Abstracts
PURPOSE: To evaluate the effect of N-acetylcysteine (NAC) combined with fluid resuscitation on pulmonary cell death in rats induced with controlled hemorrhagic shock (HS). METHODS: Two arteries (MAP calculation and exsanguination) and one vein (treatments) were catheterized in 22 anesthetized rats. Two groups of male albino rats were induced with controlled HS at 35mmHg MAP for 60 min. After this period, the RL group was resuscitated with Ringer's lactate and the RL+NAC group was resuscitated with Ringer's lactate combined with 150mg/Kg NAC. The control group animals were cannulated only. The animals were euthanized after 120 min of fluid resuscitation. Lung tissue samples were collected to evaluate the following: histopathology, TUNEL and imunohistochemical expression of caspase 3. RESULTS: RL showed a greater number of cells stained by TUNEL than RL + NAC, but there was no change in caspase 3 expression in any group. CONCLUSION: N-acetylcysteine associate to fluid resuscitation, after hemorrhagic shock, decreased cell death attenuating lung injury.
Acetylcysteine; Lung; Shock, Hemorrhagic; Cell Death; Rats
OBJETIVO: Avaliar o efeito da N-acetilcisteína (NAC) combinada ao fluido de reposição volêmica na morte celular pulmonar de ratos submetidos ao choque hemorrágico (CH) controlado. MÉTODOS: Duas artérias (cálculo da PAM e exsanguinação) e uma veia (tratamentos) foram cateterizadas em 22 ratos anestesiados. Dois grupos de ratos machos albinos foram induzidos ao CH controlado com PAM de 35mmHg por 60 min. Após este período, o grupo RL foi ressuscitado com Ringer lactato e o grupo RL+NAC foi ressuscitado com Ringer lactato associado com 150mg/Kg de NAC. O grupo controle sofreu somente o procedimento cirúrgico de cateterização. Os animais sofreram eutanásia após 120 min. da ressuscitação. Amostras de tecido pulmonar foram coletadas para histopatologia, TUNEL e a imuno-expressão da caspase 3. RESULTADOS: RL apresentou maior número de células marcadas pelo TUNEL do que RL+NAC, porém sem alteração na expressão da caspase 3 em nenhum dos grupos estudados. CONCLUSÃO: A N-acetilcisteína teve um papel protetor na morte celular em modelo de choque hemorrágico controlado.
Acetilcisteína; Pulmão; Choque Hemorrágico; Morte Celular; Ratos
8 - ORIGINAL ARTICLE
ISCHEMIA-REPERFUSION
Effect of N-acetylcysteine on pulmonary cell death in a controlled hemorrhagic shock model in rats1 1 Research performed at Laboratory of the Experimental Surgical Research (LIM-26) and Laboratory of Surgical Physiopathology (LIM-62), Faculty of Medicine, University of Sao Paulo (USP). Part of PhD thesis, Postgraduate Program in Interdisciplinary Surgical Science, Paulista School of Medicine, Federal University of Sao Paulo (UNIFESP), Brazil. Tutor: Prof. Edna Frasson de Souza Montero.
Efeito da N-acetilcisteína na morte celular pulmonar em um modelo de choque hemorrágico controlado em ratos
Paulo Fernandes SaadI; Karen Ruggeri SaadII; Luiz Dantas de Oliveira FilhoIII; Sueli Gomes FerreiraIV; Marcia Kiyomi KoikeV; Edna Frasson de Souza MonteroVI
IFellow PhD degree, Postgraduate Program in Interdisciplinary Surgical Science, Paulista Medical School, UNIFESP, Sao Paulo, Brazil. Main author. Responsible for conception, design, intellectual and scientific content of the study
IIPhD, Associate Professor of Morphology, Faculty of Medicine, Sao Francisco Valley Federal University (UNIVASF), Pernambuco, Brazil. Acquisition and interpretation of data, manuscript writing
IIIPhD, Associate Professor of Semiology, Faculty of medicine, UNIVASF, Pernambuco, Brazil. Acquisition and interpretation of data, critical revision
IVResearcher, Laboratory of Cardiovascular Surgery and Circulation Physiopathology, Faculty of Medicine, USP, Sao Paulo, Brazil. Histopathological analysis
VPhD, Researcher, Laboratory of Clinical Emergencies, Faculty of Medicine, USP, Sao Paulo, Brazil. Interpretation of data, histopathological analysis
VIPhD, Associate Professor, Laboratory of Surgical Physiopathology (LIM-62), Faculty of Medicine, USP, Sao Paulo, Brazil. Responsible for conception and critical revision of the study
Correspondence Correspondence: Edna Frasson de Souza Montero Alameda Espada, 134 Residencial Onze 06540-395 Santana de Parnaíba SP Brasil Tel.: (55 11) 3061-7216 / 9686-7933 edna.montero@gmail.com
ABSTRACT
PURPOSE: To evaluate the effect of N-acetylcysteine (NAC) combined with fluid resuscitation on pulmonary cell death in rats induced with controlled hemorrhagic shock (HS).
