Abstract
PURPOSE:
To investigate the effect of vibration therapy on the bone callus of fractured femurs and the bone quality of intact femurs in ovariectomized rats.
METHODS:
Fifty-six rats aged seven weeks were divided into four groups: control with femoral fracture (CON, n=14), ovariectomized with femoral fracture (OVX, n=14), control with femoral fracture plus vibration therapy (CON+VT, n=14), and ovariectomized with femoral fracture plus vibration therapy (OVX+VT, n=14). Three months after ovariectomy or sham surgery, a complete fracture was produced at the femoral mid-diaphysis and stabilized with a 1-mm-diameter intramedullary Kirschner wire. X-rays confirmed the fracture alignment and fixation. Three days later, the VT groups underwent vibration therapy (1 mm, 60 Hz for 20 minutes, three times per week for 14 or 28 days). The bone and callus quality were assessed by densitometry, three-dimensional microstructure, and mechanical test.
RESULTS
: Ovariectomized rats exhibited a substantial loss of bone mass and severe impairment in bone microarchitecture, both in the non-fractured femur and the bone callus. Whole-body vibration therapy exerted an important role in ameliorating the bone and fracture callus parameters in the osteoporotic bone.
CONCLUSION:
Vibration therapy improved bone quality and the quality of the fracture bone callus in ovariectomized rats.
Ovariectomy; Osteoporosis; Bony Callus; Vibration; Rats
Introduction
Throughout life the skeleton undergoes a continuous remodeling process that involves bone resorption and formation. During growth and in the young adult period, there is a positive bone balance, i.e., more bone is formed than is absorbed. Equilibrium is then reached, but after 40 years of age, more bone tissue is resorbed than created. This phenomenon may give rise to an osteopenic condition that becomes more severe with aging11. Cauley JA. Estrogen and bone health in men and women. Steroids. 2015 Jul;99(Pt A):11-5. doi: 10.1016/j.steroids.2014.12.010.
https://doi.org/10.1016/j.steroids.2014....
. In post-menopausal women, lack of estrogen is an additional factor that impairs bone quality, thus leading to an osteoporotic condition, which favors the occurrence of fractures. Treating a fracture of osteoporotic bone is a challenge, as older patients are more vulnerable to surgical and anesthetic complications. In addition, osteoporotic bone does not provide a good anchor for orthopedic implants22. Yuan Q, Zhang G, Wu J, Xing Y, Sun Y, Tian W. Clinical evaluation of the polymethylmethacrylate-augmented thoracic and lumbar pedicle screw fixation guided by the three-dimensional navigation for the osteoporosis patients. Eur Spine J. 2015;24(5):1043-50. doi: 10.1007/s00586-013-3131-8.
https://doi.org/10.1007/s00586-013-3131-...
.
Bone healing after a fracture is a specialized repair process that involves a cascade of events including inflammation, osteogenesis, callus formation, and remodeling33. Einhorn TA. The cell and molecular biology of fracture healing. Clin Orthop Relat Res. 1998 Oct;(355 Suppl):S7-21. PMID: 9917622.,44. Einhorn TA. The science of fracture healing. J Orthop Trauma. 2005 Nov-Dec;19(10 Suppl):S4-6. PMID: 16479221.. Although the literature does not show strong scientific evidence that fracture healing is impaired in the elderly, this condition may be expected, as overall metabolic activities are decreased in this population.
It has been recently demonstrated that the disregulation of estrogen receptor expression in ovariectomized animals (OVX) impairs bone healing, as evidenced by a reduction of angiogenic factors, reduction of bone formation markers, and increased bone resorption55. He YX, Zhang G, Pan XH, Liu Z, Zheng LZ, Chan CW, Lee KM, Cao YP, Li G, Wei L, Hung LK, Leung KS, Qin L. Impaired bone healing pattern in mice with ovariectomy-induced osteoporosis: a drill-hole defect model. Bone. 2011 Jun 1;48(6):1388-400. PMID: 21421090., leading to delayed healing or non-union. However, micromovements at the fracture site seems to stimulate bone formation and fracture repair66. Yamaji T, Ando K, Wolf S, Augat P, Claes L. The effect of micromovement on callus formation. J Orthop Sci. 2001;6(6):571-5. PMID: 11793181.. Therefore, controlled mechanical conditions may exert beneficial effects on bone fracture healing. Consistent with this hypothesis, several studies demonstrated an osteogenic effect of vibration therapy in humans77. Gomez-Cabello A, Gonzalez-Aguero A, Morales S, Ara I, Casajus JA, Vicente-Rodriguez G. Effects of a short-term whole body vibration intervention on bone mass and structure in elderly people. J Sci Med Sport. 2014 Mar;17(2):160-4. PMID: 23711620.,88. Liphardt AM, Schipilow J, Hanley DA, Boyd SK. Bone quality in osteopenic postmenopausal women is not improved after 12 months of whole-body vibration training. Osteoporos Int. 2015 Mar;26(3):911-20. PMID: 25567775. and experimental animals99. Rubin CT, Sommerfeldt DW, Judex S, Qin Y. Inhibition of osteopenia by low magnitude, high-frequency mechanical stimuli. Drug Discov Today. 2001 Aug 15;6(16):848-58. PMID: 11495758.,1010. Zhou Y, Guan X, Liu T, Wang X, Yu M, Yang G, Wang H. Whole body vibration improves osseointegration by up-regulating osteoblastic activity but down-regulating osteoblast-mediated osteoclastogenesis via ERK1/2 pathway. Bone. 2015 Feb;71:17-24. PMID: 25304090..
The effects of vibration therapy on the fracture healing process have not yet been fully clarified in the literature. Although previous authors have reported the effects of whole-body vibration therapy on bone osteotomy1111. Komrakova M, Sehmisch S, Tezval M, Ammon J, Lieberwirth P, Sauerhoff C, Trautmann L, Wicke M, Dullin C, Stuermer KM, Stuermer EK. Identification of a vibration regime favorable for bone healing and muscle in estrogen-deficient rats. Calcif Tissue Int. 2013 Jun;92(6):509-20. PMID: 23416966.
12. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877.-1313. Wehrle E, Liedert A, Heilmann A, Wehner T, Bindl R, Fischer L, Haffner-Luntzer M, Jacob F, Schinke T, Amling M, Ignatius A. The impact of low-magnitude high-frequency vibration on fracture healing is profoundly influenced by the oestrogen status in mice. Dis Model Mech. 2015 Jan;8(1):93-104. PMID: 25381012., whether this therapy affects bone secondary healing via callus formation remains undetermined. Therefore, the purpose of this investigation was to assess the effects of vibration therapy on bone tissue and bone fracture callus in both intact and ovariectomized rats.
Methods
Experimental design
Universidade de São Paulo
Fifty-six female 7-week-old Wistar rats (Rattus norvegicus albinus) with initial body mass of 210±10g, whose masses were significantly similar throughout groups. Rats were kept under standard laboratory conditions (room temperature 22±2ºC, humidity 55±5%, 12-h light-dark cycles) with free access to water and standard chow (Nuvilab CR-1, PR, Brazil). After one week of acclimatization, the animals were randomly assigned to one of four groups: [1] control rats with provoked femoral fracture (CON, n=21), [2] ovariectomized rats with provoked femoral fracture (OVX, n=21), [3] control rats with provoked femoral fracture that underwent whole-body vibration therapy (CON+VT, n=22), and [4] OVX rats with provoked femoral fracture that underwent whole-body vibration therapy (OVX+VT, n=21).
Ovariectomy and post-operative care
Rats were anesthetized with an intramuscular injection of xylazine (0.2 mL/kg) and ketamine (0.4 mL/kg) mixture and underwent bilateral extraction of the ovaries through a dorsolateral approach1414. Simoes PA, Zamarioli A, Bloes P, Mazzocato FC, Pereira LH, Volpon JB, Shimano AC. Effect of treadmill exercise on lumbar vertebrae in ovariectomized rats: anthropometrical and mechanical analyses. Acta Bioeng Biomech. 2008;10(2):39-41. PMID: 19031996.. Age-matched control rats were subjected to sham surgery where ovaries were surgically exposed but left undisturbed. After the operation, animals were housed in regular cages (four rats per cage) with unrestricted access to water and chow. All rats received intramuscular buprenorphine (0.03 mg/kg) twice per day over five days for post-operative pain.
Fracture operative technique
Three months after ovariectomy or sham surgery, animals were anesthetized and underwent femoral fracture followed by a surgical procedure for bone fragment stabilization. First, the right pelvic limb was shaved and disinfected with 70% alcohol. Subsequently, the limb was placed in a device especially manufactured to generate a closed fracture at the mid-femur. The animal's right thigh was firmly held on two metallic supports, and a blunt blade aimed at the mid-thigh was lowered by a lever mechanism. Force was applied until a lack of resistance occurred and abnormal movement was felt at the fracture site. Afterward, the animal was removed from the device, and topical pirrolidone iodine solution (Riodeine, RJ, Brazil) was applied to the entire pelvic limb. After surgical isolation, a mini-incision was made on the lateral part of the thigh, over the fracture level. The bone was approached through blunt dissection of the intermuscular septum, and the fracture was carefully exposed and inspected. Then a 1-mm-thick Kirschner wire (K wire) was introduced into the medullary canal of the proximal femoral fragment until its tip protruded through the skin, just above the trochanteric region. The fracture was reduced, and the distal tip of the K wire was advanced until it reached the femoral condylar region. The extra length of the proximal portion of the K wire was cut, bent, and buried beneath the muscle, close to the major trochanter. Radiographs of the hind limb (anteroposterior and profile views) were taken using the following parameters: distance 115 cm, 5 mA, 44 kV, and 5 seconds of exposure. The images were checked for fracture reduction and correctness of the implant positioning. The surgical incision was closed in layers with absorbable suture, and the skin suture was sprayed with a solution to avoid self-mutilation (Adestro, SP, Brazil).
Any fractures not consistent with the standardized (diaphyseal) location were excluded. The animals were housed in individual cages and provided water and chow ad libitum. Post-operation daily care consisted of animal stimulation, gait inspection, incision examination, and assessment of the knee range of motion. All rats received intramuscular buprenorphine (0.03 mg/kg) (Ibasa, RS, Brazil) twice a day for five days to treat post-operative pain and were weighed weekly. Radiographs were taken every week to assess bone healing (Figure 1).
- Profile X-ray view of a rat hindlimb displaying mid-diaphyseal fracture of the femur and intramedullary bone fixation.
Whole-body vibration therapy
Three days after surgery, the OVX+VT rats and their age-matched controls (CON+VT) were subjected to whole-body vibration therapy, with peak-to-peak vertical displacement of 1.0 mm at a frequency of 60.0 Hz. For this therapy, rats were placed in a special cage (20´25´10 cm) on a vibrating platform. This acrylic cage had two compartments, which allowed the treatment of two rats at the same time, but they remained physically isolated from each other1515. Falcai MJ, Zamarioli A, Okubo R, de Paula FJ, Volpon JB. The osteogenic effects of swimming, jumping, and vibration on the protection of bone quality from disuse bone loss. Scand J Med Sci Sports. 2014 Apr 30. PMID: 24779886.. Therapy was performed 3 days per week for 20 minutes per session for 14 or 28 days1616. Zamarioli A, Battaglino RA, Morse LR, Sudhakar S, Maranho DA, Okubo R, Volpon JB, Shimano AC. Standing frame and electrical stimulation therapies partially preserve bone strength in a rodent model of acute spinal cord injury. Am J Phys Med Rehabil. 2013 May;92(5):402-10. PMID: 23478455..
