Abstract
PURPOSE:
To determine the minimum volume of methylene blue (MB) to completely color the brachial plexus (BP) nerves, simulating an effective anesthetic block in cats.
METHODS:
Fifteen adult male cat cadavers were injected through subscapular approach with volumes of 2, 3, 4, 5 and 6 ml in both forelimbs, for a total of 30 brachial plexus blocks (BPB). After infusions, the specimens were carefully dissected preserving each nervous branch. The measurement of the effective area was indicated by the impregnation of MB. Nerves were divided into four segments from the origin at the spinal level until the insertion into the thoracic limb muscles. The blocks were considered effective only when all the nerves were strongly or totally colored.
RESULTS:
Volumes of 2, 3 and 4 ml were considered insufficient suggesting a failed block, however, volumes of 5 and 6 ml were associated with a successful block.
CONCLUSIONS:
The injection of methylene blue, in a volume of 6 ml, completely colored the brachial plexus. At volumes of 5 and 6 ml the brachial plexus blocks were considered a successful regional block, however, volumes of 2, 3 and 4 ml were considered a failed regional block.
Brachial Plexus; Methylene Blue; Forelimb; Shoulder; Anatomy, Regional; Cats
Introduction
Multimodal anesthesia is the administration of different drugs and/or techniques to
exploit their synergistic effects. Thereby, it is possible to meet the main
prerequisites of surgical anesthesia: hypnosis, analgesia and muscle relaxation.
Moreover, through this synergism it is possible to reduce the general anesthetic doses,
known to be responsible for the undesirable depression of the cardiovascular system11 Rioja E, Sinclair M, Chalmers H, Foster R, Monteith G. Comparison of
three techniques for paravertebral brachial plexus blockade in dogs. Vet Anaesth
Analg. 2012 Mar;39(2):190-200. doi: 10.1111/j.1467-2995.2011.00677.x.
https://doi.org/10.1111/j.1467-2995.2011...
.
In modern anesthesia, regional blocks are an important tool used by the anesthesiologists, since these techniques provide complete intraoperative analgesia and suppression the sympathetic-adrenal axis. In this context, the brachial plexus block (BPB) is the most accepted anesthetic protocol for the forelimbs and shoulder major surgeries, because the desensitization of the peripheral nerves provides analgesia for the musculoskeletal structures.
The brachial plexus (BP) is a set of nerve fibers that innervate the forelimb and
shoulder, comprising motor and sensory nerves that are present in specific areas, whose
origin is the spinal cord between segments C5 and T222 Anson A, Gil F, Laredo F, Soler M, Belda E, Ayala M, Agut A.
Comparative ultrasound anatomy of the feline brachial plexus and major nerves of the
thoracic limb. Vet Radiol Ultrasound. 2013 Mar-Apr; 54(2):185-93. doi:
10.1111/vru.12012.
https://doi.org/10.1111/vru.12012...
. In most species it usually consists of the suprascapular, subscapular,
musculocutaneous, axillary, radial, median and ulnar nerves. The radial nerve is the
largest nerve composing the BP and is responsible for the motor innervation of the
extensor muscles in the region of the arm, while the ulnar nerve is responsible for
innervating the muscles of the flexor carpi ulnar and digital flexor.
BPB involves the deposition of local anesthetic in the perineurium, leading to
electrical stimulation blockage and delayed nociceptive transmission. This regional
block is currently one of the most studied techniques in veterinary medicine because,
due to anatomical difficulties, access to the nerves that make up this complex is an
arduous task for anesthesiologists. Traditionally, the main route used by the surgeon is
to introduce the needle through the axillary or subscapular approach. Nowadays, the
effectiveness of BPB is improved with use of technological methods such as peripheral
nerve stimulatuion33 Wakoff T, Mencalha R, Souza N, Santos-Sousa C, Desterro M, Scherer P.
Bupivacaína 0,25% versus ropivacaína 0,25% no bloqueio do plexo braquial em cães da
raça beagle. Sem Cien Agra. 2013;34(3):1259-72. doi: 10.5433/1679-0359.2013.
https://doi.org/10.5433/1679-0359.2013...
and ultrasound44 Campoy L, Bezuidenhout A, Gleed R, Martin-Flores M, Raw R, Santare C,
Jay A, Wang A. Ultrasound-guided approach for axillary brachial plexus, femoral
nerve, and sciatic nerve blocks in dogs. Vet Anaesth Analg. 2010 Mar;37(2):144-53.
doi: 10.1111/j.1467-2995.2009.00518.x.
https://doi.org/10.1111/j.1467-2995.2009...
.
Because of the apparent increase of cat ownership, surgical cases of this species have
increased significantly. Thus, clinical and experimental research related to the
management of acute feline pain following the surgical cases has greatly increased55 Brondani J, Mama K, Luna S, Niyon S, Ambrosio J, Vogel P, Padovani C.