METHODS: Two arteries (MAP calculation and exsanguination) and one vein (treatments) were catheterized in 22 anesthetized rats. Two groups of male albino rats were induced with controlled HS at 35mmHg MAP for 60 min. After this period, the RL group was resuscitated with Ringer's lactate and the RL+NAC group was resuscitated with Ringer's lactate combined with 150mg/Kg NAC. The control group animals were cannulated only. The animals were euthanized after 120 min of fluid resuscitation. Lung tissue samples were collected to evaluate the following: histopathology, TUNEL and imunohistochemical expression of caspase 3.
RESULTS: RL showed a greater number of cells stained by TUNEL than RL + NAC, but there was no change in caspase 3 expression in any group.
CONCLUSION: N-acetylcysteine associate to fluid resuscitation, after hemorrhagic shock, decreased cell death attenuating lung injury.
Key words: Acetylcysteine. Lung. Shock, Hemorrhagic. Cell Death. Rats.
RESUMO
OBJETIVO: Avaliar o efeito da N-acetilcisteína (NAC) combinada ao fluido de reposição volêmica na morte celular pulmonar de ratos submetidos ao choque hemorrágico (CH) controlado.
MÉTODOS: Duas artérias (cálculo da PAM e exsanguinação) e uma veia (tratamentos) foram cateterizadas em 22 ratos anestesiados. Dois grupos de ratos machos albinos foram induzidos ao CH controlado com PAM de 35mmHg por 60 min. Após este período, o grupo RL foi ressuscitado com Ringer lactato e o grupo RL+NAC foi ressuscitado com Ringer lactato associado com 150mg/Kg de NAC. O grupo controle sofreu somente o procedimento cirúrgico de cateterização. Os animais sofreram eutanásia após 120 min. da ressuscitação. Amostras de tecido pulmonar foram coletadas para histopatologia, TUNEL e a imuno-expressão da caspase 3.
RESULTADOS: RL apresentou maior número de células marcadas pelo TUNEL do que RL+NAC, porém sem alteração na expressão da caspase 3 em nenhum dos grupos estudados.
CONCLUSÃO: A N-acetilcisteína teve um papel protetor na morte celular em modelo de choque hemorrágico controlado.
Descritores: Acetilcisteína. Pulmão. Choque Hemorrágico. Morte Celular. Ratos.
Introduction
Trauma prevails as the leading cause of death in young people in Brazil and worldwide, despite advances in medicine over the last century1.
Hemorrhagic shock is characterized by a state of tissue hypoperfusion with a reduction of oxygen at the cellular level at the expense of blood loss2. Unfortunately, direct hemorrhage control prior to fluid resuscitation is not always possible3, thus re-establishing adequate perfusion of organs and tissues is often essential, even before controlling bleeding3,4. However, multiple organ dysfunction syndrome (MODS), beginning after fluid resuscitation, mainly occurs due to injuries associated with ischemia and reperfusion (I/R), perpetuating the high mortality condition5. Although there are several factors that contribute to MODS, there is increasing evidence that acute lung injury (ALI) and acute respiratory distress syndrome (ARDS) plays a central role in its evolution6.
Despite improvement in supportive care, ALI/ARDS still carries high mortality rates up to 60%7. An improved understanding of the pathophysiology of ALI is needed in order to develop treatments that will contribute with mechanical ventilation strategies in reducing overall mortality8.
