Abstract
PURPOSE: To investigate the effect of Cenostigma macrophyllum Tul. in the tissue repair process of skin lesions in rats with induced Diabetes mellitus. METHODS: Sixty-three male Wistar rats were distributed into three groups: control (C), diabetic (D), and diabetic treated with an oil-in-water emulsion of the plant (DPL) subdivided according to time of observation (seven, 14, and 28 days). Diabetes was induced by administration of by injection of streptozotocin (50 mg/kg, after a 12-h fast) into the penile vein and confirmed by glucose levels exceeding 240 mg/dL. Held surgical lesion (2.5 cm in diameter) on the back of the animals. The DPL received topical application of group of the oil-in-water emulsion plant (0.5ml). To evaluate the levels of nitric oxide, was collected 5ml of blood from the abdominal aorta, after his euthanasia. The samples of interest were sent for routine histological processing. RESULTS:A reduction in the percentage of inflammatory cells and increased numbers of fibroblasts in the group DPL, seven days, compared to the other groups. At 14 days, the DPL group also showed a higher concentration of nitric oxide (p<0.01) than in groups C and D. CONCLUSION: The oil-in-water emulsion of C. macrophyllum Tul accelerated wound healing in diabetic rats.
Diabetes mellitus; Plants, Medicinal; Wound Healing; Nitric Oxide; Skin; Rats
7 - ORIGINAL ARTICLE
WOUND HEALING
Cenostigma macrophyllum Tul. on the healing of skin wounds in rats with Diabetes mellitus1 1 Research performed at Institute of Research and Development (IPD), Vale do Paraiba University (UNIVAP), Sao Jose dos Campos-SP and College Integral Differential (FACID), Teresina-PI, Brazil.
Nayana Pinheiro Machado de Freitas CoelhoI, Veruska Cronemberger NogueiraII, Maria Angélica Gargione CardosoIII, Luciano da Silva LopesIV, Paulo Pedro do NascimentoV, Evair dos Santos RochaVI, Caio Leôncio Probo da SilvaVI, Emília Ângela Loschiavo ArisawaVII
IFellow PhD degree, Postgraduate Program in Biomedical Engineering, Vale do Paraiba University (UNIVAP), Sao Jose dos Campos-SP, Brazil. Conception, design, intellectual and scientific of the study
IIFellow PhD degree, Postgraduate Program in Biomedical Engineering, UNIVAP, Sao Jose dos Campos-SP, Brazil. Acquisition of data
IIIAssistant Professor, IPD, UNIVAP, Sao Jose dos Campos-SP, Brazil. Conception and design of the study, critical revision
IVAssociate Professor, Department of Pharmacology, FACID, Teresina-PI, Brazil. Acquisition and interpretation of data
VAssistant Professor, Department of Pharmacology, FACID, Teresina-PI, Brazil. Histopathological examinations
VIGraduate student, FACID, Teresina-PI, Brazil. Acquisition of data
VIIAssociate Professor, IPD, UNIVAP, Sao Jose dos Campos-SP, Brazil. Scientific and intellectual content of the study, interpretation of data, critical revision
Correspondence Correspondence: Nayana Pinheiro Machado de Freitas Coelho Rua Miosótis, 303/2202 64049-536 Teresina PI Brasil Tel.: (55-86)3234-2045/9989-1114 nayanamachado@oi.com.br
ABSTRACT
PURPOSE: To investigate the effect of Cenostigma macrophyllumTul. in the tissue repair process of skin lesions in rats with induced Diabetes mellitus.
METHODS: Sixty-three male Wistar rats were distributed into three groups: control (C), diabetic (D), and diabetic treated with an oil-in-water emulsion of the plant (DPL) subdivided according to time of observation (seven, 14, and 28 days). Diabetes was induced by administration of by injection of streptozotocin (50 mg/kg, after a 12-h fast) into the penile vein and confirmed by glucose levels exceeding 240 mg/dL. Held surgical lesion (2.5 cm in diameter) on the back of the animals. The DPL received topical application of group of the oil-in-water emulsion plant (0.5ml). To evaluate the levels of nitric oxide, was collected 5ml of blood from the abdominal aorta, after his euthanasia. The samples of interest were sent for routine histological processing.