Experimental analysis
On days 14 and 28 following surgery, seven rats from each group were killed by intraperitoneal thiopental (Vetec, RJ, Brazil) overdose. The femurs were harvested and cleaned of soft tissues, taking care not to disrupt the bone callus. The K wire was removed, and the bones were subjected to determination of bone mineral density (BMD), three-dimensional microstructure analysis, and mechanical test assessment. A laparotomy was performed, and the uterus was removed and weighted.
Assessment of bone mineral density (BMD) and bone mineral content (BMC)
BMD and BMC were assessed in the femurs by dual energy x-ray absorptiometry (DXA) using a Lunar DPX-IQ Densitometer (Lunar; software version 4.7e, GE Healthcare, Chalfont St. Giles, United Kingdom). The isolated bones were scanned with the anterior surface facing up. Regions of interest (14´16 pixels) were analyzed at the distal femoral metaphysis, both on the right and left femurs. The bone callus was also assessed over the entire callus size. Scanning reproducibility assessed by root mean square coefficient of variation was 4%.
Microarchitectural analysis
A high-resolution desktop micro-CT system (SkyScan 1174v2; Bruker microCT, Kontich, Belgium) was used to quantify the BMD and the three-dimensional microarchitecture parameters in the femur. The specimens were scanned at 50 kV and 800 mA, with the aid of a 0.5-mm-thick aluminum filter to optimize the contrast, a rotation step of 1°, three-frame averaging, and an isotropic resolution of 26.7 µm. Images of each specimen were reconstructed with dedicated software (NRecon version 1.6.3; Bruker, Belgium), providing axial cross-sections of the inner structures of the samples.
In the non-fractured bone, two regions of interest were delimited, one at the femoral distal metaphysis, which mainly contains trabecular bone, and another at the mid-diaphysis, which mainly contains cortical bone. The reconstruction of the metaphysis was selected manually, starting just proximally from the distal growth plate for an extension of 3.0 mm. The reconstruction of the diaphysis was defined by a 2.0-mm region starting 8.0 mm proximal from the distal growth plate. Cortical and trabecular bone were isolated using manually drawn contouring. CTAn software (version 2.2.1; Bruker, Belgium), was used to determine the optimal threshold from the image histograms and set to exclude soft tissue but to include poorly mineralized bone. The same threshold was used in all samples, but differed between trabecular and cortical bone. The thresholded image was used as a mask to measure the BMD of the trabecular bone structures. The trabecular architecture of the distal metaphysis was characterized by determining trabecular bone volume (BV), trabecular bone volume fraction (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), and connectivity density (Conn.D). Cortical architecture was assessed in the diaphysis and characterized by cortical volume (Ct.V and Ct.BV/TV) and cortical thickness (Ct.Th). All bone morphometric measurements and nomenclature were in accordance with the recommendations of the ASBMR (American Society for Bone and Mineral Research)1717. Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Muller R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res. 2010 Jul;25(7):1468-86. PMID: 20533309.. The callus volume was analyzed using CTAn software, with the entire callus selected as the region of interest.
Mechanical tests
Bone mechanical properties were determined by testing the femurs to fracture using a mechanical testing device (EMIC, PR, Brazil) equipped with a 500-N load cell. Isolated femurs were submitted to a shear test with the anterior surface facing up. The bones were placed on two metallic supports 7.0 mm apart. The load was vertically applied at a constant displacement rate of 0.5 mm/min at the distal femur for non-fractured bones and at the center of fractured femur callus. For all tests, the load-deflection curve was obtained in real time, and load at the fracture point was assessed (TESC software, version 13.4; EMIC, PR, Brazil) to represent bone or callus failure1818. Turner CH. Bone strength: current concepts. Ann N Y Acad Sci. 2006 Apr;1068:429-46. PMID: 16831941..
Statistical analysis
All data were expressed as means ± standard deviations. All statistical analyses were carried out with IBM SPSS Statistics version 20 (Armonk, NY, USA). Comparisons among the groups were statistically assessed by non-parametric Kruskal-Wallis test (ANOVA), followed by Dunn's post hoc analysis. The level of statistical significance was set at p<0.05.
Results
Deaths
Post-operative complications were common and resulted in death (n=14) or euthanasia (n=15) in twenty-nine rats prior to the end of the study. All 14 deaths occurred under anesthesia. Six deaths occurred during ovariectomy, and eight deaths occurred during fracture production and fixation. Of the 15 cases of euthanasia, 11 were due to extensive fracture fragmentation, and four were due to K-wire migration. Fifty-six rats (n=14 for each group; seven per subgroup) were included in the final analysis.
Ovariectomy confirmation
The mean uterus mass was 0.194±0.08g in ovariectomized rats and 0.990±0.28g in sham-operated animals (p<0.001).
Bone quality
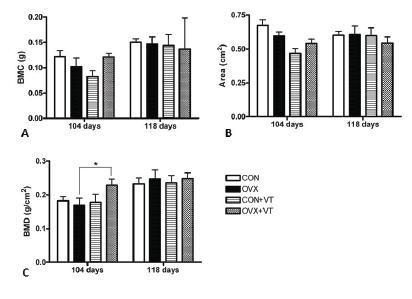
Compared with controls, the ovariectomized rats had significantly lower (p<0.05) bone density (Figure 2A) and bone content (Figure 2B) at the distal femur. Conversely, whole-body vibration therapy was partially effective at protecting against bone loss in the OVX rats, but not in controls. Vibration therapy increased BMD by 21% and BMC by 28% in the OVX rats (OVX+VT vs. OVX, both p<0.05).
- DXA measurements of bone mineral density (A) and bone mineral content (B) at the distal metaphysis both in the non-fractured (left) femur and fractured (right) femur.
