Abstracts
PURPOSE: Since Technetium-99m (99mTc) has favorable physical and chemical characteristics, it is widely used radioisotope in Nuclear Medicine. However, stannous dichloride (SnCl2) has been widely used as a reducing agent in labeling procedure of pharmaceutical with radionuclide, it has been realized that SnCl2 have genotoxic and cytotoxic effects on biological systems. In previous studies, it has been shown that some herbal extract can reduce genotoxic and cytotoxic effects of SnCl2. In the present study, it is aimed to evaluate the effect of the broccoli extract on the survival of E. coli ATCC 25922 strain against to toxic effects of SnCl2. METHODS: Broccoli was extracted with methanol extraction. HPLC and TLC analysis of broccoli extract were performed. Then antitoxicity and dose response assays were performed on bacterial strain. RESULTS: The broccoli extract had dose dependent protective effect against SnCl2 toxic effect on E. coli. CONCLUSIONS: The consumption of broccoli may alter the stannous dichloride toxicity. Broccoli extract may use as a new protective strategies against the toxic effect of SnCl2 on patients who were taken 99mTc radiopharmaceuticals.
Broccoli; Stannous dichloride; Technetium-99m; Escherichia coli; Toxicity
OBJETIVO: Em face de suas características físico-químicas, o Tecnécio-99m (99mTc) é um radiofármaco amplamente utilizado na Medicina Nuclear. Todavia, o dicloreto de estanho (SnCl2) tem sido largamente aplicado como um agente redutor no procedimento farmacêutico de marcação com radionuclídeos. Constatou-se que o SnCl2 apresenta efeitos genotóxicos e citotóxicos nos sistemas biológicos. Em estudos prévios, foi demonstrado que alguns extratos de ervas podem reduzir tais efeitos. O estudo atual objetivou avaliar os efeitos do extrato de brócolis na sobrevida da cepa E. coli ATCC 25922, exposta ao efeito tóxico do SnCl2. MÉTODOS: O extrato de brócolis foi obtido mediante extração com metanol. Analises com HPLC e TLC foram efetuadas. Avaliou-se a antitoxicidade e realizou-se um ensaio dose-resposta para uma cepa de bactérias. RESULTADOS: O extrato de brócolis mostrou um efeito protetor dose dependente para os efeitos tóxicos do SnCl2 sobre a E. coli. CONCLUSÕES: O consumo de brócolis pode alterar a toxicidade do dicloreto de estanho. O extrato de brócolis pode ser utilizado como uma nova estratégia para proteção de pacientes contra os efeitos tóxicos do SnCl2, nos quais foi administrado o radiofármaco Tecnécio-99m.
Brassica; Compostos Estanho; Tecnécio; Escherichia coli; Toxicidade
3 - ORIGINAL ARTICLE
EFFECTS OF DRUGS
Anti toxic effect of broccoli extract on stannous dichloride toxicity1 1 Research performed at Department of Nuclear Applications, Institute of Nuclear Sciences, Ege University, 35100, Bornova, Izmir-Turkey and Adnan Menderes University, Science & Technology Research & Development Central, (ADUBILTEM), 09100, Aydin-Turkey.
Efeito antitóxico do extrato de brócolis na toxicidade do dicloreto de estanho
Betul CekicI; Fazilet Zumrut Biber MuftulerII; Ayfer Yurt KilcarIII; Necati GunayIV; Serhan SakaryaV; Perihan UnakVI
IFellow Master degree, Department of Nuclear Applications, Institute of Nuclear Sciences, Ege University, Bornova, Izmir-Turkey. Carried out the study and drafted the manuscript
IIAssociate Professor, Department of Nuclear Applications, Institute of Nuclear Sciences, Ege University, Bornova, Izmir-Turkey. Responsible for conception, design, intellectual and scientific content of the study, supervised all phases of the study, manuscript writing and critical revision
IIIFellow PhD degree, Research Assistant, Department of Nuclear Applications, Institute of Nuclear Sciences, Ege University, Bornova, Izmir-Turkey. Involved in technical procedures and extract preparation
IVFellow PhD degree, Research Assistant, ADUBILTEM, Adnan Menderes University, Aydin-Turkey. Involved in technical procedures and bacteria preparation
VFull Professor, ADUBILTEM, Adnan Menderes University Aydin-Turkey. Designed the studies concerning bacteria, analysis and interpretation of data, critical revision
VIFull Professor, Department of Nuclear Applications, Institute of Nuclear Sciences, Ege University, Bornova, Izmir-Turkey. Analysis and interpretation of data, critical revision.
