Abstracts
PURPOSE: Evaluate anatomical and functional changes of the esophageal stump and gastric fundus of patients with advanced megaesophagus, submitted to laparoscopic subtotal esophagectomy. METHODS: Twenty patients with advanced megaesophagus, previously submitted to a videolaparoscopic subtotal esophagectomy , were evaluated. Were conducted: radiological evaluation of the stump esophagus with transposed stomach, electromanometric, endoscopic examination and histopathology of the esophageal stump and gastric fundus, without making gastric tube or pyloroplasty. RESULTS: It was observed that the average height and pressure of the anastomosis, in the electromanometric evaluation, were 23.45cm (±1.84cm) and 7.55mmHg (±5.65mmHg). In patients with megaesophagus III, the pressure of the anastomosis was 10.91mmHg (±6.33mmHg), and pressure from the UES, 31.89mmHg (±14.64mm Hg), were significantly higher than those in grade IV. The pathological evaluation detected mild esophagitis in 35% of patients, moderate in 20% and acanthosis glicogenica in 45%. The examination of the gastric fundus showed that 50% of patients were infected with Helicobacter pylori. Chronic gastritis occurred in 95% of the patients. CONCLUSIONS: The laparoscopic esophagectomy shown to be effective in the treatment of advanced achalasia. The cervical level anastomosis protects the esophageal stump from the aggression resulted from gastric reflux after the esophagectomy.
Laparoscopy; Esophagectomy; Esophageal Achalasia
OBJETIVO: Avaliar as alterações anatômicas e funcionais do coto esofágico e fundo gástrico de pacientes com megaesôfago avançado, submetidos à esofagectomia subtotal laparoscópica. MÉTODOS: Vinte pacientes com megaesôfago avançado, previamente submetidos à esofagectomia subtotal videolaparoscópica, foram avaliados. Foram realizados: avaliação radiológica do coto esofágico com o estômago transposto, eletromanometria endoscopia e exame histopatológico do coto esofágico e fundo gástrico,sem fazer tubo gástrico ou piloroplastia. RESULTADOS: Observou-se que a altura média e pressão da anastomose, na avaliação eletromanométrica, foram: 23,45cm (± 1,84cm) e 7,55mmHg (± 5,65mmHg), Em pacientes com megaesôfago III, a pressão da anastomose foi de 10,91mmHg (± 6,33mmHg), e a pressão do ESE, 31,89mmHg (±14,64mmHg) foram significativamente mais elevados do que aqueles em grau IV. A avaliação patológica detectou esofagite leve em 35% dos pacientes, moderada em 20% e acantose glicogênica em 45%. O exame do fundo gástrico mostrou que 50% dos pacientes foram infectados com Helicobacter pylori. Gastrite crônica ocorreu em 95% dos pacientes. CONCLUSÕES: A esofagectomia laparoscópica mostrou-se eficaz no tratamento de acalasia avançada. A anastomose em nível cervical tem um papel protetor para o coto esofágico ao evitar a agressão resultante de refluxo gástrico após a esofagectomia.
Laparoscopia; Esofagectomia; Acalasia Esofágica
11 - ORIGINAL ARTICLE
CLINICAL INVESTIGATION
Evaluation of anatomical and functional changes esophageal stump of patients with advanced megaesophagus submitted to subtotal laparoscopic esophagectomy1 1 Research performed at Department of Surgery, Federal University of Triangulo Mineiro, Uberaba-MG, Brazil.
Avaliação das alterações anatômicas e funcionais do coto esofágico de pacientes portadores de megaesôfago avançado submetidos à esofagectomia subtotal laparoscópica
Júverson Alves Terra JúniorI; Guilherme Azevedo TerraII; Alex Augusto da SilvaIII; Eduardo CremaIV
IMaster, Associate Professor, Department of Surgery, UFTM, Uberaba-MG, Brazil. Intellectual and scientific content of the study, technical procedures and critical revision
IIGraduate student, School of Medicine, UFTM, Uberaba-MG, Brazil. Scientific content of the study, English language, manuscript writing and critical revision
IIIPhD, Associate Professor, Department of Surgery, UFTM, Uberaba-MG, Brazil. Helped with technical procedures and critical revision
IVPhD, Full Professor, Department of Surgery, UFTM, Uberaba-MG, Brazil. Helped with scientific content of the study, technical procedures and critical revision
Correspondence Correspondence: Júverson Alves Terra Júnior Departamento de Cirurgia, Universidade Federal do Triângulo Mineiro Avenida Getúlio Guaritá, s/n 38025-440 Uberaba - MG Brasil Tel.: (55 34)3318-5228 juverson@terra.com.br
ABSTRACT
PURPOSE: Evaluate anatomical and functional changes of the esophageal stump and gastric fundus of patients with advanced megaesophagus, submitted to laparoscopic subtotal esophagectomy.
