Abstract
PURPOSE:
To investigate the induction of neoplastic lesions under the action of ultraviolet B radiation (UVR-B) and dimethyl benzanthracene (DMBA).
METHODS:
Forty Wistar rats were assigned to four groups (ten animals each), according to the procedure: group A received UVR-B irradiation, group B received topic DMBA, group C, UVR-B+DMBA and group D as control, observed for ten weeks. In the tenth week they went through a skin biopsy and histopathological study. The average thickness of the epidermis was calculated and evaluated statistically.
RESULTS:
Macroscopic lesions in group B were more of inflammatory kind compared to group A. Group C presented more injuries with neoplastic features than the others (p<0.01). Histologically there was a significant increase in thickness of the epidermis of all groups compared to control, however the greatest thickness measures occurred in Group C (p<0.01).
CONCLUSIONS:
The population exposed to ultraviolet B radiation is subject to suffer skin lesions that can develop into cancer. The association with hydrocarbons as the dimethyl benzanthracene increases the possibility of malignancy. May not be clinically evident determine when a solar keratosis ends and when a CEC begins. For this reason, histological study associated with health education prompting the early and irreversible injury prevention is necessary.
9,10-Dimethyl-1,2-benzanthracene; Epidermis; Solar Radiation; Skin Neoplasms; Rats
Introduction
The incidence of skin cancer has been increasing each year highlighting the basal cell carcinoma (BCC) and melanoma (MM). The sum of the incidence of these neoplasms and cutaneous squamous cell carcinomas (SCC) determines that skin cancer is the most common among all other types of cancers in the body, being a major problem of public health11. Instituto Nacional de Câncer José Alencar Gomes da Silva. General coordination of strategic actions. Coordination of prevention and awareness. 2012 estimate: incidence of cancer in Brazil. Rio de Janeiro: INCA; 2011..
The use of chemicals is more and more often and, in contact with human skin, many of them can cause skin cancer. The dimethylbenzoantracen 7.12 (DMBA) belongs to the group of polycyclic aromatic hydrocarbons and is considered a carcinogen. Is found in occupational and residential environment and is among the most dangerous air pollutants. It originates from burning agricultural plantations, metallurgical, mineral coal and burning of petroleum products, cigarette smoke, plastics, pesticides and paints. Can still be found in smoked foods, fruits, grains and vegetables, due to atmospheric deposition on them22. Oliveira LBO, Bampi VF, Garcia CF, Souza E. Angioarchitecture of squamous cell carcinoma from hamster buccal pouch: a scanning electron microscopy study. J Scan Microsc. 2009;31(5):188-94..
Biomolecular and epidemiological data suggest an association between sun exposure and skin cancer development. Recurrent exposure to UVR-B can cause cutaneous effects not only by genetic mutation, but also by caused immunodeficiency. For each new exhibition there are major changes. Photobiological effects over time can be observed on the skin, immediately or later33. Jans J, Garinis GA, Schul W, van Oudenaren A, Moorhouse M, Smid M, Yurda_Gul S. Differential role of basal keratinocytes in UV-induced immunosuppression and skin cancer. Mol Cell Biol. 2006;26(22):8515-26.
4. Ichihashi M, Ueda M, Budiyanto A, Elaine T, Oka M, Fukunaga M, Tsuru K. UV-induced skin damage. Toxicology. 2003;189(1-2):21-39.
-
55. Pfeifer GP, You YH, Besaratinia A. Mutations induced by ultraviolet light. Mut Res. 2005;571(1-2):19-31..
Early lesions are easily ignored or not identified macroscopically, but once initiated can make the area more susceptible to more aggressive changes. The increase in new cases and frequent recurrences may be related the fact that medically it may not be possible to determine when a solar keratosis ends and when a CEC begins. Cockerell66. Cockerel CJ. Histopathology of incipient intrapidermal squamous cell carcinoma ("actinic keratosis"). J Am Acad Dermatol. 2000;1(42):11-7. says that the solar keratosis and SCC represent the same injury in different stages of evolution, whereas the solar keratosis a very early stage. So, one must be aware to the evolution of keratosis which can cure spontaneously or after treatment or injury persist and evolves into an invasive skin cancer: CEC or CBC77. Almeida EMP, Wow RA, Adam R, Souza In, Metze K, Cintra ML. Photodamage in feline skin: clinical and histomorphometric analysis. Vet Pathol 2008;45(3):327-35. , 88. Dergham M, Mesquita LAF, Muraru CC, Ramos EA, Collaco, LM. Distribution of diagnosis of neoplastic and pre neoplastic skin lesions at Evangelical Hospital in Curitiba. An Bras Dermatol. 2004;79(5):555-9..
Epidemiological evidence prove CEC increase related to the morbidity coefficients in recent years, with a predominance of males over the age of 60 years with injuries more frequently located in areas exposed to the Sun being more incident in patients above 60 years99. Nasser M. Epidemiology of cancers espinoclulares-Blumenau (SC) - Brazil, from 1980 to 1999. An Bras Dermatol. 2004;79:149-55.
10. Nunes DH, Back L, Silva RV, Mann VS. Incidência do carcinoma de células escamosas da pele na cidade de Tubarão (SC) - Brasil nos anos de 2000, 2003 e 2006. An Bras Dermatol. 2009;84(5):482-8.