Validation of the English version of the UNESP-Botucatu multidimensional composite
pain scale for assessing postoperative pain in cats. BMC Vet Res. 2013 Jul 17;9:143.
doi: 10.1186/1746-6148-9-143.
https://doi.org/10.1186/1746-6148-9-143...
. However, there is still a scarcity of data in the literature regarding the
recognition and treatment of pain in this species66 Robertson S. Managing pain in feline patients. Vet Clin North Am
Small Anim Pract. 2005 Jan;35:129-46. PubMed PMID: 15627631..
The goal of this study was to determine the minimal volume of methylene blue to completely color the brachial plexus nerves (suprascapular, subscapular, musculocutaneous, axillary, radial, ulnar and median) after the application of different volumes by the subscapular approach, simulating the local anesthetic required to anesthetize all the structures involved in forelimb and shoulder surgery in cats. We hypothesized that the usual volumes used in casuistry are insufficient for thorough dyeing of these nerves.
Methods
The Institutional Animal Care Use Committee of the Rio de Janeiro Federal Rural University (UFRRJ) approved this study through process no. 23083.0053334/2009-49. A total of 15 adult male cat cadavers, weighing 3.0 ± 0.5 kg, were obtained from the Animal Anatomy Area of UFRRJ. These cadavers were previously sexed, identified and fixed with 10% formaldehyde solution.
All procedures were performed in the animal anatomy laboratory of UFRRJ. The block simulation was performed by a single veterinarian, who was considered inexperienced in the technique. This choice was made to minimize the influence of professional skill in coloring nerves, thereby giving more importance to the total volume administered.
By the subscapular approach, the brachial plexus was located between the medial scapular and proximal humerus region, at the costochondral joint of the first costal arch, between the costal arches and scapula. The best access to carry out anesthetic block is at the height of the shoulder joint. The caudal limit for needle insertion was the cranial border of the first rib.
The infusions of methylene blue (pH 7.34) 10mg/ml (Sigma-Aldrich, Inc. MO, USA), diluted in an equal volume of 0.9% saline solution, were administrated through the subscapular approach through a 23-SWG unipolar needle (Atraumatic Pencil Point Spinal Needle, Tokyo, Japan) using volumes of 2, 3, 4, 5 and 6 ml. In each mentioned volume, three animals were used for a total of six blocks per volume of test solution. All injections were performed at a pressure less than 15 psi as measured with a Bourdon aneroid gauge.
After completing the infusions, the proposed areas were carefully dissected, in both antimeres, with the naked eye, and when necessary, with a surgical microscope (Opto 200 - Opto Eletronica S/A, Sao Carlos-SP). Additionally, the sternum and then the vertebral bodies were removed until complete exposure of the spinal cord and nerve roots of the brachial plexus. Next the nerves of brachial plexus were photographed with a high definition camera and outlined manually via drawings.
The evaluation of the effective area of the brachial plexus block was indicated by the impregnation solution of methylene blue. Nerves belonging to the brachial plexus (suprascapular, subscapular I and II, musculocutaneous, axillary, radial, median and ulnar) were divided into four segments from its origin (C6, C7, C8, T1 and T2) to its insertion in the muscles of musculoskeletal forelimb. The following criteria were used to assess the dye impregnation: I- nil (no coloration); II- weak coloration (1/4 of the nerve colored); III- medium coloration (half of the nerve colored); IV- strong coloration (3/4 of the nerve colored) and V- totally colored (nerve completely colored).
The block was considered satisfactory only when all the nerves were strongly or totally impregnated by the dye.
Statistical analyses were performed using the Graphpad Prim for Mac OS X version 6.0 program to determine the effectiveness of dye impregnation. The nonparametric data present in this study were analyzed through the Kruskal Wallis Test followed by Dunn's test. These data were used to compare the efficacy of the different volumes of methylene blue administered. Significant difference was accepted when p<0.05. Nerve coloration was classified as mentioned previously.
Results
Thirteen brachial plexuses of fifteen cat cadavers with body weight (mean ± SD, 3.0 ± 0.5 kg), body length (mean 44 ± 3.0 cm) and forearm length (mean 25 ± 0.5 cm) were used in the experiment. No intraneural injections were observed during this study.
