Abstract
PURPOSE:
To describe a new experimental flap for studying skin viability in rats.
METHODS:
Twenty male Wistar rats weighing between 250-300g were divided into two groups: group A - McFarlane technique, a 4 x 10cm flap was used (McFarlane); and in group B modified McFarlane technique, a 3 x 10cm flap was used. Seven days later, the animals were sacrificed and the area of necrosis was evaluated in both groups.
RESULTS:
Group A presented necrosis in 3% of the total area of the flap (CI: 0.01-0.05), Group B presented necrosis in 37% of the total area of the flap (CI: 0.29-0.46), (p<0.001).
CONCLUSION:
The modified McFarlane flap presented a larger area of necrosis and could be an adequate experimental model of skin flap viability.
Surgical Flaps; Necrosis; Models, Animal; Rats
Introduction
Flaps have been used in plastic surgery since 800b.C. Skin flaps are basically employed for the repair of large wound defects. The techniques of skin flaps are well established and they vary according to the location of the wound and the extension of the defect11. Frederick FW, Sweeny L, Carroll WR, Peters GE, Rosenthal EL. Outcomes in head and neck reconstruction by surgical site and donor site. Laryngoscope. 2013;123(7):1612-7.. The most feared complication of a skin flap is a significant area of necrosis causing the failure of the surgical procedure. The estimated frequency of necrosis of skin flaps varies between 2 and 20%11. Frederick FW, Sweeny L, Carroll WR, Peters GE, Rosenthal EL. Outcomes in head and neck reconstruction by surgical site and donor site. Laryngoscope. 2013;123(7):1612-7. , 22. Lebas D, Wiart T, Gros C, Modiano P. Use of a rhomboid flap to repair temporal and frontotemporal cutaneous defects: 11 cases. An Dermatol Venereal. 2013;140:170-5. .
Bearing this fact in mind, a reproducible and effective experimental model of necrosis of skin flaps would be useful to evaluate different substances and devices that might increase the viability of flaps33. Acevedo-Bogado CE, Bins-Ely J, d'Acampora AJ, Neves RD. Efeito da hialuronidase na sobrevida de retalhos cutâneos em ratas. Acta Cir Bras. 2002;17(1):14-6. , 44. Reichner DR, Scholz T, Vanderkam VM, Gutierrez S, Steward E, Evans GR. Laser flap delay: comparison of Erbium: YAG and CO2 lasers. Am Surg. 2003;69(1):69-72..
McFarlane et al.55. McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82. described the dorsal cutaneous flap in rats. Since then this experimental model has been adopted as the gold standard in studies evaluating the viability of axial flaps. The original dimensions described in McFarlane's et al. study were of 4 cm across the base by 10 cm in length (i.e. a 1:2.5 ratio), taking the scapulae as the base of the flap. In the original study, the authors described necrosis of 25-50% of the flap length.
However, in the literature several studies have cited this technique as a reference, but with different dimensions. Flaps measuring 2 x 8cm66. Oh M, Chang H, Minn KW. The effects of tadalafil on axial-pattern skin flap survival in rats. Dermatol Surg. 2008;34(5):626-30., 3 x 7cm77. Barral SM, Araujo ID, Vidigal PVT, Mayrink CAC, Araujo AD, da Costa PR. Effects of sildenafil on the viability of random skin flaps. Acta Cir Bras. 2011;26(4):314-9., and 3 x 9cm88. Lin DS, Zheng X, Li ZJ, Zhang YP, Wang AY, Xie XG, Gao WY. Effect of hirudin on random skin flap survival in rats. Zhonghua Zheng Xing Wai Ke Za Zhi. 2011;27(1):35-9. have been described and attributed to McFarlane, casting doubt on the results of the original description. In addition, the location of the base described as cranial (at the level of the scapulae) has also not been uniform66. Oh M, Chang H, Minn KW. The effects of tadalafil on axial-pattern skin flap survival in rats. Dermatol Surg. 2008;34(5):626-30.