METHODS: Twenty patients with advanced megaesophagus, previously submitted to a videolaparoscopic subtotal esophagectomy , were evaluated. Were conducted: radiological evaluation of the stump esophagus with transposed stomach, electromanometric, endoscopic examination and histopathology of the esophageal stump and gastric fundus, without making gastric tube or pyloroplasty.
RESULTS: It was observed that the average height and pressure of the anastomosis, in the electromanometric evaluation, were 23.45cm (±1.84cm) and 7.55mmHg (±5.65mmHg). In patients with megaesophagus III, the pressure of the anastomosis was 10.91mmHg (±6.33mmHg), and pressure from the UES, 31.89mmHg (±14.64mm Hg), were significantly higher than those in grade IV. The pathological evaluation detected mild esophagitis in 35% of patients, moderate in 20% and acanthosis glicogenica in 45%. The examination of the gastric fundus showed that 50% of patients were infected with Helicobacter pylori. Chronic gastritis occurred in 95% of the patients.
CONCLUSIONS: The laparoscopic esophagectomy shown to be effective in the treatment of advanced achalasia. The cervical level anastomosis protects the esophageal stump from the aggression resulted from gastric reflux after the esophagectomy.
Key words: Laparoscopy. Esophagectomy. Esophageal Achalasia.
RESUMO
OBJETIVO: Avaliar as alterações anatômicas e funcionais do coto esofágico e fundo gástrico de pacientes com megaesôfago avançado, submetidos à esofagectomia subtotal laparoscópica.
MÉTODOS: Vinte pacientes com megaesôfago avançado, previamente submetidos à esofagectomia subtotal videolaparoscópica, foram avaliados. Foram realizados: avaliação radiológica do coto esofágico com o estômago transposto, eletromanometria endoscopia e exame histopatológico do coto esofágico e fundo gástrico,sem fazer tubo gástrico ou piloroplastia.
RESULTADOS: Observou-se que a altura média e pressão da anastomose, na avaliação eletromanométrica, foram: 23,45cm (± 1,84cm) e 7,55mmHg (± 5,65mmHg), Em pacientes com megaesôfago III, a pressão da anastomose foi de 10,91mmHg (± 6,33mmHg), e a pressão do ESE, 31,89mmHg (±14,64mmHg) foram significativamente mais elevados do que aqueles em grau IV. A avaliação patológica detectou esofagite leve em 35% dos pacientes, moderada em 20% e acantose glicogênica em 45%. O exame do fundo gástrico mostrou que 50% dos pacientes foram infectados com Helicobacter pylori. Gastrite crônica ocorreu em 95% dos pacientes.
CONCLUSÕES: A esofagectomia laparoscópica mostrou-se eficaz no tratamento de acalasia avançada. A anastomose em nível cervical tem um papel protetor para o coto esofágico ao evitar a agressão resultante de refluxo gástrico após a esofagectomia.
Descritores: Laparoscopia. Esofagectomia. Acalasia Esofágica.
Introduction
The megaesophagus, is a set of signs and symptoms mainly characterized by a slowly progressive dysphagia for solids, evolving to liquids, with a drop of the patient's nutritional status and that culminates in hypertrophy of the organ wall, stretching and dilation, especially due to motor alterations of the esophageal body and dysfunction in relaxation of the lower esophageal sphincter.
Despite being a disease of worldwide occurrence, the megaesophagus has large oscillations about its geographical distribution. In Brazil, Chagas disease remains a serious public health problem, especially in the states of Sao Paulo, Minas Gerais, Goias and northwest region of the country, with an estimated population of about 8 million affected, in which the prevalence of chagasic megaesophagus varies from 6.8% to 18.3%, and about 3% will develop advanced esophageal form. There is a predominance of male over female, specifically aged between 20 and 40 years1.
The chagasic megaesophagus has as its main pathological findings a strong intramural denervation that affects the almost entirely the middle and lower esophageal segments. The esophageal motor disorders arise when about 50% of neuronal structures are affected. The main complications of chagasic megaesophagus are stasis, esophagitis, malnutrition, aspiration and esophageal carcinoma2.The megaesophagus has been treates preferentially by subtotal esophagectomy by cervicotomy associated with right thoracotomy and laparotomy. Orringer et al.3 proposed esophageal resection by laparotomy and cervicotomy, abolishing the thoracotomy. With the advent of video surgery, the techniques of videolaparoscopy have been extremely useful especially in the dissection of the mediastinal esophagus.
There are several reports in the literature about the anatomical and functional changes of the remaining esophageal stump after subtotal esophagectomy, especially in the treatment of esophageal cancer4. However, for the treatment of achalasia reports are still few, which encouraged us to investigate these possible changes5.
The hypothesis of this study was that the esophagogastric anastomosis at the cervical level, associated with non-performance of pyloroplasty, prevents anatomical and functional changes due to gastro-esophageal reflux after esophagectomy for the treatment of achalasia.
Methods
This project was evaluated by the Ethics Committee in Research of the Federal University of Triangulo Mineiro and was approved under protocol n. 1307 of 2008.