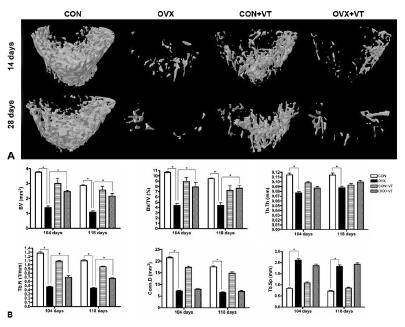
Figure 3A shows three-dimensional images of non-fractured trabecular bones. The OVX rats exhibited substantial changes to the trabecular microarchitecture of the distal metaphysis in the left non-fractured femurs. At 104 days after OVX, we observed a marked deterioration (Figure 3B) in the parameters of trabecular architecture assessed in the distal metaphysis (reduction of 63% in BV, 59% in BV/TV, 63% in Tb.N, 33% in Tb.Th, and 67% Conn.D, and increase of 150% in Tb.Sp). At 118 days after OVX, we observed a reduction of 63% in BV, 54% in BV/TV, 60% in Tb.N, 23% in Tb.Th, and 62% in Conn.D and an increase of 156% in Tb.Sp). Whole-body vibration treatment mitigated bone quality loss induced by OVX, when compared to the non-vibration-treated rats (OVX+VT vs. OVX). Fourteen days after fracture, vibration therapy increased BV by 77%, BV/TV by 81%, Tb.Th by 13%, Tb.N by 50%, and Conn.D by 10%, and decreased Tb.Sp by 12%. Twenty-eight days after fracture, vibration therapy increased BV by 101%, BV/TV by 75%, Tb.Th by 14%, Tb.N by 52%, and Conn.D by 7%.
- Trabecular bone microstructural images of distal femoral metaphysis showing a dramatic reduction of trabecular bone in OVX rats, which was ameliorated by vibration therapy (A). Quantitative analysis of trabecular bone microstructure confirmed the significant loss of trabecular volume and structure in OVX rats and the marked improvement achieved by vibration therapy (B). Asterisks indicate significant difference across groups (p<0.05).
Figure 4A shows three-dimensional images of non-fractured cortical bones. The OVX rats exhibited non-significant changes (p>0.05) in the cortical bone in left femurs, but to a lesser degree than the changes seen in trabecular bone. At 104 days after OVX, we observed a reduction of 13% in Ct.V, 14% in Ct.BV/TV, and 13% in Ct.Th (Figure 4B). At 118 days after OVX, no differences were observed between the CON and OVX groups. Whole-body vibration treatment mitigated cortical bone deterioration induced by OVX, when compared to the non-vibration-treated rats (OVX+VT vs. OVX). Fourteen days post-fracture, vibration therapy slightly increased Ct.V by 26%. At 28 days post-fracture, vibration therapy increased Ct.V by 25% and Ct.BV/TV by 10%.
- Three-dimensional images of cortical microstructure shows that cortical bone is not as affected by estrogen suppression as the trabecular bone, either by ovariectomy or vibration therapy (A). Quantitative analysis of cortical bone microstructure revealed a non-significant (p>0.05) impairment of cortical tissue due to OVX, and the improvement achieved by vibration therapy (B).
In comparison with controls, ovariectomized rats had significantly lower bone strength (p<0.05, 16%) at the distal femur (Table 1). Although the difference between the OVX and OVX+VT groups was not significant, whole-body vibration therapy improved bone strength by 13% in OVX rats. This difference was not observed in controls.
Changes in bone healing due to OVX and the osteogenic effect of vibration therapy for stimulating bone callus formation
In the early stage of bone healing (14 days post-fracture), OVX decreased bone callus content by 20% (p<0.05, Figure 5A), which is consistent with the 6% smaller callus area formed in these rats (p<0.05, Figure 5B). Whole-body vibration therapy increased the bone callus density in OVX rats by 20%, but not in controls (p<0.05, Figure 5C).
- Assessments of bone mineral content (A), callus area (B), and bone mineral density (C) at the bone callus. OVX decreases BMC and callus area, which are ameliorated by vibration therapy. Asterisks indicate significant difference (p<0.05).
Regarding the bone callus microstructure, OVX rats demonstrated a smaller callus volume at 14 and 28 days post-fracture (Figure 6). At 14 days post-fracture, callus volume was 56% smaller in OVX rats in comparison with controls (OVX vs. CON); however, vibration therapy increased the callus volume by 317% at 14 days and 128% at 28 days post-fracture (OVX+VT vs. OVX).
- Micro-CT illustrative images of the femurs, in sagittal (upper images) and axial planes (lower images) depicting the fracture site and callus formation. OVX induced decreased bone healing, with a smaller bone callus than in controls. Vibration therapy improved bone healing in OVX rats, augmenting bone callus and mineralization.
Discussion
This study confirmed the deleterious changes caused by estrogen deficiency on both trabecular and cortical bone tissue in the intact femur and also on the bone callus of fractured femur. We further demonstrated the positive effect of vibration therapy on both the bone tissue and the bone callus of ovariectomized rats.
Non-fractured bones displayed a 16% reduction of in bone mass, 4% reduction in bone strength, and extensive changes in bone microarchitecture, highlighting the 97% decrease in trabecular bone volume and 96% decrease in trabecular number and thickness, as well as the changes in cortical volume and thickness. Similarly, several authors have also demonstrated changes to bone tissue in both post-menopausal women11. Cauley JA. Estrogen and bone health in men and women. Steroids. 2015 Jul;99(Pt A):11-5. doi: 10.1016/j.steroids.2014.12.010.
https://doi.org/10.1016/j.steroids.2014....
,19 19. Khosla S, Oursler MJ, Monroe DG. Estrogen and the skeleton. Trends Endocrinol Metab. 2012 Nov;23(11):576-81. PMID: 22595550.and estrogen-depletion-induced osteoporotic animals99. Rubin CT, Sommerfeldt DW, Judex S, Qin Y. Inhibition of osteopenia by low magnitude, high-frequency mechanical stimuli. Drug Discov Today. 2001 Aug 15;6(16):848-58. PMID: 11495758.,2020. Jee WS, Yao W. Overview: animal models of osteopenia and osteoporosis. J Musculoskelet Neuronal Interact. 2001 Mar;1(3):193-207. PMID: 15758493.. These changes in bone tissue may lead to increased bone fracture risk, which in humans impairs quality of life and may increase mortality2121. Singer A, Exuzides A, Spangler L, O'Malley C, Colby C, Johnston K, Agodoa I, Baker J, Kagan R. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc. 2015 Jan;90(1):53-62. PMID: 25481833.
