Abstract
PURPOSE:
To evaluate the effects of the adipose-derived stem cells (ADSC) in the viability of random skin flap in rats.
METHODS:
Thirty five adult male Wistar rats (weight 250-300 g) were used. ADSC were isolated from adult male rats (n=5). ADSC were separated, cultured and then analyzed. A dorsal skin flap measuring 10x4 cm was raised and a plastic barrier was placed between the flap and its bed. After the surgical procedure, the animals were randomized into two groups (n=15 each group), group control and group ADSC. In all groups the procedures were performed immediately after the surgery. The percentage of flap necrosis was measured on the seventh postoperative day.
RESULTS:
The ADSC were able to replicate in our culture conditions. We also induced their adipogenic, osteogenic and chondrogenic differentiation, verifying their mesenchymal stem cells potentiality in vitro. The results were statistically significant showing that the ADSC decreased the area of necrosis (p<0.05).
CONCLUSION:
The cells demonstrated adipogenic, osteogenic and chondrogenic differentiation potential in vitro. The administration of adipose-derived stem cells was effective to increase the viability of the random skin flaps in rats.
Surgical Flaps; Rats; Adult Stem Cells; Stem Cells; Adipose Tissue
Introduction
The stem cells are characterized by their undifferentiated state and their ability to not only generate new stem cells but also specialized cells with different functions. Stem cells can be embryonic or adult11. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Apr;7(2):211-28.. Stem cells derived from adipose tissue are the pluripotent type. In this case these cells can differentiate into osteocytes, chondrocytes, adipocytes, muscle cells, neural and angiogenic lineages11. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Apr;7(2):211-28. , 22. Planat-Benard V, Silvestre JS, Cousin B, André M, Nibbelink M, Tamarat R, Clergue M, Manneville C, Saillan-Barreau C, Duriez M, Tedgui A, Levy B, Pénicaud L, Casteilla L. Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation. 2004 Feb 10;109(5):656-63..
The beginning of the study of stem cells occurred to the researchers Ernest McCulloch and James Till at the Ontario Cancer Institute in Toronto. Their research reported on the presence of self-renewing cells in bone marrow of mice, and these cells were postulated as regenerative stem cells33. Becker AJ, McCulloch EA, Till JE. Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells. Nature. 1963 Feb 2;197:452-4. , 44. Zhang J, Shehabeldin A, da Cruz LA, Butler J, Somani AK, McGavin M, Kozieradzki I, dos Santos AO, Nagy A, Grinstein S, Penninger JM, Siminovitch KA. Antigen receptor-induced activation and cytoskeletal rearrangement are impaired in Wiskott-Aldrich syndrome protein-deficient lymphocytes. J Exp Med. 1999 Nov 1;190(9):1329-42..
In 2001 stem cells derived from adipose tissue (ADSC) were added to the group of adult stem, showing that they are able to differentiate into mesodermal cells (adipocytes, chondrocytes, osteocytes, and myocytes)11. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Apr;7(2):211-28..
Nowadays it is known that the ADSCs have the ability to form consistent cells as neurons55. Kang SK, Putnam LA, Ylostalo J, Popescu IR, Dufour J, Belousov A, Bunnell BA. Neurogenesis of Rhesus adipose stromal cells. J Cell Sci. 2004 Aug 15;117(Pt 18):4289-99., Oligodendrocytes66. Safford KM, Safford SD, Gimble JM, Shetty AK, Rice HE. Characterization of neuronal/glial differentiation of murine adipose-derived adult stromal cells. Exp Neurol. 2004 Jun;187(2):319-28., Schwann cells77. Kingham PJ, Kalbermatten DF, Mahay D, Armstrong SJ, Wiberg M, Terenghi G. Adipose-derived stem cells differentiate into a Schwann cell phenotype and promote neurite outgrowth in vitro. Exp Neurol. 2007 Oct;207(2):267-74. , 88. Xu Y, Liu L, Li Y, Zhou C, Xiong F, Liu Z, Gu R, Hou X, Zhang C. Myelin-forming ability of Schwann cell-like cells induced from rat adipose-derived stem cells in vitro. Brain Res. 2008 Nov 6;1239:49-55. and epidermal cell lineage99. Trottier V, Marceau-Fortier G, Germain L, Vincent C, Fradette J. IFATS collection: Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells. 2008 Oct;26(10):2713-23..