Cell death has been demonstrated in the lung during the pathogeneses of ALI/ARDS, of which there is no consensus about the pathway involved in cell death. Apoptosis and necrosis remains prevalent in literature and new evidences suggested that this classical dichotomy is a simplification of a complex processes9,10.
Considering that using fluid resuscitation alone in treating hemorrhagic shock would not be able to prevent MODS and mortality11, current research has diversified the search for solutions to prevent this complication. One aspect is combining drugs with fluid resuscitation solutions - drug therapy4.
N-acetylcysteine (NAC) is a thiol-containing compound with antioxidant and anti-inflammatory properties, exhibiting effects on microcirculation12. NAC acts directly as an antioxidant, by deactivating reactive oxygen species, or indirectly, by replacing intracellular glutathione stores. However, despite the many benefits reported in studies using NAC in attenuating I/R damage, there are very few studies that use drug therapy for treating hemorrhagic shock13,14.
Based on the fact that NAC is a low-cost, widely available, and clinically tested, with proven benefits in treating injuries associated with I/R15,16, it was decided to test the hypothesis that NAC can modulate cell death in lung tissue induced by HS in an experimental model.
Methods
This research was approved by the Ethics Committee of Federal University of Sao Paulo (protocol number 1712/11), ratified by the Sao Paulo University (186/11). This study was conducted at the Laboratory of the Experimental Surgical Research LIM-26 (surgical procedures), and the Laboratory of Surgical Physiopathology LIM-62 (sample analyses), Faculty of Medicine, University of Sao Paulo, Brazil.
It was used 22, adult male Wistar rats (rattus norvegicus albinus), aged between 90 and 120 days, weighing between 250 and 300g. The animals were kept in a vivarium for six days for observation and adaptation, where they received water and balanced food rations ad libitum and were kept in appropriate cages (40x30x25cm), with a maximum of five animals, under controlled conditions for light and temperature.
The animals were evaluated randomly into three groups: the control group CG (n=6) was only subjected to the surgical procedure and catheterization, without induced HS., the RL group (n=8) was HS induced and for fluid resuscitation, 33 ml/kg of Ringer's lactate solution was infused, and soon after, 50% blood (previously withdrawn) was infused and the RL+NAC group (n=8) that was HS induced and for fluid resuscitation, 150 mg/kg of NAC and 33 ml/kg of Ringer's lactate solution were infused, and soon after, 50% blood (previously withdrawn) was infused.
The animals were weighed and anesthetized with a ketamine (50mg/kg)+xylazine (15mg/kg) combination by intraperitoneal injection, thus put under and unresponsive to mechanical stimuli after being anesthetized, exhibiting loss of righting and hind limb withdrawal reflexes, and palpebral reflex, after pain stimulus via gripping and lightly touching the eye, respectively. The animals were subjected to additional doses of the combination (half of the initial dose) during the procedure, when necessary. They were also kept well ventilated at room temperature.
The animals were placed on a constantly heated plate (37˚C) in supine position with hind limbs immobilized.
Trichotomy was performed on the right anterior cervical and right inguinal regions with a razor blade followed by antisepsis of the operative area with iodine polyvinylpyrrolidone tincture. Using a no. 11 scalpel blade, a cervical incision was made, with dissection and isolation of the right common carotid artery and incision in the right inguinal region with dissection and isolation of the femoral artery and vein. All animals were cannulated with Intracath® 22G (Bencton-Dicknson, Sandy, USA). The venous catheter was used for injecting heparin and fluids for resuscitation according to the experimental groups, and arterial catheters were used for collecting blood samples, exsanguination that caused the shock, and for monitoring mean arterial pressure (MAP).
For controlling MAP and effectiveness of the procedures employed, the arterial catheter was connected to a pressure transducer connected to a calibrated preamplifier and a computerized data acquisition system (Dixtal DX 2020), in which hemodynamic data were stored (MAP and heart rate). At the end of surgery, the animals were observed for 15 minutes for MAP stabilization.
To induce hemorrhagic shock, non-fractionated heparin was infused (100 IU/rat). Next, blood was taken via the arterial catheter for 10 minutes, using a previously heparinized 10mL syringe, until reaching 35 mm Hg MAP. This pressure was maintained for 60 minutes, removing or reinserting heparinized whole blood, in the case of ±5 mmHg change in MAP. After 60 minutes from the onset of hemorrhagic shock, the animals were subjected to fluid resuscitation and treatment according to the groups specified. Successful resuscitation was considered when MAP remained above 80 mmHg for at least 5 min.