To the patients to participate in this study, we reviewed the medical records of patients with advanced megaesophagus, followed in the Clinic of Digestive Surgery at Teaching Hospital of the Federal University of Triangulo Mineiro who underwent surgical treatment with laparoscopic esophagectomy with cervical esophageal-gastroplasty in the period from 1996 to 2007, these patients were invited to participate in the study.
The inclusion criteria in this study were individuals with advanced chagasic megaesophagus, with satisfactory clinical condition who underwent esophagectomy, oriented, lucid, who agreed to participate and signed an informed consent. The criteria for surgical treatment were: radiological advanced megaesophagus (grade IV), functionally advanced megaesophagus (aperistalsis or contration amplitude of the body less than 20mmHg), relapse of megaesophagus after surgery on the esophagogastric junction.
The exclusion criteria in this study were: patients with severe comorbidities that limited surgical treatment; patients with altered level of consciousness; and also those who after informed about the research did not agree to participate. Patients who gave up on the clinical follow-up or did not maintain any contact with the research group were also excluded from the study.
Data on age, gender, serologic tests for Trypanosoma cruzi, degree of megaesophagus and interval between surgery and biopsy were collect from patient charts. Three serological tests for T. cruzi were performed: Machado-Guerreiro complement fixation, indirect hemagglutination and ELISA, and were considered positives for Chagas disease patients with two or more positive tests.
Preoperative clinical evaluations particularly cardiac, pulmonary and nutritional were routinely performed as patients usually had co-morbidities and when presenting for treatment were malnourished.
Nutritional assessment was performed pre and postoperatively in all patients and consisted of: anthropometric measures (weight, height arm muscle circumference), serum proteins, albumin and transferrin and immunological evaluation (PPD and total lymphocytes).
All patients underwent preoperative visits for 7-21 days, which consisted of respiratory therapy and enteral nutritional support (1.5g protein/kg /day), through a nasogastric tube positioned in the stomach. The patients were also submitted to contrasted esophageal exam, upper endoscopy, electromanometry, prolonged esophageal pH monitoring and upper abdominal ultrasonography.
On the eve of surgery, patients were submitted to mechanical cleaning of the esophagus through oral-esophageal probe (Fouchet or Levine) with 0.9% saline solution, for cleaning the esophageal lumen and consequently reduce the presence of any potential contaminants; this procedure was associated with venous dissection of the right upper limb.
For the surgery, the patient was positioned as the laparoscopic esophageal cardiomyotomy. Patient in horizontal dorsal decubitus position with legs extended, separated and properly supported in appropriate leggings, leaving the surgeon between the legs and the assistants (camera and presentation) to the left of the patient. The left arm was kept parallel to the trunk and the right arm in abduction at 45º angle. The monitor was positioned at the right upper bedside of the operating table. Five punctures were made:
a. 10mm trocar above the navel, approximately 15cm from the xiphoid process (30 º optical);
b. 10mm trocar on the left subcostal region, on the hemi-clavicular line (right hand of the surgeon);
c. 5mm trocar on the left subcostal region, on the hemi-clavicular line (left hand of the surgeon);
d. 5mm trocar midline sub-xiphoid (auxiliary removing the left lobe);
e. 5mm trocar to the left of the scar (a traction assist transition and esophagus).
After pneumo-peritoneum of 12 mmHg, the procedure began, it consisted of opening the phreno-esophageal membrane and lesser omentum, giving access to the right branch of the right crus of the diaphragm. From this point, the peri-esophageal tissue of the terminal esophagus was released, isolating it with dissection of the esophagogastric junction, repairing the abdominal esophagus with Penrose drain or flexible retractor (endoflex). The anterior and posterior vagal trunks were identified, and were isolated and dissected until below the esophagogastric transition.
Subsequently, we kept on dissecting, by direct visualization of the esophageal body with identification of the pleura, pericardium, azygos vein and vagal trunks across thoracic extension. Hemostasis was performed with monopolar cautery and/or clipping of larger esophageal branches, preferably, with the use of harmonic scalpel, separating the surgical plan near the esophagus avoiding, thus, damage to the pleura and mediastinal structures.
For better access to the mediastinum at the time of surgical dissection of the thoracic esophagus, there was a median transection of the diaphragm and the positioning of the surgical table in Trendelenburg position, as in cervical-abdominal approach at the open surgery.
After the dissection of the thoracic and abdominal esophagus, the release of stomach greater curvature was prepared, using a harmonic scalpel for short vessels section and the gastrocolic omentum. For ligation of the gastro-epiploic and lest gastric vessels double clipping was used, preserving the arcade of large and small curvature.
After the prepation of the stomach, left cervicotomy was performed for the dissection of the cervical esophagus. Through proximal traction, making the section of the esophagogastric junction with the aid of linear cutting stapler (green load), with reinforcement of the staple line with monofilament polypropylene 3-0.
During the traction of the previously dissected esophagus, it was possible to track the passage of the esophagus and stomach through the mediastinum by direct visualization with the aid of the optical equipment positioned in the lower mediastinum.