The clinical use of this cell type may vary from angiogenesis and neurogenesis stimulation in spinal cord injury1010. Oh JS, Park IS, Kim KN, Yoon DH, Kim SH, Ha Y. Transplantation of an adipose stem cell cluster in a spinal cord injury. Neuroreport. 2012 Mar 28;23(5):277-82. to the suppression of the inflammatory response, oxidative stress, and apoptosis in rodent models of ischemia and reperfusion1111. Reichenberger MA, Heimer S, Schaefer A, Lass U, Gebhard MM, Germann G, Leimer U, Köllensperger E, Mueller W. Adipose derived stem cells protect skin flaps against ischemia-reperfusion injury. Stem Cell Rev. 2012 Sept;8(3):854-62..
The partial necrosis of the skin flaps remains a significant problem in plastic surgery. Recent studies on addition of stem cells from adipose tissue in subcutaneous tissue of rats demonstrate increased vascularity and viability of skin flaps1212. Yang M, Sheng L, Li H, Weng R, Li QF. Improvement of the skin flap survival with the bone marrow-derived mononuclear cells transplantation in a rat model. Microsurgery. 2010 May;30(4):275-81. , 1313. Lee DW, Jeon YR, Cho EJ, Kang JH, Lew DH. Optimal administration routes for adipose-derived stem cells therapy in ischaemic flaps. J Tissue Eng Regen Med. 2014 Aug;8(8):596-603.. The aim of this study was to evaluate the effects of adipose-derived stem cells on the viability of random skin flap in rats.
Methods
This project was approved by the Ethics Committee of UNIFESP - CEUA N 251501. Animals (250g-300 g) were anesthetized with an intraperitoneal injection of 60 mg/kg of ketamine and 5 mg/kg xylazine. The dorsal random skin flap, measuring 10X4 cm, following experimental model proposed by1414. MCFarlane RM, Deyoung G, Henry RA. The design of a pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965 Feb;35:177-82. was raised from the deep fascia, including the superficial fascia, panniculus carnosus, subcutaneous tissue, and skin. After flap elevation, a plastic barrier (polyester/polyethylene), with the same dimensions (10X4 cm) was placed between the skin flap and its bed. The flap was then sutured back in place with simple 4-0 nylon sutures. Subsequently, 5X1066. Safford KM, Safford SD, Gimble JM, Shetty AK, Rice HE. Characterization of neuronal/glial differentiation of murine adipose-derived adult stromal cells. Exp Neurol. 2004 Jun;187(2):319-28. ADSC in PBS (0.5 mL) were slowly injected into the caudal vein over 3 min using a insulin syringe in the group experimental. Control group of animals received only 0.5 mL of PBS.
Macroscopic analysis of necrosis percentages
The percentage of skin flap necrosis was measured on the seventh postoperative day, using the paper template method described by1515. Sasaki GH, Pang CY. Hemodynamics and viability of acute neurovascular island skin flaps in rats. Plast Reconstr Surg. 1980 Feb;65(2):152-8.. After anesthesia, each flap's limit between viable skin and necrosis was delineated with a pen. The viable tissue limit has been characterized by soft skin, pink, warm and haired, and necrotic tissue by stiff, dark cool, and hairless skin.
Isolation, culture and expansion of ADSC
ADSC were isolated from adult male Wistar rats (weight 250 - 300 g, n = 5). Rat adipose tissue from inguinal region was enzymatically dissociated for 30 min at 37 °C by 0.1 % (w/v) collagenase type I (Sigma-Aldrich). After centrifugation, the stromal cell pellet was resuspended in Dulbecco's Modified Eagle's Medium/Nutrient Mixture F-12 Ham (DMEM/F12) (Sigma-Aldrich) supplemented with 10% Fetal Bovine Serum (FBS) (Cultilab, Campinas-SP, Brazil), 100 U/ml penicillin (Sigma-Aldrich) and 0.1 mg/ml streptomycin (Sigma-Aldrich). The culture was maintained at 37ºC in humidified atmosphere of 95% O2 and 5% CO2 and passages with trypsin/EDTA (Gibco) when required. Cells at passage 3 or below were used for experimentation.