RESULTS:A reduction in the percentage of inflammatory cells and increased numbers of fibroblasts in the group DPL, seven days, compared to the other groups. At 14 days, the DPL group also showed a higher concentration of nitric oxide (p<0.01) than in groups C and D.
CONCLUSION: The oil-in-water emulsion of C. macrophyllum Tul accelerated wound healing in diabetic rats.
Key words:Diabetes mellitus. Plants, Medicinal. Wound Healing. Nitric Oxide. Skin. Rats.
Introduction
Diabetes mellitus (DM) is a metabolic disorder characterized by hyperglycemia and abnormalities in protein, lipid and carbohydrate metabolism. The disease is a public health problem due to its high incidence and prevalence, as well as social and economic repercussions resulting from premature death, work incapacity, and high costs for the control and treatment of its complications1,2. These factors have encouraged studies on the disease in recent years, particularly those trying to develop new drugs for the treatment of tissue injuries that affect patients with diabetes1.
The main alterations that occur in DM associated with suppression of the inflammatory response are reduced angiogenesis, altered proliferation of fibroblasts and endothelial cells, reduced fibroblast migration, and inadequate collagen deposition3. Another factor is the complexity of the wound repair process which starts with the formation of granulation tissue after the release of chemical mediators. This step is by the proliferation of inflammatory cells, fibroblasts, collagen fiber and extracellular matrix-producing cells, and endothelial cells to supply nutrients and oxygen to the newly formed tissue. The last step in this process, tissue remodeling, involves fibers and extracellular matrix reorganization, confering the appearance of mature tissue4,5. If for some reason alterations occur in the proliferation and migration of fibroblasts, the wound repair process will be delayed2,5.
Nitric oxide is one of the mediators that play an important role in wound healing and has been implicated in DM because reduced levels can cause alterations in vascular permeability and a reduction of capillary flow, causing oxidative stress6,7. High amounts of nitric oxide are produced under normal conditions, increasing blood flow and vascular permeability, with this mediator acting as a moderate vasodilator. In contrast, the endothelial dysfunction and decreased nitric oxide production observed in patients with DM reduce tissue perfusion and cause vasoconstriction and reduced blood flow, with a consequent delay in wound healing8.
Studies have tested new drugs for the treatment of skin wounds in patients with DM in an attempt to minimize inflammation and to accelerate the repair process by increasing the production of fibroblasts and collagen fibers, factors that are important for wound healing9. Medicinal plants such as Strychnos pseudoquina and Momordica charantia have been used for the treatment of DM as coadjuvants in the anti-inflammatory process9,10.
Commonly known as "caneleiro", Cenostigma macrophyllum Tul. var, acuminata Teles Freire belongs to the family Caesalpiniaceae and is found throughout Brazil, except for the southern region. The plants are rich in flavonoids and biosynthetically related compounds such as rotenoids and isoflavonoids that play a role in wound healing11,12. Studies using the leaf extract of Cenostigma macrophyllum Tul. have demonstrated its antitumor, antioxidant, anti-inflammatory, antibacterial and antiviral properties, as well as the presence of biflavones11.
In view of the above considerations, the objective of the present study was to evaluate the effect of an oil-in-water emulsion of Cenostigma macrophyllum Tul. var. acuminata Teles Freire on the healing of surgical wounds in rats with experimentally induced DM.
Methods
The study was approved by the Ethics Committee of College Integral Differential (FACID), Teresina-PI, Brazil (nº. 263/2009) and was conducted according to Lei Arouca No. 11.794/2008. Sixty-three male Wistar rats (Rattus norvegicus), aged 30 to 40 days and weighing 200-250 g, were used. The animals were kept in individual cages at the animal house of the institution with free access to food and water under a light/dark cycle.