-
1111. Oliveira LMC, Glauss N, Palma A. Habits of physical education teachers who work with water activities. An Bras Dermatol. 2011;86(3):445-50..
This study was conducted in the semi-arid climate of the northeast of Brazil, where the level of solar radiation is intense and almost continuous with low relative humidity throughout the year, risk factors for the development of skin cancer, especially for workers exposed to the Sun1212. Fisher MS, Kripke ML. Systemic alteration induced in mice by ultraviolet light irradiation and its relationship to ultraviolet carcinogenesis. Proc Natl Acad Sci USA. 1977;74(4):1688-92.. Aims to observe the effect of ultraviolet-B radiation and atmospheric pollutant DMBA in rats skin, comparing the clinical and histological aspects changes over time.
Methods
This study was approved by Ethics Committees of Sao Paulo Federal University (Protocol # 0648/10) and Sao Francisco Valley Federal University (Protocol # 0648/10).
This experimental study with case control used Wistar rats (n=40), male, three months of age, weighting 200±30g housed individually in polypropylene boxes. They were daily monitored for temperature and humidity, brightness control of light/dark cycles of 12 hours. The dorsal region was shaved in an area measuring 3 x 3cm, every five days.
The animals were separated in four randomized groups and observed for ten weeks. The groups A, B and C, (ten animals in each) received respectively UVR-B, DMBA and UVR-B associated with DMBA. In Group D, control (n=10) no animal have suffered any kind of intervention.
We used the radiation UVR-B transmitter device with wavelength (λ) of 312nm (Spectroline(r) E-series), 6J/s (W) power and 0.17A. The appliance has been set at 20cm wooden structure above the back of the rat that was restrained in a special box. Rats in group A and C were exposed for 30 seconds, three times per week for ten weeks1212. Fisher MS, Kripke ML. Systemic alteration induced in mice by ultraviolet light irradiation and its relationship to ultraviolet carcinogenesis. Proc Natl Acad Sci USA. 1977;74(4):1688-92. , 1313. Kligman LH, Schwartz E, Sapadin AN, Kligman AM. Collagen loss in human photoaged skin is overestimated by histochemistry. Photodermatol Photoimmunol Photomed. 2000;16:224-8.. The exposure time to UVR-B was established based on the lamp power and minimum time to achieve twice the minimum erythematous dose on the animal skin.
Through a micropipette 0.2mL of a 1% DMBA diluted in acetone 95% solution was applied, on the bald back of the rats of groups B and C, three times a week for ten weeks1414. Schwartz E. Connective tissue alterations in the skin of hairless mice irradiated ultraviolet. J Invest Dermatol. 1988;919(2):158-61..
Macroscopic analysis
The animals were photographed on week 5, 8 and 10. All photos were taken at the same daytime. The results were based upon a score (1 to 6), according to the degree of severity of the dermatological lesion: 1 = no injury; 2 = erythema; 3 = erythema with desquamation; 4 = keratosis; 5 = ulcer; 6 = crust and 7 = vegetation. The observers were unaware of the study. The numerical distribution was statistically analyzed by the McNemar test.
At the end of the 10th week we performed a skin biopsy through a 10mm circular punch; the animals were anesthetized with ketamine hydrochloride (60mg/kg of body weight) and xilasina hydrochloride (10mg/kg of body weight) and then the animals were killed. The histological preparation was performed by Hematoxylin-Eosin method.
Under optical microscopy (100-fold increase) in each plate we randomly selected four fields. In these fields the epidermis was photographed through a video camera and the image processed and analyzed by the Imagelab(r) software. In each image four distinct measures of the epidermis thickness were taken, two of the largest and two of the smallest one. From these measures It was calculated a mean thickness of the epidermis.
For macroscopic results we considered the comparison between the four groups at the 5th, 8th and 10th week. On the comparison between the ranges of times, two by two, within each group the Signal test was used. The microscopic variables were compared between the four groups by Kruskall-Wallis and Mann Whitney tests. It was considered the significance value of 5% (Statistical Package for Social Sciences (SPSS)(r) version 10.0 for Windows(r) and Statistic/Data Analys (STATA)(r) version 8.0) for all statistical procedures.