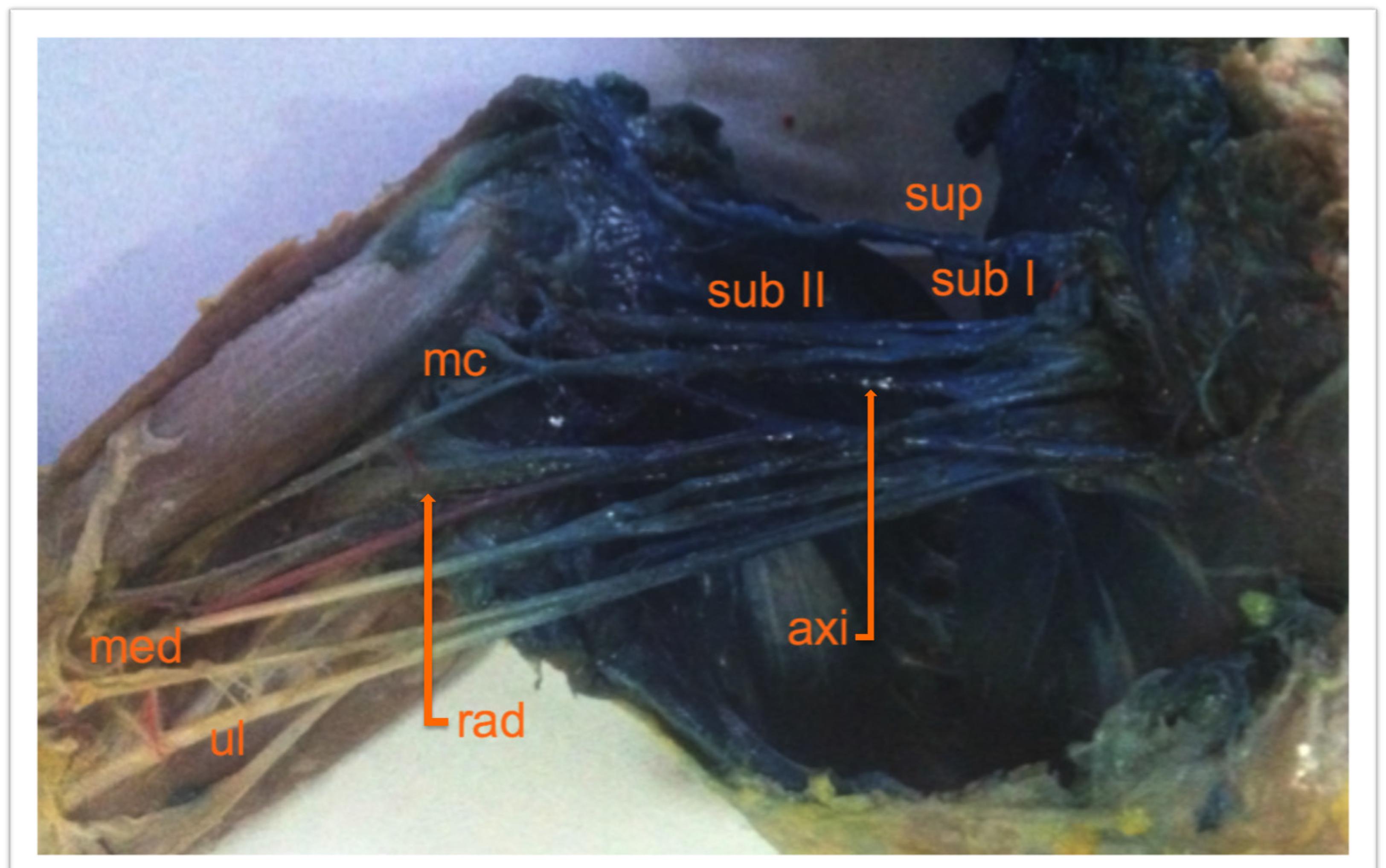
With volumes of 2, 3 and 4 ml, all nerves of the studied plexuses had nil, weak or medium coloration, denoting a failed (partial or absent) regional block (Figures 1, 2 and 3). Furthermore, with volumes of 5 and 6 ml, all the nerves of the brachial plexuses were strongly (3/4) or totally colored (4/4). Consequently, we considered these cases to be successful (total or complete) regional block simulation (Figures 4 and 5). The components of the brachial plexuses were completely colored only with a volume of 6 ml (Figure 5).
- Dissection appearance of the brachial plexus in a cat. Nerves colored with 2 ml of 1% methylene blue by subscapular approach. Note that radial (rad), median (med) and ulnar (ul) nerves were weakly colored and suprascapular (sup), subscapular I (sub I) and II (sub II), axillary (axi) and musculocutaneus (mc) were moderately colored.
- Dissection appearance of the brachial plexus in a cat. Nerves colored with 3ml of 1% methylene blue by subscapular approach. Note that radial (rad), median (med) and ulnar (ul) nerves were weakly colored; axillary (axi) and musculocutaneus (mc) were moderately colored.
- Dissection appearance of the brachial plexus in a cat. Nerves colored with 4 ml of 1% methylene blue by subscapular approach. Note that suprascapular (sup), subscapular I (sub I) and II (sub II), axillary (axi) and musculocutaneus (mc) nerves were strongly colored. Radial (rad), median (med) and ulnar (ul) nerves were moderately colored.