The esophagogastric anastomosis was performed with manual continuous suture in a single plane with 3.0 monofilament polypropylene.
A nasogastric probe was positioned below the cervical anastomosis in the duodenum, when possible, or in the antrum, which received enteral feeding 24 hours after surgery, as described for the preoperative. There was no cervical or abdominal drainage employed.
Postoperatively, patients were maintained, on average, for nine days at hospital, receiving exclusive enteral support, physiotherapy and psychological assessment. On the ninth day, a swallowing test was performed in order to exclude the presence of fistulas, and determine the height and patency of the anastomosis. Those who normal expected results had had the withdrawal of enteral feeding, were discharged with outpatient follow-up. However, those patients who had fistulas, pulmonary decompensation and any systemic regime remained in hospital until complete clinical recovery.
During late follow-up period, patients were subjected to contrasted esophageal stump, stomach and duodenum exam, being invites ingest 100ml of 100% barium suspension, followed by fluoroscopy in order to determine: the presence of stenosis, the anatomic position of the anastomosis, contrasted morphology of these organs and gastric emptying. Patients also underwent endoscopy, to determined: the height of the anastomosis; the appearance of the esophageal mucosa, with special attention to possible injury from gastroesophageal reflux; patency of the esophageal-gastric anastomosis and other changes possible. At this point, three samples of esophageal tissue and other three samples of gastric tissue were collected, about 2 cm above and 2 cm below the anastomosis, respectively, the fragments being fixed in 3.7% formalin for subsequent histological analysis.
Eletromanometry was also performed to evaluate the function of the upper esophageal sphincter and, in accordance with the possibility, to evaluate the esophageal stump and the height anastomosis. The procedure was performed after the application of topical anesthesia (2% lidocaine) in the nasopharynx. Six channels polyvinyl probe was inserted in one of the nostrils, three channels were placed in the more distal portion and the three others on the upper portion at intervals of 5 cm between them. These channels were perfused with distilled water, at constants flow rate of 0.5 ml/min and pressure of 60 mmHg. The recording of pressure changes in the area of the anastomosis, the upper esophageal sphincter (UES), as well as the extension of the UES and the gauge height of the anastomosis were obtained through a polygraph and decoded by specific software (Alacer Multiplex 2).
For the histopathological study, we made paraffin blocks of the samples collected during the endoscopy, and from each block five blades were made: one intended for staining by hematoxylin-eosin (HE) to investigate changes in esophageal and gastric stump; one stained by Warthin-Starry (WS) to search the H. pylori, and another one stained by periodic acid-Schiff (PAS) to search intestinal metaplasy. Abnormalities were found classified according Chandrasoma et al.6 and Updated Sydney System (WCOG90/94).
For statistical analysis, we prepared a database and analysis was conducted by using SigmaStat 2.03. Qualitative variables were represented in frequency (%) and semi-quantitative data were categorized as absent, mild, moderate and intense.
Kolmogorv-Smirnov test was used to verify normality. Continuous with normal distribution and homogeneity of variances were analyzed by Student's t test. The non-continuous variables with normal distribution and non-homogeneous variances, or non-normal distribution were analysed by Wilcoxon test. Fisher exact test was used to compare the degree of megaesophagus and gender. In relative comparison to gender and infection by H. pylori, we used the chi-square (χ2). Pearson correlation test was used to analyse association between extent and pressure of the UES. Results were considered statistically significant when the probability of error was less than 5% (p<0.05).
Results
After reviewing the medical records of patients with advanced megaesophagus, 60 (100%) patients were selected, 20 (33.33%) obeyed the inclusion and exclusion criteria specified, being invited to the study.
In relation to gender and age of the patients, 12 (60%) were male, mean age of 58.25 years (± 13.25 years) and eight (40%) were female, mean age of 54.25 years (± 13.83 years), and the patient's age ranged from 31 to 75 years.
The serology for Chagas's disease was positive in 17 (85%) patients, following positive results in at least two serological methods.
In determining the degree of megaesophagus, nine (45%) patients had megaesophagus grade III, six (66.67%) were male and three (33.33%) female; 11 (55%) patients had megaesophagus grade IV, six (54.54%) patients were male and five (45.45%) females, without significant statistical difference (p=0.67).
At abdominal ultrasonography, none of the patients submitted to the study had choilelithiasis associated with the megaesophagus.
During the late postoperative follow-up with contrasted study of the esophageal stump, stomach and duodenum, we observed the esophageal stump with smooth walls, satisfactory passage of contrast through the anastomosis area (Figure 1), as well as the pylorus located in the cervical region, above the first thoracic vertebra in all 20 (100%) patients.
At the endoscopic exam, it was found that the heights of the anastomosis in females was 19.88cm (± 0.35cm) and in males was 20.92cm (± 2.27cm) without significant statistical difference between both genders (p>0.05).
