ABSTRACT
PURPOSE
: To investigate the effects of thiamine pyrophosphate (TPP) against desflurane induced hepatotoxicity.
METHODS
: Thirty experimental animals were divided into groups as healthy (HG), desflurane control (DCG) , TPP and desflurane group (TDG). 20 mg/kg TPP was injected to intraperitoneally TDG. After one hour of TPP administration, desflurane was applied for two hours. After 24 hours, liver tissues of the animals killed with decapitation were removed. The oxidant/antioxidant levels and ALT, AST and LDH activities were measured. The histopathological examinations were performed in the liver tissues for all rats.
RESULTS
: Notwithstanding the levels of oxidants and liver enzymes were significantly increased (p<0.0001), antioxidant levels were significantly decreased in DCG (p<0.0001). On contrary to the antioxidant parameters were increased (p<0.05) the oxidant parameters and liver enzymes were decreased in TDG (p<0.0001). Whereas multiple prominent, congestion, hemorrhage and dilatation were observed in sinusoids and lymphocyte-rich inflammation results in the centrilobular and portal areas of liver tissue in DCG, these findings were observed less frequently in TDG.
CONCLUSİON
: Thiamine pyrophosphate prevented liver oxidative damage induced with desflurane and may be useful in prophylaxis of desflurane induced hepatotoxicity.
Key words:
Thiamine Pyrophosphate; Oxidative Stress; Rats
Introductıon
The desflurane is a halogenated ether anesthetic which has been synthesized as a result of the studies conducted by Terrel et al .11. Pandit SK, Green CR. General anesthetic techniques. Int Anesthesiol Clin. 1994;32(3):55-79. PMID: 7960175. in order to find an ideal inhalation agent. Desflurane is known to have important advantages such as ability to provide anaesthetic depth more easily and quicker recovery. Notwithstanding these important advantages, it has also serious disadvantages that may negatively affect human health; recent studies demonstrated that hepatotoxic effect emerges due to the use of halogenated anesthetics22. Safari S, Motavaf M, Seyed Siamdoust SA, Alavian SM. Hepatotoxicity of halogenated inhalational anesthetics. Iran Red Crescent Med J. 2014;16(9):e20153. doi: 10.5812/ircmj.20153.
https://doi.org/10.5812/ircmj.20153....
. Martin et al .3 reposted desflurane to have serious hepatotixic effects33. Martin JL, Plevak DJ, Flannery KD, Charlton M, Poterucha JJ, Humphreys CE, Derfus G, Pohl LR. Hepatotoxicity after desflurane anesthesia. Anesthesiology. 1995;83(5):1125-9. PMID: 7486167.. Even cases of hepatotoxicity related to desflurane and resulted in death are found in the literature33. Martin JL, Plevak DJ, Flannery KD, Charlton M, Poterucha JJ, Humphreys CE, Derfus G, Pohl LR. Hepatotoxicity after desflurane anesthesia. Anesthesiology. 1995;83(5):1125-9. PMID: 7486167.
55. Chin MW, Njoku DB, MacQuillan G, Cheng WS, Kontorinis N. Desflurane-induced acute liver failure. Med J Aust. 2008;189(5):293-4. PMID: 18759731.. In an experimental study by Arslan et al .77. Mustafa A, Berrin I, Yusuf U, Nurlu N, Zerrin O, Metin CF, Isin G, Bekir DC, Nedim C, Mustafa K. Effects of sevoflurane and desflurane on oxidant/antioxidant status of young versus old rat liver tissues. J Anim Vet Adv. 2008;7(11):1345-51. doi: javaa.2008.1345.1351., desflurane was found to produce hepatic damage in aged female rats66. Arslan M, Ozkose Z, Akyol G, Barit G. The age- and gender-dependent effects of desflurane and sevoflurane on rat liver. Exp Toxicol Pathol. 2010;62(1):35-43. doi: 10.1016/j.etp.2008.12.011.
https://doi.org/10.1016/j.etp.2008.12.01...
. Again desflurane was argued to create oxidative stress in the liver of aged male rats by increasing oxidants and decreasing antioxidants77. Mustafa A, Berrin I, Yusuf U, Nurlu N, Zerrin O, Metin CF, Isin G, Bekir DC, Nedim C, Mustafa K. Effects of sevoflurane and desflurane on oxidant/antioxidant status of young versus old rat liver tissues. J Anim Vet Adv. 2008;7(11):1345-51. doi: javaa.2008.1345.1351.. As is known, hepatocellular intergrity is indicated by the plasma activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactic dehydrogenase (LDH) and gamma glutamyl transferase (GGT). Determination of transaminase activities is one of the most commonly used tests in the diagnosis of hepatic diseases. In a case report presenting hepatotoxicity of desflurane, Martin et al .3 demonstrated that, ALT, AST, alcaline phosphatase total bilirubine, direct bilirubine and gamma glutamyl transferase were rapidly elevated.This information indicates that, oxidative stress is an important factor in hepatotoxicity caused by desflurane and antioxidant therapy may be beneficial. Thiamine pyrophosphate (TPP) that we tested against hepatotixicity of desflurane is an active metabolite of thiamine (TM). In their study, Yilmaz et al .88. Yilmaz I, Demiryilmaz I, Turan MI, Cetin N, Gul MA, Suleyman H. The effects of thiamine and thiamine pyrophosphate on alcohol-induced hepatic damage biomarkers in rats. Eur Rev Med Pharmacol Sci. 2015;19(4):664-70. PMID: 25753885. stated that TPP prevented alcohol-induced oxidative stress in the rat liver. Kisaoglu et al .99. Kisaoglu A, Ozogul B, Turan MI, Yilmaz I, Demiryilmaz I, Atamanalp SS, Bakan E, Suleyman H. Damage induced by paracetamol compared with N-acetylcysteine. J Chin Med Assoc. 2014;77(9):463-8. doi: 10.1016/j.jcma.2014.01.011.