- Dissection appearance of the brachial plexus in a cat. Nerves colored with 5 ml of 1% methylene blue by subscapular approach. Note that suprascapular (sup), median (med) and ulnar (ul) nerves were strongly colored and subscapular I (sub I) and II (sub II), radial (rad) and axillary (axi) nerves were fully colored (p<0.05).
- Dissection appearance of the brachial plexus in a cat. Nerves colored with 5ml of 1% methylene blue by subscapular approach. Note that all nerves of brachial plexus were fully colored (p<0.05). Suprascapular (sup), subscapular I (sub I), subscapular II (sub II), axillary (axi), musculocutaneus (mc), radial (rad), median (med) and ulnar (ul).
- Dye impregnation in brachial plexus after infusion of different volumes of methylene blue. Data expressed as mean.
According to the statistical treatment, the volume administered did not influence dye uptake in the suprascapular nerve, but significant differences (p<0.05) were found in coloring the subscapular I and II, musculocutaneus, axillary, radial, median and ulnar nerves. Furthermore, we observed that the volumes of 5 and 6 ml were visibly better at impregnating these nerves than 2, 3 and 4 ml (p<0.05).
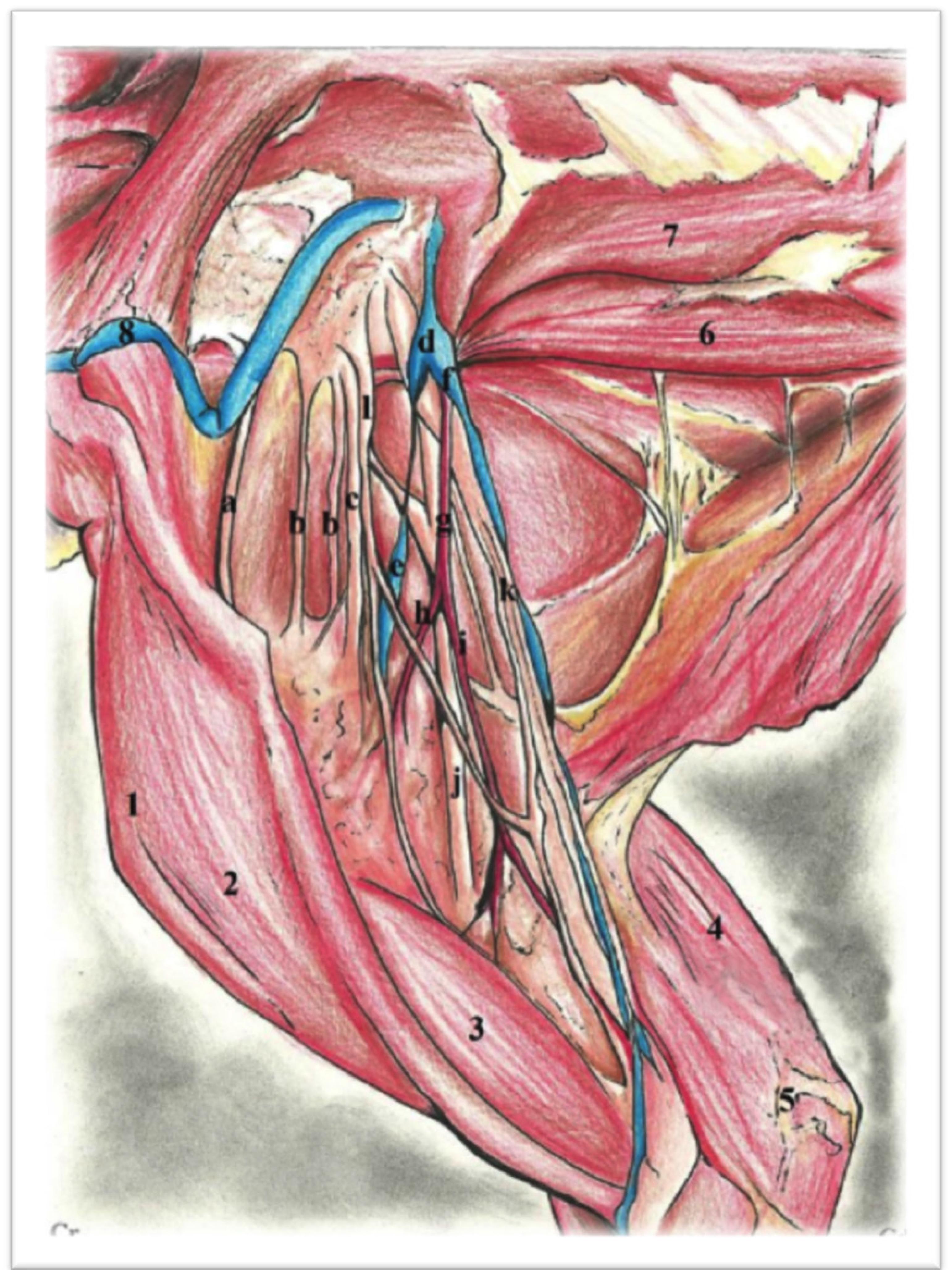
After dissection, the brachial plexus was easily detected in all cases (n=30). The plexuses in cats appeared as wide and thin tracks that reached the axillary space, pasting through the long muscles of the neck and ventral scalene, which concealed the output of the cervical nerves through the intervertebral foramina. In these animals, the brachial plexus resulted from connections established mainly between the ventral branches of the cervical spinal nerves (C5, C6, C7 and C8) and the first and second thoracic spinal nerves (T1 and T2). Their components were identified from cranial to caudal as follows: suprascapular nerve, subscapular nerve (double), axillary nerve, musculocutaneous nerve, ansa axillaris (union between the musculocutaneous and median), radial nerve, and the common trunk of median nerves and ulnaris nerves. Figure 7 shows a schematic drawing of these dissections.
