ishida K, Naka T, Iida T, Katsuda M, Tsuji T, Nakatani Y, Yamaue H.. An analysis of the factors contributing to a reduction in the incidence of pulmonary complications following an esophagectomy for esophageal cancer. Langenbecks Arch Surg. 2008;393:127-33.
Publication Dates
-
Publication in this collection
27 Mar 2012 -
Date of issue
Mar 2012
History
-
Received
18 Oct 2011 -
Accepted
20 Jan 2012 -
Reviewed
14 Dec 2011