Abstracts
PURPOSE: To investigate the effects of intravenous L-arginine (LG) infusion on liver morphology, function and proinflammatory response of cytokines during the early phase of ischemia-reperfusion injury (IRI). METHODS: Thirty rabbits were subjected to 60 minutes of hepatic ischemia and 120 minutes of reperfusion. An intravenous injection of saline or L-arginine was administered five minutes before the ischemia and five minutes before initiating the reperfusion and at the 55th and 115th minutes after the ischemia. Samples were collected for histological analysis of the liver and measurements of the serum AST, ALT and LDH and the cytokines IL-6 and TNF-alpha. RESULTS: It was observed a significant reduction of sinusoidal congestion, cytoplasmic vacuolization, infiltration of polymorphonuclear leukocyte, nuclear pyknosis, necrosis and steatosis in liver tissue, as well as AST, ALT and LDH after injection of LG in the ischemia (p <0.001). Lower levels of IL-6 and TNF-alpha were associated with LG infusion during ischemia. Higher levels these proteins were observed in animals receiving LG during reperfusion. CONCLUSION: L-arginine protects the liver against ischemia/reperfusion injury, mainly when is administered during the ischemic phase.
Nitric Oxide; Arginine; Warm Ischemia; Cytokines; Antioxidants; Interleukin-6; Liver; Rabitts
OBJETIVO: Investigar os efeitos da infusão endovenosa da L-arginina (LG) na morfologia, função e resposta de citocinas pró-inflamatórias do fígado durante a fase precoce da lesão de isquemia e reperfusão (IRI). MÉTODOS: Trinta coelhos foram submetidos a 60 minutos de isquemia hepática e 120 minutos de reperfusão. Foi administrada injecção intravenosa de solução salina ou L-arginina aos cinco minutos antes de iniciar a isquemia e cinco minutos antes de iniciar a reperfusão e aos 55 e 115 minutos após o início da isquemia. Realizou-se análise histológica do fígado e dosagens séricas de AST, ALT, LDH, citocinas IL-6 e TNF-alfa. RESULTADOS: Ocorreu redução significante da congestão sinusoidal, vacuolização citoplasmática, infiltração de leucócitos polimorfonucleares, picnose nuclear, necrose e esteatose no tecido hepático, assim como nos níveis de AST, ALT e LDH após a injeção da LG na isquemia (p<0,001). Níveis mais baixos de IL-6 e TNF-alfa foram associados com a infusão LG durante a isquemia. Níveis mais elevados dessas proteínas foram observados nos animais que receberam LG durante a reperfusão. CONCLUSÃO: A L-arginina protegeu o fígado contra a lesão de isquemia e reperfusão principalmente quando administrada durante a fase de isquemia.
Óxido Nítrico; Arginina; Isquemia Quente; Citocinas; Antioxidantes; Interleucina-6; Fígado; Coelhos
5 - ORIGINAL ARTICLE
ISCHEMIA-REPERFUSION
L-arginine in the ischemic phase protects against liver ischemia-reperfusion injury1 1 Research performed at Laboratory of Experimental Surgery and Operative Technique, Department of Morphology, Laboratory of Histology and Structural Biology, Department of Pharmacology and Department of Biochemistry, Federal University of Sao Paulo (UNIFESP), Brazil.
A L-arginina durante a fase isquêmica protege o fígado das lesões de isquemia e reperfusão
Murched Omar TahaI; Afonso Caricati-NetoII; Regiane Miranda FerreiraIII; Manuel de Jesus SimõesIV; Hugo Pequeno MonteiroV; Djalma José FagundesVI
IPhD, Associate Professor, Operative Technique and Experimental Surgery Division, Department of Surgery, UNIFESP, Sao Paulo-SP, Brazil. Main author. Responsible for conception, design, intellectual and scientific content of the study
IIPhD, BSc, Associate Professor, Department of Pharmacology, UNIFESP, Sao Paulo-SP, Brazil. Acquisition and interpretation of pharmacological data, manuscript writing
IIIPhD, BSc, Post-doctoral Fellow, Department of Pharmacology, UNIFESP, Sao Paulo-SP, Brazil. Acquisition and interpretation of pharmacological data, manuscript writing
IVPhD, Associate Professor, Histology and Structural Biology Division, Department of Morphology, UNIFESP, Sao Paulo-SP, Brazil. Interpretation of data and histophatological analysis
VPhD, BSc, Associate Professor of Biochemistry, Department of Biochemistry, UNIFESP, Sao Paulo-SP, Brazil. Interpretation of biochemistry data and manuscript writing
VIPhD, Associate Professor, Operative Technique and Experimental Surgery Division, Department of Surgery, UNIFESP, Sao Paulo-SP, Brazil. Conception and critical revision of the study.