- Schematic drawing of brachial plexus. 1. Shoulder joint; 2. Brachiocephalicus muscle; 3. Biceps brachii muscle; 4. Triceps brachii muscle; 5. Elbow joint; 6. Scalenus dorsalis muscle; 7. Retus thoracis muscle; 8. Jugularis externa vein; a. Suprascapularis nerve; b. Subscapularis nerve (double); c. Axillares nerve; d. Gross dissection of the braquial plexus; e. Subscapularis vein; f. Brachialis vein; g. Axillaris artery; h. Subscapularis artery; i. Brachialis artery; j. Radialis nerve; k. Common trunk of medianus, and ulnaris nerves; l. Musculocutaneos nerve; ansa axillaris.
Furthermore, the anatomical structures observed in the experiment were similar in all cadavers, and there is no distinction that could influence the success of the block.
Discussion
The assessment of embedded area after subscapular injection was proposed due to a
scarceness data in the veterinary literature regarding the correlation between the
volume of local anesthetic administered and efficacy of brachial plexus block77 Winnie A, Radonjic R, Akkineni S, Durrani, Z. Factors influencing
distribution of local anesthetic injected into the brachial plexus sheath. Anesth
Analg. 1979 May-Jun;58:225-34. PubMed PMID: 572160. . When it comes to feline medicine such data are even less enlightening88 Mosing M, Reich H, Moens Y. Clinical evaluation of the anaesthetic
sparing of brachial plexus block in cats. Vet Anaesth Analg. 2010 Mar;37(2):154-61.
doi: 10.1111/j.1467-2995.2009.00509.x.
https://doi.org/10.1111/j.1467-2995.2009...
, causing doubts and fears on the part of anesthesiologists. We chose the
subscapular approach due to its greater acceptance and popularity among veterinarians.
As previously mentioned, the blockades were performed by a single person, who was considered inexperienced through the definition of expertise in regional anaesthesia99 Smith A, Pope C, Goodwin D, Mort M. What defines expertise in regional anaesthesia? An observation analysis of practice. Br J Anaesth. 2006 Sep;97(3):401-7. Epub 2006 Jul 11. PubMed PMID: 16835256. . However, it is known that success in carrying out this technique requires basic knowledge of anatomy, so that the placement of the local anesthetic can be more accurate and, consequently, requiring smaller drug volumes to achieve the specific blockade of a nerve. In this context, this method was recommended to minimize the influence of professional ability to perform the blockade, so that the results would indicate the influence of the total volume of solution administered on the impregnation of the peripheral nerves.
The volume of solution injected is a relevant factor for successful anesthesia77 Winnie A, Radonjic R, Akkineni S, Durrani, Z. Factors influencing
distribution of local anesthetic injected into the brachial plexus sheath. Anesth
Analg. 1979 May-Jun;58:225-34. PubMed PMID: 572160.
8 Mosing M, Reich H, Moens Y. Clinical evaluation of the anaesthetic
sparing of brachial plexus block in cats. Vet Anaesth Analg. 2010 Mar;37(2):154-61.
doi: 10.1111/j.1467-2995.2009.00509.x.
https://doi.org/10.1111/j.1467-2995.2009...
9 Smith A, Pope C, Goodwin D, Mort M. What defines expertise in
regional anaesthesia? An observation analysis of practice. Br J Anaesth. 2006
Sep;97(3):401-7. Epub 2006 Jul 11. PubMed PMID: 16835256.
-
1010 Harper G, Stafford M, Hill D. Minimum volume of local anaesthetic
required to surround each of the constituent nerves of the axillary brachial plexus,
using ultrasound guidance: a pilot study. Br J Anaesth. 2010 May;104(5):633-6. doi:
10.1093/bja/aeq050.
https://doi.org/10.1093/bja/aeq050...
. In the majority of the medical literature, the dose of local anesthetic chosen
for brachial plexus block is mainly associated with the drug concentration and volume.