22. Zofkova I, Nemcikova P, Kuklik M. Polymorphisms associated with low bone mass and high risk of atraumatic fracture. Physiol Res. 2015 Mar 24. PMID: 25804099.-2323. Schilcher J, Koeppen V, Aspenberg P, Michaelsson K. Risk of atypical femoral fracture during and after bisphosphonate use. N Engl J Med. 2014 Sep 4;371(10):974-6. PMID: 25184886..
Antiresorptive agents are known to have the potential to delay or impair bone healing in postmenopausal women; however such complications have not been clinically evidenced in the scientific literature2424. Adami S, Libanati C, Boonen S, Cummings SR, Ho PR, Wang A, Siris E, Lane J, FREEDOM Fracture-Healing Writing Group, Adachi JD, Bhandari M, de Gregorio L, Gilchrist N, Lyritis G, Moller G, Palacios S, Pavelka K, Heirinch R, Roux C, Uebelhart D. Denosumab treatment in postmenopausal women with osteoporosis does not interfere with fracture-healing: results from the FREEDOM trial. J Bone Joint Surg Am. volume. 2012 Dec 5;94(23):2113-9. PMID: 23097066.. By contrast, estrogen depletion is known to delay or impair bone fracture healing in ovariectomized animals55. He YX, Zhang G, Pan XH, Liu Z, Zheng LZ, Chan CW, Lee KM, Cao YP, Li G, Wei L, Hung LK, Leung KS, Qin L. Impaired bone healing pattern in mice with ovariectomy-induced osteoporosis: a drill-hole defect model. Bone. 2011 Jun 1;48(6):1388-400. PMID: 21421090.,1111. Komrakova M, Sehmisch S, Tezval M, Ammon J, Lieberwirth P, Sauerhoff C, Trautmann L, Wicke M, Dullin C, Stuermer KM, Stuermer EK. Identification of a vibration regime favorable for bone healing and muscle in estrogen-deficient rats. Calcif Tissue Int. 2013 Jun;92(6):509-20. PMID: 23416966.
12. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877.-1313. Wehrle E, Liedert A, Heilmann A, Wehner T, Bindl R, Fischer L, Haffner-Luntzer M, Jacob F, Schinke T, Amling M, Ignatius A. The impact of low-magnitude high-frequency vibration on fracture healing is profoundly influenced by the oestrogen status in mice. Dis Model Mech. 2015 Jan;8(1):93-104. PMID: 25381012.. He et al. (2011), Komrakova et al.1111. Komrakova M, Sehmisch S, Tezval M, Ammon J, Lieberwirth P, Sauerhoff C, Trautmann L, Wicke M, Dullin C, Stuermer KM, Stuermer EK. Identification of a vibration regime favorable for bone healing and muscle in estrogen-deficient rats. Calcif Tissue Int. 2013 Jun;92(6):509-20. PMID: 23416966., Stuermer et al.1212. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877., and Wehrle et al.1313. Wehrle E, Liedert A, Heilmann A, Wehner T, Bindl R, Fischer L, Haffner-Luntzer M, Jacob F, Schinke T, Amling M, Ignatius A. The impact of low-magnitude high-frequency vibration on fracture healing is profoundly influenced by the oestrogen status in mice. Dis Model Mech. 2015 Jan;8(1):93-104. PMID: 25381012. have previously studied the effects of estrogen depletion on the bone healing of ovariectomized rodents. However, unlike these authors, who studied bone healing in osteotomy or drill-hole55. He YX, Zhang G, Pan XH, Liu Z, Zheng LZ, Chan CW, Lee KM, Cao YP, Li G, Wei L, Hung LK, Leung KS, Qin L. Impaired bone healing pattern in mice with ovariectomy-induced osteoporosis: a drill-hole defect model. Bone. 2011 Jun 1;48(6):1388-400. PMID: 21421090. models, we performed a closed femoral fracture, which better mimics the osteoporotic fractures in postmenopausal women. Leung et al.2525. Leung KS, Shi HF, Cheung WH, Qin L, Ng WK, Tam KF, Tang N. Low-magnitude high-frequency vibration accelerates callus formation, mineralization, and fracture healing in rats. J Orthop Res. 2009 Apr;27(4):458-65. PMID: 18924140. used the closed femoral shaft fracture model, but they did not study bone healing in an estrogen deficiency condition. Similarly to our study, Shi et al.2626. Shi HF, Cheung WH, Qin L, Leung AH, Leung KS. Low-magnitude high-frequency vibration treatment augments fracture healing in ovariectomy-induced osteoporotic bone. Bone. 2010 May;46(5):1299-305. PMID: 19961960. also assessed the effects of OVX on bone secondary healing in rats and concluded that bone healing was severely damaged by estrogen deficiency. Here, we demonstrated that OVX reduced bone callus content by 19% and callus area by 6%, as well as deleterious changes in callus mineralization and volume.
The effects of mechanical loading on bone tissue are difficult to be clinically investigated due to the limited assessment tools currently available. Therefore, the use of experimental animals in such studies is of great importance. Although large animals most closely resemble the human bone metabolism, they offer disadvantages such as greater logistical, financial and ethical considerations2727. Hatsushika D, Muneta T, Nakamura T, Horie M, Koga H, Nakagawa Y, Tsuji K, Hishikawa S, Kobayashi E, Sekiya I. Repetitive allogeneic intraarticular injections of synovial mesenchymal stem cells promote meniscus regeneration in a porcine massive meniscus defect model. Osteoarthritis cartilage. 2014;22(7):941-50. doi: 10.1016/j.joca.2014.04.028.
https://doi.org/10.1016/j.joca.2014.04.0...