Correspondence Correspondence: Murched Omar Taha Rua Joaquim Távora, 1599/161 04015-003 São Paulo - Brasil Tel.: (55 11)5084-0100 / 9982-0001 Fax: (55 11)5571-0233 taha@uol.com.br
ABSTRACT
PURPOSE: To investigate the effects of intravenous L-arginine (LG) infusion on liver morphology, function and proinflammatory response of cytokines during the early phase of ischemia-reperfusion injury (IRI).
METHODS: Thirty rabbits were subjected to 60 minutes of hepatic ischemia and 120 minutes of reperfusion. An intravenous injection of saline or L-arginine was administered five minutes before the ischemia and five minutes before initiating the reperfusion and at the 55th and 115th minutes after the ischemia. Samples were collected for histological analysis of the liver and measurements of the serum AST, ALT and LDH and the cytokines IL-6 and TNF-alpha.
RESULTS: It was observed a significant reduction of sinusoidal congestion, cytoplasmic vacuolization, infiltration of polymorphonuclear leukocyte, nuclear pyknosis, necrosis and steatosis in liver tissue, as well as AST, ALT and LDH after injection of LG in the ischemia (p <0.001). Lower levels of IL-6 and TNF-alpha were associated with LG infusion during ischemia. Higher levels these proteins were observed in animals receiving LG during reperfusion.
CONCLUSION: L-arginine protects the liver against ischemia/reperfusion injury, mainly when is administered during the ischemic phase.
Key words: Nitric Oxide. Arginine. Warm Ischemia. Cytokines. Antioxidants. Interleukin-6. Liver. Rabitts.
RESUMO
OBJETIVO: Investigar os efeitos da infusão endovenosa da L-arginina (LG) na morfologia, função e resposta de citocinas pró-inflamatórias do fígado durante a fase precoce da lesão de isquemia e reperfusão (IRI).
MÉTODOS: Trinta coelhos foram submetidos a 60 minutos de isquemia hepática e 120 minutos de reperfusão. Foi administrada injecção intravenosa de solução salina ou L-arginina aos cinco minutos antes de iniciar a isquemia e cinco minutos antes de iniciar a reperfusão e aos 55 e 115 minutos após o início da isquemia. Realizou-se análise histológica do fígado e dosagens séricas de AST, ALT, LDH, citocinas IL-6 e TNF-alfa.
RESULTADOS: Ocorreu redução significante da congestão sinusoidal, vacuolização citoplasmática, infiltração de leucócitos polimorfonucleares, picnose nuclear, necrose e esteatose no tecido hepático, assim como nos níveis de AST, ALT e LDH após a injeção da LG na isquemia (p<0,001). Níveis mais baixos de IL-6 e TNF-alfa foram associados com a infusão LG durante a isquemia. Níveis mais elevados dessas proteínas foram observados nos animais que receberam LG durante a reperfusão.
CONCLUSÃO: A L-arginina protegeu o fígado contra a lesão de isquemia e reperfusão principalmente quando administrada durante a fase de isquemia.
Descritores: Óxido Nítrico. Arginina. Isquemia Quente. Citocinas. Antioxidantes. Interleucina-6. Fígado. Coelhos.
Introduction
Ischemia-reperfusion injury (IRI) is associated with multiple events that affect liver function following transient episodes of hepatic ischemia, such as solid organ transplantation, trauma, hypovolemic shock, and elective liver resection, when inflow occlusion or total vascular exclusion is used to minimize blood loss1. Alterations in the hepatic microcirculation and a decrease in hepatic flow can cause local hypoxia and, consequently, liver damage. The liver is further compromised following the reintroduction of blood flow and oxygen delivery to the damaged areas2.