In this study, the minimum volume of methylene blue (10mg/ml) used was 2 ml and the
maximum of 6 ml, in cat cadavers that did not weigh more than 3.5 kg, and the total
effectiveness of imbibition was observed with 6 ml.
With volumes of 5 and 6 ml, all the nerves of the brachial plexus were strongly or totally colored. Consequently, we considered a successful (total or complete) regional block simulation. We correlated the impregnation level of methylene blue with the anesthetic quantity to be deposited on the peripheral nerves. Therefore, a more extensive coloring of the nerves presumably would be directly related to the effectiveness of anesthesia. At the same time, with volumes of 2, 3 and 4 ml, all nerves of the studied plexuses showed no, weak of medium coloration, denoting a failed (partial or absent) regional block, for the same reasons mentioned above. This assessment parameter was included in this study due to data obtained in a study conducted by electromyographic evaluation of ulnar nerve block. In that paper, the authors reported that 2% lidocaine produced faster onset of action and longer duration than 1% lidocaine1111 Atanassoff P, Kelly D, Ayoub C, Brull S. Electromyographic assessment of ulnar nerve motor block induced by lidocaine. J Clin Anesth. 1998 Dec;10:641-5. PubMed PMID: 9873964.. It is believed that the concentration of the local anesthetic used has an influence on the absorption capacity at the perineural level, obeying Fick's principle1212 Löfström B. Aspects of the pharmacology of local anaesthetic agents. Br J Anaesth. 1970 Mar;42:194-206. PubMed PMID: 4316769..
In addition, the quantitative relationship between the concentration of local anesthetic, the length of nerve exposed, and degree of conduction blockade was studied by others authors1313 Raymond S, Steffensen S, Gugino D, Strichartz R. The role of length of nerve exposed to local anesthetics in impulse blocking action. Anesth Analg. 1989 May;68:563-70. PubMed PMID: 2785778. . In summary, the study indicated that recordings of the compound action potential and of single axons established that small variations in the length of nerve exposed to local anesthetic strongly modulate conduction block even at exposure lengths in excess of 2 centimeters. Therefore, exposure length is a significant factor in determining blocking potency, and only at very high concentrations of local anesthetics, where voltage-dependent sodium conductance is almost completely blocked, is less critical exposure period of three nodes of Ranvier. The results of this study are consistent with the hypothesis of decrease conduction, where a partial active response in nodes exposed to marginal blocking concentrations extends the decline of the action potential along the axon and do not support the interpretation that lengths of several centimeters affect blocking concentration because such distances increase the probability that three nodes will be blocked in succession. This study contradicts the broader common assumption that beyond three nodes, the length of nerve exposed is not a relevant factor with local anesthetics.
Based of their small body weight in relation to other animals, cases of local anesthetic
intoxication in cats are not uncommon1414 Ford D, Singh P, Watters C, Raj P. Duration and toxicity of
bupivacaine for topical anesthesia of the airway in the cat. Anesth Analg. 1984
Nov;63:1001-4. PubMed PMID: 6496968.. In this way, the use of high volumes of this class of drugs should be
associated with dilution, i.e., being administered at lower concentrations than
usual15 15 Yang S, Abrahams M, Hurn P, Grafe M, Kirsch J. Local anesthestic
Schwann cell toxicity is time and concentration dependent. Reg Anesth Pain Med. 2011
Sep-Oct;36(5):444-51. doi: 10.1097/AAP.0b013e318228c835.
https://doi.org/10.1097/AAP.0b013e318228...
to minimize the risks of intoxication. Additionally, it is possible that in the
light of these lower concentrations, the effective blockade is related only to sensitive
fibers in function of a more slender caliber in detriment the motor fibers, while still
maintaining the analgesic quality1616 Bilgen S, Koner O, Menda F, Karacay S, Kaspar E, Sozubir S. A
comparison of two different doses of bupivacaine in caudal anesthesia for neonatal
circumcision. A randomized clinical trial. Middle East J Anesthesiol. 2013 Feb;22:
93-8. PubMed PMID: 23833857..
According to our study, the total volume administered in the brachial plexus influenced
dye uptake in subscapular I and II, musculocutaneus, axillary, radial, median and ulnar
nerves. These data corroborate recent studies on human and dog cadavers, which associate
complete dyeing of major nerve clusters with high volumes of solution1717 Joanne G, Detlev G. A cadaver study to determine the minimum volume
of methylene blue or black naphthol required to completely color the nerves relevant
for anesthesia during breast surgery. Clin Anat. 2011
Mar;24(2):202-8.
-
1818 Campoy L, Martin-Flores M, Looney A, Erb H, Ludders J, Stewart J,
Gleed R, Asakawa M. Distribution of lidocaine-methylene blue solution staining in
brachial plexus, lumbar plexus and sciatic nerve blocks in the dog. Vet Anaesth
Analg. 2008 Jul;35(4):348-54. doi: 10.1111/j.1467-2995.2007.00390.x.
https://doi.org/10.1111/j.1467-2995.2007...