. Taking all into consideration, the use of small rodents may be still considered the most suitable animal for bone researches with several advantages including inexpensiveness, they are easy to be housed in the animals facility, their use in research is widely accepted and public is accustomed to it2828. Turner AS. Animal models of osteoporosis--necessity and limitations. Eur Cells Mater. 2001 Jun 22;1:66-81. PMID: 14562261..
Considering the well-documented effect of mechanical stimuli on bone metabolism, several authors have documented the osteogenic effects of whole-body vibration on the bone tissue99. Rubin CT, Sommerfeldt DW, Judex S, Qin Y. Inhibition of osteopenia by low magnitude, high-frequency mechanical stimuli. Drug Discov Today. 2001 Aug 15;6(16):848-58. PMID: 11495758.,1010. Zhou Y, Guan X, Liu T, Wang X, Yu M, Yang G, Wang H. Whole body vibration improves osseointegration by up-regulating osteoblastic activity but down-regulating osteoblast-mediated osteoclastogenesis via ERK1/2 pathway. Bone. 2015 Feb;71:17-24. PMID: 25304090.,29 29. Nowak A, Lochynski D, Pawlak M, Romanowski W, Krutki P. High-magnitude whole-body vibration effects on bone resorption in adult rats. Aviat Space Environ Med. 2014 May;85(5):518-21. PMID: 24834565.and on the bone callus of rats1111. Komrakova M, Sehmisch S, Tezval M, Ammon J, Lieberwirth P, Sauerhoff C, Trautmann L, Wicke M, Dullin C, Stuermer KM, Stuermer EK. Identification of a vibration regime favorable for bone healing and muscle in estrogen-deficient rats. Calcif Tissue Int. 2013 Jun;92(6):509-20. PMID: 23416966.
12. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877.-1313. Wehrle E, Liedert A, Heilmann A, Wehner T, Bindl R, Fischer L, Haffner-Luntzer M, Jacob F, Schinke T, Amling M, Ignatius A. The impact of low-magnitude high-frequency vibration on fracture healing is profoundly influenced by the oestrogen status in mice. Dis Model Mech. 2015 Jan;8(1):93-104. PMID: 25381012.,2525. Leung KS, Shi HF, Cheung WH, Qin L, Ng WK, Tam KF, Tang N. Low-magnitude high-frequency vibration accelerates callus formation, mineralization, and fracture healing in rats. J Orthop Res. 2009 Apr;27(4):458-65. PMID: 18924140.,2626. Shi HF, Cheung WH, Qin L, Leung AH, Leung KS. Low-magnitude high-frequency vibration treatment augments fracture healing in ovariectomy-induced osteoporotic bone. Bone. 2010 May;46(5):1299-305. PMID: 19961960.. Nevertheless, differing loading procedures and parameters of vibration (frequency, acceleration, and amplitude) have been used in animal and human studies, making the comparison across experiments difficult3030. de Oliveira ML, Bergamaschi CT, Silva OL, Nonaka KO, Wang CC, Carvalho AB, Jorgetti V, Campos RR, Lazaretti-Castro M. Mechanical vibration preserves bone structure in rats treated with glucocorticoids. Bone. 2010 Jun;46(6):1516-21. PMID: 20152951.,3131. Edwards JH, Reilly GC. Vibration stimuli and the differentiation of musculoskeletal progenitor cells: Review of results in vitro and in vivo. World J Stem Cells. 2015 Apr 26;7(3):568-82. PMID: 25914764.. Although the ideal frequency or intensity of stimuli has yet to be established, frequencies ranging from 15 to 90 Hz are known to be strongly anabolic3030. de Oliveira ML, Bergamaschi CT, Silva OL, Nonaka KO, Wang CC, Carvalho AB, Jorgetti V, Campos RR, Lazaretti-Castro M. Mechanical vibration preserves bone structure in rats treated with glucocorticoids. Bone. 2010 Jun;46(6):1516-21. PMID: 20152951., whereas higher frequencies appear to be more effective3131. Edwards JH, Reilly GC. Vibration stimuli and the differentiation of musculoskeletal progenitor cells: Review of results in vitro and in vivo. World J Stem Cells. 2015 Apr 26;7(3):568-82. PMID: 25914764..
Nowak et al.2929. Nowak A, Lochynski D, Pawlak M, Romanowski W, Krutki P. High-magnitude whole-body vibration effects on bone resorption in adult rats. Aviat Space Environ Med. 2014 May;85(5):518-21. PMID: 24834565. studied the effects of whole-body vibration therapy on non-fractured bone tissue in adult rats and found a decrease in the level of bone resorption markers, but no changes to bone formation markers or bone mass. In the present study, we did not observe substantive changes in the intact bone tissue of control rats. By contrast, we demonstrated that whole-body vibration therapy was very effective at enhancing the intact bone quality of osteoporotic rats. This finding was also reported by previous authors, who consistently showed that vibration exerted a greater effect on the osteoporotic bone of rats than on control bone1212. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877.,3232. Sehmisch S, Galal R, Kolios L, Tezval M, Dullin C, Zimmer S, Stuermer KM, Stuermer EK. Effects of low-magnitude, high-frequency mechanical stimulation in the rat osteopenia model. Osteoporos Int. 2009 Dec;20(12):1999-2008. PMID: 19283328.. We found that vibration therapy increased non-fractured bone density by 27%, bone strength by 13%, trabecular volume almost two fold, trabecular thickness by 33%, and cortical volume by 24%.
Stuermer et al.1212. Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877. studied the effects of whole-body vibration therapy on post-osteotomy bone healing in ovariectomized rats and found significant increases in the cortical density and width of vibration-treated OVX rats. Furthermore, expression of the bone formation marker osteocalcin was higher in vibration-treated OVX rats than in the non-vibration-treated OVX group. Shi et al.2626. Shi HF, Cheung WH, Qin L, Leung AH, Leung KS. Low-magnitude high-frequency vibration treatment augments fracture healing in ovariectomy-induced osteoporotic bone. Bone. 2010 May;46(5):1299-305. PMID: 19961960. also found osteogenic effects of whole-body vibration therapy to enhance bone healing in ovariectomized rats. The authors, however, employed the vibration therapy five times per week, which may not realistically reflect a regular rehabilitation program in humans. Our findings demonstrated that vibration therapy three times per week increased bone mineral density and content by 35% and 19%, respectively, and augmented bone callus volume by 317%.