Experimental groups
The animals were randomly distributed into three groups of 21 animals each according to treatment: control, experimentally induced DM (DM), and experimentally induced DM treated with an oil-in-water emulsion of Cenostigma macrophyllum (DPL). The animals were subdivided according to time of observation seven, 14 and 28 days (n = 7 per group). Control animals received no treatment. Diabetes was induced in animals of groups DM and DPL. The wounds of animals of the DPL group were treated with 0.5 mL of the oil-in-water emulsion for seven, 14, and 28 days.
Induction of Diabetes mellitus
For the induction of diabetes, a single dose (50 mg/kg body weight) of the diabetogenic drug streptozotocin (Sigma Chemical Company, St. Louis, MO, USA), dissolved in 0.01 M citrate buffer (pH 4.5), was injected through the penile vein after a fast of at least 12 h3. Diabetes was confirmed by an increase of glucose levels measured with an Accu-Check® glucose meter on day 21 after induction. Animals with glucose levels > 240 mg/dL were defined as diabetic.
Surgical procedure
The animals were anesthetized by intramuscular administration of 10% ketamine hydrochloride (0.1mL/100g/kg) and the same dose of 2% xylazine hydrochloride. An area in the dorsolateral region measuring 6 x 4 cm was shaved and cleaned with 4% iodinated alcohol. Next, a circular injury measuring 2.5 cm in diameter was created in the center of the area with a circular instrument and nº4 scalpel blade until complete removal of the tissue.
After surgery, the animals received antibiotic prophylaxis consisting of deep intramuscular injection of a single dose of 0.02 mL/100 g of a broad-spectrum antibiotic (Pentabiótico, Fort Dodge®).The procedure was carried out in a silent environment with minimal handling to avoid stress to the animal.
Preparation of the oil-in-water emulsion of Cenostigma macrophyllum
First, seeds were collected from the plant and dried and their shells were removed. The seeds were triturated and extracted with hexane in the dark for 30 min under occasional shaking. The hexane extract was filtered and concentrated in a rotary evaporator under reduced pressure at 45ºC. The oil of the seeds was stored in an amber flask in a refrigerator until the time of use. The emulsion was prepared using standard procedures13, which cannot be provided because of a patent application process.
Experimental procedure
Animals of the control and DM groups were not submitted to any treatment. In the DPL group, the animals received a topical application of 0.5 mL of the oil-in-water emulsion of C. macrophyllum once a day for seven, 14, and 28 days. It should be noted that 21 days were necessary for the confirmation of DM since a significant increase of glucose levels and stability of the diabetic syndrome were only observed after this period.
Macroscopic evaluation
For macroscopic evaluation of the wound area, digital images were captured with a Nikon Coolpix P100 digital camera (resolution of 10.6 Megapixels) mounted on a tripod at a constant distance of 30 cm from the surgical wound. All images were analyzed with the ImageJ® software, delimiting the wound periphery and comparing the initial and final wound area in all groups at the different time points studied. The percent reduction in wound size was calculated using the following formula:
Initial wound area final wound area / initial wound area x 100.
Histological and histomorphometric analysis
The animals were sacrificed with an overdose of the anesthetic (60 mg/kg sodium pentobarbital). A specimen of interest was removed using a sterile knife with a margin of 1 cm around the wound, identified, fixed in 10% formalin, and submitted to routine histological processing. Semi-serial sections (0.5 mm) were cut and stained with hematoxylin-eosin. Histological and histomorphometric analysis was performed using a Leica DM 1000 light microscope coupled to a digital camera system. Images were acquired with the Leica Qwin program from three sites of the wound per animal (right and left margin and center) using a x40 objective. The ImageJ® program in the Cell Counter function was used to determine the number of inflammatory cells. For qualitative assessment, the presence of inflammatory cells, fibroblasts and collagen fibers was evaluated in the histological sections at the different time points.