. Furthermore, this correlation was not observed in dye uptake in suprascapular
nerves. We believe that the anatomic proximity of the nerve above the route of
administration of the dye solution may have contributed to a greater facility of
staining.
The results of our study indicate that the anesthetic block of the median and ulnar nerves is associated only with larger volumes of solution, so it is easy to explain the major flaws of this block when it comes to forearm, hand and finger surgery. Thus, the complete block of the brachial plexus when performed blindly was associated with high anesthetic volumes.
Accidental puncture of the jugular vein, axillary artery, axillary vein, brachial plexus
caudal branches, needle insertion in the chest or the pleural space, intraneural
injection and neuropraxia are the complications most often related to this block1919 Jeng C, Rosenblatt M. Intraneural injections and regional
anesthesia: the known and the unknown. Minerva Anesthesiol. 2011 Jan;77:54-8. PubMed
PMID: 21273966.
-
2020 Steinfeldt T, Werner T, Nimphius W, Wiesmann T, Kill C, Muller H,
Wulf H, Graf J. Histological analysis after peripheral nerve puncture with
pencil-point or tuohy needletip. Anesth Analg. 2011 Feb;112(2):465-70. doi:
10.1213/ANE.0b013e318202cb9c.
https://doi.org/10.1213/ANE.0b013e318202...
. In this study we did not observe any of these complications.
After anatomic dissection, the brachial plexus was best visualized using an axillary
approach, while the major nerves of the thoracic limb were best visualized using a
humeral approach. These data corroborate a recent study in the same species, which
focused on ultrasonographic anatomy22 Anson A, Gil F, Laredo F, Soler M, Belda E, Ayala M, Agut A.
Comparative ultrasound anatomy of the feline brachial plexus and major nerves of the
thoracic limb. Vet Radiol Ultrasound. 2013 Mar-Apr; 54(2):185-93. doi:
10.1111/vru.12012.
https://doi.org/10.1111/vru.12012...
.
Nowadays with the advance of sonoanatomy study and image resources, techniques of nerve
block can be guided by ultrasonography. These blocks are based on the direct
visualization of innervation, anesthetic needle, and adjacent anatomical structures11 Rioja E, Sinclair M, Chalmers H, Foster R, Monteith G. Comparison of
three techniques for paravertebral brachial plexus blockade in dogs. Vet Anaesth
Analg. 2012 Mar;39(2):190-200. doi: 10.1111/j.1467-2995.2011.00677.x.
https://doi.org/10.1111/j.1467-2995.2011...
.
Conclusions
The injection of methylene blue, in a volume of 6 ml, completely colored the brachial plexus. At volumes of 5 and 6 ml the brachial plexus block was considered a successful regional block (total or complete), but volumes of 2, 3 and 4 ml were considered failed regional blocks (absent or partial). In this context, considering the effective anesthetic block for shoulder and forelimb surgery is necessary the use of larger volumes of local anesthetic when the aforementioned technique is performed without neurostimulation or ultrasound guidance.
Acknowledgement
To Brunna Veloso Palmeira dos Santos for making the schematic drawing of the brachial plexus.
References
-
1Rioja E, Sinclair M, Chalmers H, Foster R, Monteith G. Comparison of three techniques for paravertebral brachial plexus blockade in dogs. Vet Anaesth Analg. 2012 Mar;39(2):190-200. doi: 10.1111/j.1467-2995.2011.00677.x.
» https://doi.org/10.1111/j.1467-2995.2011.00677.x -
2Anson A, Gil F, Laredo F, Soler M, Belda E, Ayala M, Agut A. Comparative ultrasound anatomy of the feline brachial plexus and major nerves of the thoracic limb. Vet Radiol Ultrasound. 2013 Mar-Apr; 54(2):185-93. doi: 10.1111/vru.12012.
» https://doi.org/10.1111/vru.12012 -
3Wakoff T, Mencalha R, Souza N, Santos-Sousa C, Desterro M, Scherer P. Bupivacaína 0,25% versus ropivacaína 0,25% no bloqueio do plexo braquial em cães da raça beagle. Sem Cien Agra. 2013;34(3):1259-72. doi: 10.5433/1679-0359.2013.
» https://doi.org/10.5433/1679-0359.2013 -
4Campoy L, Bezuidenhout A, Gleed R, Martin-Flores M, Raw R, Santare C, Jay A, Wang A. Ultrasound-guided approach for axillary brachial plexus, femoral nerve, and sciatic nerve blocks in dogs. Vet Anaesth Analg. 2010 Mar;37(2):144-53. doi: 10.1111/j.1467-2995.2009.00518.x.