https://doi.org/10.1016/j.jcma.2014.01.0...
argued that, TPP protected the liver tissue against oxidative damage by inhibiting increase of the oxidant and decrease of the antioxidant parameters in the liver tissue related to paracetamol. The significant antioxidant feature of TPP suggests that it may protect the liver tissue against desflurane induced damages. No information was found in the literature screening about the protective effects of TTP against hepatotoxicity with desflurane in rats. Therefore, objective of this study was to investigate biochemically and histopathologically effects of TTP on the desflurane induced hepatotoxicity in rats.
Methods
The animal experiments were performed in accordance with the National Guidelines for the Use and Care of Laboratory Animals and were approved by the Local Animal Ethics Committee of Recep Tayyip Erdogan University, Rize, Turkey (Ethics Committee Number: 2015/28, Dated: 27.04.2015). The experimental animals were obtained from the Recep Tayyip Erdoğan University, Medical Experimental Research and Application Center.
A total of 30 male albino Wistar rats weighed between 235 or 245g and aged between four or five months, were randomly selected for the experiment. The animals were housed and fed in the pharmacology laboratory at normal room temperature (220C) for one week before the experiment in order to provide adaptation to their environment.
Chemical substances
Of the chemical substances used for the experiments, thiopental sodium was provided by IE
Ulagay-Turkey. Thiamine and thiamine pyrophosphate were obtained from Biopharma-
Russia. Desflurane were obtained from Eczacibasi, Istanbul, Turkey.
Experimental groups
The experimental animals were composed from three group as healthy (HG; n:10), desflurane control (DCG; n:10) and TPP + desflurane administration (TDG; n:10) rat groups.
Experimental procedure
The rats had free access to food and water until 2 h before anaesthesia procedure and were kept in a room at 20-24°C with a rhythm of 12 h light and 12 h darkness. The anesthetic gas vaporizer was calibrated before beginning of the study. In performing the experiment, TDG group was intraperitoneally (ip) injected 20 mg/kg TPP1010. Turan MI, Cayir A, Cetin N, Suleyman H, Siltelioglu Turan I, Tan H. An investigation of the effect of thiamine pyrophosphate on cisplatin-induced oxidative stress and DNA damage in rat brain tissue compared with thiamine: thiamine and thiamine pyrophosphate effects on cisplatin neurotoxicity. Hum Exp Toxicol. 2014;33(1):14-21. doi: 10.1177/0960327113485251.
https://doi.org/10.1177/0960327113485251...
1111. Yapca OE, Turan MI, Borekci B, Akcay F, Suleyman H. Bilateral ovarian ischemia/reperfusion injury and treatment options in rats with an induced model of diabetes. Iran J Basic Med Sci. 2014;17(4):294-302. PMID: 24904723.. The HG and DCG groups were administered distilled water through the same route. After one hour of TPP and distilled water administration, anesthesia procedure was applied on the TDG and DCG rats in transparent plastic box with sizes of 40x40x70 cm.The container that allowed observation of the rats, were connected to the anesthesia machine with half open inlet using fixed hoses. For the maintenance of the minimum alveolar concentration (MAC) 1, Desflurane (Suprane(r)) was administered at 6% volume inspiratory concentration, in a flowrate of 6 L min-1 100% O2 for 2 hours. Anesthetic gas was released in the cage containing 100% oxygen. The healthy group was not subjected to any procedures The blood samples were collected 24 hours after anesthesia. Then the liver tissues of the animals killed with decapitation were removed. ALT, AST and LDH activities were measured in the blood samples collected. In addition, oxidant/antioxidant levels were studied in the liver tissue and all livers tissues of histopathological examinations were performed.
Biochemical analysis
Determination of malondialdehide (MDA)
The kit uses a double-antibody sandwich enzyme-linked immunosorbent assay (Elisa) to assay the level of rat MDA in samples (Elisa kit, Eastbiopharm Co. Ltd, China). Add malondialchehyche (MDA) to monoclonal antibody enzyme well which is pre-coated with rat malondialchehyche (MDA) monoclonal antibody, incubation; then, add malondialchehyche (MDA) antibodies labeled with biotin, and combined with streptavidin-hrp to form immune complex; then carry out incubation and washing again to remove the uncombined enzyme. Then add chromogen solution a, b, the color of the liquid changes into the blue, and at the effect of acid, the color finally becomes yellow. The chroma of color and the concentration of the rat substance malondialchehyche (MDA) of sample were positively correlated.
Determination of myeloperoxidase (MPO) activity
Potassium phosphate buffer of pH=6 which contained 0.5% HDTMAB (0.5% hexadecyl trimethyl ammonium bromide) was prepared in order to determine MPO in the liver tissue homogenates. The mixture obtained then was centrifuged at 10,000 rpm for 15 minutes at +4 °C. Supernatant part was used as the analysis sample. Oxidation reaction with MPO mediated H2O2 which included 4-amino antipyrin/ phenol solution as substrate was used in determination of the activity of MPO enzyme1212. Bradley PP, Priebat DA, Christensen RD, Rothstein G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol. 1982;78(3):206-9. PMID: 6276474..