One of the earliest events associated with the reperfusion of an ischemic liver is endothelial dysfunction that is characterized by a decreased production of endothelial cell-derived nitric oxide (eNO)3. This rapid post-ischemic decrease in NO bioavailability appears to be due to a decrease in the synthesis of NO, enhanced inactivation of NO via the overproduction of superoxide, or both phenomena3,4.
NO availability has been shown to be beneficial in various experimental models5-10.Organ infusion with NO donors or supplementation with L-arginine (LG), a substrate for NO synthases (NOS), minimizes hepatic injury by improving the microcirculation11. In animal models of IRI, inhibitors of NOS promote tissue injury11. However, high tissue levels of NO may also have harmful effects12-16. Collectively, these observations suggest a balance between the local NO concentration and the time of NO exposure in determining the outcome of liver IRI8. Irrespective of the precise mechanisms involved, increased inflammation and cytotoxicity are key components in hepatocellular dysfunction during the pathogenesis of liver IRI16-20.
The development of new therapeutic approaches to control IRI may be aided by a better understanding of the defense mechanisms that occur in the liver when it is subjected to ischemic insults 21. The LG-NOS pathway has been critically implicated in liver IRI, and most studies have suggested that intravenous LG infusion increases NO levels and protects the liver against damage due to IRI8-20.
The purpose of the present study was to evaluate the effects of intravenous LG infusion on liver morphology and function and to evaluate the proinflammatory response of cytokines during the early phase of IRI in an experimental model using rabbits.
Methods
The experimental protocol (1815/08) was approved by the Ethics Committee of the Federal University of Sao Paulo (UNIFESP), Brazil, on according to the recommendations of the international legislations on animal protection. The study was designed as a randomized controlled trial with a blinded assessment of the outcome.
Thirty male New Zealand white rabbits (Center for the Development of Experimental Models for Medicine and Biology - CEDEME-UNIFESP) that weighed 2.500 to 3.000g were housed under temperature - and light - controlled environmental conditions with a 12-hour light-dark cycle. The animals had free access to water and standard pellet chow until six hours prior to the surgical procedures. All procedures were conducted in the laboratories of Experimental Surgery, Department of Surgery (UNIFESP).
The animals were randomly assigned to one of four groups (Figure 1): the ischemia plus saline (I-SS) group (n=6), the ischemia and reperfusion plus saline (IR-SS) group (n=6), the ischemia plus LG (I-LG) group (n=6), and the ischemia and reperfusion plus LG (IR-LG) group (n=6). All animals were subjected to 60 minutes of lobar ischemia and 120 minutes of reperfusion. In the I-SS and I-LG groups, an intravenous administration of saline or LG, respectively, was performed both five minutes (T-5) before starting the ischemia and five minutes (T55) before starting the reperfusion. In the IR-SS or IR-LG groups, saline or L-arginine, respectively, was administered at both the 55th (T115) and 115th (T175) minutes during the reperfusion. At 120 minutes after starting the reperfusion (T-180), the animals were euthanized.
Anesthesia and euthanasia procedures
After 6h of fasting for a solid diet and 4h for a liquid diet, the animals received a combination of 80 mg/kg of ketamine i.m. (KetalarTM, Medical Division of Pfizer do Brazil) and 10 mg/kg of xylazine i.m. (RompumTM, Bayer, Sao Paulo, Brazil). Anesthesia was maintained connected to a volume-controlled ventilator (Harvard ventilator, Harvard Apparatus, USA). The animals were ventilated at a rate of 30 breaths/min with a tidal volume of 10 mL/kg, an inspired oxygen fraction of 1.0, and a positive end-expiratory pressure of 2 cmH2O. The marginal ear artery and vein were catheterized, and the systemic arterial pressure, oxygen saturation (SpO2), and heart rate were continuously monitored (Datex Ohmeda Type F-CU8, Helsinki, Finland). Sodium heparin (500 IU/kg) was administered via the peripheral ear vein. Body temperature was maintained at 37.8ºC using a rabbit homeothermic soft blanket (40 x 70 cm) with a YSI 401 rectal temperature probe.
After the samples were collected, the animals received a combination of intravenous (0.3 ml/Kg) embutamide, mebezone and tetracain (T61TM, Schering Plough, Sao Paulo, Brazil), which promotes rapid death.