At the end of the resuscitation process, the animals were kept for 120 min and submitted thoracotomy to remove a fragment of left lung. They were euthanized by exsanguination while still under anesthesia.
Samples of lung were processed for Hematoxylin and Eosin (H.E) staining, TUNEL and immunohistochemistry for caspase 3.
The histological slides were qualitatively evaluated under an optical microscope (Zeiss Axio Image A2, Oberkochen, Germany) by an experienced researcher kept "blind" to the different treatment groups. At least 20 fields of each slide were randomly chosen and analyzed to assess areas with morphological characteristics consistent with necrosis.
Another lung fragments were processed for imunohistochemical labeling of caspase 3 and its expression as a measure of lung cell apoptosis using the avidin-biotin-peroxidase. The imunohistochemical expression of caspase 3 was identifies and quantified by a computer program (AxionVision Rel 4.2 Carl Zeiss) and adopted as positivity the appearance of brown color in the core, at the region of the boundary cell or the cell cytoplasm. The rate of apoptosis was calculated using the percentage of labeled cells per area.
The DNA fragmentation analysis in lung cells was evaluated by TUNEL method with the In situ cell death detection kit, POD (Roche, Germany). The slides were evaluated by optical microscopy, where 10 fields (10x10μm) from each section were used to count positive and negative cells.
The data were analyzed using SigmaStat Statistical program, version 3.1 (Systat Software, San Jose, USA).
The groups were compared by Kruskal-Wallis One Way Analysis of Variance on ranks and complemented by appropriate post-hoc test (Dunn tests). It was considered 95% level of significance.
Results
There were no aspects suggestive of necrosis in lung tissue evaluated by optical microscopy in HE staining (Figure 1).
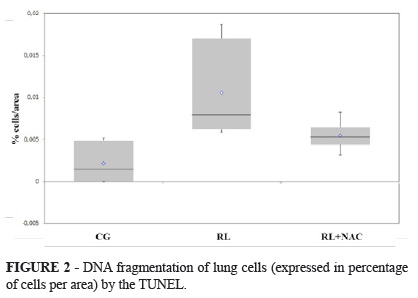
Cell death evaluated by TUNEL (Figure 2) showed decreased number of positive cells when NAC was associate to fluid resuscitation (Kruskal-Wallis test and Dunn): GC<RL; RL+NAC<RL (p<0.05). However, the activity of caspase 3 was not significantly modified by any treatment (p=0.304) (Figure 3).
Discussion
NAC plays a promising role in drug therapy combined with fluid resuscitation in treating hemorrhagic shock by attenuating cell death in the lung tissue, according these results. To the best of our knowledge, this is the first study evaluating the effect of NAC on pulmonary cell death in a controlled hemorrhagic shock model in rats.
Despite the influence of inflammatory mediators in ALI development, we emphasize that this injury's pathophysiology is complex and multifactorial, involving oxidative stress, inflammatory response, alterations of the microcirculation and cell death.
There are few studies14,17 that evaluated the effects of NAC in lung tissue in models of hemorrhagic shock and none of them assesses cellular death. However, there is evidence that NAC is able to attenuate apoptosis of lung epithelial cells in experimental models of sepsis18.
The histopathology assessment of the lung showed no lesions compatible with necrosis in all groups studied. This result can be explained by experimental protocol adopted in this study in which observation time was not sufficient for detection the necrosis morphological characteristics.
There are many immunological and biochemical methods to characterize cell deaths. The TUNEL (terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick-end labeling) has the advantage of allowing diagnosis of DNA fragmentation prior to cell disintegration, irreversible and final phase of the cell death process, thus revealing the possibility of diagnosing apoptosis or other forms of cell death19. In the recent past, the TUNEL assay was used as a marker of apoptosis and that fact generated confusion in the literature about the type of cell death process involved20,21.
In this study using the TUNEL, RL showed a significant increase in count of positive cells compared to CG. These results demonstrate a higher incidence of DNA fragmentation, suggesting an increase in cell death associated with the injury mechanism. However, there was no significant difference in comparing CG and RL+NAC, indicating NAC have a protective effect against this injury mechanism. Other studies also confirm the protective role of NAC in lung cell death in experimental models of sepsis18,22,23.