Differentiation assays
Differentiation assays was done according to the method described by Gaiba et al 2012. Osteogenic, adipogenic and chondrogenic differentiation were performed to ascertain multipotency of isolated cells. The cells cultured in differentiation media for 21 days. After that, the cultures were stained by a solution of Alizarin, Oil Red O and Toluidine Blue for osteogenic, adipogenic and chondrogenic differentiation, respectively. The fixed and dyed cells were observed using Nikon Ti-U optical microscope and photographed using the NIS-Elements - 3.2 Software (Nikon Instruments INC, New York).
Statistical analysis
The results are expressed as mean ± SD. Comparison between two means was performed by unpaired Student's t-test. All data were analyzed using GraphPad Prism 3.0 software. Statistically significance was accepted when P<0.05.
Results
Macroscopic analysis of necrosis percentages
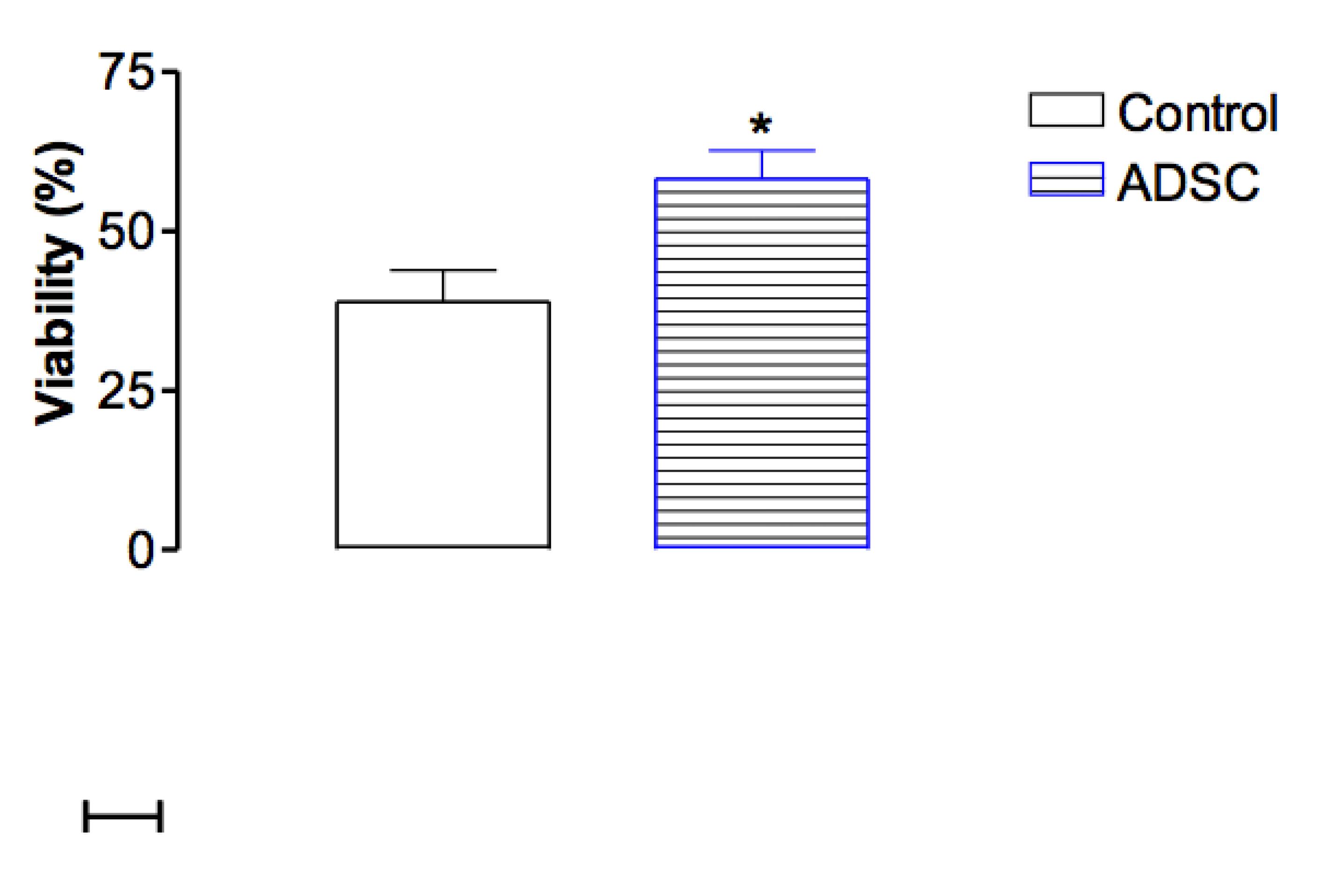
The regions of survival and necrosis were clearly demarcated in every flap at 7th day post operation. Figure 1 presents means and distribution of data obtained for percentages of flap viability in the groups. The percentages of viability area (mean ± standard deviation) in the ADSC and control groups were (58.14 ± 4.460)% and (38.86 ± 5.021)%, respectively.