Correspondence Correspondence: Fazilet Zumrut Biber Muftuler Department of Nuclear Applications, Institute of Nuclear Sciences Ege University 35100 Bornova, Izmir, Turkey Tel.: +90 232 311 3467 Fax: +90-232-311 3433 fazilet.zumrut.biber@ege.edu.tr, fazilet.zumrut.biber@gmail.com
ABSTRACT
PURPOSE: Since Technetium-99m (99mTc) has favorable physical and chemical characteristics, it is widely used radioisotope in Nuclear Medicine. However, stannous dichloride (SnCl2) has been widely used as a reducing agent in labeling procedure of pharmaceutical with radionuclide, it has been realized that SnCl2 have genotoxic and cytotoxic effects on biological systems. In previous studies, it has been shown that some herbal extract can reduce genotoxic and cytotoxic effects of SnCl2. In the present study, it is aimed to evaluate the effect of the broccoli extract on the survival of E. coli ATCC 25922 strain against to toxic effects of SnCl2.
METHODS: Broccoli was extracted with methanol extraction. HPLC and TLC analysis of broccoli extract were performed. Then antitoxicity and dose response assays were performed on bacterial strain.
RESULTS: The broccoli extract had dose dependent protective effect against SnCl2 toxic effect on E. coli.
CONCLUSIONS: The consumption of broccoli may alter the stannous dichloride toxicity. Broccoli extract may use as a new protective strategies against the toxic effect of SnCl2 on patients who were taken 99mTc radiopharmaceuticals.
Key words: Broccoli. Stannous dichloride. Technetium-99m. Escherichia coli. Toxicity.
RESUMO
OBJETIVO: Em face de suas características físico-químicas, o Tecnécio-99m (99mTc) é um radiofármaco amplamente utilizado na Medicina Nuclear. Todavia, o dicloreto de estanho (SnCl2) tem sido largamente aplicado como um agente redutor no procedimento farmacêutico de marcação com radionuclídeos. Constatou-se que o SnCl2 apresenta efeitos genotóxicos e citotóxicos nos sistemas biológicos. Em estudos prévios, foi demonstrado que alguns extratos de ervas podem reduzir tais efeitos. O estudo atual objetivou avaliar os efeitos do extrato de brócolis na sobrevida da cepa E. coli ATCC 25922, exposta ao efeito tóxico do SnCl2.
MÉTODOS: O extrato de brócolis foi obtido mediante extração com metanol. Analises com HPLC e TLC foram efetuadas. Avaliou-se a antitoxicidade e realizou-se um ensaio dose-resposta para uma cepa de bactérias.
RESULTADOS: O extrato de brócolis mostrou um efeito protetor dose dependente para os efeitos tóxicos do SnCl2 sobre a E. coli.
CONCLUSÕES: O consumo de brócolis pode alterar a toxicidade do dicloreto de estanho. O extrato de brócolis pode ser utilizado como uma nova estratégia para proteção de pacientes contra os efeitos tóxicos do SnCl2, nos quais foi administradooradiofármaco Tecnécio-99m.
Descritores: Brassica. Compostos Estanho. Tecnécio. Escherichia coli. Toxicidade.