Based on the knowledge of major apoptosis pathways, considering caspase activity defined as a relevant characteristic of apoptosis, we evaluated it by immunohistochemistry, caspase-3 expression, of which activation is considered the most exclusive characteristic of apoptosis19,24. However, our data of caspase-3 expression in lung tissue showed that there was no significant difference among groups. RL and RL+NAC groups, despite being HS-induced, did not exhibit caspase-3 expression, similarly to CG. These data corroborate Mura et al.25 findings in a model of extra pulmonary I/R injury. The authors evidenced cell death in the lung, by TUNEL, in an experimental model of MODS, but not observed an increase of cellular expression of caspase 3. They performed electron microscopy with showed that structural changes in lung tissue were compatible with oncotic cell death25. Based on these evidences, the mechanism of cell death could be explained by apoptosis non-mediated by caspase or another type of cell death.
Despite of the benefit of reducing cell death in lung injury by associating NAC to the fluid resuscitation post hemorrhagic shock, other studies should be conducted to better explain the protective role of NAC in this experimental model.
Conclusion
N-acetylcysteine associate to fluid resuscitation, after hemorrhagic shock, decreased cell death attenuating lung injury.
Acknowledgments
The authors thank Professor Dr. Luiz Francisco Poli de Figueiredo (in memoriam) for collaborating with this research by discussing and offering his lab for the surgical procedures (Laboratory of the Experimental Surgical Research LIM-26), We also thank the technicians from LIM-26, especially, Mrs Junko Takano Osaka and Mr. Elias Aparecido Marcelino; and LIM-62, especially, Mr. Marco de Luna and Mr. Mario Itinoshe
Received: March 22, 2012
Review: May 23, 2012
Accepted: June 21, 2012
Conflict of interest: none
Financial source: National Council of Technological and Scientific Development (CNPq)
- 1. Demetriades D, Kimbrell B, Salim A, Velmahos G, Rhee P, Preston C, Gruzinski G, Chan L. Trauma deaths in a mature urban trauma system: is "trimodal" distribution a valid concept? J Am Coll Surg. 2005;201(3):343-8.
- 2. Vincenzi R, Cepeda LA, Pirani WM, Sannomyia P, Rocha-E-Silva M, Cruz RJ Jr. Small volume resuscitation with 3% hypertonic saline solution decrease inflammatory response and attenuates end organ damage after controlled hemorrhagic shock. Am J Surg. 2009;198(3):407-14.
- 3. Alam HB, Rhee P. New developments in fluid resuscitation. Surg Clin North Am. 2007;87(1):55-72.
- 4. Santry H P, Alam H B. Fluid Ressuscitation: past, present and future. Shock. 2010;33(3):229-41.
- 5. Heckbert SR, Vedder NB, Hoffman W, Winn RK, Hudson LD, Jurkovich GJ, Copass MK, Harlan JM, Rice CL, Maier RV. Outcome after hemorrhagic shock in trauma patients. J Trauma.1998;45(3):5459.
- 6. Moore FA, McKinley BA, Moore EE. The next generation in shock resuscitation. Lancet. 2004;363(9425);1988-96.
- 7. Tang PS, Mura M, Seth R, Liu M. Acute lung injury and cell death: how many ways can cells die? Am J Physiol Lung Cell Mol Physiol. 2008;294(4):L632-41.
- 8. Martin TR, Hagimoto N, Nakamura M, Matute-Bello G. Apoptosis and epithelial injury in the lungs. Proc Am Thorac Soc. 2005;2(3):214-20.
- 9. Kitanaka C, Kuchino Y. Caspase-independent programmed cell death with necrotic morphology. Cell Death Differ. 1999;6(6):508-15.
- 10. Jaeschke H, Lemasters JJ. Apoptosis versus oncotic necrosis in hepatic ischemia/reperfusion injury. Gastroenterology. 2003;125(4):1246-57.
- 11. Liaudet L, Soriano FG, Szabó E, Virág L, Mabley JG, Salzman AL, Szabo C. Protection against hemorrhagic shock in mice genetically deficient in poly (ADP-ribose) polymerase. Proc Natl Acad Sci USA. 2000;97(18):10203-8.