Nitric oxide (NO) analysis
Tissue NO levels were measured as total nitrite + nitrate levels with the use of the Griess reagent as previously described. The Griess reagent consists of sulfanilamide and N -(1-napthyl)-ethylenediamine. The method is based on a two-step process. The first step is the conversion of nitrate into nitrite using a nitrate reductase. The second step is the addition of the Griess reagent, which converts nitrite into a deep purple azo compound; photometric measurement of absorbance at 540 nm is due to the fact that this azo chromophore accurately determines nitrite concentration. NO levels were expressed as μmol/g protein1313. Moshage H, Kok B, Huizenga JR, Jansen PL. Nitrite and nitrate determinations in plasma: a critical evaluation. Clin Chem. 1995;41(6):892-6. PMID: 7768008..
Determination of total glutathione (tGSH) levels
This kit uses enzyme-linked immune sorbent assay (Elisa) based on the biotin double antibody sandwich technology to assay the rat total glutathione (Elisa Kit, Eastbiopharm Co. Ltd, China). Add total glutathione to the wells, which are pre-coated with total glutathione monoclonal antibody and then incubate. After that, add anti glutathione antibodies labeled with biotin to unite with streptavidin-hrp, which forms immune complex. Remove unbound enzymes after incubation and washing. Add substrate a and b. Then the solution will turn blue and change into yellow with the effect of acid. The shades of solution and the concentration of rat total glutathione are positively correlated.
Determination of glutathione peroxidase (GSHPX) activity
GSHPX was determined by monitoring NADP+ production at 340 nm and 25 0C. The assay mixture contained 10 mm magnesium chloride, 0.2 mm NADPH, 0.1 u/ml gr and 0.1 m GSH in 100 mm tris-hydrochloride buffer solution at ph 8.0. Assays were carried out in triplicate and the activities were followed up for 60 s. One unit of activity (u) is defined as the amount of enzyme required to reduce 1 μmol/min of NADPH under the assay conditions. The activity of Gpx was calculated using the extinction coefficient of 6.22 mm-1 cm-1.1414. Beutler E. General aspects of erythrocyte physiology and biochemistry. Exp Eye Res. 1971;11(3):261-3. PMID: 5166266..
Determination of glutathione reductase (GSHRd) activity
GSHRD enzyme activity was measured by Beutler's method. One enzyme unit is defined as the oxidation of 1 mmol NADPH per min under the assay condition (25 0C, ph 8.0)1414. Beutler E. General aspects of erythrocyte physiology and biochemistry. Exp Eye Res. 1971;11(3):261-3. PMID: 5166266..
Determination of cathalase (CAT) activity
The CAT activity was measured by the Aebi method. In this method, 20 ml enzyme solution was added to the 1 ml 10mm H2O2 in 20 mm potassium phosphate buffer (ph 7.0) and incubated at 25 0C for 1 min. Initial reaction rate was measured from the decrease in absorbance at 240 nm1515. Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121-6. PMID: 6727660..
Determination of superoxide dismutase (SOD) activity
SOD activity was based on the generation of superoxide radicals produced by xanthine and xanthine oxidase, which reacts with nitro blue tetrazolium to form formazan dye. SOD activity was then measured at 560 nm by the degree of inhibition of this reaction and is expressed as EU/ml1616. Sun Y, Oberley LW, Li Y. A simple method for clinical assay of superoxide dismutase.. Clin Chem 1988;34(3):497-500. PMID: 3349599..
Alanine transaminase (ALT), aspartate aminotransferase (AST) analysis
Venous blood samples were collected into tubes without anticoagulant. Serum was separated by centrifugation after clotting and stored at -80ºC until assayed. Serum AST and ALT activities were measured spectrophotometrically as liver function tests, and LDH activity, as a marker of tissue injury, using a Cobas 8000 (Roche) autoanalyzer with commercially available kits (Roche Diagnostics, GmBH, Mannheim, Germany).
ALT analysis
Quantitative determination of the serum ALT (Alanine Aminotransferase) was studied by spectrophotometric method using Roche cobas 8000 autoanalyser. According to the International Federation of Clinical Chemistry (IFCC), pyridoxal 5'-phosphate method catalizes the reaction between 3,4 ALT L-alanine and 2 - oxoglutarate. The pyruvate formed is reduced by NADH with a reaction catalized by lactate dehydrogenase (LDH) in which L-lactate and NAD+ are formed. Pyridoxal phosphate functions as a coenzyme in the amino transfer reaction. It enables enzyme activation to be complete. L-Alanine + 2- oxoglutarate → (ALT) pyruvate + L-glutamate. Pyruvate + NADH + H+ → (LDH) L-lactate + NAD +. Rate of the NADH oxidization is proportional to the ALT activity.
AST analysis
Quantitative determination of the serum AST (Aspartate Aminotransferase) was studied by spectrophotometric method using Roche cobas 8000 autoanalyser. According to the International Federation of Clinical Chemistry (IFCC), pyridoxal 5'-phosphate method catalizes the transfer of an amino acid group between L-aspartate and 2- oxoglutarate in order to form 3,4 AST oxaloacetate and L-glutamate in the sample. Than oxaloacetate reacts with NADH in the presence of malate dehydrogenase (MDH) in order to provide formation of NAD+. Pyridoxal phosphate functions as a coenzyme in the amino transfer reaction. L-Aspartate + 2- oxoglutarate → (AST) oxaloacetate + L-glutamate. Oxaloacetate + NADH + H+→ (MDH) L-malate + NAD+. Rate of the NADH oxidization is proportional to the AST activity.