Measurement of nitric oxide
Nitric oxide was measured in all groups at the different time points (seven, 14, and 28 days) and immediately before sacrifice of the animals. For this purpose, 5 mL blood was collected from the abdominal artery of anesthetized animals and the serum was obtained by centrifugation at 2000 rpm for 15 min. For nitric oxide determination 100 µL of animal serum was mixed with 100 µL of Griess reagent (Sigma-AldrichTM) in a multiwall plate (TPP-Switzerland) in duplicate. The plate was incubated for 10 min at room temperature and absorbance was read in a microplate spectrophotometer (µQuantTM, BioTek® Instruments Inc., USA) at 540 nm5. A standard curve was constructed using sodium nitrate at concentrations of 1.5 to 200µM.
Statistical analysis
The histomorphometric and nitric oxide results were analyzed statistically by ANOVA, followed by the Tukey test, adopting a 95% confidence interval and a p<0.05. Statistical analysis was performed using the GraphPad Prism 5.0 program.
Results
Comparison of the percent reduction in wound size showed a significant difference between the C and D groups on day 7 after wounding (p<0.001) and between the D and DPL groups. At 14 days, wound size reduction differed significantly between the control and D groups and between the D and DPL groups (p<0.01 for both) (Figure 1).
Qualitative analysis of the inflammatory process in specimens of the DPL group on day 7 after wounding revealed a moderate amount of granulation tissue, a larger number of blood vessels, presence of fibroblasts and collagen fiber deposition when compared to the D group which presented a larger number of inflammatory cells. At 14 days, fibroblasts predominated in the DPL group, the inflammatory infiltrate had disappeared, and wound reepithelization had started (Figure 2).
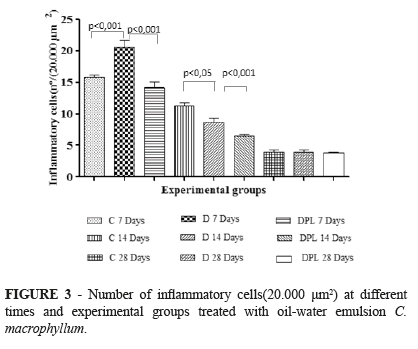
Comparison of the number of inflammatory cells showed significant differences between the C and D groups and between the D and DPL group on day 7 (p<0.01 for both, Tukey test). At 14 days, the DPL group presented a significant difference when compared to the control and D groups (p<0.001 and p<0.05, respectively). However, no significant differences between groups were observed on day 28 (Figure 3).
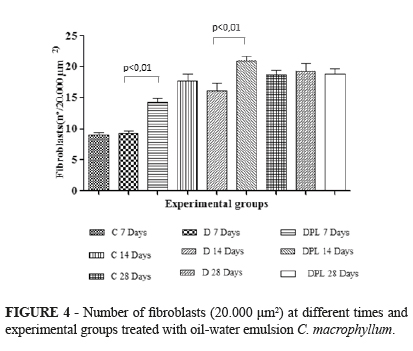
The number of fibroblasts differed significantly between the D and DPL groups on day 7 (p<0.01). The same was observed on day 14, with a larger number of fibroblasts in animals of the DPL group (Figure 4).
Nitric oxide levels differed significantly between the C and DPL groups on day 14 after wounding (p<0.05), i.e., specimens of the DPL group at 14 days presented the highest concentrations of nitric oxide among the groups studied (Figure 5).
Discussion
Under physiological conditions, fibroblasts are stimulated to migrate to the injured area during tissue repair and to produce collagen fibers necessary for wound healing1. In contrast, in addition to elevated glucose levels, patients with DM present reduced vascular permeability and blood flow that alters the tissue repair process, influencing both the formation and remodeling of collagen fibers2. The steps of tissue repair can be evaluated in skin wounds by qualitative and quantitative histological analysis of the main features of this process, including the number of inflammatory cells, number of newly formed blood vessels (angiogenesis), and number of fibroblasts14,15.