Results
Macroscopic results
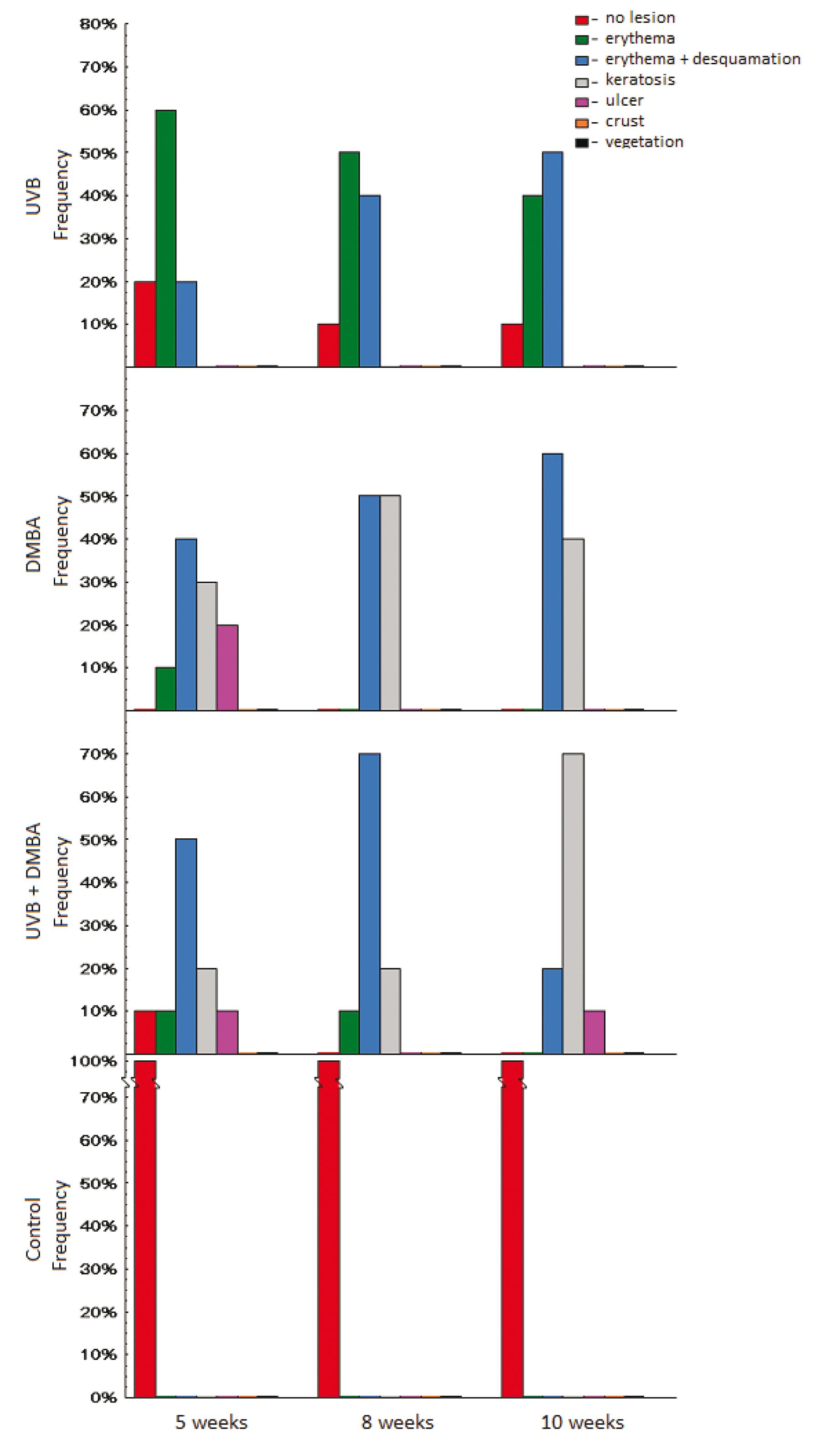
Severity of the lesions ranged from grade 1 to 5 (Figure 1). There was no difference inside each group in each time as tested by McNemar test (Table 1).
Skin lesions at weeks 5, 8 and 10 in groups RUV (A), DMBA (B), DMBA + UVR (C) and control (D) (Modal score).
The results are presented by group of animals UVR, DMBA, RUV/DMBA and control, according to the percentages of macroscopic lesions (Figure 2). As one can observe, no lesions were found in the control group (red bar) at any time. In the first week, 20% of the animals in group A, 0% in group B and 10% in group C had no lesions; erythema (green bar) was present in 60%, 10% and 10% of the animals respectively in group A, B and C; erythema plus desquamation (blue bar) was present respectively in 20%, 40% and 50% of the animals; incidence of keratosis (grey bar) was 0%, 30% and 20% respectively; ulcers (pink bar) were found in 0%, 20% and 10% of the animals. We found no crusts or vegetation lesions (orange and black bars respectively). By week eight, the distribution in percentage of the lesions was 10, 50, 40, 0, 0, 0, 0 (group A), 0, 0, 50, 50, 0, 0, 0 (group B) and 0, 10, 70, 20, 0, 0, 0 (group C). By the tenth week, these results were respectively 10, 40, 50, 0, 0, 0, 0 (group A), 0, 0, 60, 40, 0, 0, 0 (group B) and 0, 0, 20, 70, 10, 0, 0 (group C).
Distribution (frequency) of dermatological lesions on week 5, 8 and 10. Comparison between groups must be done from top to bottom for each week.