Lactate dehydrogenase (LDH) analysis
Quantitative determination of serum LDH (Lactate Dehydrogenase (P-L)) was studied with spectrophotometric method in Roche cobas 8000 autoanalyzer. This is a standard method optimized according to Deutsche Gesellschaft für Klinische Chemie (DGKC). LDH catalizes the reaction between pyruvate and NADH for formation of NAD+ and L-lactate.
Pyruvate + NADH + H+ →yr(LDH) L-laktat + NAD+. Initial rate of NADH oxidation is proportionate to the catalytical LDH activity. Reduction in the absorbance was determined by measurement at 340 nm.
Histopathological analysis
The liver tissues removed from rats were fixed in 10% buffered formalin solution. Sections of 5 µm thickness were obtained from the paraffin blocks that were obtained following routine tissue processes and stained with hematoxylin and eosin (H&E). All the sections were coded and examined under light microscope (Olympus CX 51, Tokyo, Japan) by the same pathologist who was blind to the applied protocol.
Statistical analysis
The results obtained from the experiments were expressed as "mean ± standard error of the mean" (x ± SEM). The statistical analysis was conducted using analysis of variance (ANOVA) followed by post-hoc comparisions (Tukey-B). All the statistical processes were carried out with "IBM SPSS Statistics Version 20" statistical software and p< 0.05 values were considered statistically significant.
Results
Biochemical results
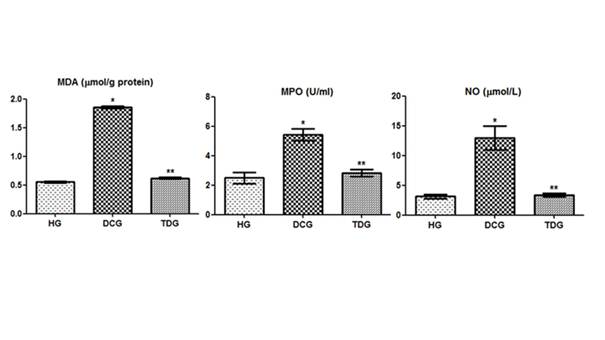
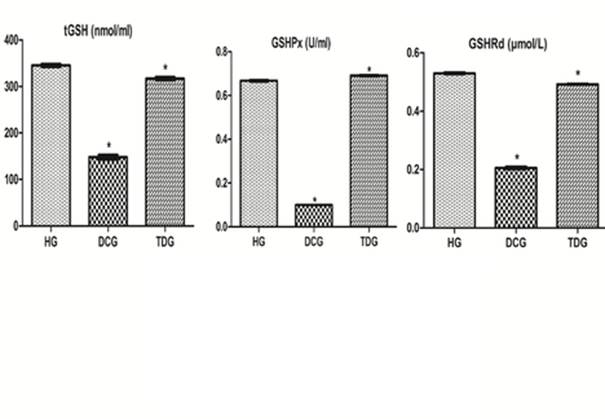
As it is seen in Figure 1, levels of MDA, MPO and NO were respectively measured as 0.55 ± 0.04 nmol/ml, 2.5 ± 0.37 u/ml and 3.1 ± 0.34 µmol/l in the liver tissue of healthy animals. However, these values were increased as 1.85 ± 0.05 nmol/ml, 5.4 ± 0.39 u/ml and 13 ± 2 µmol/l in the animals exposed to desflurane inhalation for 2 hours (p<0.0001). In TDG these values were respectively found as 0.63 ± 0,03 nmol/ml, 2.9 ± 0,24 u/ml and 3.38 ± 0.32 µmol/l. Therefore when we compared HG to DCG and TDG the difference between HG and TDG was found statistically insignificant (p>0.05). Despite of the fact that the levels of tGSH, GSHPx and GSHRd were respectively found as 345 ± 7.8 nmol/ml, 0.667 ± 0.009 u/ml and 0.529 ± 0.007 µmol/l in the healthy group, these values decreased to respectively 147.83 ± 12.2 nmol/ml, 0.993 ± 0.004 u/ml and 0.204 ± 0.009 µmol/l levels in DCG. The differences were statistically significant (p<0.0001). However TPP prevented the reduction of the tGSH, GSHPx and GSHRd levels with desflurane as the respective levels of 317 ± 8.4 nmol/ml, 0.690 ± 0.006 u/ml and 0.492 ± 0.003 µmol/l (Figure 2). Again the difference between HG and TDG was found statistically significant (p<0.0001).
The effects of thiamine pyrophosphate on MDA, MPO and NO levels in rats administered desflurane. *p˂0.0001 according to HG group; **p˃0.05 according to HG group
The effects of thiamine pyrophosphate on tGSH, GSHPx and GSHRd levels in rats administered desflurane. *p˂0.0001 according to HG group
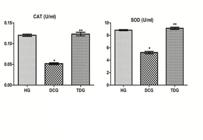
CAT and SOD values were respectively found as 0.120 ± 0.006 u/ml and 8.8 ± 0.21 u/ml in the healthy group. While desflurane increased the activities of CAT to 0.51 ± 0.05 u/ml (p<0.001), SOD value decreased to 5.2 ± 0.47 u/ml (p<0.0001) , The activity of CAT as 0.123 ± 0.01 u/ml and activity of SOD as 9.1 ± 0.44 u/ml were found in TDG (Figure 3).When we compared HG to TDG, these differences were statistically insignificant (p>0.05).