Distribution of the percentage of viability area of the groups. These values were analyzed using unpaired Student's t test and statistical significance was obtained (p <0.05).
Isolation, culture and expansion of ADSC
Upon applying multilineage differentiation (adipogenic, osteogeneic and chondrogenic) the cells showed accumulated intracellular lipid droplets as revealed by Oil Red O staining (Figure 2A) and displayed extracellular calcium precipitates, which were identified by Alizarin red staining (Figure 2B) and Chondrogenic differentiation demonstrated by Toluidin blue stain (Figure 2C). Indicates that these cells can differentiate into adipocytes, osteoblasts and chondroblasts.
Multilineage differentiation. ADSC are typical fibroblast-like cells with fusiform shape. (A) Adipogenic differentiation demonstrated by Oil Red O staining after 21 days (positive intracellular lipid droplets). (B) Osteogenic differentiation demonstrated by Alizarin red stain after 21 days induction (positive staining of calcium nodule formation). (C) Chondrogenic differentiation demonstrated by Toluidin blue stain 21 days induction.
Discussion
The use of ADSCs capable of differentiate into mesodermal cells is made since 200111. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Apr;7(2):211-28.. Their applicability in experimental models is increasing and it is approaching clinical practice1616. Wei X, Yang X, Han ZP, Qu FF, Shao L, Shi YF. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013 Jun;34(6):747-54..
In plastic surgery this type of cell has also been studied and increasingly used, for example, to increase the success rate of viability in the grafts1717. Beahm EK, Walton RL, Patrick CW Jr. Progress in adipose tissue construct development. Clin Plast Surg. 2003 Oct;30(4):547-58. and small defects in fat grafting1818. DiGirolamo M, Fine JB, Tagra K, Rossmanith R. Qualitative regional differences in adipose tissue growth and cellularity in male Wistar rats fed ad libitum. Am J Physiol. 1998 May;274(5 Pt 2):R1460-7.. Studies have been done in rats comparing qualitatively peritoneal and inguinal region tissues1818. DiGirolamo M, Fine JB, Tagra K, Rossmanith R. Qualitative regional differences in adipose tissue growth and cellularity in male Wistar rats fed ad libitum. Am J Physiol. 1998 May;274(5 Pt 2):R1460-7., however there are no studies that quantitatively compare the number of ADSC in these regions. Thus, interpreting the results we can infer that the number of stem cells in the inguinal region predominates over the peritoneal region (data not shown).
The peritoneal fat, have lower gain of adipose tissue mass compared to lower regions of the body, as the inguinal region, due to a protective mechanism that aims to reduce the metabolic consequences of weight gain1919. Tchoukalova YD, Koutsari C, Votruba SB, Tchkonia T, Giorgadze N, Thomou T, Kirkland JL, Jensen MD. Sex- and depot-dependent differences in adipogenesis in normal-weight humans. Obesity (Silver Spring). 2010 Oct;18(10):1875-80.. The statistically significant results comparing both collected areas directs the ADSC extraction from the inguinal region, ensuring greater concentration of cells collected in comparision to the peritoneal region, which in turn can be useful in designing future studies aimed at testing the properties of ADSC, as done in this work, which envisaged its closest application to clinical practice.
Regarding the clinical applicability of stem cells, the cutaneous flap is a common and valuable procedure in plastic surgery, such as the repair of retractions of burns and reconstructions after oncologic resections. However, there are factors such as ischemia and necrosis, which may damage its development, justifying the need to investigate possibilities to reduce these risks and increase the viability of the flap2020. Harder Y, Amon M, Erni D, Menger MD. Evolution of ischemic tissue injury in a random pattern flap: a new mouse model using intravital microscopy. J Surg Res. 2004 Oct;121(2):197-205.
21. Abla LE, Gomes HC, Percario S, Ferreira LM. Acetylcysteine in random skin flap in rats. Acta Cir Bras. 2005 Mar-Apr;20(2):121-3.
