Abstracts
PURPOSE: Investigate the morphological effects of chronic exposure to tobacco smoke inhalation and alcohol consumption on the lungs and on the growth of rats. METHODS: Sixty male Wistar rats were divided into four groups: control, tobacco, alcohol, tobacco + alcohol, for a period of study 260 days. Morphological analysis was conducted by optical and electron microscopy. Rat growth was investigated by measuring the snout-anus length, body mass index and body weight. RESULTS: The three groups exposed to the drugs presented lower growth and lower weight than the control group. The percentages of alveolitis, bronchiolitis and the mean alveolar diameter were greater, particularly in the groups exposed to tobacco smoke, but were not significantly different from the control group. Electron microscopy revealed more intense apoptotic and degenerative lesions in the smoking group, while degenerative lesions in the lamellar bodies were more intense with the association of both drugs. CONCLUSIONS: This experimental model showed morphological alterations observed by electron microscopy, principally due to tobacco smoke exposure. Alcohol and tobacco hindered the growth of rats, such that tobacco showed a greater effect on body length and alcohol on body weight.
Alcoholism; Smoking; Lung Diseases; Rats
OBJETIVO: Investigar os efeitos morfológicos da exposição crônica à inalação de fumaça do tabaco e o do consumo de álcool nos pulmões e no crescimento de ratos. MÉTODOS: Sessenta ratos Wistar machos foram distribuídos em quatro grupos: controle, tabaco, álcool e tabaco + álcool, e acompanhados por um período de 260 dias. No final do periodo foi realizada análise morfológica dos pulmões por microscopia óptica e eletrônica. O crescimento dos ratos foi investigado através da medição do comprimento focinho-ânus, peso corporal e índice de massa corporal. RESULTADOS: Os três grupos expostos às drogas apresentaram peso e comprimento significativamente menores que os do grupo controle. As percentagens de bronquiolite e alveolite, e o diâmetro alveolar médio foram maiores nos grupos expostos à fumaça do tabaco, mas sem significancia estatística quando comparadas ao grupo controle. A microscopia eletrônica revelou apoptose mais intensa e lesões degenerativas no grupo de fumantes, enquanto lesões degenerativas nos corpos lamelares foram mais intensas com a associação de ambas as drogas. CONCLUSÕES: Este modelo experimental mostrou alterações morfológicas observadas por microscopia eletrônica, principalmente devido à exposição ao tabaco. Tanto o alcool como o tabaco prejudicaram o crescimento dos animais, o tabaco mostrando um efeito maior sobre o comprimento e o álcool sobre o peso corporal.
Alcoolismo; Tabagismo; Pneumopatias; Ratos
4 - ORIGINAL ARTICLE
MODELS, BIOLOGICAL
Lung morphology and growth of rats exposed to tobacco smoke and alcohol1 1 Research performed at Postgraduate Program in General Basis of Surgery. Botucatu School of Medicine, Sao Paulo State University (UNESP), Brazil. Part of PhD thesis degree. Tutor: Antonio Jose Maria Cataneo, Co-Tutor: Daniele Cristina Cataneo.
Estudo morfológico dos pulmões e crescimento de ratos expostos à fumaça do cigarro e ao álcool
Karla Luciana MagnaniI; Daniele Cristina CataneoII; Vera Luiza CapelozziIII; Julio DefaveriIV; Erica Nishida HasimotoI; Antônio José Maria CataneoV
IFellow PhD degree, Postgraduate Program in General Basis of Surgery, Botucatu School of Medicine, UNESP, Sao Paulo-SP, Brazil. Acquisition and interpretation of data, manuscript writing
IIPhD, Assistant Professor, Division of Thoracic Surgery, UNESP, Sao Paulo-SP, Brazil. Conception and design of the study
IIIPhD, Associate Professor, Department of Pathology, University of Sao Paulo (USP), Brazil. Acquisition and interpretation of data
IVPhD, Associate Professor, Department of Pathology, UNESP, Sao Paulo-SP, Brazil. Acquisition and interpretation of data
VPhD, Chairman, Full Professor, Division of Thoracic Surgery, UNESP, Sao Paulo-SP, Brazil. Conception, design, intellectual and scientific content of the study
Correspondence Correspondence: Antônio José Maria Cataneo Programa de Pós-Graduação em Bases Gerais da Cirurgia Faculdade de Medicina de Botucatu, UNESP 18618-970 Sao Paulo Brasil Tel.: (55 14)9775-3452 Fax: (55 14)3815-7615 acataneo@fmb.unesp.br
ABSTRACT
PURPOSE: Investigate the morphological effects of chronic exposure to tobacco smoke inhalation and alcohol consumption on the lungs and on the growth of rats.