The effects of thiamine pyrophosphate on CAT and SOD levels in rats administered desflurane. *p˂0.0001 according to HG group; **p˃0.05 according to HG group
The activities of AST, ALT and LDH that are used to evaluate the liver function were respectively found as 29 ± 1.25 u/ml, 20 ± 1.39 u/ml and 151 ± 2,96 u/ml in the blood samples of the healthy group, desflurane changed the activities of AST, ALT and LDH as respective 274 ± 8,20 u/ml, 157 ± 13,35 u/ml and 630 ± 12,71 u/ml. Whereas these activities were found as 194 ± 4,62 u/ml, 86 ± 4,22 u/ml and 333 ± 16,71 u/ml in the blood samples of the animals treated with TPP (Figure 4). The differences between compared groups were statistically significant (p<0.0001).
The effects of thiamine pyrophosphate on AST, ALT and LDH levels in rats administered desflurane. *p˂0.0001 according to HG group
Histopathological results
The histopathological evaluations that were performed for all animals, were completely subjective up to the pathologist's evaluation. In Figure 5a, normal histological appearance of the liver tissues of the HG is monitored (H&E, x200). In Figure 5b, limited mild congestion, hemorrhage (long star), dilatation (star) and lymphocytic inflammation areas were observed in the liver tissue of the animals treated with TPP. However, there are multiple marked congestion, hemorrhage (long arrow) and dilatation (star) areas in the sinusoids of the liver tissues of DCG group which received desflurane (H&E, x200). Lymphocyte-rich inflammation signs (short arrow) are observed in the centrilobular and portal areas in Figure 5c. (H&E, x200).
Histopathological image of the liver tissues of the HG (a), TDG (b) and DCG (c) rat groups (H&E, x200)
Dıscussıon
In this study, the protective effect of TPP against hepatotoxicity induced with desflurane in rats, was biochemically and histopathologically investigated. The liver is the organ that is most affected by the toxic effects. The informations from the literature indicates that the incidence of desflurane induced hepatotoxicity appears to directly correlated with anesthetic metabolism catalysed by cytochrome P 450 2E1 to trifluoroacetylated proteins1717. Njoku D, Laster MJ, Gong DH, Eger EI, Reed GF, Martin JL. Biotransformation of halothane, enflurane, isoflurane, and desflurane to trifluoroacetylated liver proteins: association between protein acylation and hepatic injury. Anesth Analg. 1997; 84 (1):173-8. PMID: 8989020.. Still desflurane also has been reported to produce hepatotoxic effect by decreasing the amounts of vitamines that have antioxidant activity1818. Naziroglu M, Gunay C. The levels of some antioxidant vitamins, glutathione peroxidase and lipoperoxidase during the anaesthesia of dogs. Cell Biochem Funct. 1999;17(3):207-12. PMID: 10451542.. Eroglu et al .1919. Eroglu F, Yavuz L, Ceylan BG, Yilmaz F, Eroglu E, Delibas N, Naziroglu M. New volatile anesthetic, desflurane, reduces vitamin E level in blood of operative patients via oxidative stress. Cell Biochem Funct. 2010,28 (3):211-6. doi: 10.1002/cbf.1641.
https://doi.org/10.1002/cbf.1641...
reported that, desflurane causes oxidative stress by decreasing the value of vitamine- E in blood plasma. Erbas et al .2020. Erbas M, Demiraran Y, Yildirim HA, Sezen G, Iskender A, Karagoz I, Kandis H. Comparison of effects on the oxidant/antioxidat system of sevoflurane, desflurane and propofol infusion during general anesthesia. Rev Bras Anestesiol. 2015 Jan-Feb;65(1):68-72. doi: 10.1016/j.bjan. 2014.05.002.
https://doi.org/10.1016/j.bjan.2014.05.0...
stated that, total oxidant capacity is elevated in the patients who received desflurane. Sivaci R et al .2121. Sivaci R, Kahraman A, Serteser M, Sahin DA, Dilek ON. Cytotoxic effects of volatile anesthetics with free radicals undergoing laparoscopic surgery. Clin Biochem. 2006;39(3):293-8. doi: 10.1016/j.clinbiochem.2006.01.001.
https://doi.org/10.1016/j.clinbiochem.20...
found that oxidant and antioxidant defense mechanisms were resulted in superiority of oxidants in desflurane induced anesthesia. Similar to these studies the biochemical experimental results of our study demonstrated that desflurane increased the values of oxidants such as MDA, MPO and NO whereas reduced the values of antioxidants as tGSH, GSHPx, GSHRd, CAT and SOD in the liver tissues. These results showed that desflurane led to oxidative damage in the liver tissue.
Aslan et al .66. Arslan M, Ozkose Z, Akyol G, Barit G. The age- and gender-dependent effects of desflurane and sevoflurane on rat liver. Exp Toxicol Pathol. 2010;62(1):35-43. doi: 10.1016/j.etp.2008.12.011.
https://doi.org/10.1016/j.etp.2008.12.01...
reported that desflurane leads to oxidative stress in the rat liver by increasing lipid peroxidation. As is known, MDA is the final product of lipid peroxidation and the MDA value increases when the liver is damaged byoxidative stress99. Kisaoglu A, Ozogul B, Turan MI, Yilmaz I, Demiryilmaz I, Atamanalp SS, Bakan E, Suleyman H. Damage induced by paracetamol compared with N-acetylcysteine. J Chin Med Assoc. 2014;77(9):463-8. doi: 10.1016/j.jcma.2014.01.011.
https://doi.org/10.1016/j.jcma.2014.01.0...