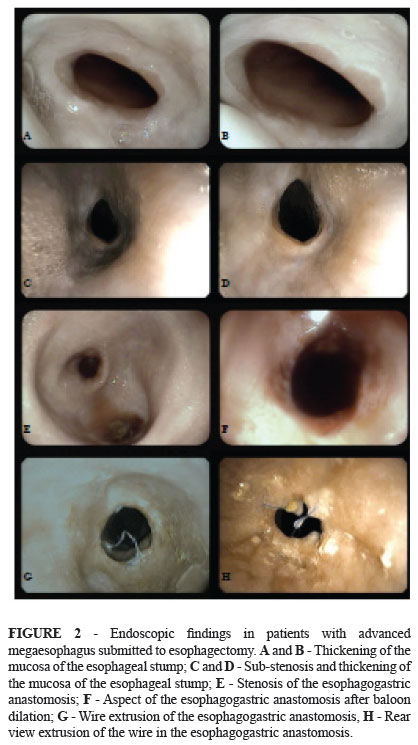
Also in the endoscopic examination, it was observed: thickening of the esophageal stump mucosa in seven (35%) patient; extrusion of the wire for the light of the anastomosis in six (30%) patients; extrusion of the wire associated with sub-stenosis of the anastomosis in three (15%); sub-stenosis of the anastomosis in two (10%) patients; in one (5%) patient it was observed total stenosis of the anastomosis, which required pneumatic balloon dilation and in another patient (5%) it was observed the occurrence of gastric reflux (Figure 2).
In eletromanometric study, we observed relaxation of the upper esophageal sphincter (UES) and that the height of the anastomosis and its mean mean pressure in the group studied were 23.45cm (±1.84cm) and 7.55mmHg (±5.65mmHg) respectively. The length and mean pressure of the UES were 2.47cm (±0.65cm) and 24.84mmHg (±12mmHg). When comparing the groups of patients with magaesophagus grade III and IV, we observed that in grade III group the pressure of the anastomosis, 10.91mmHg (±6.33mmHg), and the pressure of the UES, 31.8mmHg (±14.64mmHg) were significantly higher than in grade IV (p=0.012; p=0.0128). However, when correlated with UES length and pressure the results were not statically significant (r=0.30, p=0.18; r=0.148, p=0.53).
When comparing the mean of the height of the anastomosis observed by manometric exam and endoscopy, it was observed that in the manometry, the height of the anastomosis was significantly greater, 23.5cm (20-27cm) and 20cm (18-25cm), respectively (W=60.000, p<0.001).
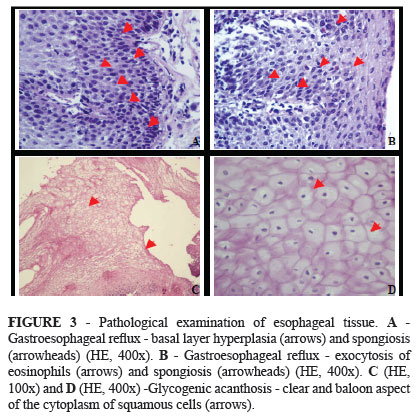
In the pathological study of the esophageal stump, mild esophagitis was detected in seven (35%) patients, moderate in four (20%) patients and glycogenic acanthosis in nine (45%) patients (Figure 3).
On examination of the fundus using Warthin-Starry stain (WS), observed that ten (50%) patients were infected with H. pylori, seven (35%) men and three (15%) women, without association between positivity to H. pylori and gender (p=0.65).
Moreover, considering parameters such as the occurrence of ulceration, fibrosis, inflammatory infiltrate, atrophy of glandular bodies, intestinal metaplasy and reactive gastropathy, we observed the occurrence of chronic gastritis in 19 (95%) patients, it was classified as intense in three (15%) patients, moderate in six (30%) patients and mild in ten (50%) patients. In one (5%) patient we did not observe the occurrence of gastritis (Figure 4).
Discussion
The advanced megaesophagus had almost the same distribution between both genders and was also more frequent in the age group 61-70 years (56.66 years). Oliveira et al.7, studying the epidemiological profile of 385 patients with achalasia operated between 1989 and 2005 showed that 55% of patients were men and 45% women, mean age of 47.1 years. Moreover, the age group with highest incidence of megaesophagus was 40 to 60 years. The difference between the two studies is probably related to the fact that the patients included in that study had megaesophagus ranging from grade I to IV, with higher incidence of grade II, whereas our study only followed patients with advanced disease, which results in higher mean age.
The serology for Chagas's disease was positive in 85% of patients, confirmed by at least two serological methods, as recommended in the literature8, probably due to the fact that patients are coming from an endemic area showing a high prevalence of disease in our region.
In the radiological classification of megaesophagus, 45% of patients had megaesophagus grade III and grade IV 55%, however, in patients with megaesophagus grade III manometric examination showed contraction waves of the esophageal body less than 20 mmHg, being this one of the indications for esophagectomy9. Thus, this study is in accordance with Crema et al.1 who comparing the radiological classification of megaesophagus with manometric evaluation found 35% of patients with megaesophagus grade III with waves of contraction of the esophageal body less than 20 mmHg, showing that these patients do not benefit from performing cardiomyotomy associated with performance of anti-reflux valve10 due to the fact that the anti-reflux valves present a mean pressure of 25 mmHg, which favors the maintenance of dysphagia1.