METHODS: Sixty male Wistar rats were divided into four groups: control, tobacco, alcohol, tobacco + alcohol, for a period of study 260 days. Morphological analysis was conducted by optical and electron microscopy. Rat growth was investigated by measuring the snout-anus length, body mass index and body weight.
RESULTS: The three groups exposed to the drugs presented lower growth and lower weight than the control group. The percentages of alveolitis, bronchiolitis and the mean alveolar diameter were greater, particularly in the groups exposed to tobacco smoke, but were not significantly different from the control group. Electron microscopy revealed more intense apoptotic and degenerative lesions in the smoking group, while degenerative lesions in the lamellar bodies were more intense with the association of both drugs.
CONCLUSIONS: This experimental model showed morphological alterations observed by electron microscopy, principally due to tobacco smoke exposure. Alcohol and tobacco hindered the growth of rats, such that tobacco showed a greater effect on body length and alcohol on body weight.
Key words: Alcoholism. Smoking. Lung Diseases. Rats.
RESUMO
OBJETIVO: Investigar os efeitos morfológicos da exposição crônica à inalação de fumaça do tabaco e o do consumo de álcool nos pulmões e no crescimento de ratos.
MÉTODOS: Sessenta ratos Wistar machos foram distribuídos em quatro grupos: controle, tabaco, álcool e tabaco + álcool, e acompanhados por um período de 260 dias. No final do periodo foi realizada análise morfológica dos pulmões por microscopia óptica e eletrônica. O crescimento dos ratos foi investigado através da medição do comprimento focinho-ânus, peso corporal e índice de massa corporal.
RESULTADOS: Os três grupos expostos às drogas apresentaram peso e comprimento significativamente menores que os do grupo controle. As percentagens de bronquiolite e alveolite, e o diâmetro alveolar médio foram maiores nos grupos expostos à fumaça do tabaco, mas sem significancia estatística quando comparadas ao grupo controle. A microscopia eletrônica revelou apoptose mais intensa e lesões degenerativas no grupo de fumantes, enquanto lesões degenerativas nos corpos lamelares foram mais intensas com a associação de ambas as drogas.
CONCLUSÕES: Este modelo experimental mostrou alterações morfológicas observadas por microscopia eletrônica, principalmente devido à exposição ao tabaco. Tanto o alcool como o tabaco prejudicaram o crescimento dos animais, o tabaco mostrando um efeito maior sobre o comprimento e o álcool sobre o peso corporal.
Descritores: Alcoolismo.Tabagismo. Pneumopatias. Ratos.
Introduction
Smoking is clearly associated with a chronic inflammation of the lung parenchyma, involving neutrophils, lymphocytes and macrophages. Excessive and inappropriate stimulation of this cells in experimental models has demonstrated destructive pathological changes in the lung. Alveolar destruction results in histological alterations that can be observed using certain basic morphometric techniques. The mean alveolar diameter is increased, which represents perhaps the most sensitive indicator of the emphysematous process. Although the high incidence of chronic lung disease in alcoholics has been accepted and documented, little is known about the effects of alcohol on the lungs. The interrelation between smoking and body weight1 and alcohol consumption and body weight2,3 has been consolidated by several studies; however, none of these have considered the combination of both drugs. Given that 80 to 95% of alcohol consumers are also smokers and that smoking is clearly associated with the increased incidence and severity of diseases in various organs, experimental models that study the harmful effects of chronic alcohol consumption should include groups that combine alcohol and tobacco consumption, thus enabling such groups to more accurately mimic the effects of chronic alcohol abuse in humans.
Thus, this study aimed to investigate the harmful effects of chronic use of alcohol and tobacco produced in rat lung, using morphological assessments, and the effects on the growth of the rats.
Methods
The experimental protocol was performed in accordance with the "Guide to procedures and use of laboratory animals" published by the U.S. National Institute of Health and approved by the Research Ethics and Animal Experimentation Committee of the Botucatu Medical School, Sao Paulo State University (UNESP). Sixty male adult Wistar rats were used, weighing approximately 180-200g, provided by the Central Animal Facility of the UNESP.