Surgical procedure
Under aseptic conditions, all 30 animals underwent a midline laparotomy. The hepatic hilum was carefully dissected with identification of the hepatic artery, portal vein, and bile duct. The left and median hepatic lobes were occluded with a small vascular clamp. Ischemia was confirmed by observing the pale appearance of the clamped liver lobes. The appearance of the intestines and signs of portal compression were also observed. A SpO2 after the clamp was removed, reperfusion was evaluated based on immediate color recovery. In cases in which an absence of ischemic color or a lack of response to the reperfusion was observed, the animals were excluded from further analyses. The surgical wounds remained covered with wet gauze wrappings throughout the experiment to minimize evaporative loss. Sham animals underwent anesthesia, laparotomy, and exposure of the portal triad without hepatic ischemia.
Serum assays
Blood samples were collected after 120 minutes of reperfusion. The samples were centrifuged, and the serum was separated. Hepatic damage was evaluated (units per liter) based on the serum levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and lactate dehydrogenase (LDH), which were measured using an in vitro diagnostic chemistry reagent kit (Catachem Inc., Bridgeport, Conn).
Histological processing
Liver samples were immersed in 10% buffered formalin. After 24 hours, the samples were embedded in paraffin, cut into 5-µm sections and stained with hematoxylin and eosin. All histological analyses were performed by a single pathologist who was blinded to the treatment of the groups. We used a computerized image analysis system consisting of a NikonTM FDX - 35 video camera coupled to a Nikon optic microscope (Eclipse 800, Tokyo, Japan) with multicolor objectives that transmitted images to a Pentium MMX MHz Vaio PC (Sony, Tokyo, Japan) equipped with a digitizing board and Image Pro-PlusTM software (Version 4.5) (Media Cybernetics, Inc., Bethesda, MD, USA).
Sinusoidal congestion, cytoplasmic vacuolation, infiltrating polymorphonuclear leukocytes (PMNLs), nuclear pyknosis, hepatocyte necrosis and steatosis were all assessed using a colorimetric scale (red, green and blue). Each parameter was marked individually and expressed as a percentage of the total area of each slide.
Real-time PCR for TNF-alpha and IL-6
Frozen liver tissues were stored liquid nitrogen to evaluate the gene expression of TNF-alpha (F:5′-GCTCCTC ACTCACACTGTCAG-3′ and R:5′-GCAATGATCCCAAAG TAGAC-3′), IL-6 (F:5′-GCTGAGGAAAGAGATGTGTGAC-3′ and R:5′ GTCTTGATCAGGTGTTGGATGT-3′), and the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (F:5′-GATTTGGCCGCATTGG-3′ and R:5′-ACATCCACTTTGCCAGAGTTAA-3′).
Total RNA was prepared from the frozen liver tissues using the TRIzol/chloroform extraction method (Invitrogen, Paisley, UK), reverse transcribed into cDNA using an iScript cDNA Synthesis Kit, and subjected to quantitative real-time PCR (RT-PCR) using Platinum SYBR Green qPCR SuperMix-UDG (Invitrogen Inc., NY, USA) and the primers listed above with an iCycler (Bio-Rad) instrument and the associated software. All of the PCR oligonucleotide primer pairs were designed using Bio-Rad Beacon Designer 2.0 software and purchased from Invitrogen. The δδCT values were calculated by subtracting the CT values for the gene of interest from those obtained for GAPDH and then subtracting the CT value for each gene from those determined for the non-operated control rabbits. The fold induction was calculated by normalizing the values to those obtained for the control rabbits.
Total RNA was treated with DNase (Invitrogen Inc., NY, USA), and separated by gel electrophoresis. Reverse-transcribed cDNA was then subjected to real time PCR analysis using the ABI PRISM 7000 Sequence Detection System (Applied Biosystems, CA, USA). The reaction mixtures had a final volume of 20 µL and contained 1 µL of cDNA (per sample) and 9 µL of 2X Platinum SYBR Green qPCR SuperMix-UDG (Invitrogen Inc., NY, USA) (40 mM Tris-HCl (pH 8.4), 100 mM KCl, 6 mM MgCl2, 400 µM dATP, 400 µM dCTP, 400 µM dGTP, 800 µM dUTP, 40 units/mL UDG, 60 units/mL Platinum Taq DNA Polymerase, and stabilizers)22-24. The gene fragments were amplified by 30 cycles of PCR, with each consisting of 15 s at 94 C, 30 s at 55ºC, and 1 min at 72ºC.