22. Liebano RE, Abla LE, Ferreira LM. Effect of low-frequency transcutaneous electrical nerve stimulation (TENS) on the viability of ischemic skin flaps in the rat: an amplitude study. Wound Repair Regen. 2008 Jan-Feb;16(1):65-9.
23. Tacani PM, Liebano RE, Pinfildi CE, Gomes HC, Arias VE, Ferreira LM. Mechanical stimulation improves survival in random-pattern skin flaps in rats. Ultrasound Med Biol. 2010 Dec;36(12):2048-56. - 2424. Nishioka MA, Pinfildi CE, Sheliga TR, Arias VE, Gomes HC, Ferreira LM. LED (660 nm) and laser (670 nm) use on skin flap viability: angiogenesis and mast cells on transition line. Lasers Med Sci. 2012 Sept;27(5):1045-50..
Studies with models of grafts2525. Zografou A, Papadopoulos O, Tsigris C, Kavantzas N, Michalopoulos E, Chatzistamatiou T, Papassavas A, Stavropoulou-Gioka C, Dontas I, Perrea D. Autologous transplantation of adipose-derived stem cells enhances skin graft survival and wound healing in diabetic rats. Ann Plast Surg. 2013 Aug;71(2):225-32., and flaps2626. Yue Y, Zhang P, Liu D, Yang JF, Nie C, Yang D. Hypoxia preconditioning enhances the viability of ADSCs to increase the survival rate of ischemic skin flaps in rats. Aesthetic Plast Surg. 2013 Feb;37(1):159-70. using the inguinal region ADSC showed increased viability of the necrotic area, however the route of administration of ADSC was subcutaneously. In the present study, the route of administration was intravenous, finding similar results to those mentioned, which show an increase in flap viability with the use of ADSC.
The statistically significant results regarding the use of ADSC from the inguinal region, decreasing skin flap necrosis, contribute to approximate the experimental use to clinical practice. However, further studies are needed to complement these results, such as the realization of immunohistochemical markers seeking whether there is an increased local vascularization of the flap and the presence of stem cells applied in the flap area.
Conclusion
The cells demonstrated adipogenic, osteogenic and chondrogenic differentiation potential in vitro. The administration of adipose-derived stem cells was effective to increase the viability of the random skin flaps in rats.
References
-
1Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001 Apr;7(2):211-28.
-
2Planat-Benard V, Silvestre JS, Cousin B, André M, Nibbelink M, Tamarat R, Clergue M, Manneville C, Saillan-Barreau C, Duriez M, Tedgui A, Levy B, Pénicaud L, Casteilla L. Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation. 2004 Feb 10;109(5):656-63.
-
3Becker AJ, McCulloch EA, Till JE. Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells. Nature. 1963 Feb 2;197:452-4.
-
4Zhang J, Shehabeldin A, da Cruz LA, Butler J, Somani AK, McGavin M, Kozieradzki I, dos Santos AO, Nagy A, Grinstein S, Penninger JM, Siminovitch KA. Antigen receptor-induced activation and cytoskeletal rearrangement are impaired in Wiskott-Aldrich syndrome protein-deficient lymphocytes. J Exp Med. 1999 Nov 1;190(9):1329-42.
-
5Kang SK, Putnam LA, Ylostalo J, Popescu IR, Dufour J, Belousov A, Bunnell BA. Neurogenesis of Rhesus adipose stromal cells. J Cell Sci. 2004 Aug 15;117(Pt 18):4289-99.
-
6Safford KM, Safford SD, Gimble JM, Shetty AK, Rice HE. Characterization of neuronal/glial differentiation of murine adipose-derived adult stromal cells. Exp Neurol. 2004 Jun;187(2):319-28.
-
7Kingham PJ, Kalbermatten DF, Mahay D, Armstrong SJ, Wiberg M, Terenghi G. Adipose-derived stem cells differentiate into a Schwann cell phenotype and promote neurite outgrowth in vitro. Exp Neurol. 2007 Oct;207(2):267-74.
-
8Xu Y, Liu L, Li Y, Zhou C, Xiong F, Liu Z, Gu R, Hou X, Zhang C. Myelin-forming ability of Schwann cell-like cells induced from rat adipose-derived stem cells in vitro. Brain Res. 2008 Nov 6;1239:49-55.
-
9Trottier V, Marceau-Fortier G, Germain L, Vincent C, Fradette J. IFATS collection: Using human adipose-derived stem/stromal cells for the production of new skin substitutes. Stem Cells. 2008 Oct;26(10):2713-23.