The great biodiversity found in Brazil offers to researchers a variety of substances with therapeutic effect on different diseases, including natural compounds with wound healing properties8,9,13. Within this context, Cenostigma macrophyllum Tul. is common in different regions of Brazil and is the tree symbol of the city of Teresina, State of Piaui. Cenostigma macrophyllum Tul. var. acuminata Teles Freire, which belongs to the family Caesalpiniaceae, shows biochemical, anti-inflammatory and healing properties. Therefore, the study of its effect on the healing of wounds created in animals with experimentally induced diabetes may result in the application of this plant in the health area.
Different natural compounds have been tested for tissue repair in an attempt to accelerate this process and to obtain better final outcomes. For example, the aqueous extract of Orbignya phalerata, commonly known as babassu, has been shown to significantly reduce the inflammatory process in rat skin after seven days of treatment13. Similar results have been reported for the application of Rafflesia hasselti to rat skin wounds, with the identification of components that modulate the inflammatory phase of wound healing16. The oil of Caryocar brasiliense (Pequi) is another substance that has been studied for the natural healing of skin wounds. This plant also has healing properties and the results of experimental studies have shown a reduction in the number of polymorphonuclear cells on day 3 after wounding, as well as an increase in angiogenesis and in the number of fibroblasts on day 717.
However, there is no rule that phytotherapeutic agents have similar effects. In this respect, delayed reepithelization of surgical wounds has been observed in a study investigating the effect of a hydroalcoholic extract of Aroeira on the treatment of skin wounds in rats18. In contrast, in the present study wounds created in healthy and diabetic rats that were treated with the oil-in-water emulsion of C. macrophyllum showed the best wound healing outcomes.
Collagen fiber production starts around day 7 after wounding and increases gradually until day 14. This process is accompanied by a reduction of the inflammatory infiltrate. The final phase of wound healing begins at about day 21 and is characterized by complete reepithelization of the wound area9,14-16. In contrast, in patients with DM, the inflammatory infiltrate persists in acute and chronic wounds and mainly consists of neutrophils and macrophages. In addition, collagen fiber deposition is reduced, a fact contributing to delayed wound healing. Therefore, studies on the use of natural products for wound treatment in animals with experimentally induced DM investigating the action of these compounds on the inflammatory process may increase the range of products available for clinical application9. In this respect, medicinal plants have been applied to wound treatment in animals with experimentally induced DM. Studies using Strychnos pseudoquina and Momordica charantia demonstrated that these plants exert wound healing activity, reduce the inflammatory process, and increase collagen production8,9.
In the present study, a higher percentage of wound size reduction was observed in specimens of the groups treated with C. macrophyllum on days 7 and 14 after wounding. Similarly, the aqueous extract of Strychnos pseudoquina was found to reduce the area of diabetic wounds after 14 days of topical treatment8. Topical application of the oil-in-water emulsion of C. macrophyllum Tul. to skin wounds surgically created in rats with experimentally induced DM also reduced the number of inflammatory cells and increased fibroplasia, demonstrating the positive effects of this extract on wound healing in diabetic rats.
In view of the importance of tissue nutrition for the inflammatory response and tissue repair, the analysis of nitric oxide, a known vasodilator that regulates different activities including the synthesis and remodeling of collagen, permits to evaluate the progression of wound healing19,20. Therefore, this study investigated whether animals with experimentally induced DM have deficient production of nitric oxide as observed in humans. This deficiency is due to low blood levels of nitric oxide synthase, the enzyme catalyzing the production of this chemical mediator of inflammation. In addition, the concentration of L-arginine, an amino acid that plays an important role in the activation of nitric oxide synthase, is reduced20. Studies have used extracts of medicinal plants to investigate their effect on the production of nitric oxide20,21. A study using Helianthus annus showed a close relationship between nitric oxide production and higher activation of L-arginine when the substance was applied to wounds20. A similar observation was made with the use of Carica papaya extract on diabetic wounds, which increased the concentration of nitric oxide20.