The rats were maintained in the Animal Laboratory of Surgical Technique and Experimental Surgery throughout the experimental period, in a closed room at 23 ± 2°C and humidity of 60 ± 5%, controlled by air conditioning, under a 12-hour light-dark period. The rats were housed in polyurethane cages with five rats per cage and received standard commercial chow (Purina®, Brazil) and water ad libitum during a 7-day acclimation period before the onset of the experiment. The cages were cleaned daily and the rats were weighed weekly (Mettler Toledo® scale, model Spider 2).
Following the acclimatization period, the rats were randomly assigned to one of four groups: control, alcohol alone, tobacco alone, alcohol + tobacco. Next, the following protocols were initiated: exposure to ethanol and tobacco smoke for 260 days, in which the concentration of ethanol in the water provided for the rats, was progressively increased by 10°GL per week to 30°GL and then maintained until the end of the experimental period. Alcohol concentration was measured with an alcoholometer. Exposure to cigarette smoke was performed using a method previously standardized in our laboratory experiment4.
The rats were placed in a transparent chamber with an approximate volume of 95x80x65 cm, connected to a smoking device. Puffs of smoke were drawn from the cigarette by vacuum through the smoking device and into the chamber for 30 min. After this period the smoke was exhausted. During the first week, smoke was released at a rate of five cigarettes once a day, seven days a week in the morning. From the second week up to the end of the experimental period, the number of cigarettes was increased to a rate of ten cigarettes. The cigarette used was a commercial brand with the following composition: 1.1 mg nicotine, 14 mg of tar and 15 mg of carbon monoxide.
After completion of the experiment, the rats were fasted for 12h and anesthetized with intraperitoneal injection of sodium pentobarbital (30mg/kg). They were weighed, the snout-anus length was measured and BMI (weight in kg/snout-anus length2) was determined. Cervical-thoracic and abdominal trichotomy, antisepsis with 2% iodine alcohol and xiphopubic laparotomy were all performed. Next, complete section of the inferior vena cava and the abdominal aorta were performed for sacrifice by bleeding.
Optical microscopy (hematoxylin/eosin)
A longitudinal sample of the right and left lungs of seven rats randomly selected from each subgroup were processed for paraffin embedding, sectioned (three microns) and stained with hematoxylin-eosin for light microscopy, using an Axiostar plus microscope (Carl Zeiss do Brasil Ltda.). The slides were evaluated by a pathologist with no prior knowledge of the group they belonged to. The presence of inflammatory infiltrates in the bronchial, bronchiolar and perialveolar regions was evaluated, such that these alterations were recorded as present or absent, and the percentages were estimated.
Mean alveolar diameter
Quantification of pulmonary emphysema was verified by the presence of alveolar destruction determined by measuring the mean alveolar diameter in micrometers (Lm). This technique consists in determining the number of times that gas exchange structures in the parenchyma intersect a series of grid lines5. When emphysema is present, the number of intercepts of the alveolar structures with a system of lines is fewer, indicating alveolar destruction. Lm is determined by the equation: Lm = Ltot / Li, where Ltot is the total length of the lines in the microscopic field (1.250 µm) and Li is the number of intercepts of alveolar structures with the lines of the reticulum. To perform morphometry, 10 randomly selected, noncoincident microscopic fields (Nikon YS2) of samples from seven rats from each group were examined with the aid of an ocular integrator at 400x magnification.
Electronic microscopy
Small fragments of different areas of the lungs of two rats from each group were fixed in glutaraldehyde to examine the alveolar septa by electron microscopy.
Statistical analysis
The results of the variables bronchiolitis, alveolitis, alveolar diameter, snout-anus length, body weight and BMI were expressed as means and standard deviation and statistically tested by factorial 2-way analysis (ANOVA). The post hoc Tukey multiple comparison test was used to investigate which of the means were different to assess the interaction between tobacco smoke exposure and alcohol consumption. All the analyses were performed using SPSS statistical software (SPSS, Inc., Chicago, IL, USA). Statistical significance was set at p<0.05. To study rat growth, the logistic function was used. Estimates of the parameters were calculated for each group in each experiment, together with 95% confidence interval for each parameter.
Results
Of the 60 rats selected for the study, 57 were euthanized. One rat from each of the respective groups died: control, alcohol and tobacco.