. Again in our study desflurane was found to be increased the MDA value similar to the studies mentioned above.
There was no information in the literature about whether desflurane changes the activity of MPO in the liver or not. However, there are studies indicating that the agents which produce liver damage increases the activity of MPO99. Kisaoglu A, Ozogul B, Turan MI, Yilmaz I, Demiryilmaz I, Atamanalp SS, Bakan E, Suleyman H. Damage induced by paracetamol compared with N-acetylcysteine. J Chin Med Assoc. 2014;77(9):463-8. doi: 10.1016/j.jcma.2014.01.011.
https://doi.org/10.1016/j.jcma.2014.01.0...
1919. Eroglu F, Yavuz L, Ceylan BG, Yilmaz F, Eroglu E, Delibas N, Naziroglu M. New volatile anesthetic, desflurane, reduces vitamin E level in blood of operative patients via oxidative stress. Cell Biochem Funct. 2010,28 (3):211-6. doi: 10.1002/cbf.1641.
https://doi.org/10.1002/cbf.1641...
2222. Kunak CS, Kukula O, Mutlu E, Genç F, GüleçPeker G, Kuyrukluyıldız U, Binici O, Altuner D, Alp HH. The effect of etoricoxib on hepatic ıschemia-reperfusion ınjury in rats. Oxid Med Cell Longev. 2015;2015:598162. doi:10.1155/2015/598162.
https://doi.org/10.1155/2015/598162...
. Therefore we found that the desflurane increased the MPO activity in the liver tissue too.
Another parameter which increased in the liver tissue of the animals given desflurane is NO. NO reacts with ROS, producing peroxynitrite (ONOOH) which is a potent oxidant and this in turn lead to the formation of HO• radical through advanced decomposition2323. Cochrane CG. Cellular injury by oxidants. Am J Med. 1991;91(3C):23S-30. PMID: 1928208.. It has not been demonstrated whether NO has a role in the pathogenesis of the desflurane hepatotoxicity. However, it has been explained in the literature with experimental data that NO can cause hepatotoxicity2424. Khan MW, Priyamvada S, Khan SA, Khan S, Gangopadhyay A, Yusufi A. Fish/flaxseed oil protect against nitric oxide-induced hepatotoxicity and cell death in the rat liver.. Hum Exp Toxicol 2016 Mar;35(3):302-11. doi: 10.1177/0960327115586207
https://doi.org/10.1177/0960327115586207...
. The NO has been reported to initiate lipid peroxidation in the environment in which superoxide takes place2525. Hogg N, Kalyanaraman B. Nitric oxide and lipid peroxidation. Biochim Biophys Acta. 1999;1411:(2-3):378-84. PMID: 10320670.. We interpreted the increased NO value as an indication of oxidative stress in liver tissue like the other increased oxidants. Finally our experimental results and this informations from the literature demonstrate that, desflurane occured oxidative stress in the liver tissue. The levels of tGSH, GSHPx, GSHRd, CAT and SOD were found to be decreased in desflurane group and we reported that desflurane changed oxidant/antioxidant balance in the liver tissue in favour of oxidants. Consequently it is understood from the studies mentioned above that desflurane has negative effect on various antioxidant systems and these knowledges are consistenting with our results.
No studies were found in the literature screening about the protective effect of TPP against desflurane hepatotoxicity. However, TPP has been emphasized to protect the liver against paracetamol induced oxidative damage by increasing the values of GSH, GPO, GRx, GST, SOD and CAT and by decreasing the amounts of MDA and NO in the liver tissue99. Kisaoglu A, Ozogul B, Turan MI, Yilmaz I, Demiryilmaz I, Atamanalp SS, Bakan E, Suleyman H. Damage induced by paracetamol compared with N-acetylcysteine. J Chin Med Assoc. 2014;77(9):463-8. doi: 10.1016/j.jcma.2014.01.011.
https://doi.org/10.1016/j.jcma.2014.01.0...
. Another study reported that TPP significantly decreased MPO value which was increased with cisplatine in the cerebral tissue, in a dose dependent manner1010. Turan MI, Cayir A, Cetin N, Suleyman H, Siltelioglu Turan I, Tan H. An investigation of the effect of thiamine pyrophosphate on cisplatin-induced oxidative stress and DNA damage in rat brain tissue compared with thiamine: thiamine and thiamine pyrophosphate effects on cisplatin neurotoxicity. Hum Exp Toxicol. 2014;33(1):14-21. doi: 10.1177/0960327113485251.
https://doi.org/10.1177/0960327113485251...
. We found that the values of these antioxidant parameters increased in TPP administered group. Consequently, TTP whose protective effect was tested against desflurane hepatotoxicity, changed the oxidant/antioxidant balance in favour of antioxidants in the liver tissue.
According to the reducing of antioxidants in the desflurane group, activities of AST, ALT and LDH were found to be increased. These enzymes are the most recognized and used ones in order to evaluate the liver functions2626. Yabe Y, Kobayashi N, Nishihashi T, Takahashi R, Nishikawa M, Takakura Y, Hashida M. Prevention of neutrophil-mediated hepatic ischemia/reperfusion injury by superoxide dismutase and catalase derivatives. J Pharmacol Exp Ther. 2001;298(3):894-9. PMID: 11504782.. However these values reduced in TPP administration group and we interpreted these result as an improvement of liver tissue by preventing oxidative damage.