Statistical analysis
Parametric results are expressed as the mean ± standard deviation, and the Shapiro-Wilks test was performed to check the normality of the data prior to statistical evaluation. ANOVA (one-way or two-way factorial variance for comparisons of ischemia versus reperfusion) and Tukey's post-hoc test were used to identify the two-way variance in the same group (SPSS ver. 18.0, SPSS Inc., Chicago, IL, USA). A p<0.05 was considered significant.
Results
Histological analysis
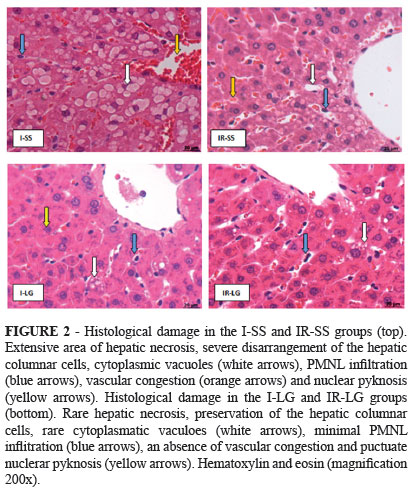
Histological analysis of the liver revealed multiple, extensive areas of hepatocyte necrosis that were randomly distributed throughout the parenchyma in the IR-SS group (Figure 2, top). The results were similar in the I-SS group (Figure 2 top), but the areas of necrosis were slightly less frequent. Furthermore, sinusoidal congestion and cytoplasmic vacuolation were randomly disseminated in the latter group. In the I-LG and IR-LG groups (Figure 2, bottom), the morphological structure of the liver was preserved, and an absence of columnar disorder of the hepatocytes was observed. In the IR-LG group, however, cytoplasmic vacuolation and PMNL infiltration were frequently observed. The greatest reductions in areas (µm2) of sinusoidal congestion, cytoplasmic vacuolation, infiltrating PMNLs, nuclear pyknosis, hepatocyte necrosis, and steatosis were observed in the I-LG group followed sequentially by the IR-LG group, the I-SS group and the IR-SS group (Table 1).
Biochemical assessment
Consistent with the morphological analysis, the biochemical assessment of liver cytolysis showed that LG administration during the ischemic phase reduced the serum levels of AST, ALT and LDH (Figure 3). This reduction was significantly greater when LG was administered during the ischemic phase compared to during the reperfusion phase (p<0.001).
Cytokine expression
The highest level of TNF-alpha expression was detected in the IR-SS group, and a significantly lower level of expression was measured in the I-SS group (p=0.001) (Figure 4). LG infusion during the ischemic phase resulted in the lowest level of TNF-alpha expression and demonstrated a value that was significantly lower than that detected in the group that received LG during the reperfusion phase (p<0.05).
The highest expression levels of the proinflammatory cytokine IL-6 were also observed in the IR-SS group followed by the I-SS group (p<0.001) (Figure 4). LG infusion during the ischemic phase resulted in the lowest expression level of IL-6 and demonstrated a value that was significantly lower than that detected in the group that received LG during the reperfusion phase (p<0.001).
Discussion
The present results showed that intravenous LG infusion during the pre-ischemic and ischemic phases promoted greater protection against IRI damage in hepatocytes compared to LG infusion during the reperfusion phase. We demonstrated the protective effects of intravenous LG on the parenchymal integrity of the liver, reduced serum levels of enzymes associated with liver cytolysis, and inhibition of the expression of the proinflammatory cytokines TNF-alpha and IL-6.
The experimental IRI model used herein was reliable with respect to the promotion of parenchymal and functional liver damage. IRI was established by clamping the vessels to the left lateral and median hepatic lobes, which account for 70% of the rabbit liver mass25. In other animal models, it has been reported that hepatic insult is similar to the clinical situation in which the liver is rendered ischemic during total vascular exclusion for liver resection25-28. Indeed, in the present study, 60 minutes of hepatic warm ischemia followed by 120 minutes of reperfusion caused severe liver injury in rabbits, as demonstrated by the structural damage to the liver and the increased serum levels of AST, ALT, and LDH. In addition, liver IRI increased the expression levels of the proinflammatory cytokines TNF-alpha and IL-6.