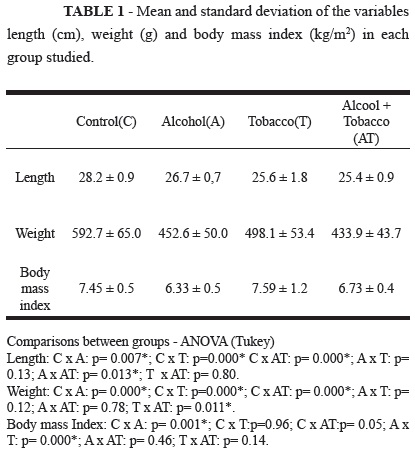
Rat growth
The results of the variables weight, length and BMI are presented in Table 1. Observation verified that the variables body weight and length from the three experimental groups showed lower values than the control group. Rats exposed to tobacco smoke showed the greatest impairment to body length, while those exposed to alcohol showed lower weight gain. Concerning BMI, only the groups exposed to alcohol showed lower values.
Morphological alterations
Bronchiolitis (optical microscopy: hematoxylin/eosin)
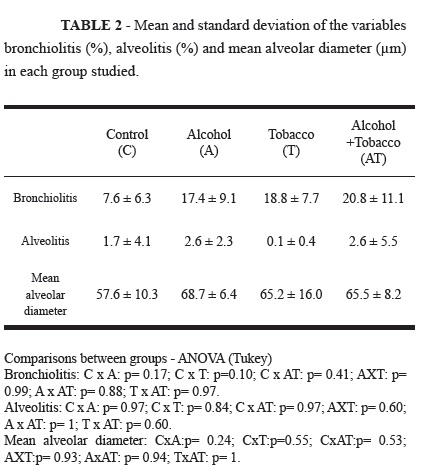
The results of this variable are presented in Table 2. Although, the mean percentages of bronchiolitis were higher in the three groups exposed to the drugs, no statistically significant differences were observed compared with the control group. It should be highlighted that analyses of this variable revealed a predominance of mononuclear cells. Figure 1 shows a bronchiole in a rat from the alcohol + tobacco group.
Alveolitis (optical microscopy: hematoxylin/eosin)
The results of this variable are presented in Table 2. Although the mean percentages of alveolitis were higher in both groups exposed to alcohol, no statistically significant differences were verified compared with the control group. Figure 2 shows lung tissue without alveolitis (2A) and with intense alveolitis (2B).
Mean alveolar diameter
The results of this variable are presented in Table 2. Although the mean alveolar diameters were greater in the three experimental groups, no statistically significant differences were observed compared with the control group.
Electronic microscopy
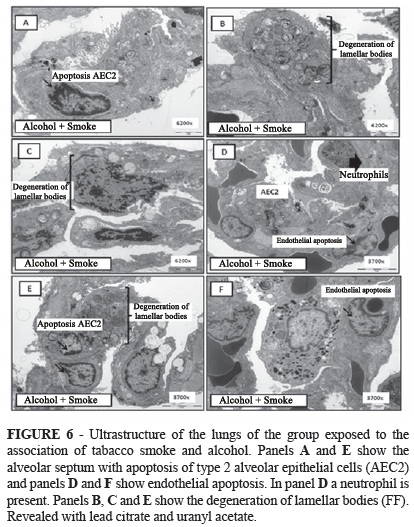
In the alveolar septa of the lungs of control rats, rare apoptotic cells were identified, particularly type 2 alveolar epithelial cells (AEC2) showing the preservation of lamellar bodies (Figure 3).
In the alveolar septa of the lungs of rats from the alcohol (Figure 4) and tobacco groups (Figure 5), besides the increase in apoptosis of AEC2, overlapping alveolar walls were also identified.
AEC2 exhibited organelle degeneration, particularly lamellar bodies. Fibroblasts were also observed in normal activity in the alveolar septa, with the production of ultrastructurally fine collagen fibers forming aggregates in different areas of the interstitium. Apoptotic and degenerative lesions were more intense in the smoking group (Figure 5). The association of both drugs resulted in more intense degenerative alterations in the lamellar bodies (Figure 6).