Fisher et al .2727. Fisher MR, Neiman HL. Periportal sinusoidal dilatation associated with pregnancy. Cardiovasc Intervent Radiol. 1984;7(6):299-302. PMID: 6529732. stated that, sinusoidal dilatation is a vascular lesion of the liver which is developed due to medicines2727. Fisher MR, Neiman HL. Periportal sinusoidal dilatation associated with pregnancy. Cardiovasc Intervent Radiol. 1984;7(6):299-302. PMID: 6529732.. Arslan et al .66. Arslan M, Ozkose Z, Akyol G, Barit G. The age- and gender-dependent effects of desflurane and sevoflurane on rat liver. Exp Toxicol Pathol. 2010;62(1):35-43. doi: 10.1016/j.etp.2008.12.011.
https://doi.org/10.1016/j.etp.2008.12.01...
found that, neutrophil and leucocyte infiltration was increased in the liver damage caused by desflurane. As mentioned above, no information was found in the literature demonstrating about the protective effect of TPP against oxidative damage by desflurane in the liver tissue. However, it has been stated that, TPP improved the histopathological damage in the rat liver such as dilatation, inflammation, necrosis and apoptosis caused by methotrexate2828. Demiryilmaz I, Sener E, Cetin N, Altuner D, Akcay F, Suleyman H. A comparative investigation of biochemical and histopathological effects of thiamine and thiamine pyrophosphate on ischemia-reperfusion induced oxidative damage in rat ovarian tissue. Arch Pharm Res. 2013;36(9):1133-9. doi: 10.1007/s12272-013-0173-8.
https://doi.org/10.1007/s12272-013-0173-...
. The intense congestion, hemorrhage and dilatation areas were observed in the liver sinusoids and lymphocyte-rich inflammation in the centrilobular and portal areas for desflurane group with changed oxidant/antioxidant balance in favour of oxidant. These histopathological findings indicate a vascular damage occurred in the liver tissue. Whereas the use of TPP has led to improvement in histopathological findings. This is understandable when demonstrated Figure 5b and those results are consistent with literature.
Conclusions
The desflurane leads to oxidative stress that causes liver damage of rats. Thiamine pyrophosphate can prevent desflurane induced oxidative stress and the liver dysfunction. Consequently TPP may be useful for the prophylaxis of hepatotoxicity induced with desflurane.
References
-
1Pandit SK, Green CR. General anesthetic techniques. Int Anesthesiol Clin. 1994;32(3):55-79. PMID: 7960175.
-
2Safari S, Motavaf M, Seyed Siamdoust SA, Alavian SM. Hepatotoxicity of halogenated inhalational anesthetics. Iran Red Crescent Med J. 2014;16(9):e20153. doi: 10.5812/ircmj.20153.
» https://doi.org/10.5812/ircmj.20153. -
3Martin JL, Plevak DJ, Flannery KD, Charlton M, Poterucha JJ, Humphreys CE, Derfus G, Pohl LR. Hepatotoxicity after desflurane anesthesia. Anesthesiology. 1995;83(5):1125-9. PMID: 7486167.
-
4Tung D, Yoshida EM, Wang CS, Steinbrecher UP. Severe desflurane hepatotoxicity after colon surgery in an elderly patient. Can J Anaesth. 2005;52(2):133-6. doi: 10.1007/BF03027717.
» https://doi.org/10.1007/BF03027717. -
5Chin MW, Njoku DB, MacQuillan G, Cheng WS, Kontorinis N. Desflurane-induced acute liver failure. Med J Aust. 2008;189(5):293-4. PMID: 18759731.
-
6Arslan M, Ozkose Z, Akyol G, Barit G. The age- and gender-dependent effects of desflurane and sevoflurane on rat liver. Exp Toxicol Pathol. 2010;62(1):35-43. doi: 10.1016/j.etp.2008.12.011.
» https://doi.org/10.1016/j.etp.2008.12.011. -
7Mustafa A, Berrin I, Yusuf U, Nurlu N, Zerrin O, Metin CF, Isin G, Bekir DC, Nedim C, Mustafa K. Effects of sevoflurane and desflurane on oxidant/antioxidant status of young versus old rat liver tissues. J Anim Vet Adv. 2008;7(11):1345-51. doi: javaa.2008.1345.1351.
-
8Yilmaz I, Demiryilmaz I, Turan MI, Cetin N, Gul MA, Suleyman H. The effects of thiamine and thiamine pyrophosphate on alcohol-induced hepatic damage biomarkers in rats. Eur Rev Med Pharmacol Sci. 2015;19(4):664-70. PMID: 25753885.
-
9Kisaoglu A, Ozogul B, Turan MI, Yilmaz I, Demiryilmaz I, Atamanalp SS, Bakan E, Suleyman H. Damage induced by paracetamol compared with N-acetylcysteine. J Chin Med Assoc. 2014;77(9):463-8. doi: 10.1016/j.jcma.2014.01.011.
» https://doi.org/10.1016/j.jcma.2014.01.011. -
10Turan MI, Cayir A, Cetin N, Suleyman H, Siltelioglu Turan I, Tan H. An investigation of the effect of thiamine pyrophosphate on cisplatin-induced oxidative stress and DNA damage in rat brain tissue compared with thiamine: thiamine and thiamine pyrophosphate effects on cisplatin neurotoxicity. Hum Exp Toxicol. 2014;33(1):14-21. doi: 10.1177/0960327113485251.