-
10Oh JS, Park IS, Kim KN, Yoon DH, Kim SH, Ha Y. Transplantation of an adipose stem cell cluster in a spinal cord injury. Neuroreport. 2012 Mar 28;23(5):277-82.
-
11Reichenberger MA, Heimer S, Schaefer A, Lass U, Gebhard MM, Germann G, Leimer U, Köllensperger E, Mueller W. Adipose derived stem cells protect skin flaps against ischemia-reperfusion injury. Stem Cell Rev. 2012 Sept;8(3):854-62.
-
12Yang M, Sheng L, Li H, Weng R, Li QF. Improvement of the skin flap survival with the bone marrow-derived mononuclear cells transplantation in a rat model. Microsurgery. 2010 May;30(4):275-81.
-
13Lee DW, Jeon YR, Cho EJ, Kang JH, Lew DH. Optimal administration routes for adipose-derived stem cells therapy in ischaemic flaps. J Tissue Eng Regen Med. 2014 Aug;8(8):596-603.
-
14MCFarlane RM, Deyoung G, Henry RA. The design of a pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965 Feb;35:177-82.
-
15Sasaki GH, Pang CY. Hemodynamics and viability of acute neurovascular island skin flaps in rats. Plast Reconstr Surg. 1980 Feb;65(2):152-8.
-
16Wei X, Yang X, Han ZP, Qu FF, Shao L, Shi YF. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013 Jun;34(6):747-54.
-
17Beahm EK, Walton RL, Patrick CW Jr. Progress in adipose tissue construct development. Clin Plast Surg. 2003 Oct;30(4):547-58.
-
18DiGirolamo M, Fine JB, Tagra K, Rossmanith R. Qualitative regional differences in adipose tissue growth and cellularity in male Wistar rats fed ad libitum. Am J Physiol. 1998 May;274(5 Pt 2):R1460-7.
-
19Tchoukalova YD, Koutsari C, Votruba SB, Tchkonia T, Giorgadze N, Thomou T, Kirkland JL, Jensen MD. Sex- and depot-dependent differences in adipogenesis in normal-weight humans. Obesity (Silver Spring). 2010 Oct;18(10):1875-80.
-
20Harder Y, Amon M, Erni D, Menger MD. Evolution of ischemic tissue injury in a random pattern flap: a new mouse model using intravital microscopy. J Surg Res. 2004 Oct;121(2):197-205.
-
21Abla LE, Gomes HC, Percario S, Ferreira LM. Acetylcysteine in random skin flap in rats. Acta Cir Bras. 2005 Mar-Apr;20(2):121-3.
-
22Liebano RE, Abla LE, Ferreira LM. Effect of low-frequency transcutaneous electrical nerve stimulation (TENS) on the viability of ischemic skin flaps in the rat: an amplitude study. Wound Repair Regen. 2008 Jan-Feb;16(1):65-9.
-
23Tacani PM, Liebano RE, Pinfildi CE, Gomes HC, Arias VE, Ferreira LM. Mechanical stimulation improves survival in random-pattern skin flaps in rats. Ultrasound Med Biol. 2010 Dec;36(12):2048-56.
-
24Nishioka MA, Pinfildi CE, Sheliga TR, Arias VE, Gomes HC, Ferreira LM. LED (660 nm) and laser (670 nm) use on skin flap viability: angiogenesis and mast cells on transition line. Lasers Med Sci. 2012 Sept;27(5):1045-50.
-
25Zografou A, Papadopoulos O, Tsigris C, Kavantzas N, Michalopoulos E, Chatzistamatiou T, Papassavas A, Stavropoulou-Gioka C, Dontas I, Perrea D. Autologous transplantation of adipose-derived stem cells enhances skin graft survival and wound healing in diabetic rats. Ann Plast Surg. 2013 Aug;71(2):225-32.
-
26Yue Y, Zhang P, Liu D, Yang JF, Nie C, Yang D. Hypoxia preconditioning enhances the viability of ADSCs to increase the survival rate of ischemic skin flaps in rats. Aesthetic Plast Surg. 2013 Feb;37(1):159-70.
-
Financial sources: CAPES, CNPq (nº 312356/2009-9)
-
1
Research performed at Plastic Surgery Division, Federal University of Sao Paulo (UNIFESP), Brazil.
Publication Dates
-
Publication in this collection
2014