Discussion
Rat growth
In this study, analysis of the results revealed that exposure to tobacco and alcohol consumption contributed to the impaired growth of the rats. The interrelation between smoking and body weight has been consolidated by several studies1. The effect of tobacco on body weight appears to be mediated by nicotine. Studies on humans and mices indicate that the administration of nicotine induces body weight loss, probably as a result of appetite suppression by acute elevation of the neurotransmitters dopamine and serotonin6. In humans, smoking increases adrenergic activity, which induces thermogenesis and the consequent weight loss7. Another aspect that should be considered is the direct effect of nicotine on the metabolism of adipose tissue, causing greater oxidation of lipids in smokers, which helps explain lower BMI values compared with nonsmokers8,9. In the rat, we were unable to demonstrate lower BMI in those exposed to tobacco; however, rats exposed to ethanol presented a reduction.
In terms of caloric content, ethanol is the only drug which when under oxidization by alcohol dehydrogenase produces 7.1 kcal per gram of oxidized ethanol. This supply of energy to the body causes the individual not feel the need to eat. This could have occurred in our rats, explaining both the difficulty in achieving weight gain and the BMI values of rats exposed to this drug. In addition to the known toxic effects of ethanol on the absorption and synthesis of essential nutrients10, some metabolic abnormalities have also been described. Pirola and Lieber11 reported that activation of the microsomal ethanol oxidizing system in hepatocytes of alcoholics may consume 1/3 of the ethanol caloric content and could compromise adenosine triphosphate (ATP) synthesis. The body uses six molecules of ATP per gram of oxidized ethanol12.
Experimental studies are consistent in reporting diminished body weight in rats exposed to ethanol. Both low (5% v/v) and high concentrations of ethanol (40% v/v) have been related to lower weight gain2. Similar results were verified with concentrations of 20%. Analysis of the results obtained in this study highlight that both tobacco and alcohol interfered negatively in the growth of the rats studied. Transposing the logic of these results to humans emphasizes the importance of deterring the use of tobacco and alcohol by children and/or teenagers who are still growing.
Morphological study
While evaluating lung morphology, the only significant alterations observed were revealed by electron microscopy. In the rats exposed to tobacco smoke and alcohol consumption, the mean percentages of bronchiolitis (all groups) and alveolitis (all, except the tobacco group) were higher than the control group, though these differences were not significant.
Despite not having been able to show significant differences between the groups using optical microscopy, the inflammatory cells identified in the experimental groups were predominantly mononuclear cells. Of these, macrophages were increased in patients with chronic obstructive pulmonary disease (COPD) due to the increased need for phagocytosis in these individuals, or increased need for the production of cytokines to mediate the inflammatory response. The release of macrophage arachidonic acid metabolites to mediate the inflammatory response can also generate reactive oxygen species (ROS) and cellular damage. The study of these ROS forms part of the continuation of this work. In general, macrophages represent 90% of the cells aspirated during bronchoalveolar lavage. In smokers, this percentage can be maintained; however, the absolute number of cells is usually four to five times greater13.
Regarding alcohol consumption, experimental studies in mices have shown that alcohol metabolites accumulate in the lungs and that intraperitoneal injection of alcohol can produce histochemical changes in lung tissue14. It has also been reported that alcohol reduces the ability to clear inhaled bacteria, which increases susceptibility to respiratory infections15. Although it is generally believed that alcohol makes the lungs more susceptible to the effects of injury induced by smoking, the model used here was unable to verify this phenomenon.
In this study, we verified that although the mean alveolar diameters of the rats of the experimental groups were greater than the control group, no significant differences were observed and emphysema could not be proven. This was an unexpected result, since the period of tabacco smoke exposure was longer than that recommended in the literature, ranging from four to six months16 of exposure for the development of emphysema.
However, it should be emphasized that the negative influence of tobacco exposure, whether alone or in combination with alcohol, was evident from the electron microscopy results.
The literature is unanimous in reporting the effects of smoking on the pathogenesis of COPD, but it remains unclear whether or not alcohol has any influence on this disease. In an extensive review of the effects of alcohol on the respiratory system, Sisson17 stated that it is difficult to define the influence of alcohol on the pathogenesis of COPD based on studies conducted in the last five decades, because different studies have often reported conflicting results. According to the author, severe alcohol consumption exacerbates the risk factors related to tobacco in the development of COPD; however, moderate consumption of alcohol may reduce the risk of death and the development of severe pulmonary dysfunction related to COPD18.