Regarding the time elapsed between the esophagectomy and endoscopy with biopsy, it was observed that the overall median age was 19.5 months, whereas for females the median (16.5 months) was smaller than males (23.5 months), reinforcing the observation that women are more concerned about their clinical monitoring and follow-up time, it is also in agreement with studies that emphasize the importance of clinical follow-up of at least 18 months to evaluate the appearance of metaplasy11. However, in respect to inflammatory changes compatible with reflux esophagitis, following the esophagitis degree classification of Cecconelo et al.4 reported that three months postoperative biopsies already have varying degrees of injury.
Researchers suggest that the systematic ultrasound of the upper abdomen should be performed in patients with megaesophagus to be submitted to surgical treatment, since denervation occurs throughout the digestive tract in patients with the digestive form of Chagas disease2.
In our study, at abdominal ultrasonography, none of the patients had gallstones associated with megaesophagus. However, studies have shown that the presence of cholecystitis varies 12-28% in the population of patients with Chagas' disease9. As justification for this occurrence, Crema et al.1 demonstrated with scintigraphic study of the biliary tract and comparison with the neuronal counts of the gallbladder, the Chagas patients showed dyskinesia of the gallbladder and the total number of neurons was decreased when compared to normal population, which may cause more biliary stasis providing the appearance of cholelithiasis. We believe that the sample size did not allow observation of the occurrence of cholelithiasis.
In the postoperative control, we found anastomosis in cervical position in all patients. There are few reports on satisfactorily results in avoiding reflux with upper thoracic level anastomosis12. Okuyama et al.13 comparing the esophagogastric anastomosis in the cervical and thoracic level, found no significant difference between the two methods, however, found a higher number of recurrent laryngeal nerve injury when performing the anastomosis at cervical level. Kim et al.14 have shown that cervical anastomosis has lower risk of reflux esophagitis, which improves the patients' quality of life. Despite the controversy in the literature, we are in agreement with Kim et al.14 and we believe that the position of the anastomosis at cervical level may decrease the degree of injury of the gastro-esophageal-stump, as it is positioned in a positive pressure area. In addition, most of studies were conducted for the treatment of esophageal cancer and not achalasia.
Williams et al.15 classified the severity score of dysphagia according to the diameter of stenosis presented at the anastomosis as: minimal (>12mm), light (9-12mm), moderate (5-8mm) and severe (<5mm). These authors also reported the occurrence of stenosis in 66% of patients submitted to esophagectomy with cervical anastomosis, which was most frequent (87.5%) in cases where the anastomosis was performed manually. However, in our study, we did not perform the measurement of the diameter of the stenosis, although, we used subjective parameters and classified the degree of stenosis (sub-stenosis or stricture) according to the resistance offered to the passage of endoscopic tube. Thus, in the cases of sub-stenosis (25%) despite a certain resistance, it was possible to pass the tube, in the stenosis cases (5%), this passage was not possible, requiring the balloon dilatation, even performing the anastomoses by hand and using non-absorbable wires. Other studies show a variation from 27 to 42% or more in the occurrence of anastomotic stenosis, esophageal-gastric patients undergoing esophagectomy16, showing that the incidence observed in our study is in agreement with the literature.
In eletromanometric study, we observed that the average pressure of the anastomosis in the patients studied was 7.55mmHg, while Yalav and Ercan4 showed an average pressure of 14.2mmHg in cases of intra-thoracic anastomosis with performance of anti-reflux valve in order to reduce aggression to the esophageal stump. In our study, although the lower average, the anastomosis is near the upper esophageal sphincter, which showed an average pressure of 24.83mmHg which seems to favor the prevention of the occurrence of gastroesophageal reflux.
When comparing groups of patients with megaesophagus grade III and IV, it was observed that in patients with grade III, the pressure of the anastomosis (10.91mmHg) and UES (31.89mmHg) were significantly higher, being in agreement with Dantas et al.17 who showed that the pressure of the UES was higher in patients with advanced non-radiological megaesophagus.
We observed a positive correlation, although not significant, between the length and pressure of the UES, as well as between the extent of the UES and the pressure at the anastomosis, and we believe that this has occurred probably due to the proximity between the UES and the anastomosis.
We also observed that the height of the anastomosis in esophageal manometry was significantly higher than in endoscopy, demonstrating the importance and accuracy of the eletromanometric method for monitoring the esophageal stump and esophagogastric anastomosis.
At pathological examination of the esophageal stump, we observed that 55% of patients had mild to moderate esophagitis, which is consistent with D'Journo et al.18 who report that approximately 50% of patients submitted to esophagectomy evolve with esophagitis caused by exposure to acid and bile. Rocha et al.5 show that the postoperatively, the incidence of esophagitis in the remaining stump varies from 45.9% to 70% in 1-10 years of follow-up.