Microscopic results
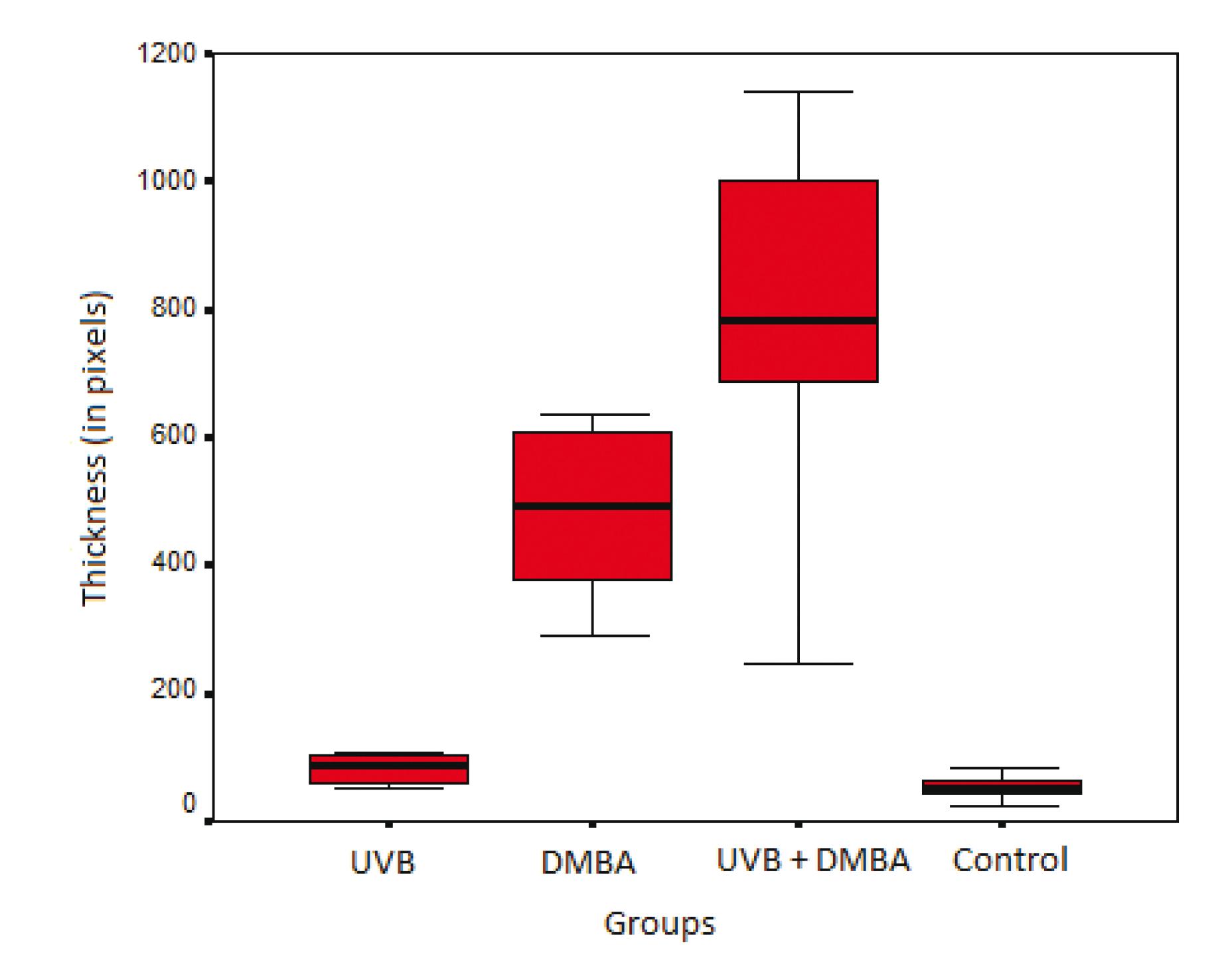
The microscopic analysis took place on the tenth week comparing groups DMBA, RUV UVR+DMBA and control. We measured the epidermal thickness and calculated an average (median ± SD) for each group (Figure 3). The comparison between groups was calculated by the Kruskal-Wallis test (Table 2).
Variations on epidermal thickness in all groups at the 10th week of the study (median ± SD).
Comparing macroscopic and microscopic results
Analyzing microscopic results compared to macroscopic results realizes that the more skin exposure to UVR-B and DMBA, macroscopic injury more serious is that the greater the thickness of the epidermis (Figure 4).
Photomicrography (100-fold increase) showing differences in thickness of the epidermis on the 10th week of the study for groups A, B, C and D.
Discussion
Macroscopical analysis
Clinical evidence and experimental findings have supported the idea of UVR-B as a causal inductor and carcinogenesis promoter. Discussions suggest that this is not only due to transformative action on the cell genome, but also by immunodeficiency. Participant elements of an inflammatory reaction as interleukins, prostaglandins, Langerhans cells, lymphocytes and macrophages are involved in this process44. Ichihashi M, Ueda M, Budiyanto A, Elaine T, Oka M, Fukunaga M, Tsuru K. UV-induced skin damage. Toxicology. 2003;189(1-2):21-39. , 1515. Santos I, Mello RJV, Santos RA, Santos IB. Quantitative study of Langerhans cells in basal cell carcinoma with higher and lower potential for aggression. An Bras Dermatol. 2010;85(2):165-71..
Almeida77. Almeida EMP, Wow RA, Adam R, Souza In, Metze K, Cintra ML. Photodamage in feline skin: clinical and histomorphometric analysis. Vet Pathol 2008;45(3):327-35. showed that in cats´ skin, the macroscopic skin lesions caused by UVR-B, begin as erythema and can evolve gradually to form scales, keratosis and crusts, in accordance with the stimulus to the skin and increase of the exposure time.
The occurrence of CBC in caucasian humans over 70 years old can be associated with the cumulative effect of the solar radiation after long periods of exposure1616. Mantese SAO, Berbert ALCW, Gomides MDA, Rocha A. Basal cell carcinoma-analysis of 300 cases observed in Uberlandia-MG. An Bras Dermatol. 2006;81(2):136-42..