IRI has been shown to occur during an early phase of reperfusion that begins following reoxygenation and during a delayed phase. The early phase is associated with hepatocellular damage during the period from 2 to 6h after reperfusion (reoxygenation), whereas the delayed phase occurs from 18 to 24h after reperfusion and is accompanied by a massive infiltration of PMNLs4,25. Liver IRI is known to be associated with microcirculatory flow disturbances, endothelial cell adhesion, and leukocyte tethering and infiltration. Local leukocyte infiltration depends on both extravasation across the vascular endothelium and migration through the extracellular matrix8. Leukocytes play an important role at the interface of the portal blood flow and exposure to pathogenic and nonpathogenic antigens. Moreover, they are an important component involved in liver IRI and function both to amplify the molecular pathways associated with IRI and to cause cellular damage directly8. After two hours of reperfusion, our IRI model displayed areas of severe necrosis and PMNL infiltration.
Together with the above-mentioned changes, the serum levels of LDH, ALT, and LDH were significantly increased in rabbits that were exposed to hepatic IRI, which indicated that cellular injury had been induced in the liver. PMNL-independent injury is thought to be initiated by changes in the redox state of the liver that produce a more oxidative environment within the cells4.
IRI is associated with a remarkable decrease in the bioavailability of NO, which represents an important initiating event in the pathophysiology of post-ischemic injury in a variety of different tissues, including the liver, heart, kidney, and gut4.
Several pharmacological agents have been used to attenuate IRI in the liver. These agents may directly inhibit the progression of the lesion, or they may induce a low level of stress in the liver cells, which in turn initiates a cellular defense mechanism against more aggressive insults during the late phase11.
NO is a gaseous free radical that exerts protective effects during IRI. It is produced by NOS enzymes that use LG and oxygen as substrates to generate L-citrulline and NO. NO is an important messenger molecule in a variety of physiological systems and serves many physiological purposes, such as the promotion of vasodilatation and the mediation of communication between cells in the nervous system1. In addition to its physiological effects, excessive levels of NO may cause cellular damage via a phenomenon known as nitrosative stress7. Thus, the role of NO in IRI is controversial. NO has been reported to have beneficial effects in attenuating organ damage due to IRI, but it may also exert harmful effects8,9. NO production via iNOS in macrophages is known to cause oxidative damage by interacting with the superoxide anion produced by the enzymatic complex NADPH oxidase. This process leads to the production of peroxynitrite, which is a potent oxidant and induces cell death10. Peroxynitrite has dual effects; it can be harmful8, or it can promote hepatoprotection via a decrease in leukocyte adhesion and infiltration13. Our results suggest that the delivery of NO is protective, depending on the phase during which it enters the cascade of events in liver IRI. Intravenous LG injection during early stages of ischemia showed a stronger protective effect than that observed during the late phase of reperfusion.
Hepatoprotection via the LG-NOS pathway has been previously established in animal models of IRI18,19. Given that the LG-NOS pathway has been reported to play critical roles during inflammation, our data support the hypothesis that the administration of LG during the ischemic phase is beneficial by attenuating liver injury in a model of warm hepatic IRI.
The transcription factor NF-κB plays a major role in inflammation and in survival signaling pathways. It participates in both protective mechanisms and in generating the inflammatory cytokines TNF-alpha and IL-6, the balance of which ultimately controls the fate of the liver during IRI29. The activation of inflammatory cytokines is mediated by reactive oxygen species and is associated with the induction of protective genes that are essential for the maintenance of liver functions29. Cytokines are key mediators within the complex interplay of intrahepatic immune cells and hepatocytes because they can activate the effector functions of immune cells and the intracellular hepatocyte signaling pathways that control cellular homeostasis. Kupffer cells and liver-infiltrating monocyte-derived macrophages are primary sources of cytokines such as TNF-alpha and IL-630. Although we did not specifically investigate the activity of Kupffer cells, our results revealed a direct relationship between the morphological and functional impairment of hepatocytes and the levels of TNF-alpha and IL-6. L-arginine infusion significantly decreased the expression of TNF-alpha and IL-6, which suggested a protective effect of LG against liver IRI during the early stage of reperfusion until 120 minutes after reperfusion.
Liver IRI comprises a number of complex processes that involve multiple cellular and molecular pathways. However, NO depletion during the early stage of reperfusion is associated with a biochemical cascade of events that results in an impairment of liver function. The administration of an exogenous NOS substrate via intravenous LG injection was effective in protecting the liver against IRI.