7. Barral SM, Araujo ID, Vidigal PVT, Mayrink CAC, Araujo AD, da Costa PR. Effects of sildenafil on the viability of random skin flaps. Acta Cir Bras. 2011;26(4):314-9.
-
88. Lin DS, Zheng X, Li ZJ, Zhang YP, Wang AY, Xie XG, Gao WY. Effect of hirudin on random skin flap survival in rats. Zhonghua Zheng Xing Wai Ke Za Zhi. 2011;27(1):35-9..
In the light of the lack of consensus, and because of the need for an effective and reproducible experimental model99. Moura TD, Marques Ade A, Bernal SO, Gagliocca GD, Gemperli R, Ferreira MC. Study of the effect of streptokinase and allopurinol in island skin flaps submitted to prolonged ischemia: experimental study in rats. Rev Assoc Med Bras. 2009;55(5):601-5. of skin flap necrosis, we decided to conduct a comparative study between the flap described by McFarlane and a skin flap with different dimensions.
Methods
This project was approved by the Ethics Committee, School of Medicine, University of Sao Paulo. All the experiments complied with regulations on animal experimentation described by the Council for international Organization of Medical Sciences (CIOMS).
Twenty male Wistar rats weighing between 250-300 g were divided into two groups:
- Group A (10 animals) - McFarlane flap (4 x 10cm);
- Group B (10 animals) - modified flap (3 x 10cm).
All the animals were housed in separate cages, with ad libitum offer of food and water, 25°C temperature and controlled day and night cycling.
The animals were anesthetized by inhalation of a mixture of 20% isoflorane and oxygen and positioned in ventral decubitus. The dorsal region was shaved. Aqueous 0.5% chlorhexidine was used for antisepsis of the surgical field.
Flap design
Group A - McFarlane
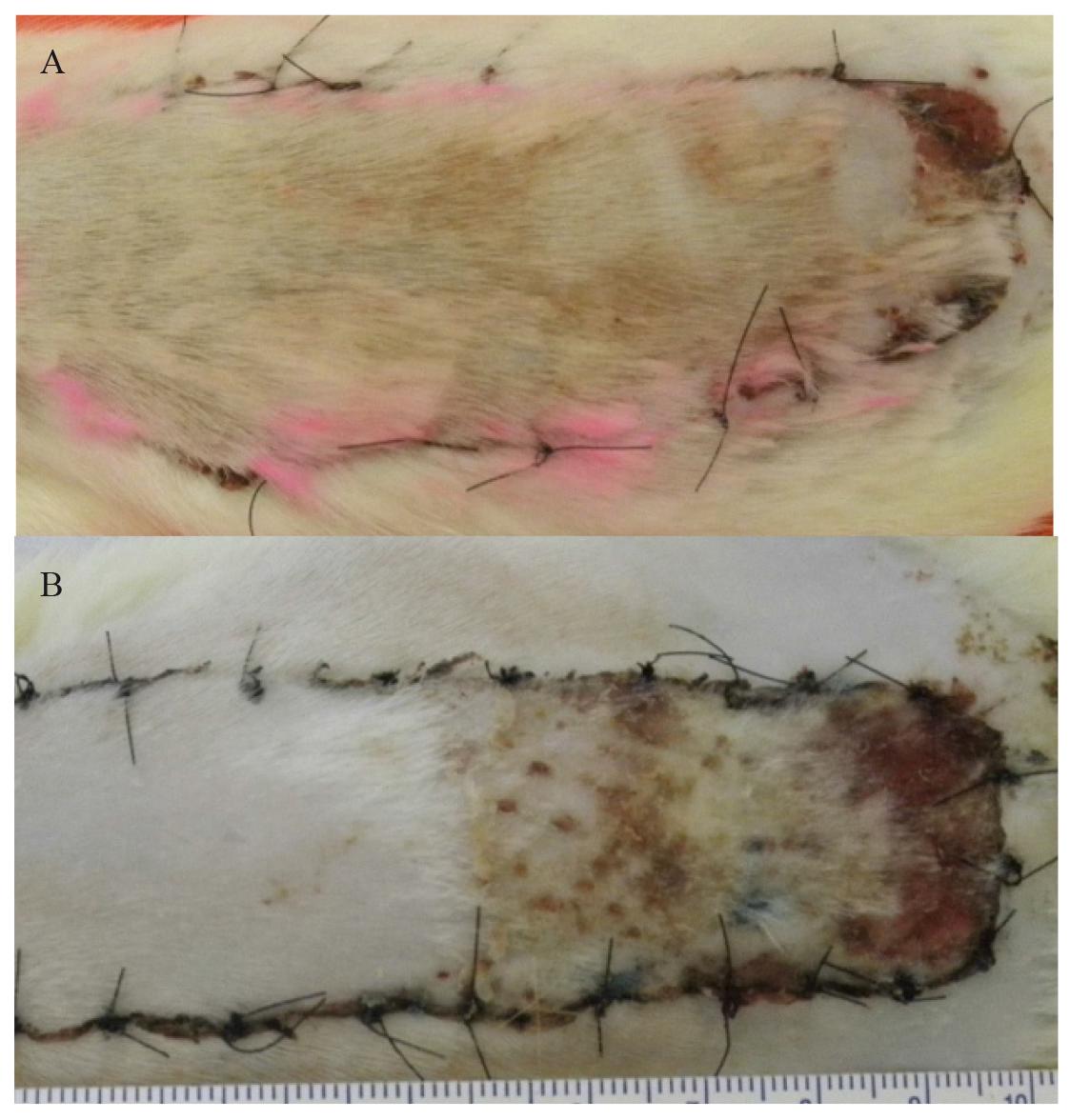
Starting from the rat's midline, two equidistant points were marked out using a marker pen, 2.0 cm on each side of the midline, at the level of the base of the scapulae. This was defined as the base of the flap. The length of the flap was marked out as two parallel lines 10 cm long, starting from the previously established base. Thus, the flap final dimensions were 4 x 10cm (Figure 1A).