The determination of nitric oxide levels in patients with DM is important because of the effect of this mediator on vascular permeability, oxygen transport, and collagen deposition8,19. In the present study, higher nitric oxide production was observed on day 14 after wounding in animals of the DPL group. This finding suggests that topical application of C. macrophyllum Tul. acts directly on the production of nitric oxide and also on calcium channels, increasing the production of nitric oxide synthase and, consequently, nitric oxide synthesis. The consequent arteriolar vasodilatation increases tissue nutrition and the formation of collagen fibers by fibroblasts and accelerates wound healing in animals with experimentally induced DM.
In summary, the oil-in-water emulsion of C. macrophyllum accelerated wound healing in animals with experimentally induced DM. However, further studies are needed to identify the mechanism of action underlying the increased production of nitric oxide and the active ingredient of the plant associated with this response.
Conclusion
The oil-in-water emulsion of C. macrophyllum Tul. Var. acuminata Teles Freire accelerated the healing of surgical wounds in rats with experimentally induced Diabetes mellitus, promoting a reduction in wound size and in the inflammatory process and increasing the number of fibroblasts and nitric oxide production.
Acknowledgments
To Talvany Luis de Barros for experimental procedures and researchers Mariana Helena Chaves for providing the materials for this research and Charllyton Luis Sena da Costa for statistical analysis.
Received: April 18, 2013
Review: June 20, 2013
Accepted: July 22, 2013
Conflict of interest: none
Financial source: Piaui Research Foundation (FAPEPI) and College Integral Differential (FACID).
- 1. Carvalho PTC, Silva IS, Reis FA, Pereira DM, Aydos RD. Influence of InGaAlP laser on the healing of skin wounds in diabetic rats. Acta Cir Bras. 2010;25(1):71-9.
- 2. Silva M, Lima WG, Silva ME, Pedrosa ML. Effect of streptozotocin on the glycemic and lipid profiles and oxidative stress in hamsters. Arq Bras Endocrinol Metab. 2011;55(1):46-53.
- 3. Ebaid H, Salem A, Sayed A, Metwalli A. Whey protein enhances normal inflammatory responses during cutaneous wound healing in diabetic rats. Lipids Health Dis. 2011;10:235.
- 4. Wilgus TA, Bergdall VK, DiPietro LA, Oberyszyn TM. The effects of hydrogen peroxide on fetal wound repair. Wound Repair Regen. 2008;13(2):28-48.
- 5. Ochoa O, Torres FM, Shireman PK. Chemokines and diabetic wound healing. Vascular. 2007;15(6):350-5.
- 6. D'Ávila VGFC, Sousa Júnior NB, Sousa FB, Guillo LA. Avaliação da produção de óxido nítrico em ratos, submetidos aos exercícios aeróbio e anaeróbio. Rev Bras Cienc Farm. 2005;44(4):755-61.
- 7. Hsu WT, Tsai LY, Lin SK, Hsiao K, Chen BH. Effects of diabetes duration and glycemic control on free radicals in children with type 1 diabetes mellitus. Ann Clin Lab Sci. 2006;36(2):174-8.
- 8. Honório-França AC, Marins CMF, Boldrini F, França EL. Evaluation of hypoglycemic activity and healing of extract from amongst bark of "Quina do Cerrado" (Strychnos pseudoquina ST. HILL). Acta Cir Bras. 2008;23(6):504-10.
- 9. Harinantenaina L, Tanaka M, Takaoka S, Oda M, Mogami O, Uchida M, Asakawa Y. Momordica charantia constituents and antidiabetic screening of the isolated major compounds. Chem Pharm Bull. 2006;54(7):1017-21.
- 10. Silva HR, Silva CCM, Caland Neto, LB, Lopes JAD, Citó AMGL, Chaves MH. Constituintes químicos das cascas do caule de Cenostigma macrophyllum: ocorrência de colesterol. Quim Nova. 2007;30(8):1877-81.