Introduction
Technetium-99m radiopharmaceuticals have favorable physical and chemical characteristics1,2 and widely used in Nuclear Medicine practice1,3. Since stannous dichloride (SnCl2) is the most common reducing agent in 99mTc radiopharmaceuticals preparation, it has been shown that severe biological effects on central nervous system and oral mucosa in animal studies1,4. Also it has been reported that SnCl2 had lethal effect on Escherichia coli (E. coli) with cytotoxic and genotoxic effects5-8. Genotoxic effect of SnCl2 may be directly on deoxyribonucleic acid (DNA) or indirectly by generating free radicals9.
In environment oxygen and stannous (II) may carry out some reactions and these reactions, leads to mutations in the DNA and it caused death of the cell by the formation of hydrogen peroxide (H2O2), as follows10-12.
Reaction I: Stannous (II) + O2→ Stannous (III) + O2-
Reaction II: Stannous (III) + O2- + 2H+ → Stannous (IV) + H2O2
Reaction III: O2 + Stannous (II) + 2H+ → H2O2 + Stannous (IV)
In recent studies, some authors have also described that genotoxic and cytotoxic effects of SnCl2 can be altered by herbal extract, chemicals and pharmaceutics on E. coli5,6,8-13.
Lima et al.6 demonstrated that cauliflower from Brassicacea family was abolished the lethal effect of SnCl2 on the E. coli strains.
In our study we used broccoli extract which is widely consumed in Turkey14. Broccoli (Brassica oleracea italica) from the Brassicaceae family is a nutrient source of bioactive components including glucosinolates, flavonoids, minerals and antioxidants15-17. In the current study we aimed to evaluate the effect of the broccoli extract on the survival of E. coli ATCC 25922 strains against to toxic effect of SnCl2.
Methods
Broccoli was extracted with methanol extraction. HPLC and TLC analysis of broccoli extract were performed. Then antitoxicity and dose response assays were performed on bacterial strain.
Extraction of broccoli
A similar procedure to that for the extraction of broccoli has been applied as described previously19. Broccoli was purchased from local market, dried at room temperature and powdered. The samples (250 mg) were extracted with 5 mL of methanol/water (60:40, v/v) using ultrasonic bath (Ceia P104) for 60 min. at room temperature. The slurry mixture was centrifuged at 2500 rpm for 15 min. The supernatant was collected and 5 mL of methanol/water (60:40, v/v) was added in the remaining parts in the tube. All procedure was repeated 3 times. All the supernatants collected and kept at -20 0C until the experiments.
High performance liquid chromatography (HPLC)
The procedure used for HPLC for broccoli extract was similar to that reported in other studies19. A low pressure gradient HPLC system (LC-10ATvp quaternary pump, SPD-10A/V UV detector, RF-10AXL Fluorescence detector, RAD501 single channel analyzer, a syringe injector equipped with a 1 mL loop and 7-µm VP 250/21 Nucleosil 100-7 C18 column (Macherey - Nagel)) was used for the analytical experiments.
HPLC of Broccoli Extract: Fluorescence was monitored with a model RF-10AXL Shimadzu Fluorescence Detector. The samples were analyzed at a flow rate of 1 mL/min and injection volume was 20 µL. The excitation was monitored at 290 nm and emission at 360 nm. The mobile phase was consisted of two solvents which were water-formic acid (0.33%) (A) and methanol (B). The elution gradient profile used was as 10-17% B in 30 min., 17-40% B in 10 min., 40-100% B in 1 min., 100% B in 5 min. Retention time (Rt) values were given in Figure 1.
Thin layer chromatography (TLC)
Five mL of broccoli extract was applied on ITLC-SG (Merck-5554) plates. As mobile phase, 60% methanol in water was used. Then UV lamp was used to determine location of components in extract. Relative front (Rf) values of the components were calculated.
Antitoxicity assay
Bacterial strain
The E. coli ATCC 25922 strain was provided from Adnan Menderes University Science and Technology Research and Development Center (ADUBILTEM Aydin, Turkey) Epidemiology Laboratory and used in all experiments. The bacteria concentration of ~108 cfu/mL was monitored spectrophotometrically as optical density of 0.1 at A625 nm (Thermolabsystems Multiscan Spectrum).