Aster1717. Astner S, Wu A, Chen J, Philips N, Rius-Diaz F, Parrado C, Mihm MC. Dietary lutein/zeaxanthin partially reduces photoaging and photocarcinogenesis in chronically UVB-irradiated skh-1 hairless mice. Skin Pharmacol Physiol. 2007;20:283-91. used hairless mice and investigated different doses of UVR-B. In a group aiming to observe chronic photodamage he used 16.000mJ/cm2 and in another group aiming to achieve photocarcinogenesis, he used 30.200mJ/cm2; these doses were applied daily to achieve the desired effect and the results were analyzed weekly. Both groups needed no more than one week. In the photoaging group it was observed: skin thickness, thickness of suprapapilar plate, number of mast cells and dermal desmosina content. In the photocarcinogenesis group larger tumors with approximately 2mm were observed. The experiment also used a group to which was offered a special diet with lutein zeaxanthin (carotenoids with antioxidant property), and in this group the cutaneous effects were lower.
Sharman and Katiyar1818. Sharman SD, Katiyar SK. Dietary grape seed proanthocyanidins inhibit UVB-induced cyclooxygenase-2 expression and other inflammatory mediators in UVB-exposed skin and skin tumors of SKH-1 hairless mice. Pharm Res. 2010;27(6):1092-102. used a protocol for induction of carcinogenesis in nude rats´ skin, aiming to the chemopreventive mechanisms of protocianidins found in grape seed. The RUV-B dose was 180mJ/cm2, three times a week for 24 weeks. At the end of the experiment they found skin tumor and advanced inflammatory process in the group to which has not been offered diet with antioxidant substance.
In experiments to the photocarcinogenesis induction with human skin graft using skin of the foreskin of newborn babies in immunocompromised rat skin with DMBA and/or UVR-B at 40mJ/cm2 over 10 months with two to three times a week, it was observed the formation of epidermal cysts, squamous cell carcinomas, melanocytic hyperplasia and melanoma. When used skin of the chest and the face of white human adults to exposure of the same dose of DMBA and/or RUVB, Actinic keratosis was observed in 30% in animals exposed to UVR + 10% and DMBA in animals exposed only the UVR-B. The group in which it was used was not observed DMBA only Actinic keratosis. It was noticed that the younger is more serious human injury appeal presents itself when exposed to UVR and/or DMBA1919. Berking C, Takemoto R, Binder RL, Hartman SM, Ruiter DJ, Gallagher PM, Lessin SR. Photocarcinogenesis in human adult skin grafts. Carcinogenesis. 2002;23(1):181-7..
In our experiment, the elementary lesion skin first and that maintains its frequency in weeks 5, 8 and 10, when irradiated animal's skin with UVR-B, is Erythema (Figure 1). With the continued exposure of the skin to irradiation of UVR-B, the elementary lesion with greater severity that presented itself was not being shown peeling with erythema, in this way, more aggressive lesions.
The initial lesion (erythema) suggests that the body reacts to UVR-B with the same process of an initial inflammatory reaction, or the dissemination of inflammatory mediators, such as cytokines released from harmed keratinocytes, associated with the fact that the UVR-B can depress the immune response of the skin33. Jans J, Garinis GA, Schul W, van Oudenaren A, Moorhouse M, Smid M, Yurda_Gul S. Differential role of basal keratinocytes in UV-induced immunosuppression and skin cancer. Mol Cell Biol. 2006;26(22):8515-26. , 44. Ichihashi M, Ueda M, Budiyanto A, Elaine T, Oka M, Fukunaga M, Tsuru K. UV-induced skin damage. Toxicology. 2003;189(1-2):21-39. , 2020. Maverakis E, Miyamura Y, Bowen MP, Correa G, Ono Y, Goodarzi H. Light, including ultraviolet. J Autoimmu. 2010;34(3):J247-57..
One can infer from this study that UVR-B seems to induce its harmful effects on the skin cells by causing acute inflammatory reaction with the expressive presentation of erythema in three times and although, in general, the inflammatory response constitutes an organic process very important defensive for numerous physiological events, in many cases the persistent inflammatory reaction can cause irreversible cell damage, contributing to photoaging33. Jans J, Garinis GA, Schul W, van Oudenaren A, Moorhouse M, Smid M, Yurda_Gul S. Differential role of basal keratinocytes in UV-induced immunosuppression and skin cancer. Mol Cell Biol. 2006;26(22):8515-26. , 2121. Syed DN, Afaq F, Mukhtar H. Differential activation of signaling pathways by UVA and UVB radiation in normal human epidermal keratinocytes. Photochem Photobiol. 2012;88(5):1184-90..
Whereas keratosis are part of a continuous process that begins with DNA damage, mutation and neoplastic transformation66. Cockerel CJ. Histopathology of incipient intrapidermal squamous cell carcinoma ("actinic keratosis"). J Am Acad Dermatol. 2000;1(42):11-7., realizing that in this work when used only DMBA, the elementary lesion presented keratosis in nearly 50% of the sample, in the three studied times, associated with the fact that this chemical was always aggressive to the skin of the animal, because in no time the skin performed without injury, it was realized that the DMBA induction neoplastic features presented more evident when compared to the effects of UVR-B in the skin of the animal.