- 12. Atkuri KR, Mantovani JJ, Herzenberg LA, Herzenberg LA. N-Acetylcysteine - a safe antidote for cysteine/glutathione deficiency. Curr Opin Pharmacol. 2007;7(4):355-9.
- 13. Galhardo MA, Quireze Jr C, Riboli Navarro PG, Morello RJ, Simões MJ, Montero EFS. Liver and lung late alterations following hepatic reperfusion associated to ischemic preconditioning or N-acetylcysteine. Microsurgery. 2007;27(4):295-9.
- 14. Alkan A, Eroğlu F, Eroğlu E, Ergin C, Cerçi C, Alsancak G. Protective effects of N-acetylcysteine and erdosteine on hemorrhagic shock induced acute lung injury. Eur J Emerg Med. 2006;13(5):281-5.
- 15. Castro AP, Castro Junior MA, Lauz S, Facin E, Simões MJ, Fagundes DJ. The role of N-acetyl-cysteine in the lung remote injury after hepatic ischemia and reperfusion in rabbits. Acta Cir Bras. 2012;27(1):49-55.
- 16. Sotoudeh A, Takhtfooladi M A, Jahanshahi A, Asl AHK, Takhtfooladi HA, Khansari M. Effect of N-acetylcysteine on lung injury induced by skeletal muscle ischemia-reperfusion: histopathological study in rat model. Acta Cir Bras. 2012;27(2):168-71.
- 17. Portella AO, Montero EF, Poli de Figueiredo LF, Bueno AS, Thurow AA, Rodrigues FG. Effects of N-acetylcysteine in hepatic ischemia-reperfusion injury during hemorrhagic shock. Transplant Proc. 2004(4);36:846-8.
- 18. Ozdulger A, Cinel I, Koksel O, Cinel L, Avlan D, Unlu A, Okcu H, Dikmengil M, Oral U. The protective effect on N-acetylcysteine on apoptotic lung injury in cecal ligation and puncture-induced sepsis model. Shock.2003;19(4):366-72.
- 19. Schulze-Bergkamen H, Schuchmann M, Fleischer B, Galle PR. The role of apoptosis versus oncotic necrosis in liver injury: facts or faith? J Hepatol. 2006;44(5):984-93.
- 20. Jernigan TW, Croce MA, Fabian TC. Apoptosis and necrosis in the development of acute lung injury after hemorrhagic shock. Am Surg. 2004;70(12):1094-8.
- 21. Deb S, Sun L, Martin B, Talens E, Burris D, Kaufmann C, Rich N, Rhee P. Lactated ringer's solution and hetastarch but not plasma resuscitation after rat hemorrhagic shock is associated with immediate lung apoptosis by the up-regulation of the Bax protein. J Trauma. 2000;49(1):47-53.
- 22. Demiralay R, Gürsan N, Ozbilin G, Erdogan G, Dermici E.Comparison of the effects of endorsteine and N-aceylcysteine on apoptosis regulation in endotoxin-induced acute lung injury. J Appl Toxicol. 2006;26(4):301-8.
- 23. Oh SH, Lim SC. A rapid and transient ROS generation by cadmium triggers apoptosis via caspase-dependent pathway in HepG2 cells and this is inhibited through N-acetylcysteine-mediated catalase upregulation. Toxicol Appl Pharmacol. 2006;212(3):212-23.
- 24. Schwab BL, Guerini D, Didszun C, Bano D, Ferrando-May E, Fava E, Tam J, Xu D, Xanthoudakis S, Nicholson DW, Carafoli E, Nicotera P. Cleavage of plasma membrane calcium pumps by caspases: a link between apoptosis and necrosis. Cell Death Differ. 2002;9(8):818-31.
- 25. Mura M, Andrade CF, Han B, Seth R, Zhang Y, Bai XH, Waddell TK, Hwang D, Keshavjee S, Liu M. Intestinal ischemia-reperfusion-induced acute lung injury and oncotic cell death in multiple organs. Shock. 2007;28(2):227-38.
Correspondence:
Publication Dates
-
Publication in this collection
25 July 2012 -
Date of issue
Aug 2012
History
-
Received
22 Mar 2012 -
Accepted
21 June 2012 -
Reviewed
23 May 2012