Vibration therapy is a low-impact exercise, in which the mechanical signals are sensed by osteocytes as mechanical loading, thus stimulating bone formation and suppressing bone resorption, as explained by Wolff's Law99. Rubin CT, Sommerfeldt DW, Judex S, Qin Y. Inhibition of osteopenia by low magnitude, high-frequency mechanical stimuli. Drug Discov Today. 2001 Aug 15;6(16):848-58. PMID: 11495758., which states that a bone becomes stronger when it is submitted to mechanical loading. Many cells responsible for producing and maintaining the tissues in the musculoskeletal system come from a lineage of progenitor cells within the bone marrow, the stem cells, which are capable of giving rise to several lineages including the osteogenic cells involved in bone formation. It has been previously established that mechanical stimuli can promote the differentiation of stem cells into osteogenic lineages3131. Edwards JH, Reilly GC. Vibration stimuli and the differentiation of musculoskeletal progenitor cells: Review of results in vitro and in vivo. World J Stem Cells. 2015 Apr 26;7(3):568-82. PMID: 25914764. through a mechanotransduction mechanism. Based on our results, we believe that whole-body vibration therapy may have the potential effect of increasing bone quality and callus formation in osteoporotic bones and may form part of rehabilitative programs to enhance bone healing.
Conclusions
Ovariectomized rats exhibited a substantial loss of bone mass and severe impairment in bone microarchitecture, both to intact bone tissue and the bone fracture callus. Whole-body vibration therapy exerted an important role in ameliorating bone mass and bone and improving trabecular bone microstructure in the estrogen-deficient rats. Moreover, vibration therapy improved bone callus formation in ovariectomized rats, as indicated by the enhancement of bone callus density, strength, and volume.
References
-
1Cauley JA. Estrogen and bone health in men and women. Steroids. 2015 Jul;99(Pt A):11-5. doi: 10.1016/j.steroids.2014.12.010.
» https://doi.org/10.1016/j.steroids.2014.12.010 -
2Yuan Q, Zhang G, Wu J, Xing Y, Sun Y, Tian W. Clinical evaluation of the polymethylmethacrylate-augmented thoracic and lumbar pedicle screw fixation guided by the three-dimensional navigation for the osteoporosis patients. Eur Spine J. 2015;24(5):1043-50. doi: 10.1007/s00586-013-3131-8.
» https://doi.org/10.1007/s00586-013-3131-8 -
3Einhorn TA. The cell and molecular biology of fracture healing. Clin Orthop Relat Res. 1998 Oct;(355 Suppl):S7-21. PMID: 9917622.
-
4Einhorn TA. The science of fracture healing. J Orthop Trauma. 2005 Nov-Dec;19(10 Suppl):S4-6. PMID: 16479221.
-
5He YX, Zhang G, Pan XH, Liu Z, Zheng LZ, Chan CW, Lee KM, Cao YP, Li G, Wei L, Hung LK, Leung KS, Qin L. Impaired bone healing pattern in mice with ovariectomy-induced osteoporosis: a drill-hole defect model. Bone. 2011 Jun 1;48(6):1388-400. PMID: 21421090.
-
6Yamaji T, Ando K, Wolf S, Augat P, Claes L. The effect of micromovement on callus formation. J Orthop Sci. 2001;6(6):571-5. PMID: 11793181.
-
7Gomez-Cabello A, Gonzalez-Aguero A, Morales S, Ara I, Casajus JA, Vicente-Rodriguez G. Effects of a short-term whole body vibration intervention on bone mass and structure in elderly people. J Sci Med Sport. 2014 Mar;17(2):160-4. PMID: 23711620.
-
8Liphardt AM, Schipilow J, Hanley DA, Boyd SK. Bone quality in osteopenic postmenopausal women is not improved after 12 months of whole-body vibration training. Osteoporos Int. 2015 Mar;26(3):911-20. PMID: 25567775.
-
9Rubin CT, Sommerfeldt DW, Judex S, Qin Y. Inhibition of osteopenia by low magnitude, high-frequency mechanical stimuli. Drug Discov Today. 2001 Aug 15;6(16):848-58. PMID: 11495758.
-
10Zhou Y, Guan X, Liu T, Wang X, Yu M, Yang G, Wang H. Whole body vibration improves osseointegration by up-regulating osteoblastic activity but down-regulating osteoblast-mediated osteoclastogenesis via ERK1/2 pathway. Bone. 2015 Feb;71:17-24. PMID: 25304090.
-
11Komrakova M, Sehmisch S, Tezval M, Ammon J, Lieberwirth P, Sauerhoff C, Trautmann L, Wicke M, Dullin C, Stuermer KM, Stuermer EK. Identification of a vibration regime favorable for bone healing and muscle in estrogen-deficient rats. Calcif Tissue Int. 2013 Jun;92(6):509-20. PMID: 23416966.
-
12Stuermer EK, Komrakova M, Werner C, Wicke M, Kolios L, Sehmisch S, Tezval M, Utesch C, Mangal O, Zimmer S, Dullin C, Stuermer KM. Musculoskeletal response to whole-body vibration during fracture healing in intact and ovariectomized rats. Calcif Tissue Int. 2010 Aug;87(2):168-80. PMID: 20532877.
-
13Wehrle E, Liedert A, Heilmann A, Wehner T, Bindl R, Fischer L, Haffner-Luntzer M, Jacob F, Schinke T, Amling M, Ignatius A. The impact of low-magnitude high-frequency vibration on fracture healing is profoundly influenced by the oestrogen status in mice. Dis Model Mech. 2015 Jan;8(1):93-104. PMID: 25381012.