A. McFarlane, with 4 x 10cm dimensions. B. Modified dorsal skin flap, with 3 x 10cm dimensions.
Group B - Modified flap
Starting from the rat's midline, a flap was delimited by marking out points 1.5 cm on each side of the midline, at the level of the base of the scapulae, and this was taken as the base of the flap. The remaining markings were the same as described for group A (Figure 1B).
The flap dissection and suture
After demarcating the flap, the skin was incised. The flap was raised by dissecting the areolar tissue at the level of the panniculus carnosus and deep fascia of the rat's dorsal musculature, as far as the interscapular base.
The flap was repositioned in its original position and was sutured with 4-0 monofilament nylon single stitches at 0.5cm intervals.
After recovery from anesthesia, the animals were kept in separate cages with ad libitum offer of food and water for seven days. Paracetamol (40mg) was added to 100ml of water, in conformity with the recommendations for laboratory animal care. All the animals were observed daily.
Evaluation of skin flap viability
On the seventh postoperative day, the flaps were photographed with a ruler in centimeters that was placed on the animal back. This image was then digitalized using the Image J(r) software (NIH; http://rsbweb.nih.gov/ij). All the photographs were calibrated for readings in mm2. By using the area function of the software it was possible to measure the total area of the flap and the area of necrosis.
Viable skin was defined as a pink, warm, soft on touch tissue (Figure 2A). Necrotic area was defined as a brown-black, cold and hard to manipulate skin (Figure 2B).
A. Viable flap with a small area of necrosis (arrows); B. Flap with a significant area of necrosis (arrows).
Excel software (Windows7(r)) was employed to calculate the area of necrosis to the total area ratio.