Conclusion
L-arginine protects the liver against ischemia/reperfusion injury, mainly when is administered during the ischemic phase.
Acknowledgment
The authors thank Mrs. Maria do Carmo Negrini Fagundes for assistance with translation and formatting of the article.
Received: April 16, 2012
Review: June 14, 2012
Accepted: July 17, 2012
Conflict of interest: none
Financial source: FAPESP
- 1. Jeyabalan G, Klune JR, Nakao A, Martik N, Wu G, Tsung A, Geller DA. Arginase blockade protects against hepatic damage in warm ischemia-reperfusion. Nitric Oxide. 2008;19:29-35.
- 2. Lanteri R, Acquaviva R, Di Giacomo C, Caltabiano R, Li Destri G, Vanella L, Santangelo M, Lanzafame S, Di Cataldo A. Heme oxygenase 1 expression in postischemic reperfusion liver damage: effect of L-arginine Microsurgery. 2006;26:25-32.
- 3. Emre MH, Erdogan H, Fadillioglu E. Effect of BQ-123 and nitric oxide inhibition on liver in rats after renal ischemia-reperfusion injury. Gen Physiol Biophys. 2006;25:192-205.
- 4. Abe Y, Hines I, Zibari G, Grisham MB. Hepatocellular protection by nitric oxide or nitrite in ischemia and reperfusion injury. Arch Biochem Biophys. 2009;484:232-7.
- 5. Liu P, Xu B, Quilley J, Wong PY. Peroxynitrite attenuates hepatic ischemia-reperfusion injury. Am J Physiol Cell Physiol. 2000;279:C1970-7.
- 6. Kanwar JR, Kanwar RK, Burrow H, Baratchi S. Recent advances on the roles of NO in cancer and chronic inflammatory disorders. Curr Med Chem. 2009;16:2373-94.
- 7. Knott AB, Bossy-Wetzel E. Nitric oxide in health and disease of the nervous system. Antioxid Redox Signal. 2009;11:541-54.
- 8. Vardanian AJ, Busuttil RW, Kupiec-Weglinski JW. Molecular mediators of liver ischemia and reperfusion injury: a brief review. Mol Med. 2008;14:337-45.
- 9. Kaizu T, Ikeda A, Nakao A, Takahashi Y, Tsung A, Kohmoto J, Toyokawa H, Shao L, Bucher BT, Tomiyama K, Nalesnik MA, Murase N, Geller DA. Donor graft adenoviral iNOS gene transfer ameliorates rat liver transplant preservation injury and improves survival. Hepatology (Baltimore). 2006;43:464-73.
- 10. Schwentker A, Billiar TR. Inducible nitric oxide synthase: from cloning to therapeutic applications. World J Surg. 2002;26:772-8.
- 11. Giovanardi RO, Rhoden EL, Cerski CT, Salvador M, Kalil AN. Pharmacological preconditioning using intraportal infusion of l-arginine protects against hepatic ischemia reperfusion. Inj J Surg Res. 2009;155:244-53.
- 12. Liu P, Xu B, Quilley J, Wong PY. Peroxynitrite attenuates hepatic ischemia-reperfusion injury. Am J Physiol Cell Physiol. 2000;279:C1970-7.
- 13. Kimura H, Katsuramaki T, Isobe M, Nagayama M, Meguro M, Kukita K, Nui A, Hirata K. Role of inducible nitric oxide synthase in pig liver transplantation. J Surg Res. 2003;111:28-37.
- 14. Koeppel TA, Mihaljevic N, Kraenzlin B, Loehr M, Jesenofsky R, Post S, Palma P. Enhanced iNOS gene expression in the steatotic rat liver after normothermic ischemia. Eur Surg Res. 2007;39:303-11.
- 15. Tsuchihashi S, Kaldas F, Chida N, Sudo Y, Tamura K, Zhai Y, Qiao B, Busuttil RW, Kupiec-Weglinski JW. FK330, a novel inducible nitric oxide synthase inhibitor, prevents ischemia and reperfusion injury in rat liver transplantation. Am J Transplant. 2006;6:2013-22.