- 11. Sousa CMM, Silva HR, Vieira Júnior GM, Ayres MCC, Costa CLS, Araújo DS, Cavalcante LCD, Barros EDS, Araújo PBM, Brandão MS, Chaves MH. Fenóis totais e atividade antioxidante de cinco plantas medicinais. Quim Nova. 2007;30:351-7.
- 12. Frange RCC, Garcia MTJ. Desenvolvimento de emulsões óleo de oliva/água: avaliação da estabilidade física. Rev Ciênc Farm Básica Apl. 2009;30(3):263-371.
- 13. Martins NLP, Malafaia O, Ribas-Filho JM, Heibel M, Baldez RN, Vasconcelos PRL, Moreira H, Mazza M, Nassif PAN, Wallbach TZ. Análise comparativa da cicatrização da pele com o uso intraperitoneal de extrato aquoso de Orbignya phalerata (babaçu). Estudo controlado em ratos. Acta Cir Bras. 2006;21(Suppl 3):66-75.
- 14. Oliveira SHSO, Soares MJGO, Rocha PS. Uso de cobertura com colágeno e aloe vera no tratamento de ferida isquêmica: estudo de caso. Rev Esc Enferm. 2010;44(2):346-51.
- 15. Garros IC, Campos ACL, Tâmbara EM, Tenório SB, Torres OJM, Agulham MA, Araújo ACF, Sains-Isolan PMB, Oliveira EM, Arruda ECM. Extrato de Passiflora edulis na cicatrização de feridas cutâneas abertas em ratos: estudo morfológico e histológico. Acta Cir Bras. 2006;21(Suppl 3):55-65.
- 16. Abdulla MA, Ahmed KA, Ali HM, Noor SM, Ismail S. Wound healing activities of Rafflesia hasselti extract in rats. J Clin Biochem Nutr. 2009;45(3):304-8.
- 17. Batista JS, Silva AE, Rodrigues CMF, Costa KMFM, Oliveira AF, Paiva ES, F. Nunes FVA, Olinda RG. Avaliação da atividade cicatrizante do óleo de pequi (Caryocar coriaceum Wittm) em feridas cutâneas produzidas experimentalmente em ratos. Arq Inst Biol. 2010;77(3):441-7.
- 18. Branco-Neto MLC, Ribas-Filho JM, Malafaia O, Oliveira-Filho MA, Czeczko NG, Aoki S, Cunha R, Fonseca VR, Teixeira HM, Aguiar LRF. Avaliação do extrato hidroalcoólico de Aroeira (Schinus terebinthifolius Raddi) no processo de cicatrização de feridas em pele de ratos. Acta Cir Bras. 2006;(Suppl 2):17-22.
- 19. Gurdol F, Cimsit M, Oner-Iyidogan Y, Kocak H, Sengun S, Yalcinkaya-Demirsoz S. Collagen synthesis, nitric oxide and asymmetric dimethylarginine in diabetic subjects undergoing hyperbaric oxygen therapy. Physiol Res. 2010;59:423-9.
- 20. Anuar NS, Zahari SS, Taib IA, Rahman MT. Effect of green and ripe Carica papaya epicarp extracts on wound healing and during pregnancy. Food Chem Toxicol. 2008;46(7):2384-9.
- 21. Chaki M, Valderrama R, Fernandez-Ocanã AM, Carreras A, Gómez-Rodríguez MV, Pedrajas JR, Begara-Morales JC, Sánchez-Calvo B, Luque F, Leterrier M, Corpas FJ, Barroso JB. Mechanical wounding induces a nitrosative stress by down-regulation of GSNO reductase and an increase in S-nitrosothiols in sunflower (Helianthus annuus) seedlings. J Ex Bot. 2011;62(6): 180313.
Publication Dates
-
Publication in this collection
26 July 2013 -
Date of issue
Aug 2013
History
-
Received
18 Apr 2013 -
Accepted
22 July 2013 -
Reviewed
20 June 2013