Bacterial viability assay
For determining the effect of SnCl2 and extract on bacteria viability, bacteria were resuspended in PBS at 108 CFU/ml and incubated with i. SnCl2 ii. Extract + SnCl2 and iii. Extract (as control group) with the 1, 3, 10, 30, 100 µg concentrations for both SnCl2 and extract for 3h at 35ºC. After the incubation bacteria was washed twice in PBS and resuspended. Samples were diluted appropriately and plated on Mueller Hinton Agar (Merck). Following 24h incubation at 35ºC, the total number of bacteria for each dilution was counted and colony-forming units per milliliter (cfu/mL) were determined. The survival fraction (N/N0) was calculated as previously described6.
Dose response of extract
To determine the protective effect of extract; amount of extract was increased 100 µg and 300 µg while SnCl2 concentrations remain stable at 30 µg. Bacterial assay were performed as described above.
Statistical analysis
All experiments were repeated three times and analysis of variance (ANOVA) was used to compare the mean responses among experimental and control groups. Probability values p<0.05 were considered as significant.
Results
HPLC and TLC studies of broccoli extract were performed. The relevant chromatogram was given in Figure 1. According to HPLC chromatogram, Rt values of broccoli extract were 3.00, 11.50, 14.50 respectively.
We have concluded that both of the Rt values were similar (as seen in Figure 1). Table 1 was showed Rf values of broccoli extract by TLC methods.
Figure 2 represents survival fractions of bacteria in the presence of broccoli extract. In the control group (Extract), there was no difference in survival fraction values for all concentrations. Survival fraction of E. coli was decreased after 3 µg/mL SnCl2 with future dose-dependent decrements at 10 µg/mL as seen in Figure 2.
When we compare SnCl2 group with Extract+SnCL2 group, similarly survival fraction was decreased after 3 µg/mL SnCl2. In Extract + SnCl2 group, E. coli survival fraction is 0.005 at 100 µg. It was seen, at 100 µg/mL, survival fraction of E. coli in Extract + SnCl2 group was more than SnCl2 group. These data suggest that, SnCl2 had toxic effect on E. coli. However, in Extract+ SnCl2 group survival fraction was expected increasing after 100 µg/mL.
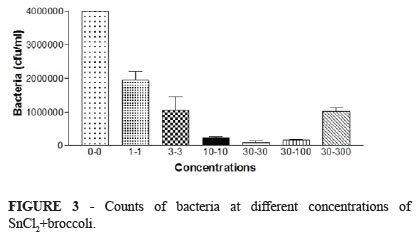
Also, survival fraction values of all groups were the same up to 3 µg. Survival fraction value of Extract + SnCl2 group (0.005) was higher than group of SnCl2 (0.00) at 100 µg concentration (p<0.05). These data thought that extract protect E. coli against toxic effect of SnCl2 at high concentrations. So, we decided to do a dose response study for to determine extract protective effect with high concentrations (Figure 3). For this aim, 30 µg SnCl2 + 100 µg extract and 30 µg SnCl2+ 300 µg extract group were prepared addition to 1-1, 3-3, 10-10 and 30-30 µg concentrations were used in bacteria viability assay. As shown in Figure 3, viable bacteria in 30 µg SnCl2 + 300 µg extract group was 10.82 fold (p=0.001) high compared to 30 µg SnCl2 + 30 µg extract group.
Discussion
Our HPLC results were confirmed with Lin et al.20 LC-MS study results with phenolic components of colloid greens, kale and Chinese broccoli. The results of HPLC and TLC analysis were compatible.
Although its toxic effects were well documented, SnCl2 has been used as 99mTc reducing agent in majority of 99mTc radiopharmaceutical kits. In previous studies it was reported that genotoxic effect mediated by the production of reactive oxygen species on pro- and eukaryotic microbial test systems1,7. So SnCl2 may have harmful effect on E. coli.