However, the elementary lesion erythema with crust maintains its high and constant frequency of the fifth to the tenth week, suggesting that the DMBA induced constantly the formation of lesions with inflammatory feature while induced lesions with neoplastic features. In addition to the inflammatory action of DMBA was more evident when using DMBA that UVR-B, because the UVR-B presented erythema more frequently than erythema with crust.
Different experiments using DMBA as neoplastic inductor in skin of animals and the clinical effects are diverse, vary from no found until the formation of melanoma. Surh2222. Surh I, Rundhaug J, Pavone A, Mikulec C, Abel E, Fischer SM. Upregulation of the EP1 receptor for prostaglandin E2 promotes skin tumorprogression. Mol Carcinog. 2011;50(6):458-68. caused cancer in mice skin, used the study of DMBA Berking1919. Berking C, Takemoto R, Binder RL, Hartman SM, Ruiter DJ, Gallagher PM, Lessin SR. Photocarcinogenesis in human adult skin grafts. Carcinogenesis. 2002;23(1):181-7. human skin graft in rats when used DMBA haven't noticed any cancer formation. In the present study it was observed macroscopically in the entire sample, the emergence of keratosis and ulcerated lesions, considered pre-neoplastic lesions.
Experiments relating RUV-B applications to different chemical carcinogens inducers have caused interest in many researchers, as well as experiments with products for photoproctetion. Berking1919. Berking C, Takemoto R, Binder RL, Hartman SM, Ruiter DJ, Gallagher PM, Lessin SR. Photocarcinogenesis in human adult skin grafts. Carcinogenesis. 2002;23(1):181-7. showed that with a single application of DMBA to skin of mice, with subsequent exposures to UVR-B and/or UVR-A has resulted in the formation of melanoma, lymphoma and CEC, indicating that the DMBA initiated cell injury and the UVR-A and UVR-B promoted the development of lesions.
Monteiro-Riviere2323. Monteiro-Riviere N, Inman A, Hedgpeth V, Mosteller B, Piedrahita J. Dermatological effects of chronic exposure to 7,12-dimethylbenz[A]anthracene (DMBA) or N-methyl-N-nitrosoguanidine (MNNG) in swine. Cutan Toxicol. 2006;25 (2):103-19., using skin from nuts exposed to chronic action of various doses to MNNG (methyl-N-Nitroglycerin) and DMBA with or without association to UVR-B for 30 weeks, caused mild to severe dermatological changes without developing skin carcinomas.
In a study with rats and murine using 12-tetradecanoylphorbol-13-acetate (TPA) and DMBA with or without the UVR-B associated, mediators of inflammation were 4 times more present when using DMBA than TPA22 22. Surh I, Rundhaug J, Pavone A, Mikulec C, Abel E, Fischer SM. Upregulation of the EP1 receptor for prostaglandin E2 promotes skin tumorprogression. Mol Carcinog. 2011;50(6):458-68..
Noting a slight increase in the incidence of melanoma in employees of an oil refinery, researchers tried to reproduce this condition experimentally. The experiment got satisfactory outcome for skin melanoma in rats, hamsters, and Guinea pigs, but only when DMBA and UVR-B were applied in high doses, disregarding the relationship of refinery workers exposure with high incidence of melanoma, because researchers found that the workers were exposed only a small fraction of thishydrocarbon2424. Ingram AJ. Review of chemical and UV light-induced melanomas in experimental animals in relation to human melanoma incidence. J Appl Toxicol. 1992;12(1):39-43.. Yang2525. Yang J-H, Lee C-H, Monteiro-Riviere NA, Riviere JE, Tsang C-L, Chou C-C. Toxicity of aliphatic and aromatic hydrocarbon jet fuel mixtures on human epidermal Keratinocytes: evaluation based on in vitro cytotoxicity and interleukin-8 release. Arch Toxicol. 2006;80:508-23. reports high toxicity of polycyclic aromatic hydrocarbons in fuels human skin.
It was evident in this work that the skin contact with hydrocarbons associated with exposure to UVR-B cell lesions caused early and late, late injuries being more aggressive with apparent cumulative effect on the skin of animals.
In this study, application of UVR-B associated with DMBA simultaneously in the skin of the animal, there was from the beginning a significant inflammatory reaction with the presentation of the elementary lesion erythema and desquamation in weeks 5 and 8 and week 10 for the highest percentage of solar keratosis among all groups within three days. These findings suggest that the DMBA associated with UVR-B was potentiated the effect of induce lesions with inflammatory characteristics being also powered the effect to induce lesions with neoplastic features (Figure 1).
Microscopic analysis
Monteiro-Riviere23 23. Monteiro-Riviere N, Inman A, Hedgpeth V, Mosteller B, Piedrahita J. Dermatological effects of chronic exposure to 7,12-dimethylbenz[A]anthracene (DMBA) or N-methyl-N-nitrosoguanidine (MNNG) in swine. Cutan Toxicol. 2006;25 (2):103-19.combining the action of UVR-B, DMBA and MNNG obtained significant effect in increasing the thickness of the epidermis and the number of layers of cells showing their highest values from 20 weeks, when used UVR-B associated with MNNG. Using only MNNG epidermal changes were intracellular and intercellular edema, and edema and dermal inflammation. However when used DMBA, associated with the no to UVR-B morphological changes were less severe and with lower values of thickness of the epidermis than when used MNNG.