We did not observe the occurrence of intestinal metaplasy while Rocha et al.5 report its occurrence in patients with achalasia submitted to esophagectomy, ranging from 10.9 to 57.5% in 1-10 years or more of follow-up. Lerut and van Lanschot19 also point out that bile and/or acid reflux can result in persistent esophagitis and even in the development of metaplasy. However, D'Journo et al.20 also reported the higher occurrence of metaplasy in patients with thoracic level anastomosis, pointing out that its prevalence is not related to the magnitude of the reflux, but to its duration, and also that the cervical anastomosis favors less gastric reflux symptoms, less visualized damage and delays the development of mucosal damage over time. Palmes et al.21 studied groups of patients submitted to esophagectomy with and without pyloroplasty, observing that pyloroplasty did not improve gastric emptying and that it may favor the appearance of reflux esophagitis. Thus, we believe that metaplasy was not observed because it was not performed pyloroplasty, what probably contained the duodenogastric reflux, but the follow-up time may be important in the detection of metaplastic changes consequent to prolonged exposure to stomach acid.
We observed the occurrence of glycogenic acanthosis in 45% of patients, but also that some authors22 believe that this occurrence may be related to gastroesophageal reflux, although due to the lack reliability of pH monitoring data, it was not possible to make any association.
On histological examination of the gastric fundus, we observed that 50% of patients were infected with H. pylori. Takemura et al.23 analyzing the infection by H. pylori in the gastric tube in outpatients found 56% positivity. Authors report the negative influence of infection by H. pylori in gastric acid secretion in pre-and postoperative esophagectomy24. Fischbach et al.25 draw attention to the protective role of infection by H. pylori in development of intestinal metaplasy at the esophageal stump, demonstrating that, probably, the high incidence of infection by H. pylori is a protective factor for esophageal stump as it decreased acid secretion.
On examination of the stomach, we observed the occurrence of chronic gastritis in 95% of patients, showing higher rates than those reported in the literature. Rocha et al.5 observed gastritis ranging from 20.4 to 40% of patients with achalasia submitted to esophagectomy, during a follow-up period ranging from 1-10 years or more. Maier et al.26 found gastritis in 83.33% of patients submitted to esophagectomy for treatment of esophageal cancer. Due to the high incidence of gastritis these patients, Okuyama et al.13 suggest the prophylactic use of proton pump inhibitors already in the early postoperative in order to prevent and alleviate symptoms related to acid secretion.
Conclusions
The laparoscopic esophagectomy shown to be effective in the treatment of advanced achalasia. The cervical level anastomosis protects the esophageal stump from the aggression resulted from gastric reflux after the esophagectomy.
Received: April 18, 2012
Review: June 20, 2012
Accepted: July 19, 2012
Conflict of interest: none
Financial source: National Council of Technological and Scientific Development (CNPq)
- 1. Crema E, Cruvinel LA, Werneck AM, Oliveira RM, Silva AA. Manometric and radiologic aspects of Chagas' megaesophagus: the importance to its surgical treatment. Rev Soc Bras Med Trop. 2003;36:665-9.
- 2. Adad SJ, Cancado CG, Etchebehere, RM, Teixeira VP, Gomes UA, Chapadeiro E, Lopes ER. Neuron count re-evaluation in the myenteric plexus of chagasic megacolon after morphometric neuron analysis. Virchows Arch. 2001;438:254-8.
- 3. Dubecz A, Kun L, Stadlhuber RJ, Peters JH, Schwartz SI. The origins of an operation: a brief history of transhiatal esophagectomy. Ann Surg. 2009;249:535-40.
- 4. Yalav E, Ercan S. Reservoir and globe-type antireflux surgical techniques in intrathoracic esophagogastrostomies. J Int Soc Dis Esophagus. 2000;13:282-7.
- 5. Rocha JR, Ribeiro Jr U, Sallum RA, Szachnowicz S, Cecconello I. Barrett's esophagus and carcinoma in the esophageal stump after esophagectomy with gastric pull-up in achalasia patients: a study based on 10 years follow-up. Ann Surg Oncol. 2008;15:2903-9.
- 6. Chandrasoma P, Wijetunge S, Ma Y, Demeester S, Hagen J, Demeester T. The dilated distal esophagus: a new entity that is the pathologic basis of early gastroesophageal reflux disease. Am J Surg Pathol. 2011;35:1873-81.
- 7. Oliveira GC, Lopes LR, Andreollo N, Coelho Neto JS. Surgically treated megaesophagus: epidemiological profile of patients operated in the Clinical Hospital of the State University of Campinas between 1989 and 2005. Rev Soc Bras Med Trop. 2008;41:183-8.
- 8. Gascón J, Albajar P, Cañas E, Flores M, Gómez I, Prat J, Herrera RN, Lafuente CA, Luciardi HL, Moncayo A, Molina L, Muñoz J, Puente S, Sanz G, Treviño B, Sergiosalles X. Diagnosis, management and treatment of chronic Chagas' heart disease in areas where Trypanosoma cruzi infection is not endemic. Enferm Infecc Microbiol Clin. 2008;26:99-106.