In recent studies, it was reported; the genotoxic and cytotoxic effects of SnCl2 can be altered by herbal extract, chemicals and pharmaceutics on E. coli5,8,10-13. One of these studies was performed by using cauliflower which is in the same family with broccoli. In this study, E. coli AB1157 strain was treated with SnCl2 in the presence of cauliflower and it abolished the lethal effect of SnCl2 on the E. coli strains6.
In another study, it was evaluated the influence of the Cymbopogon citratus, Maytenus ilicifolia and Baccharis genistelloides crude extracts on the survival of E. coli AB1157 (wild type) strain. Maytenus ilicifolia extract had high protection effect on E. coli survival compared to other extracts and this protection was highly related with antioxidant and/or oxidant properties of the extracts11. Similarly, the Ganoderma lucidum (reishi) extract had protective effect on E. coli against the oxidative effect of SnCl2, and the chemical compounds in reishi extract had redox/chelating activity5. Although, it has been shown that some other herbal extracts have been eliminated the toxic effects of SnCl25,8,9,11, action of mechanisms are not well known yet.
Broccoli has nutritional antioxidants, such as vitamins C and E, but also a great quantity of non-nutritional antioxidants, such as flavonoids, flavones, and other polyphenolic compounds16,21. In our study broccoli extract had dose dependent protective effect against SnCl2 toxic effect on E. coli. We suggest that consumption of broccoli may alter the SnCl2 toxicity.
Conclusions
The consumption of broccoli may alter the stannous dichloride toxicity. Broccoli extract may use as a new protective strategies against the toxic effect of SnCl2 on patients who were taken 99mTc radiopharmaceuticals.
Acknowledgement
The authors would like to thank to PhD student Hasan Zora for helping.
Received: April 25, 2012
Review: June 22, 2012
Accepted: July 24, 2012
Conflict of interest: none
Financial source: Ege University Research Fund (contract no 2010 NBE 007)
- 1. Guedes AP, Cardosoc VN, Mattosa JCPD, Dantasa FJS, Matosa VC, Silva JCF, Bezerra RJAC, Caldeira-de-Araujo A. Cytotoxic and genotoxic effects induced by stannous chloride associated to nuclear medicine kits. Nucl Med Biol. 2006;33:915-21.
- 2. Hladik WB, Saha GB, Study KT. Essentials of nuclear medicine science. London: Williams and Wilkins; 1987.
- 3. Paoli S, Dias APM, Capriles PVSZ, Costa TEMM, Fonseca AS, Bernardo-Filho M. Effects of a tomato (Solanum lycopersicum) extract on the labeling of blood constituents with technetium-99m. Rev Bras Farmacogn. 2008;18:190-6.
- 4. Silva CR, Oliveria MBN, Melo SF, Dantas FJS, Mattos JCP, Bezerra RJAC, Caldeira-de-Araujo A, Duatti A, Bernardo-Filho M. Biological effects of stannous chloride, a substance that can produce stimulation or depression of the central nervous system. Brain Res Bull. 2002;59:213-6.
- 5. Agostinho RT, Santos-Filho SD, Fonseca AS, Missailidis S, Bernardo-Filho M. The effect of an extract from Ganoderma lucidum (Reishi) on the labeling of blood constituents with Technetium-99m and on the survival of Escherichia coli. Braz Arch Biol Technol. 2008;51:157-62.
- 6. Lima EAC, Dire G, Mattos DMM, Freitas RS, Gomes ML, Oliveira MBN, Faria MVC, Jales RL, Bernardo-Filho M. Effect of an extract of cauliflower (leaf) on the labeling of blood elements with technetium-99m and on the survival of Escherichia coli AB1157 submitted to the treatment with stannous chloride. Food Chem Toxicol. 2002;40:919-23.
- 7. Pungartnik C, Viaua C, Picada J, Caldeira-de-Araujo A, Henriques JAP, Brendel M. Genotoxicity of stannous chloride in yeast and bacteria. Mutat Res. 2005;583:146-57.