-
14Simoes PA, Zamarioli A, Bloes P, Mazzocato FC, Pereira LH, Volpon JB, Shimano AC. Effect of treadmill exercise on lumbar vertebrae in ovariectomized rats: anthropometrical and mechanical analyses. Acta Bioeng Biomech. 2008;10(2):39-41. PMID: 19031996.
-
15Falcai MJ, Zamarioli A, Okubo R, de Paula FJ, Volpon JB. The osteogenic effects of swimming, jumping, and vibration on the protection of bone quality from disuse bone loss. Scand J Med Sci Sports. 2014 Apr 30. PMID: 24779886.
-
16Zamarioli A, Battaglino RA, Morse LR, Sudhakar S, Maranho DA, Okubo R, Volpon JB, Shimano AC. Standing frame and electrical stimulation therapies partially preserve bone strength in a rodent model of acute spinal cord injury. Am J Phys Med Rehabil. 2013 May;92(5):402-10. PMID: 23478455.
-
17Bouxsein ML, Boyd SK, Christiansen BA, Guldberg RE, Jepsen KJ, Muller R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J Bone Miner Res. 2010 Jul;25(7):1468-86. PMID: 20533309.
-
18Turner CH. Bone strength: current concepts. Ann N Y Acad Sci. 2006 Apr;1068:429-46. PMID: 16831941.
-
19Khosla S, Oursler MJ, Monroe DG. Estrogen and the skeleton. Trends Endocrinol Metab. 2012 Nov;23(11):576-81. PMID: 22595550.
-
20Jee WS, Yao W. Overview: animal models of osteopenia and osteoporosis. J Musculoskelet Neuronal Interact. 2001 Mar;1(3):193-207. PMID: 15758493.
-
21Singer A, Exuzides A, Spangler L, O'Malley C, Colby C, Johnston K, Agodoa I, Baker J, Kagan R. Burden of illness for osteoporotic fractures compared with other serious diseases among postmenopausal women in the United States. Mayo Clin Proc. 2015 Jan;90(1):53-62. PMID: 25481833.
-
22Zofkova I, Nemcikova P, Kuklik M. Polymorphisms associated with low bone mass and high risk of atraumatic fracture. Physiol Res. 2015 Mar 24. PMID: 25804099.
-
23Schilcher J, Koeppen V, Aspenberg P, Michaelsson K. Risk of atypical femoral fracture during and after bisphosphonate use. N Engl J Med. 2014 Sep 4;371(10):974-6. PMID: 25184886.
-
24Adami S, Libanati C, Boonen S, Cummings SR, Ho PR, Wang A, Siris E, Lane J, FREEDOM Fracture-Healing Writing Group, Adachi JD, Bhandari M, de Gregorio L, Gilchrist N, Lyritis G, Moller G, Palacios S, Pavelka K, Heirinch R, Roux C, Uebelhart D. Denosumab treatment in postmenopausal women with osteoporosis does not interfere with fracture-healing: results from the FREEDOM trial. J Bone Joint Surg Am. volume. 2012 Dec 5;94(23):2113-9. PMID: 23097066.
-
25Leung KS, Shi HF, Cheung WH, Qin L, Ng WK, Tam KF, Tang N. Low-magnitude high-frequency vibration accelerates callus formation, mineralization, and fracture healing in rats. J Orthop Res. 2009 Apr;27(4):458-65. PMID: 18924140.
-
26Shi HF, Cheung WH, Qin L, Leung AH, Leung KS. Low-magnitude high-frequency vibration treatment augments fracture healing in ovariectomy-induced osteoporotic bone. Bone. 2010 May;46(5):1299-305. PMID: 19961960.
-
27Hatsushika D, Muneta T, Nakamura T, Horie M, Koga H, Nakagawa Y, Tsuji K, Hishikawa S, Kobayashi E, Sekiya I. Repetitive allogeneic intraarticular injections of synovial mesenchymal stem cells promote meniscus regeneration in a porcine massive meniscus defect model. Osteoarthritis cartilage. 2014;22(7):941-50. doi: 10.1016/j.joca.2014.04.028.
» https://doi.org/10.1016/j.joca.2014.04.028 -
28Turner AS. Animal models of osteoporosis--necessity and limitations. Eur Cells Mater. 2001 Jun 22;1:66-81. PMID: 14562261.
-
29Nowak A, Lochynski D, Pawlak M, Romanowski W, Krutki P. High-magnitude whole-body vibration effects on bone resorption in adult rats. Aviat Space Environ Med. 2014 May;85(5):518-21. PMID: 24834565.
-
30de Oliveira ML, Bergamaschi CT, Silva OL, Nonaka KO, Wang CC, Carvalho AB, Jorgetti V, Campos RR, Lazaretti-Castro M. Mechanical vibration preserves bone structure in rats treated with glucocorticoids. Bone. 2010 Jun;46(6):1516-21. PMID: 20152951.
-
31Edwards JH, Reilly GC. Vibration stimuli and the differentiation of musculoskeletal progenitor cells: Review of results in vitro and in vivo. World J Stem Cells. 2015 Apr 26;7(3):568-82. PMID: 25914764.
-
32Sehmisch S, Galal R, Kolios L, Tezval M, Dullin C, Zimmer S, Stuermer KM, Stuermer EK. Effects of low-magnitude, high-frequency mechanical stimulation in the rat osteopenia model. Osteoporos Int. 2009 Dec;20(12):1999-2008. PMID: 19283328.
-
Financial source: CAPES
-
1Research performed at Laboratory of Bioengineering, Department of Biomechanics, Medicine and Rehabilitation, Faculty of Medicine of Ribeirao Preto, Universidade de Sao Paulo, Ribeirao Preto-SP, Brazil. Part of Master degree thesis, Postgraduate Program in Health Sciences Applied to the Locomotor System. Tutor: José Batista Volpon.
Publication Dates
-
Publication in this collection
Nov 2015
History
-
Received
07 July 2015 -
Reviewed
09 Sept 2015 -
Accepted
05 Oct 2015