Conclusions
This experimental model showed morphological alterations only observed by electron microscopy, principally resulting from exposure to tobacco smoke. It was not possible to determine any significant differences using optical microscopy. The effects of both alcohol and tobacco were evident in the compromised body growth of the rats, such that damage by tobacco had a greater effect on body length and by alcohol, on body weight.
Acknowledgment
We thank the Research Support Group of the Botucatu School of Medicine, UNESP, for language revision.
Conflict of interest: none
Financial source: FAPESP
Received: May 17, 2012
Review: July 20, 2012
Accepted: August 21, 2012
- 1. Klesges RC, Ward KD, Ray JW, Cutter G, Jacobs DR Jr, Wagenknecht LE. The prospective relationships between smoking and weight in a young, biracial cohort: the Coronary Artery Risk Development in Young Adults Study. J Consult Clin Psychol. 1998;66(6):987-93.
- 2. Macieira MS, Almeida WG, Silva EA, Schenberg LC, Nakamura- Palacios EM. Alcohol dependence induced in rats by semivoluntary intermittent intake. Braz J Med Biol Res. 1997;30:1107-11.
- 3. Pereira RSC, Hasimoto CN, Pelafsky L, Llanos JC, Cataneo DC, Spadella CT, Minossi JG. Intestinal healing in rats submitted to ethanol ingestion. Acta Cir Bras. 2012;27(3):236-43.
- 4. Paiva SAR, Zornoff LAM, Okoshi MP, Okoshi K; Cicogna AC, Campana AO. Behavior of cardiac variables in animals exposed to cigarette smoke. Arq Bras Cardiol. 2003;81(3): 221-4.
- 5. Weibel ER. Principles and methods for the morphometric study of the lung and other organs. Lab Invest. 1963;12:131-55.
- 6. Chen H, Vlahos R, Bozinovski S, Jones J, Anderson GP, Morris MJ. Effect of short-term cigarette smoke exposure on body weight, appetite and brain neuropeptide Y in mice. Neuropsychopharmacology. 2005;30(4):713-9.
- 7. Pisinger C, Jorgensen T. Waist circumference and weight following smoking cessation in a general population: the Inter99 study. Prev Med. 2007;44(4):290-5.
- 8. Filozof C, Fernandez-Pinilla MC, Fernandez-Cruz A. Smoking cessation and weight gain. Obes Rev. 2004;5(2):95-103.
- 9. Ferrara CM, Kumar M, Nicklas B, McCrone S, Goldberg AP. Weight gain and adipose tissue metabolism after smoking cessation in women. Int J Obes Relat Metab Disord. 2001;25(9):1322-6.
- 10. Lieber, CS. Perspectives: do alcohol calories count? Am J Clin Nutr.1991 54:976-82.
- 11. Pirola RC, Lieber CS. Hypothesis: energy wastage in alcoholism and drug abuse: possible role of hepatic microsomal enzymes. Am J Clin Nutr. 1976;29(1):90-3.
- 12. Lands WEM, Zakhari S. The case of missing calories. Am J Clin Nutr. 1991;54(1):47-8.
- 13. Finlay GA, O'Driscoll LR, Russell KJ, D'Arcy EM, Masterson JB, Fitz Gerald MX, O'Connor CM. Matrix metalloproteinase expression and production by alveolar macrophages in emphysema. Am J Resp Crit Care Med. 1997;156(1):240-7.
- 14. Burch GE, DePasquale N. Alcoholic lung disease: a hypothesis. Am Heart J. 1966;73:147-48.
- 15. Green G, Kass E. Factors influencing the clearance of bacteria by the lung. J Clin Invest. 1964;43:769-78.
- 16. Taraseviciene-Stewart L, Voelkel NF. Molecular pathogenesis of emphysema. J Clin Invest. 2008;118(2):394-402.
- 17. Sisson JH. Alcohol and airways function in health and disease. Alcohol. 2007;41(5):293-307.
- 18. Tabak C, Smit HA, Heederik D, Ocke MC, Kromhout D. Diet and chronic obstructive pulmonary disease: independent beneficial effects of fruits, whole grains, and alcohol (the MORGEN study). Clin Exp Allergy. 2001;31:747-5.
Publication Dates
-
Publication in this collection
27 Sept 2012 -
Date of issue
Oct 2012
History
-
Received
17 May 2012 -
Accepted
21 Aug 2012 -
Reviewed
20 July 2012