» https://doi.org/10.1111/j.1467-2995.2009.00518.x -
5Brondani J, Mama K, Luna S, Niyon S, Ambrosio J, Vogel P, Padovani C. Validation of the English version of the UNESP-Botucatu multidimensional composite pain scale for assessing postoperative pain in cats. BMC Vet Res. 2013 Jul 17;9:143. doi: 10.1186/1746-6148-9-143.
» https://doi.org/10.1186/1746-6148-9-143 -
6Robertson S. Managing pain in feline patients. Vet Clin North Am Small Anim Pract. 2005 Jan;35:129-46. PubMed PMID: 15627631.
-
7Winnie A, Radonjic R, Akkineni S, Durrani, Z. Factors influencing distribution of local anesthetic injected into the brachial plexus sheath. Anesth Analg. 1979 May-Jun;58:225-34. PubMed PMID: 572160.
-
8Mosing M, Reich H, Moens Y. Clinical evaluation of the anaesthetic sparing of brachial plexus block in cats. Vet Anaesth Analg. 2010 Mar;37(2):154-61. doi: 10.1111/j.1467-2995.2009.00509.x.
» https://doi.org/10.1111/j.1467-2995.2009.00509.x -
9Smith A, Pope C, Goodwin D, Mort M. What defines expertise in regional anaesthesia? An observation analysis of practice. Br J Anaesth. 2006 Sep;97(3):401-7. Epub 2006 Jul 11. PubMed PMID: 16835256.
-
10Harper G, Stafford M, Hill D. Minimum volume of local anaesthetic required to surround each of the constituent nerves of the axillary brachial plexus, using ultrasound guidance: a pilot study. Br J Anaesth. 2010 May;104(5):633-6. doi: 10.1093/bja/aeq050.
» https://doi.org/10.1093/bja/aeq050 -
11Atanassoff P, Kelly D, Ayoub C, Brull S. Electromyographic assessment of ulnar nerve motor block induced by lidocaine. J Clin Anesth. 1998 Dec;10:641-5. PubMed PMID: 9873964.
-
12Löfström B. Aspects of the pharmacology of local anaesthetic agents. Br J Anaesth. 1970 Mar;42:194-206. PubMed PMID: 4316769.
-
13Raymond S, Steffensen S, Gugino D, Strichartz R. The role of length of nerve exposed to local anesthetics in impulse blocking action. Anesth Analg. 1989 May;68:563-70. PubMed PMID: 2785778.
-
14Ford D, Singh P, Watters C, Raj P. Duration and toxicity of bupivacaine for topical anesthesia of the airway in the cat. Anesth Analg. 1984 Nov;63:1001-4. PubMed PMID: 6496968.
-
15Yang S, Abrahams M, Hurn P, Grafe M, Kirsch J. Local anesthestic Schwann cell toxicity is time and concentration dependent. Reg Anesth Pain Med. 2011 Sep-Oct;36(5):444-51. doi: 10.1097/AAP.0b013e318228c835.
» https://doi.org/10.1097/AAP.0b013e318228c835 -
16Bilgen S, Koner O, Menda F, Karacay S, Kaspar E, Sozubir S. A comparison of two different doses of bupivacaine in caudal anesthesia for neonatal circumcision. A randomized clinical trial. Middle East J Anesthesiol. 2013 Feb;22: 93-8. PubMed PMID: 23833857.
-
17Joanne G, Detlev G. A cadaver study to determine the minimum volume of methylene blue or black naphthol required to completely color the nerves relevant for anesthesia during breast surgery. Clin Anat. 2011 Mar;24(2):202-8.
-
18Campoy L, Martin-Flores M, Looney A, Erb H, Ludders J, Stewart J, Gleed R, Asakawa M. Distribution of lidocaine-methylene blue solution staining in brachial plexus, lumbar plexus and sciatic nerve blocks in the dog. Vet Anaesth Analg. 2008 Jul;35(4):348-54. doi: 10.1111/j.1467-2995.2007.00390.x.
» https://doi.org/10.1111/j.1467-2995.2007.00390.x -
19Jeng C, Rosenblatt M. Intraneural injections and regional anesthesia: the known and the unknown. Minerva Anesthesiol. 2011 Jan;77:54-8. PubMed PMID: 21273966.
-
20Steinfeldt T, Werner T, Nimphius W, Wiesmann T, Kill C, Muller H, Wulf H, Graf J. Histological analysis after peripheral nerve puncture with pencil-point or tuohy needletip. Anesth Analg. 2011 Feb;112(2):465-70. doi: 10.1213/ANE.0b013e318202cb9c.
» https://doi.org/10.1213/ANE.0b013e318202cb9c
-
1
Research performed at Animal Anatomy Laboratory, Department of Biology, Faculty of Veterinary Medicine, Federal Rural University of Rio de Janeiro (UFRRJ), Seropedica-RJ, Brazil.
Publication Dates
-
Publication in this collection
June 2014
History
-
Received
16 Jan 2014 -
Reviewed
18 Mar 2014 -
Accepted
22 Apr 2014