Statistical analysis
The necrosis/total flap area ratio was compared between groups A and B by using the Mann-Whitney test. The tests were calculated using the IBM SPSS statistical software (Statistical Package for the Social Sciences), version 21.0. p<0.05 was considered significant.
Results
After measuring the areas on the flap, the total area, area of necrosis and ratio of the area of necrosis/total area were calculated for both groups, as shown in Tables 1 and 2.
McFarlane flap (group A): total area, area of necrosis and ratio of the area of necrosis/total area.
Modified flap (group B): total area, area of necrosis and ratio of the area of necrosis/total area.
The mean ratios of the area of necrosis/total area were 3% and 37% for the McFarlane and modified flap groups respectively (Figure 3). The comparison between groups is demonstrated in Table 3.
Discussion
Although the flap described by McFarlane et al.55. McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82. is considered the gold standard for experimental studies on skin viability, it has been observed that there is not uniformity in this technique. Based on the literature, we noted that several studies cited this technique as a reference, but many of them had not used the dimensions established by McFarlane. In addition, it was seen that several authors who had used the original design (4x10cm) described areas of necrosis which varied from 10%1010. Tosun Z, Bite U. A comparative study of the influence of smoking on skin flap survival in pregnant rats. Br J Plastic Surg. 2005;58(6):812-6. to 60%1111. Abla LEF, Gomes HG, Percario S, Ferreira LM. Acetylcysteine in random skin flap in rats. Acta Cir Bras. 2005;20(2):121-3..
Another point to be considered is that there was some variation in the location of the base among the flaps attributed to McFarlane et al.55. McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82.. Some were elevated with the base in the caudal part of the animal's back66. Oh M, Chang H, Minn KW. The effects of tadalafil on axial-pattern skin flap survival in rats. Dermatol Surg. 2008;34(5):626-30., which others had the base in the cranial part, at the level of the scapulae77. Barral SM, Araujo ID, Vidigal PVT, Mayrink CAC, Araujo AD, da Costa PR. Effects of sildenafil on the viability of random skin flaps. Acta Cir Bras. 2011;26(4):314-9..
In view of the lack of consensus, we decided to conduct a study on a new flap with 3x10cm dimension, with a cranial base, comparing the area of necrosis at the seventh postoperative day with the original flap proposed by McFarlane et al.55. McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82.
The cranial base was chosen because the base of the animal's scapulae forms a rigid anatomical parameter. Moreover, in all dissections, it was noted that in this anatomical position there is a small vascular bundle that follows along the length of this flap, which makes it an axial flap.
However, this positioning can be criticized. Briggs et al.1212. Briggs PC. The McFarlane flap. Plast Reconstr Surg. 1987;80(3):472-3. reported that the position of the forelimbs caused variations in the location of the flap when the base was positioned at the level of the scapulae. Depending on the angle formed between the forelimbs and trunk, the base of the flap could change because the scapulae could shift up to one centimeter. This was not confirmed in the present study because the animal was positioned on a wooden board that was appropriate for this purpose and the animal's forelimbs were kept fixed, i.e. always at the same angle, without changing the level of the scapulae.
Following these simple rules, we can say that all the flaps were drawn up and the incisions were made correctly and in a reproducible manner because rigid anatomical parameters were employed.
We utilized the same parameters as those describe by Gemperli et al.1313. Gemperli R, Munhoz AM. The influence of type of vascular pedicle occlusion on the viability of skin Island flaps. A postoperative quantitative assessment of flap survival in an experimental model in rats. Acta Cir Bras. 2013;28(7):487-93. to define the macroscopic criteria for flap viability. Viable area was defined as a pink, warm and soft-to- touch skin on the 7th postoperative day. Necrotic area was defined as a brown-black, rigid-to-touch skin. And the area that was between these two presentations was classified as epithelyosis. For statistical purpose, only necrotic and total areas were evaluated. In clinical practice, most of epithelyosis areas regenerate with conservative measures such as.