» https://doi.org/10.1177/0960327113485251. -
11Yapca OE, Turan MI, Borekci B, Akcay F, Suleyman H. Bilateral ovarian ischemia/reperfusion injury and treatment options in rats with an induced model of diabetes. Iran J Basic Med Sci. 2014;17(4):294-302. PMID: 24904723.
-
12Bradley PP, Priebat DA, Christensen RD, Rothstein G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol. 1982;78(3):206-9. PMID: 6276474.
-
13Moshage H, Kok B, Huizenga JR, Jansen PL. Nitrite and nitrate determinations in plasma: a critical evaluation. Clin Chem. 1995;41(6):892-6. PMID: 7768008.
-
14Beutler E. General aspects of erythrocyte physiology and biochemistry. Exp Eye Res. 1971;11(3):261-3. PMID: 5166266.
-
15Aebi H. Catalase in vitro. Methods Enzymol. 1984;105:121-6. PMID: 6727660.
-
16Sun Y, Oberley LW, Li Y. A simple method for clinical assay of superoxide dismutase.. Clin Chem 1988;34(3):497-500. PMID: 3349599.
-
17Njoku D, Laster MJ, Gong DH, Eger EI, Reed GF, Martin JL. Biotransformation of halothane, enflurane, isoflurane, and desflurane to trifluoroacetylated liver proteins: association between protein acylation and hepatic injury. Anesth Analg. 1997; 84 (1):173-8. PMID: 8989020.
-
18Naziroglu M, Gunay C. The levels of some antioxidant vitamins, glutathione peroxidase and lipoperoxidase during the anaesthesia of dogs. Cell Biochem Funct. 1999;17(3):207-12. PMID: 10451542.
-
19Eroglu F, Yavuz L, Ceylan BG, Yilmaz F, Eroglu E, Delibas N, Naziroglu M. New volatile anesthetic, desflurane, reduces vitamin E level in blood of operative patients via oxidative stress. Cell Biochem Funct. 2010,28 (3):211-6. doi: 10.1002/cbf.1641.
» https://doi.org/10.1002/cbf.1641 -
20Erbas M, Demiraran Y, Yildirim HA, Sezen G, Iskender A, Karagoz I, Kandis H. Comparison of effects on the oxidant/antioxidat system of sevoflurane, desflurane and propofol infusion during general anesthesia. Rev Bras Anestesiol. 2015 Jan-Feb;65(1):68-72. doi: 10.1016/j.bjan. 2014.05.002.
» https://doi.org/10.1016/j.bjan.2014.05.002 -
21Sivaci R, Kahraman A, Serteser M, Sahin DA, Dilek ON. Cytotoxic effects of volatile anesthetics with free radicals undergoing laparoscopic surgery. Clin Biochem. 2006;39(3):293-8. doi: 10.1016/j.clinbiochem.2006.01.001.
» https://doi.org/10.1016/j.clinbiochem.2006.01.001. -
22Kunak CS, Kukula O, Mutlu E, Genç F, GüleçPeker G, Kuyrukluyıldız U, Binici O, Altuner D, Alp HH. The effect of etoricoxib on hepatic ıschemia-reperfusion ınjury in rats. Oxid Med Cell Longev. 2015;2015:598162. doi:10.1155/2015/598162.
» https://doi.org/10.1155/2015/598162 -
23Cochrane CG. Cellular injury by oxidants. Am J Med. 1991;91(3C):23S-30. PMID: 1928208.
-
24Khan MW, Priyamvada S, Khan SA, Khan S, Gangopadhyay A, Yusufi A. Fish/flaxseed oil protect against nitric oxide-induced hepatotoxicity and cell death in the rat liver.. Hum Exp Toxicol 2016 Mar;35(3):302-11. doi: 10.1177/0960327115586207
» https://doi.org/10.1177/0960327115586207 -
25Hogg N, Kalyanaraman B. Nitric oxide and lipid peroxidation. Biochim Biophys Acta. 1999;1411:(2-3):378-84. PMID: 10320670.
-
26Yabe Y, Kobayashi N, Nishihashi T, Takahashi R, Nishikawa M, Takakura Y, Hashida M. Prevention of neutrophil-mediated hepatic ischemia/reperfusion injury by superoxide dismutase and catalase derivatives. J Pharmacol Exp Ther. 2001;298(3):894-9. PMID: 11504782.
-
27Fisher MR, Neiman HL. Periportal sinusoidal dilatation associated with pregnancy. Cardiovasc Intervent Radiol. 1984;7(6):299-302. PMID: 6529732.
-
28Demiryilmaz I, Sener E, Cetin N, Altuner D, Akcay F, Suleyman H. A comparative investigation of biochemical and histopathological effects of thiamine and thiamine pyrophosphate on ischemia-reperfusion induced oxidative damage in rat ovarian tissue. Arch Pharm Res. 2013;36(9):1133-9. doi: 10.1007/s12272-013-0173-8.
» https://doi.org/10.1007/s12272-013-0173-8.
-
Financial source: Erzincan University Scientific Research Projects Department (Project nº SAG-B-080715-0173)
-
1
Research performed at Recep Tayyip Erdoğan University, Medical Experimental Research and Application Center, Rize, Turkey
Publication Dates
-
Publication in this collection
Mar 2016
History
-
Received
21 Nov 2015 -
Reviewed
20 Jan 2016 -
Accepted
19 Feb 2016