- 8. Reiniger IW, Silva CR, Felzenszwalb I, Mattos JCP, Oliveira JF, Dantas FJS, Bezerra RJAC, Caldeira-de-Araujo A, Bernardo-Filho M. Boldine action against the stannous chloride effect. J Ethnopharmacol. 1999;68:345-8.
- 9. Soares SF, Brito LC, Souza DE, Bernardo LC, Oliveira JF, Bernardo-Filho M. Potential pitfalls in the nuclear medicine imaging: experimental models to evaluate the effect of natural products on the radiolabeling of blood constituents, bioavailability of radiopharmaceutical and on the survival of Escherichia coli strains submitted to the treatment with stannous ion. Nucl Instrum Meth A. 2006;569:505-8.
- 10. Assis MLB, Neto JBC, Souza JEQ, Caldeira-de-Araujo A, Bernardo-Filho M. Stannous chloride and the glucoheptonic acid effect: study of a kit used in nuclear medicine. Cancer Lett. 1998;130:127-31.
- 11. Melo S.F., Soares S.F., Costa R.F., Silva C.R., Oliveira M.B.N., Bezerra R.J.A.C., Caldeira-de-Araújo A, Bernardo-Filho M. Effect of the Cymbopogon citratus, Maytenus ilicifolia and Baccharis genistelloides extracts against the stannous chloride oxidative damage in Escherichia coli. Mutat Res. 2001;496:33-8.
- 12. Dantas FJS, Moraes MO, Mattos JCP, Bezerra RJAC, Carvalho EF, Bernardo-Filho M, Caldeira de Araujo A. Stannous chloride mediates single strand breaks in plasmid DNA through reactive oxygen species formation. Toxicol Lett. 1999;110:129-36.
- 13. Santos-Filho SD, Diniz CL, Carmo FS, Fonseca AS, Bernardo-Filho M. Influence of an extract of Juglans regia on the growth of Escherichia coli, on the electrophoretic profile of plasmid DNA and on the radiolabeling of blood constituents. Braz Arch Biol Technol. 2008;51:163-8.
- 14. Sarikamis G, Balkaya A, Yanmaz R. Glucosinolates in Kale Genotypes from the Blacksea Region of Turkey. Biotechnol Biotec Eq. 2008;22:942-6.
- 15. Price KR, Casuscelli F, Colquhoun IJ, Rhodes MJC. Composition and content of flavonol glycosides in broccoli florets (Brassica olearacea) and their fate during cooking. J Sci Food Agric. 1998;77:468-72.
- 16. Vallejo F, Tomas-Barberan FA, Garcia-Viguera C. Potential bioactive compounds in health promotion from broccoli cultivars grown in Spain. J Sci Food Agric. 2002;82:1293-7.
- 17. Moreno DA, Carvajal M, Lopez-Berenguer C, Garcia-Viguera C. Chemical and biological characterisation of nutraceutical compounds of broccoli. J Pharm Biomed Anal. 2006;41:1508-22.
- 18. Ferreres F, Valentao P, Llorach R, Pınheıro C, Cardoso L, Pereıra JA, Sousa C, Seabra RM, Andrade PB. Phenolic compounds in external leaves of Tronchuda cabbage (Brassica oleracea L. var. costata DC). J Agric Food Chem. 2005;53:2901-7.
- 19. Cekic B, Biber Muftuler FZ, Yurt Kilcar A, Ichedef C, Unak P. Effects of broccoli extract on biodistribution and labeling blood components with 99mTc-GH. Acta Cir Bras. 2011;26:339-45.
- 20. Lin LZ, Harnly JM. Identification of the phenolic components of collard greens, kale, and Chinese broccoli. J Agric Food Chem. 2009;57:7401-8.
- 21. Lin CH, Chang CY. Textural change and antioxidant properties of broccoli under different cooking treatments. Food Chem. 2005;90:9-15.
Publication Dates
-
Publication in this collection
27 Aug 2012 -
Date of issue
Sept 2012
History
-
Received
25 Apr 2012 -
Accepted
24 July 2012 -
Reviewed
22 June 2012