- 9. Crema E, Silva ECRR, Franciscon PM; Rodrigues Júnior V, Martins Júnior A, Teles CJO, Silva AA. Prevalence of cholelithiasis in patients with chagasic megaesophagus. Rev Soc Bras Med Trop. 2011;44:324-6.
- 10. Campos G, Vittinghoff E, Rabl C, Takata M, Gadenstätter M, Lin F, Ciovica R. Endoscopic and surgical treatments for achalasia: a systematic review and meta-analysis. Ann Surg. 2009;249:45-57.
- 11. Rocha JR, Cecconello I, Zilberstein B, Sallum RA, Sakai P, Shioka S, Pinotti HW. Barrett esophagus in the esophageal stump after subtotal esophagectomy with cervical esophagogastroplasty. Rev Hosp Clin. 1992;47:69-70.
- 12. Kayani B, Jarral OA, Athanasiou T, Zacharakis E. Should oesophagectomy be performed with cervical or intrathoracic anastomosis? Interact Cardiovasc Thorac Surg. 2012;14(6):821-6.
- 13. Okuyama M, Motoyama S, Maruyama K, Sasaki K, Sato Y, Ogawa J. Proton pump inhibitors relieve and prevent symptoms related to gastric acidity after esophagectomy. World J Surg. 2008;32:246-54.
- 14. Kim HK, Choi YH, Shim JH, Cho YH, Baek MJ, Sohn YS, Kim HJ. Endoscopic evaluation of the quality of the anastomosis after esophagectomy with gastric tube reconstruction. World J Surg. 2008;32:2010-4.
- 15. Williams VA, Watson TJ, Zhovtis S, Gellersen O, Raymond D, Jones C, Peters JH. Endoscopic and symptomatic assessment of anastomotic strictures following esophagectomy and cervical esophagogastrostomy. Surg Endosc. 2008;22:1470-6.
- 16. Barthel JS, Kelley ST, Klapman JB. Management of persistent gastroesophageal anastomotic strictures with removable self-expandable polyester silicon-covered (Polyflex) stents: an alternative to serial dilation. Gastrointest Endosc. 2008;67:546-52.
- 17. Dantas RO, Alves LMT, Nascimento WY. Effect of bolus volume on proximal esophageal contractions of patients with Chagas' disease and patients with idiopathic achalasia. Dis Esophagus. 2010;23:670-4.
- 18. D'Journo XB, Martin J, Ferraro P, Duranceau A. The esophageal remnant after gastric interposition. J Int Soc Dis Esophagus. 2008;21:377-88.
- 19. Lerut TE, van Lanschot JJ. Chronic symptoms after subtotal or partial esophagectomy: diagnosis and treatment. Best Pract Res Clin Gastroenterol. 2004;18:901-15.
- 20. D'Journo XB, Martin J, Rakovich G, Brigand C, Gaboury L, Ferraro P, Duranceau A. Mucosal damage in the esophageal remnant after esophagectomy and gastric transposition. Ann Surg. 2009;249:262-18.
- 21. Palmes D, Weilinghoff M, Colombo-Benkmann M, Senninger N, Bruewer M. Effect of pyloric drainage procedures on gastric passage and bile reflux after esophagectomy with gastric conduit reconstruction. Langenbeck's Arch Surg. 2007;392:135-41.
- 22. Vieth M. Contribution of histology to the diagnosis of reflux disease. Best Pract Res Clin Gastroenterol. 2008;22:625-38.
- 23. Takemura M, Higashino M, Osugi H, Tokuhara T, Fujiwara K, Kinoshita H. Five cases of peptic ulcer of gastric tube after radical esophagectomy for esophageal carcinoma and analysis of Helicobacter pylori infection at gastric tube. Nihon Kyōbu Geka Gakkai. 1997;45:1992-7.
- 24. Mori N, Fujita H, Sueyoshi S, Aoyama Y, Yanagawa T, Shirouzu K. Helicobacter pylori infection influences the acidity in the gastric tube as an esophageal substitute after esophagectomy. J Int Soc Dis Esophagus. 2007;20:333-40.
- 25. Fischbach LA, Nordenstedt H, Kramer JR, Gandhi S, Dick-Onuoha S, Lewis A, El-Serag HB. The association between Barrett's esophagus and Helicobacter pylori infection: a meta-analysis. Helicobacter. 2012;17:163-75.
- 26. Maier A, Tomaselli F, Sankin O, Anegg U, Fell B, Renner H, Pinter H, Friehs GB, Smolle-Jüttner FM. Acid-related diseases following retrosternal stomach interposition. Hepatogastroenterol. 2001;48:899-902.
Publication Dates
-
Publication in this collection
27 Aug 2012 -
Date of issue
Sept 2012
History
-
Received
18 Apr 2012 -
Accepted
19 July 2012 -
Reviewed
20 June 2012