In a clinical study, evaluating the epidermal lesion the ear of cats, Daniel77. Almeida EMP, Wow RA, Adam R, Souza In, Metze K, Cintra ML. Photodamage in feline skin: clinical and histomorphometric analysis. Vet Pathol 2008;45(3):327-35. showed that when there is production of solar keratosis (evaluated by clinical) the thickness of the epidermis increases, from the initial stage of cutaneous lesion. Comments although the macroscopic events are immediate result of inter-and intracellular edema and with prolonged exposure, there is a true cell hyperplasia (acanthosis), keratosis of all epidermal layers, from the Bush to the cornea with the exception of basal. For each new exposure, the greater thickness of the epidermis.
Clinical epidemiological study in humans showed a high frequency of CBC with more of an injury in different areas of the body in women as the cephalic region, nasal dorsum and men had prevalence in the area of the trunk, neck and ear. All areas are more easily exposed to solar radiation1616. Mantese SAO, Berbert ALCW, Gomides MDA, Rocha A. Basal cell carcinoma-analysis of 300 cases observed in Uberlandia-MG. An Bras Dermatol. 2006;81(2):136-42..
Experiments with human skin graft in mice skin using DMBA and/or UVR-B showed development actinic keratosis, in accordance with point mutations in codon 61 of human gene Ha-ras1919. Berking C, Takemoto R, Binder RL, Hartman SM, Ruiter DJ, Gallagher PM, Lessin SR. Photocarcinogenesis in human adult skin grafts. Carcinogenesis. 2002;23(1):181-7..
Noting the findings of the present study, when taking into consideration changes in histological and epidermal thickness parameter, comparing the groups with the control group, the group that demonstrated greater epidermal thickness, was the group that was exposed to UVR-B and DMBA attached (Figure 1).
These findings were similar to the results of Monteiro-Riviere23 23. Monteiro-Riviere N, Inman A, Hedgpeth V, Mosteller B, Piedrahita J. Dermatological effects of chronic exposure to 7,12-dimethylbenz[A]anthracene (DMBA) or N-methyl-N-nitrosoguanidine (MNNG) in swine. Cutan Toxicol. 2006;25 (2):103-19.and Daniel77. Almeida EMP, Wow RA, Adam R, Souza In, Metze K, Cintra ML. Photodamage in feline skin: clinical and histomorphometric analysis. Vet Pathol 2008;45(3):327-35. suggesting that the increase in epidermal thickness of this layer is formed cells increase (Figure 1). Marevacs2020. Maverakis E, Miyamura Y, Bowen MP, Correa G, Ono Y, Goodarzi H. Light, including ultraviolet. J Autoimmu. 2010;34(3):J247-57. reports on their experimental findings that UVR-B can activate immune system components, generating by distinct inflammatory response mechanisms, such as: direct activation of keratinocites and other cells that release inflammatory mediators and redistribution and release of kidnapped auto-antigens of cells damaged by UVR.
Associating the macroscopic to microscopic findings it seems that UVR-B and DMBA concomitant exposure leads to an induction of more serious injuries such as keratosis; the epidermis becomes thicker, suggesting neoplastic changes (Figure 4).
Early lesions are easily overlooked, making early diagnosis difficult. Accurate clinical examination associated with histological study are indispensable factors for prevention of irreversible lesions. Yet educational campaigns warning of the photoprotection and early and irreversible injury prevention also are key2626. Montagner S, Costa A. Biomolecular basis of photoaging. An Bras Dermatol. 2009;84(3):263-9. , 2727. Balogh TS, Velasco MVR, Pedriali CA, Kaneko TM, Baby AR. Ultraviolet radiation protection: resources available today in photoprotection. An Bras Dermatol. 2011;86(4):732-42..
Conclusions
The population exposed to ultraviolet B radiation is subject to suffer skin lesions that can develop into cancer. The association with hydrocarbons as the dimethyl benzanthracene increases the possibility of malignancy. May not be clinically evident determine when a solar keratosis ends and when a CEC begins. For this reason, histological study associated with health education prompting the early and irreversible injury prevention is necessary.
References
-
1Instituto Nacional de Câncer José Alencar Gomes da Silva. General coordination of strategic actions. Coordination of prevention and awareness. 2012 estimate: incidence of cancer in Brazil. Rio de Janeiro: INCA; 2011.
-
2Oliveira LBO, Bampi VF, Garcia CF, Souza E. Angioarchitecture of squamous cell carcinoma from hamster buccal pouch: a scanning electron microscopy study. J Scan Microsc. 2009;31(5):188-94.
-
3Jans J, Garinis GA, Schul W, van Oudenaren A, Moorhouse M, Smid M, Yurda_Gul S. Differential role of basal keratinocytes in UV-induced immunosuppression and skin cancer. Mol Cell Biol. 2006;26(22):8515-26.
-
4Ichihashi M, Ueda M, Budiyanto A, Elaine T, Oka M, Fukunaga M, Tsuru K. UV-induced skin damage. Toxicology. 2003;189(1-2):21-39.
-
5Pfeifer GP, You YH, Besaratinia A. Mutations induced by ultraviolet light. Mut Res. 2005;571(1-2):19-31.
-
6Cockerel CJ. Histopathology of incipient intrapidermal squamous cell carcinoma ("actinic keratosis"). J Am Acad Dermatol. 2000;1(42):11-7.