In this study, necrosis was observed on almost 40% of the area of the modified flap, more than 10x when compared with the necrosis observed in the McFarlane group. These findings confirm that the flap described by McFarlane et al.55. McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82., in the form that was proposed, does not produce a significant area of necrosis. Therefore the McFarlane flap does not seem to be suitable for studying strategies that might change the viability of dorsal skin flaps. On the other hand, the modified flap with a shorter cranial base gave rise to an area of necrosis that was compatible with the expectations for an experimental model that would be suitable for studying skin viability.
Conclusion
The modified flap with dimensions of 3x10cm, with a cranial base, is more appropriate for experimental studies for the viability of skin flaps.
References
-
1Frederick FW, Sweeny L, Carroll WR, Peters GE, Rosenthal EL. Outcomes in head and neck reconstruction by surgical site and donor site. Laryngoscope. 2013;123(7):1612-7.
-
2Lebas D, Wiart T, Gros C, Modiano P. Use of a rhomboid flap to repair temporal and frontotemporal cutaneous defects: 11 cases. An Dermatol Venereal. 2013;140:170-5.
-
3Acevedo-Bogado CE, Bins-Ely J, d'Acampora AJ, Neves RD. Efeito da hialuronidase na sobrevida de retalhos cutâneos em ratas. Acta Cir Bras. 2002;17(1):14-6.
-
4Reichner DR, Scholz T, Vanderkam VM, Gutierrez S, Steward E, Evans GR. Laser flap delay: comparison of Erbium: YAG and CO2 lasers. Am Surg. 2003;69(1):69-72.
-
5McFarlane RM, DeYoung G, Henry RA. The design of pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965;35(2):177-82.
-
6Oh M, Chang H, Minn KW. The effects of tadalafil on axial-pattern skin flap survival in rats. Dermatol Surg. 2008;34(5):626-30.
-
7Barral SM, Araujo ID, Vidigal PVT, Mayrink CAC, Araujo AD, da Costa PR. Effects of sildenafil on the viability of random skin flaps. Acta Cir Bras. 2011;26(4):314-9.
-
8Lin DS, Zheng X, Li ZJ, Zhang YP, Wang AY, Xie XG, Gao WY. Effect of hirudin on random skin flap survival in rats. Zhonghua Zheng Xing Wai Ke Za Zhi. 2011;27(1):35-9.
-
9Moura TD, Marques Ade A, Bernal SO, Gagliocca GD, Gemperli R, Ferreira MC. Study of the effect of streptokinase and allopurinol in island skin flaps submitted to prolonged ischemia: experimental study in rats. Rev Assoc Med Bras. 2009;55(5):601-5.
-
10Tosun Z, Bite U. A comparative study of the influence of smoking on skin flap survival in pregnant rats. Br J Plastic Surg. 2005;58(6):812-6.
-
11Abla LEF, Gomes HG, Percario S, Ferreira LM. Acetylcysteine in random skin flap in rats. Acta Cir Bras. 2005;20(2):121-3.
-
12Briggs PC. The McFarlane flap. Plast Reconstr Surg. 1987;80(3):472-3.
-
13Gemperli R, Munhoz AM. The influence of type of vascular pedicle occlusion on the viability of skin Island flaps. A postoperative quantitative assessment of flap survival in an experimental model in rats. Acta Cir Bras. 2013;28(7):487-93.
-
1
Research performed at Laboratory of Descriptive and Topographic Anatomy Division, Department of Surgery, School of Medicine, University of Sao Paulo (USP), Brazil. Part of PhD degree thesis, Postgraduate Program in Medicine Surgical Clinics. Tutor: Rolf Gemperli.
Publication Dates
-
Publication in this collection
Mar 2014
History
-
Received
18 Nov 2013 -
Reviewed
20 Jan 2014 -
Accepted
21 Feb 2014