- 16. Meguro M, Katsuramaki T, Nagayama M, Kimura H, Isobe M, Kimura Y, Matsuno T, Nui A, Hirata K.A novel inhibitor of inducible nitric oxide synthase (ONO-1714) prevents critical warm ischemia-reperfusion injury in the pig liver. Transplantation. 2003;273:1439-46.
- 17. Shiva S, Sack MN, Greer JJ, Duranski M, Ringwood LA, Burwell L, Wang X, MacArthur PH, Shoja A, Raghavachari N, Calvert JW, Brookes PS, Lefer DJ, Gladwin MT. Nitrite augments tolerance to ischemia/reperfusion injury via the modulation of mitochondrial electron transfer. J Exp Med. 2007;204:2089-102.
- 18. Yagnik GP, Takahashi Y, Tsoulfas G, Reid K, Murase N, Geller DA. Blockade of the L-arginine/NO synthase pathway worsens hepatic apoptosis and liver transplant preservation injury. Hepatology (Baltimore). 2002;36:573-81.
- 19. Reid KM, Tsung A, Kaizu T, Jeyabalan G, Ikeda A, Shao L, Wu G, Murase N, Geller DA. Liver I/R injury is improved by the arginase inhibitor, N (omega)-hydroxy- nor-L-arginine (nor-NOHA). Am J Physiol Gastrointest Liver Physiol. 2007;292:G512-7.
- 20. Lang JD Jr, Teng X, Chumley P, Crawford JH, Isbell TS, Chacko BK, Liu Y, Jhala N, Crowe DR, Smith AB, Cross RC, Frenette L, Kelley EE, Wilhite DW, Hall CR, Page GP, Fallon MB, Bynon JS, Eckhoff DE, Patel RP. NO accelerates restoration of liver function in adults following orthotopic liver transplantation. J Clin Invest. 2007;117:2583-91.
- 21. Iñiguez M, Berasain C, Martinez-Ansó E, Bustos M, Fortes P, Pennica D, Avila MA, Prieto J. Cardiotrophin-1 defends the liver against ischemia-reperfusion injury and mediates the protective effect of ischemic preconditioning. J Exp Med. 2006;203:2809-15.
- 22. Higuchi R, Fockler C, Walsh PS, Griffith R. Simultaneous amplification and detection of specific DNA sequences. Biotechnology. 1992;10:413-7.
- 23. Higuchi R, Fockler C, Dollinger G, Watson R. Kinetic PCR analysis: real-time monitoring of DNA amplification reactions. Biotechnology. 1993;11:1026-30.
- 24. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-Delta Delta C (T) method. Methods. 2001;25:402-8.
- 25. Arab HA, Sasani F, Rafiee MH, Fatemi A, Javaheri A. Histological and biochemical alterations in early-stage lobar ischemia-reperfusion in rat liver. World J Gastroenterol. 2009;15:1951-7.
- 26. Noh J, Behrends M, Choi S, Bedolli MA, Yardi J, Hirose R, Niemann CU. Glutamine does not protect against hepatic warm ischemia/reperfusion injury in rats. J Gastrointest Surg. 2006;10:234-9.
- 27. Hirsch J, Hansen KC, Choi S, Noh J, Hirose R, Roberts JP, Matthay MA, Burlingame AL, Maher JJ, Niemann CU. Warm ischemia induced alterations in oxidative and inflammatory proteins in hepatic Kupffer cells in rats. Mol Cell Proteomics. 2006;5:979-86.
- 28. Gujral JS, Bucci TJ, Farhood A, Jaeschke H. Mechanism of cell death during warm hepatic ischemia-reperfusion in rats: apoptosis or necrosis? Hepatology. 2001;33:1555-7.
- 29. Llacuna L, Marí M, Lluis JM, García-Ruiz C, Fernández-Checa JC, Morales A. Reactive oxygen species mediate liver injury through parenchymal nuclear factor-b inactivation in prolonged ischemia/reperfusion. Am J Pathol. 2009;174:1776-85.
- 30. Tacke F, Luedde T, Trautwein C. Inflammatory pathways in liver homeostasis and liver injury. Clin Rev Allergy Immunol. 2009;36:4-12.
Publication Dates
-
Publication in this collection
27 Aug 2012 -
Date of issue
Sept 2012
History
-
Received
16 Apr 2012 -
Accepted
17 July 2012 -
Reviewed
14 June 2012