-
7Almeida EMP, Wow RA, Adam R, Souza In, Metze K, Cintra ML. Photodamage in feline skin: clinical and histomorphometric analysis. Vet Pathol 2008;45(3):327-35.
-
8Dergham M, Mesquita LAF, Muraru CC, Ramos EA, Collaco, LM. Distribution of diagnosis of neoplastic and pre neoplastic skin lesions at Evangelical Hospital in Curitiba. An Bras Dermatol. 2004;79(5):555-9.
-
9Nasser M. Epidemiology of cancers espinoclulares-Blumenau (SC) - Brazil, from 1980 to 1999. An Bras Dermatol. 2004;79:149-55.
-
10Nunes DH, Back L, Silva RV, Mann VS. Incidência do carcinoma de células escamosas da pele na cidade de Tubarão (SC) - Brasil nos anos de 2000, 2003 e 2006. An Bras Dermatol. 2009;84(5):482-8.
-
11Oliveira LMC, Glauss N, Palma A. Habits of physical education teachers who work with water activities. An Bras Dermatol. 2011;86(3):445-50.
-
12Fisher MS, Kripke ML. Systemic alteration induced in mice by ultraviolet light irradiation and its relationship to ultraviolet carcinogenesis. Proc Natl Acad Sci USA. 1977;74(4):1688-92.
-
13Kligman LH, Schwartz E, Sapadin AN, Kligman AM. Collagen loss in human photoaged skin is overestimated by histochemistry. Photodermatol Photoimmunol Photomed. 2000;16:224-8.
-
14Schwartz E. Connective tissue alterations in the skin of hairless mice irradiated ultraviolet. J Invest Dermatol. 1988;919(2):158-61.
-
15Santos I, Mello RJV, Santos RA, Santos IB. Quantitative study of Langerhans cells in basal cell carcinoma with higher and lower potential for aggression. An Bras Dermatol. 2010;85(2):165-71.
-
16Mantese SAO, Berbert ALCW, Gomides MDA, Rocha A. Basal cell carcinoma-analysis of 300 cases observed in Uberlandia-MG. An Bras Dermatol. 2006;81(2):136-42.
-
17Astner S, Wu A, Chen J, Philips N, Rius-Diaz F, Parrado C, Mihm MC. Dietary lutein/zeaxanthin partially reduces photoaging and photocarcinogenesis in chronically UVB-irradiated skh-1 hairless mice. Skin Pharmacol Physiol. 2007;20:283-91.
-
18Sharman SD, Katiyar SK. Dietary grape seed proanthocyanidins inhibit UVB-induced cyclooxygenase-2 expression and other inflammatory mediators in UVB-exposed skin and skin tumors of SKH-1 hairless mice. Pharm Res. 2010;27(6):1092-102.
-
19Berking C, Takemoto R, Binder RL, Hartman SM, Ruiter DJ, Gallagher PM, Lessin SR. Photocarcinogenesis in human adult skin grafts. Carcinogenesis. 2002;23(1):181-7.
-
20Maverakis E, Miyamura Y, Bowen MP, Correa G, Ono Y, Goodarzi H. Light, including ultraviolet. J Autoimmu. 2010;34(3):J247-57.
-
21Syed DN, Afaq F, Mukhtar H. Differential activation of signaling pathways by UVA and UVB radiation in normal human epidermal keratinocytes. Photochem Photobiol. 2012;88(5):1184-90.
-
22Surh I, Rundhaug J, Pavone A, Mikulec C, Abel E, Fischer SM. Upregulation of the EP1 receptor for prostaglandin E2 promotes skin tumorprogression. Mol Carcinog. 2011;50(6):458-68.
-
23Monteiro-Riviere N, Inman A, Hedgpeth V, Mosteller B, Piedrahita J. Dermatological effects of chronic exposure to 7,12-dimethylbenz[A]anthracene (DMBA) or N-methyl-N-nitrosoguanidine (MNNG) in swine. Cutan Toxicol. 2006;25 (2):103-19.
-
24Ingram AJ. Review of chemical and UV light-induced melanomas in experimental animals in relation to human melanoma incidence. J Appl Toxicol. 1992;12(1):39-43.
-
25Yang J-H, Lee C-H, Monteiro-Riviere NA, Riviere JE, Tsang C-L, Chou C-C. Toxicity of aliphatic and aromatic hydrocarbon jet fuel mixtures on human epidermal Keratinocytes: evaluation based on in vitro cytotoxicity and interleukin-8 release. Arch Toxicol. 2006;80:508-23.
-
26Montagner S, Costa A. Biomolecular basis of photoaging. An Bras Dermatol. 2009;84(3):263-9.
-
27Balogh TS, Velasco MVR, Pedriali CA, Kaneko TM, Baby AR. Ultraviolet radiation protection: resources available today in photoprotection. An Bras Dermatol. 2011;86(4):732-42.
-
1
Research performed at Nursing Department, Sao Francisco Valley Federal University (UNIVASF), Petrolina-PE, Brazil. Part of Master degree thesis, Postgraduate Program in Interdisciplinary Surgical Science, Sao Paulo Federal University. Tutor: Hélio Plapler.
Publication Dates
-
Publication in this collection
Feb 2014
History
-
Received
17 Oct 2013 -
Reviewed
18 Dec 2013 -
Accepted
20 Jan 2014